Abstract
Excessive perioperative bleeding remains a substantial problem. Factor XIII (FXIII) contributes to clot stability, and it has therefore been suggested that supplementation with FXIII concentrate may improve perioperative hemostasis. We evaluated the effects of increasing doses of FXIII, alone or in combination with fibrinogen or platelet concentrate, in blood samples from 2 considerably different groups of surgical patients: cardiac and scoliosis surgery patients. Whole-blood samples were collected immediately after operation from cardiac and scoliosis surgery patients. The samples were supplemented with 3 clinically relevant doses of FXIII concentrate (+20%, +40%, and +60%), alone or in combination with a fixed dose of fibrinogen concentrate (+1.0 g/L) or fresh apheresis platelets (+92 × 109/L). Clot formation was assessed with rotational thromboelastometry (ROTEM). When the highest dose of FXIII concentrate was added, EXTEM clotting time was shortened by 10% in both cardiac and scoliosis surgery patients (95% confidence intervals: 2.4%-17% and 3.3%-17%, respectively), and FIBTEM maximum clot firmness was increased by 25% (9.3%-41%) in cardiac patients, relative to baseline. When fibrinogen was added, the dose-dependent effect of FXIII on clot stability was maintained, but the total effect was markedly greater than with FXIII alone, +150% (100%-200%) and +160% (130%-200%) for the highest FXIII dose in cardiac and scoliosis patients, respectively. Ex vivo supplementation with clinically relevant doses of FXIII improved clot formation moderately in blood samples from cardiac and scoliosis surgery patients, both alone and when given in combination with fibrinogen or platelet concentrate.
Introduction
Excessive blood loss remains a significant problem during and after major surgical procedures such as spine and cardiac surgery. 1 –3 The bleeding is multifactorial and may depend on patient factors such as poor liver and kidney function and antithrombotic medication, procedural factors, and impaired perioperative hemostasis caused by large bleeding and fluid replacement with crystalloids and colloids, that is, hemodilution. In cardiac surgery, the use of cardiopulmonary bypass (CPB) with exposure of blood to nonendothelial surfaces may also contribute.
Bleeding and hemodilution reduce the plasma concentration of coagulation factors. It has previously been shown that fibrinogen is the first coagulation factor to reach critically low plasma levels in patients with ongoing bleeding, 4 but the concentration of other coagulation factors may also be insufficient when the hemostasis is challenged from major bleeding. We, and others, have previously shown a significant correlation between coagulation factor XIII (FXIII) plasma activity and bleeding volume after cardiac surgery, 5 –7 and it has been demonstrated that FXIII activity is reduced in surgical patients with unexpected bleeding. 8 In another study, patients were randomized to receive either FXIII substitution or placebo at the start of gastrointestinal cancer surgery. The decrease in clot stability and the blood loss during surgery were significantly less in the group that received FXIII concentrate. 9 Accordingly, current guidelines on the treatment of perioperative bleeding state that FXIII supplementation can be considered in patients with ongoing bleeding. 10,11
However, little is known about the functional effects of FXIII supplementation in surgical patients and the relationship between dose and response. Furthermore, it is not known whether potential effects are reduced, maintained, or enhanced when FXIII is combined with other prohemostatic blood products such as fibrinogen and platelet concentrates. The aim of the present ex vivo study was to investigate the effects of FXIII concentrate on clot formation in blood samples collected immediately after surgery in 2 groups of surgical patients with major differences, but both of which have a relatively high risk of large perioperative bleeding—cardiac and scoliosis surgery patients.
Materials and Methods
Patients
Nine patients undergoing cardiac surgery with CPB and 10 patients undergoing scoliosis surgery were included from February to October 2012 and from February to May 2014. Patient characteristics are given in Table 1. All patients gave written informed consent. The inclusion criteria were elective coronary artery bypass grafting procedure for the cardiac patient group and idiopathic scoliosis in otherwise healthy individuals for the scoliosis patient group. Exclusion criteria for the cardiac patients were preoperative medication affecting coagulation, P2Y12 platelet inhibition within 5 days before surgery, liver or kidney dysfunction, and age <18. Exclusion criterion for the scoliosis patients was ongoing medication affecting hemostasis. The study was approved by the regional research ethics committee and performed according to the World Medical Association Declaration of Helsinki.
