Abstract
Platelets may selectively execute apoptosis (PL-Apo), activation (PL-Act), and both or no responses when exposed to different chemical agents, shear stresses, and stored under blood banking conditions. Appropriate diagnosis of PL-Apo is an important issue of platelet physiology investigations. However, in diagnosing PL-Apo, there is a risk of a
Introduction
Appropriate diagnosis of PL-Apo and distinguishing PL-Apo from PL-Act is an important issue of platelet physiology investigations. However, in diagnosing PL-Apo, there is a risk of
Two difficulties complicate an appropriate PL-Apo diagnosis. First, anucleate platelets appear in the circulation as a result of fragmentation from megakaryocytes, a process that may involve apoptosis in platelet precursor cells megakaryocytes. 1 –3 Second, exposure of phosphatidylserine (PS) on the platelet surface may be involved in 2 platelet reactions: PL-Act facilitating blood coagulation and PL-Apo facilitating platelet clearance from the circulation by phagocytosis (reviewed in Leytin and Gyulkhandanyan et al 4,5 ). The goal of the current article is, based on analysis of relevant publications, to present recommendations that may help to avoid incorrect PL-Apo diagnosis.
We will discuss the following questions relevant to diagnosis of PL-Apo in unknown experimental and clinical settings: (1) what platelet preparation should be used for minimizing artificial stimulation of PL-Apo during purification of platelets from red and white blood cells; (2) how to distinguish PL-Apo from PL-Act and what markers should be used for characterizing
Platelet Isolation Procedure
The platelet isolation procedure may potentially lead to a
Experimental and Clinically Relevant Cases of Selective or Concurrent Stimulation of PL-Apo and PL-Act
The cases described in Figure 1 indicate clinical and experimental conditions for diagnosing PL-Apo, distinguishing it from PL-Act. This figure demonstrates the considerable diversity of PL-Apo and PL-Act responses in the following 7 cases.
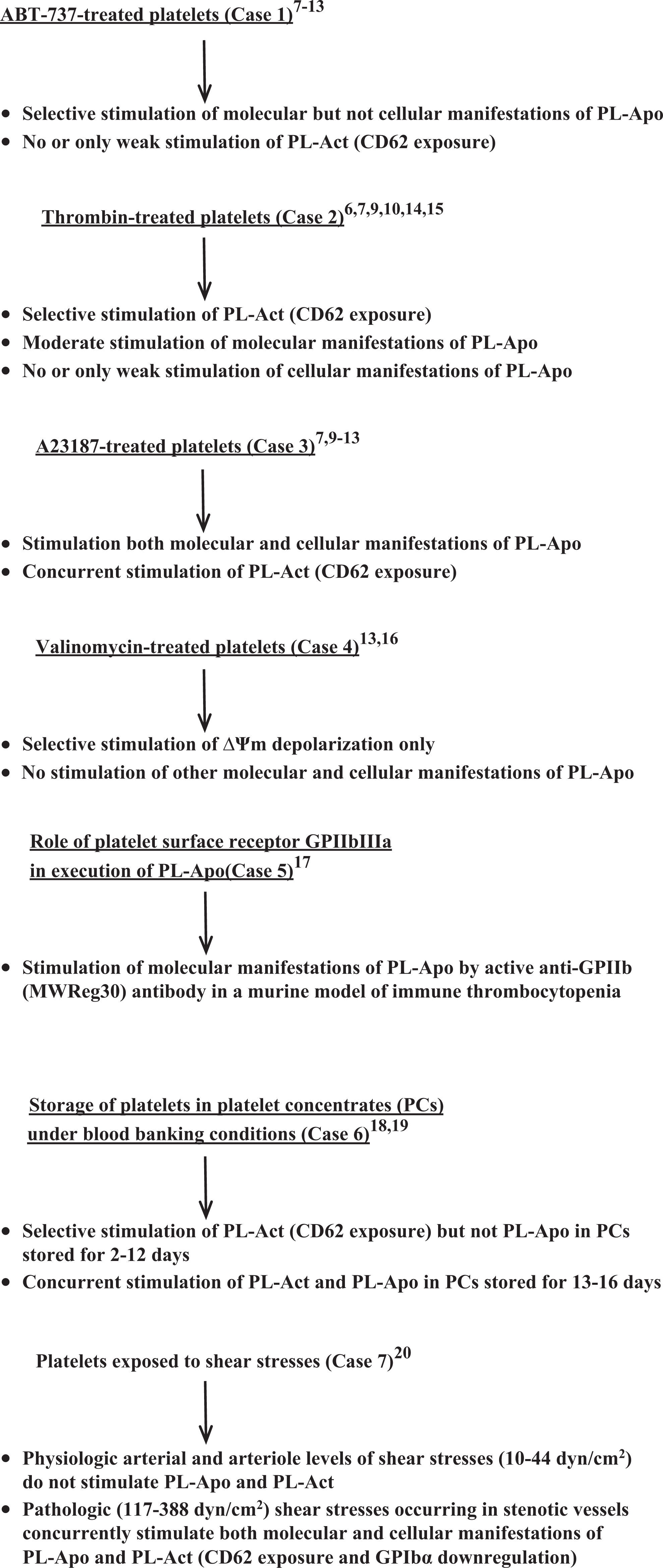
Stimulation of platelet apoptosis (PL-Apo), platelet activation (PL-Act), and both platelet responses: illustrative experimental and clinically relevant cases. In the
In cases 1 to 4, platelets were treated in vitro with chemical agents (proapoptotic BH3 mimetic ABT-737, platelet agonist thrombin, calcium ionophore A23187, and potassium ionophore valinomycin) and analyzed at different doses, time, and temperature conditions. Cases 5 to 7 are illustration of clinically relevant cases.