Patient Demographics and Results of Preoperative and Postoperative Blood Analyses.a
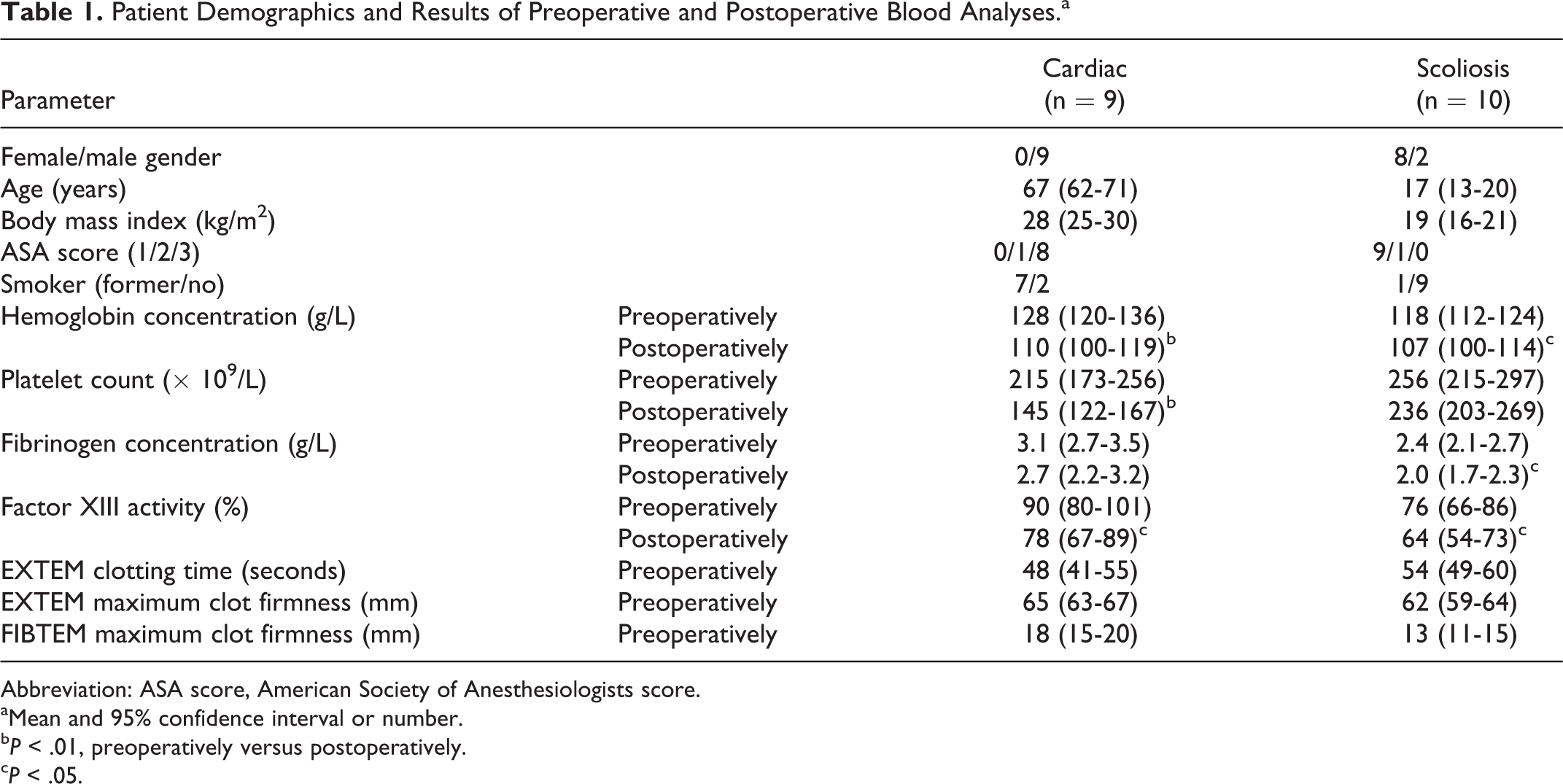
Abbreviation: ASA score, American Society of Anesthesiologists score.
aMean and 95% confidence interval or number.
b
c
Clinical Management
Cardiac patients
All cardiac surgery patients were operated with CPB. Before cannulation, heparin (Lövens, Ballerup, Denmark), 300 IU/kg, was given and supplemented as required to maintain an activated clotting time of more than 480 seconds. Cardiopulmonary bypass was performed with a hollow fiber membrane oxygenator. All patients received tranexamic acid, 2 g before surgery and 2 g after skin closure. Aprotinin was not used in any patient. Clopidogrel or ticagrelor was discontinued at least 5 days before surgery. The operations were performed with standard nonpulsatile CPB technique in normothermia and hemodilution (hematocrit 20%-30%). Cardioprotection was achieved with cold blood cardioplegia. Weaning off CPB was performed at a temperature of at least 36°C. Protamine (1 mg protamine per 100 U of heparin) was given to reverse the effect of heparin.
Scoliosis patients
All scoliosis surgery patients were operated under total intravenous anesthesia. None of the patients were on any medication that would influence coagulation or platelet function. Five patients received tranexamic acid perioperatively. All patients received thrombosis prophylaxis with dalteparin (Pfizer Inc, New York), 5000 IU, with the first dose being given the evening before surgery.
Study Design
Preoperative blood samples from cardiac surgery patients were collected after the induction of anesthesia but before the initiation of surgery, and postoperative samples were collected when the patient was weaned from CPB and after heparin had been neutralized with protamine. Blood samples from scoliosis surgery patients were collected preoperatively after the induction of anesthesia but before the initiation of surgery and postoperatively immediately after surgery was completed. All samples were collected from an arterial line, except in 2 scoliosis surgery patients where sampling was performed from a peripheral vein catheter. Samples were collected in citrated tubes (0.129 M citrate, volume 2.7 mL) for analysis of fibrinogen plasma concentration and FXIII activity. The blood was centrifuged for 20 minutes at 2000
Preoperatively, clot formation was analyzed in citrated whole blood (0.129 M citrate) without any additives. Postoperatively, 10 different samples were prepared for each study patient: 1 baseline and 3 with increasing doses of FXIII concentrate (Fibrogammin; CSL Behring, Marburg, Germany; +20%, +40%, and +60%), alone or in combination with a fixed dose of fibrinogen concentrate (+1.0 g/L, Riastap; CSL Behring) or freshly prepared apheresis platelets (+92 × 109/L) from the institutional blood bank. The doses of FXIII, fibrinogen, and platelet concentrates correspond to clinically relevant doses of approximately 1050 to 3150 IU FXIII concentrate, 3 g fibrinogen concentrate, and 3 U single-donor apheresis platelets to a patient weighing 70 kg. 12 Phosphate-buffered saline (140 mM NaCl, 10 mM Na3PO4, pH 7.4) was used in different volumes to maintain the same hemodilution in the samples. All samples for clot formation analysis had a total volume of 987 µL, 760 µL of which was citrated whole blood.
Analyses
Clot formation was assessed with rotational thromboelastometry (ROTEM; Pentapharm GmbH, Munich, Germany). Technical details have been given elsewhere. 13 The ROTEM assays, EXTEM and FIBTEM, were used for each sample. EXTEM analyzes clot formation activated with tissue factor. FIBTEM uses the same activator but eliminates the platelet contribution by adding cytochalasin D to the sample, in order to highlight the effect of fibrin polymerization. Clotting time and maximum clot firmness are reported for the EXTEM analysis, and maximum clot firmness is reported for the FIBTEM analysis.
Fibrinogen concentration and FXIII activity were analyzed at the accredited coagulation laboratory at Sahlgrenska University Hospital. The laboratory participates in the external quality assessment program of the ECAT Foundation (www.ecat.nl). Plasma concentration of fibrinogen (reference range: 2.0-4.5 g/L) was measured by the modified method of Clauss. 14 Plasma activity of FXIII (reference range: 70%-140%) was analyzed with the Berichrom FXIII assay (Dade Behring, Marburg, Germany) on the BCS XP instrument (Siemens Healthcare GmbH, Erlangen, Germany). Hemoglobin concentration, hematocrit, and platelet count were analyzed using standard clinical methods.
Statistics
A sample size of 8 patients would give us a power of 80% with a 2-sided paired test to detect a difference of 2 mm in FIBTEM maximum clot firmness, with a significance level of 0.05 and a standard deviation of 2 mm. A 2-mm increase corresponds to an increase of approximately 20% in FIBTEM maximum clot firmness in postoperative samples from cardiac surgery patients.