ABT-737-Treated Platelets (Case 1)
Treatment of platelets with ABT-737 is strongly dose-, time-, and temperature-dependent. At optimal 30 μmol/L ABT-737 dose,
8
prolonged treatment for 90 minutes at 37oC, rather than at room temperature (RT), is required to achieve the maximal level of
Thrombin-Treated Platelets (Case 2)
In contrast, in thrombin-treated platelets, PL-Act (CD62 exposure), are selectively induced, compared to PL-Apo, in a wide range of thrombin doses from 0.05 to 1 U/mL, and this selective induction of PL-Act is persistent for 15 to 90 minutes at either 37oC or RT.
6,7,9,10,14
However, a thrombin dose titration study up to a very high dose of 10 U/mL has demonstrated that the percentage of cells with depolarized ΔΨm, activated caspase-3, and exposed PS is progressively increased up to 30% to 40%, whereas CD62 exposure is reached to the maximal level of 87% to 93% at the lowest thrombin doses 0.05 to 0.10 U/mL when only 4% to 12% platelets undergo apoptosis.
14
Thrombin (1.0 U/mL) also induces proapoptotic Bax and Bak expression and activation
6,15
but does not induce or only weakly induces
A23187-Treated Platelets (Case 3)
Optimal conditions for A23187-treated platelets are dose- dependent but time- and temperature-independent. At saturating A23187 dose of 10 μmol/L,
10
and incubation for 15 to 90 minutes at 37oC or RT both
Valinomycin-Treated Platelets (Case 4)
Case 4 is a notable example when the treatment of platelets with potassium ionophore valinomycin at optimal dose of 1 μmol/L induces strong ΔΨm depolarization in almost all cells after short-term 15-minute incubation of platelets at RT, whereas other
Role of GPIIbIIIa in Execution of PL-Apo (Case 5)
Case 5 illustrates the role of platelet surface receptor glycoprotein IIbIIIa (GPIIbIIIa) in PL-Apo. Injection of active agonistic anti-GPIIb antibody (MWReg30) significantly induces
Platelet Storage under Blood Banking Conditions (Case 6)
Platelet storage lesion is a serious problem limiting the clinical use of platelet concentrates (PCs). 18 Case 6 elucidates the contribution of PL-Act and PL-Apo to the platelet storage lesion in PCs stored at 22oC under blood banking conditions. PL-Act (CD62 exposure) is selectively induced in comparison to PL-Apo in prestorage-leukoreduced PCs prepared by PRP method during conventional (2-5 days), extended (6-8 days), and long-term (11-12 days) storage of PCs with the mean percentage of CD62-positive cells 38.6%, 66.5%, and 92.0%, respectively. In contrast, PL-Apo markers (ΔΨm depolarization, caspase-3 activation, PS exposure, and microparticle formation) are not induced or only slightly (5%-11%) induced during platelet storage for 2 to 12 days. However, a long-term storage for 13 to 16 days results in concurrent stimulation of PL-Act and PL-Apo. 19 These data indicate that during platelet storage, PL-Act is triggered much earlier and stronger than PL-Apo, and PL-Act rather than PL-Apo mostly contributes to the platelet storage lesion during conventional and extended storage of PCs. Notably, if storage of PCs is limited to 12 days without subsequent storage for 13 to 16 days, it may lead to erroneous PL-Apo diagnosis and only determination of PL-Apo and PL-Act markers for all storage days helps to avoid false-negative and false-positive PL-Apo diagnosis.
Platelets Exposed to Shear Stresses (Case 7)
Case 7 describes the study in which human PRP was exposed to shear stresses for 90 seconds at 37oC generated by cone-and-plate device.
20
Platelets subjected to physiologic levels of shear stresses occurring in arteries and arterioles (10-44 dyn/cm2) and pathologic high levels characteristic for stenotic vessels (117-388 dyn/cm2) have been examined for
Conclusions
In summary, we recommend the following procedures for avoiding
(1) Platelet-rich plasma, rather than isolated platelets, should be used to minimize artificial enhancement of PL-Apo induced by platelet isolation stress, which may result in a
(2) Since different doses, times, temperatures, and shear stresses are required for obtaining the maximal levels of PL-Apo and/or PL-Act induced by different triggers, a thorough elaboration of optimal conditions for stimulation of PL-Apo and PL-Act, separately or concurrently, is required by performing dose, time, temperature, and shear stress titrations in wide titration ranges. Detection of PL-Apo in nonoptimized conditions may lead to a
(3) Diagnosis of PL-Apo
(4) Finally, A23187-treated and diluent buffer-treated platelets, and appropriate case-specific positive and negative controls should be used for quantification of PL-Apo and PL-Act responses.
Footnotes
Acknowledgments
The authors thank D.J. Allen, S. Mykhaylov, A. Mutlu, J.W. Semple, A.H. Lazarus, E. Lyubimov, H. Ni and B. Garvey for research cooperation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the grant to VL from the Platelet Research Fund of Ronya Beskin, Israel.