15,16
Patient characteristics, baseline parameters, and thromboelastometry results are presented as mean with 95% confidence interval, unless stated otherwise. Related-samples Wilcoxon signed rank test was used for comparisons of baseline parameters within groups. To evaluate the difference in thromboelastometry results between the blood samples prepared with various concentrations of FXIII concentrate, alone or together with fibrinogen or platelet concentrate, a mixed model was used. Treatment and baseline value were defined as fixed effects, and dose was included as a random effect. Hence, the repeated effect of dose was estimated with a random slope. Any
Results
General
All patients recovered as expected after surgery. None of the patients received intraoperative transfusion of blood products. The median amount of perioperative bleeding was 880 mL (range: 510-1410 mL) in the cardiac group and 695 mL (range: 470-1185 mL) in the scoliosis group.
Baseline Variables
Hemoglobin concentration and FXIII activity were reduced in both groups of patients after surgery (Table 1). Platelet count was only significantly reduced in cardiac surgery patients, and fibrinogen concentration was only significantly reduced in scoliosis surgery patients.
Factor XIII Supplementation
Cardiac surgery patients—Clotting time
In the mixed-model analysis, supplementation with FXIII concentrate alone resulted in a shortened EXTEM clotting time for the highest FXIII dose by 8.2 seconds (95% confidence interval: 1.9-15 seconds;
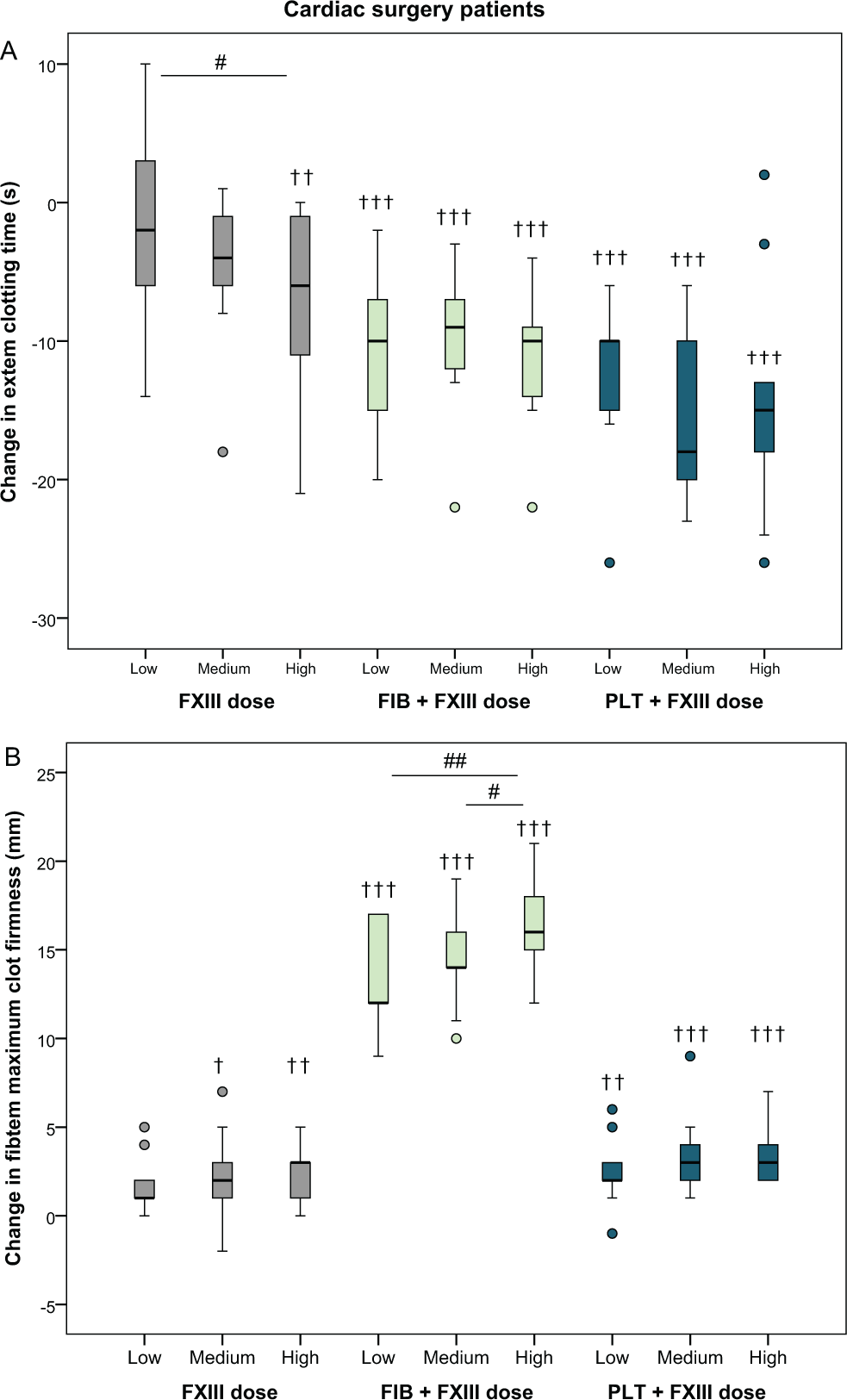
Absolute changes in EXTEM clotting time and FIBTEM maximum clot firmness after the addition of increasing doses of factor XIII (+20%, +40%, and +60%) in blood samples from cardiac surgery patients (n = 9), alone or in combination with a fixed dose of fibrinogen or platelets. See Methods section for details. †
Results of Clot Formation Analyses of Blood Samples From Cardiac and Scoliosis Surgery Patients.a
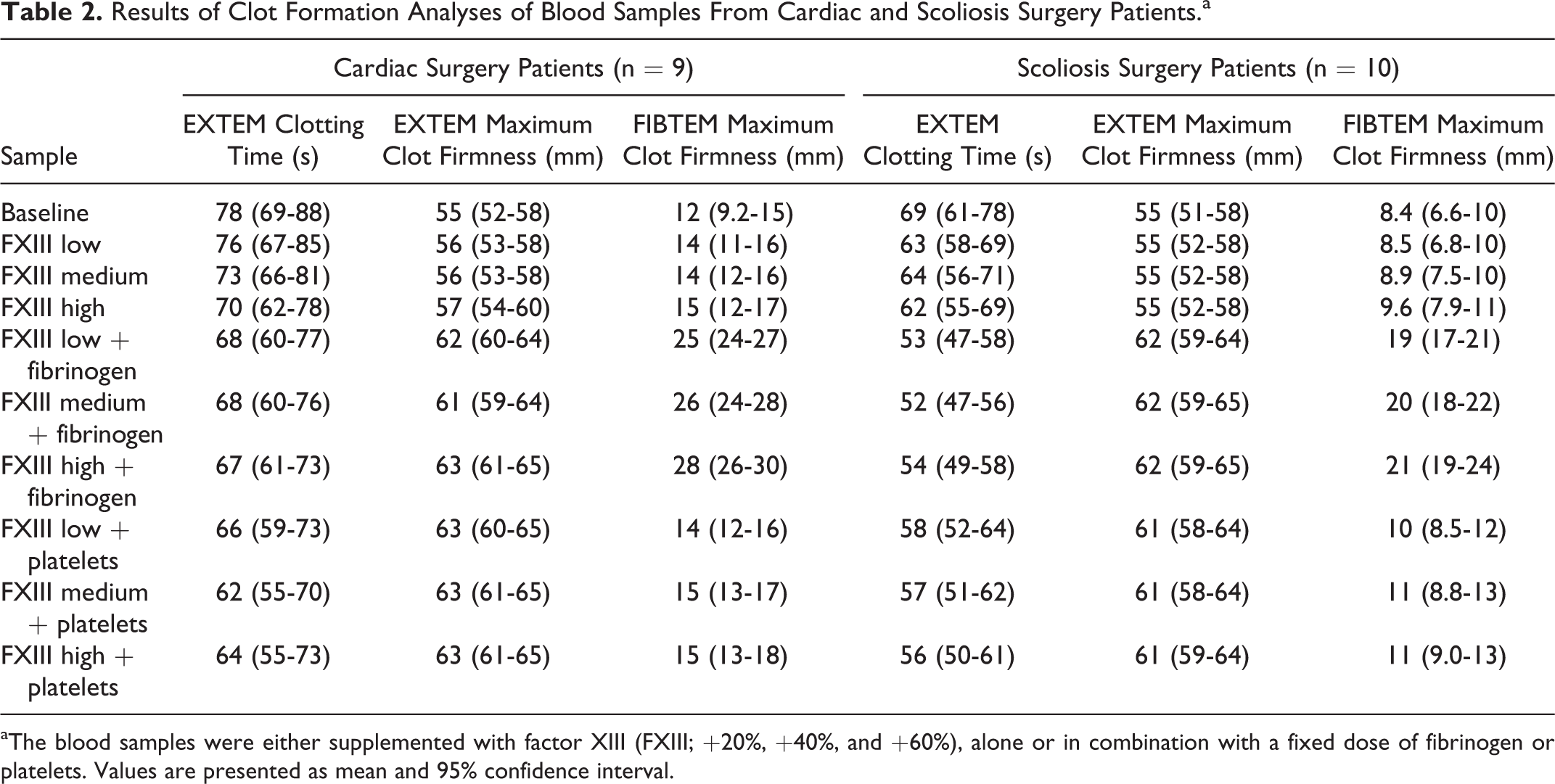
aThe blood samples were either supplemented with factor XIII (FXIII; +20%, +40%, and +60%), alone or in combination with a fixed dose of fibrinogen or platelets. Values are presented as mean and 95% confidence interval.
Cardiac surgery patients—Clot stability
There was a significant increase in FIBTEM maximum clot firmness for the medium dose and the highest dose of FXIII alone compared to baseline (+2.0 mm [−0.14 to 4.1 mm],
Scoliosis surgery patients—Clotting time
Supplementation with increasing doses of FXIII concentrate resulted in a significant decrease in EXTEM clotting time of 7.5 seconds (1.8-13 seconds,
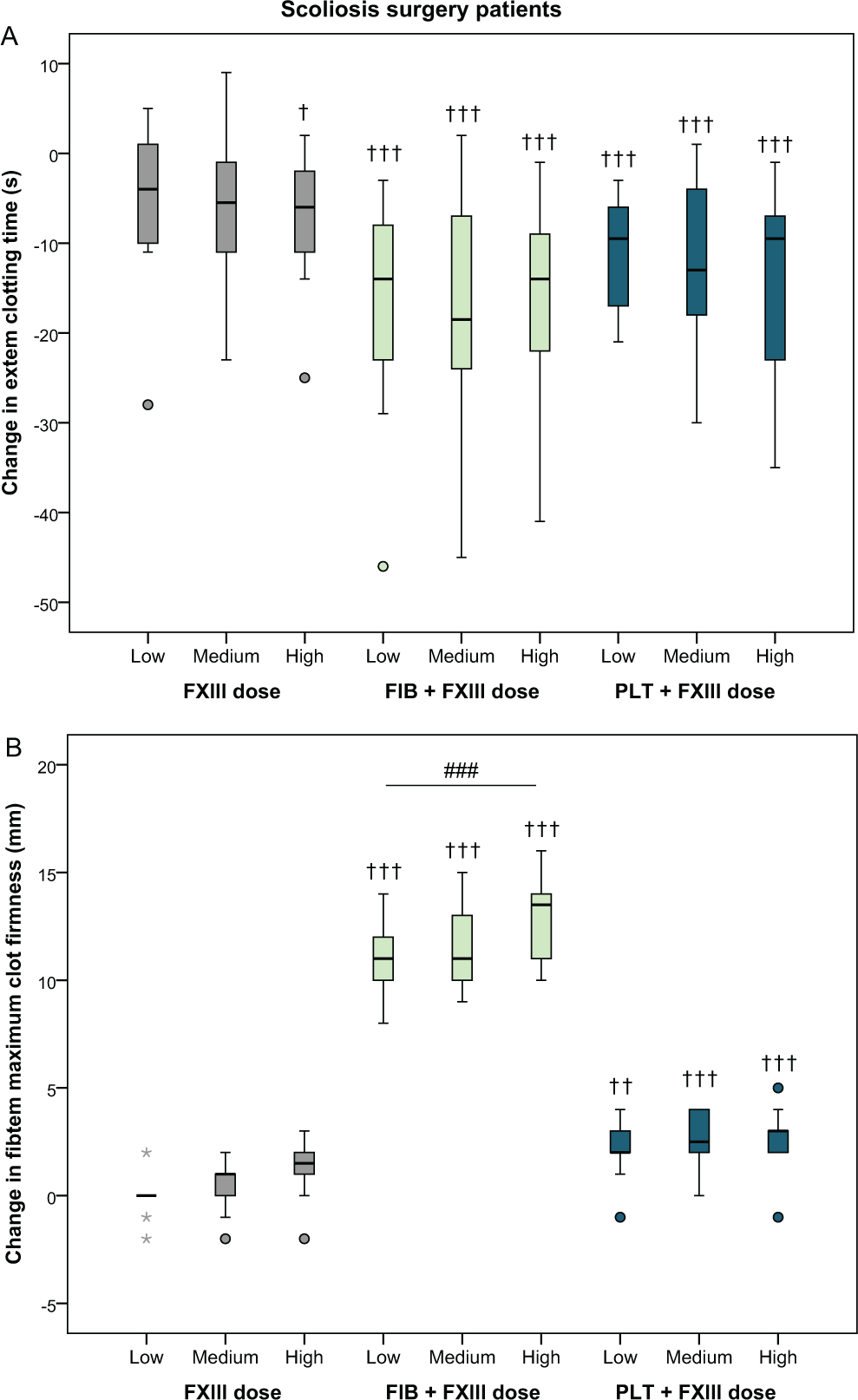
Absolute changes in EXTEM clotting time and FIBTEM maximum clot firmness after the addition of increasing doses of factor XIII (+20%, +40%, and +60%) in blood samples from scoliosis surgery patients (n = 10), alone or in combination with a fixed dose of fibrinogen or platelets. See Methods section for details. †
Scoliosis surgery patients—Clot stability
In FIBTEM maximum clot firmness, fibrinogen enhanced the effect of FXIII compared to FXIII alone (Figure 2B and Table 2). There was no significant change between the lowest and highest dose of FXIII alone (+1.1 mm [0.1-2.1 mm],
Discussion
The main findings of the present study were as follows. (1) Clinically relevant doses of FXIII improved clot formation in blood samples from cardiac surgery patients and scoliosis surgery patients. (2) The effects of FXIII alone were markedly lower than when FXIII was combined with fibrinogen or platelet concentrate. (3) The dose-dependent effect of FXIII on FIBTEM maximum clot firmness was enhanced when increasing doses of FXIII were added together with fibrinogen.
Several previous studies have investigated the effect of FXIII supplementation on clot formation in whole-blood samples. 17 –31 The present study differs from the previous studies in 2 important aspects. Firstly, we used whole-blood samples from 2 categories of patients with a high risk of bleeding, cardiac surgery patients and scoliosis surgery patients. The samples were collected immediately after surgery, that is, when the coagulation would be expected to be most disturbed. Most previous studies have used samples from healthy volunteers or blood donors and then diluted the samples with crystalloids or colloids to induce coagulopathy—and/or added tissue plasminogen activator to cause fibrinolysis. 18,20 –24,26,27,29,31 Secondly, we tested clinically relevant doses of FXIII, +20% to +60%, added alone or in combination with 2 other hemostatic agents, fibrinogen and platelets. These are blood products that may be used in the clinical setting to treat bleeding complications when impairment of clot stability is confirmed or suspected. This setup differs from other studies where supraphysiological doses of FXIII concentrate have been added to blood samples from intensive care patients 17 or to diluted samples from healthy volunteers. 18,22,23,27
The previous studies that have investigated the effect of FXIII supplementation of blood samples on clot formation have shown divergent results. Some studies have shown a positive effect on clotting time or clot stability with FXIII alone 17,20,23 –25 or an improved effect when FXIII was added simultaneously with fibrinogen as compared to fibrinogen alone. 18,22,26,27,29 Other studies have not been able to demonstrate any effect of FXIII on these parameters. 19,21,28,30,31 One possible explanation for such variation could be dissimilarities in the baseline activity level of FXIII in the blood sample. If the FXIII activity is normal, the addition of more FXIII may not be able to improve clot formation further. Other possible explanations might be the large differences in study design, grade and type of hemodilution, and analysis device used.
In some of the studies already mentioned, the influence of FXIII supplementation of blood samples on fibrinolysis was investigated. 17,23,25,27,28,31 These 6 studies demonstrated that the addition of FXIII concentrate had a protective effect on fibrinolysis by improving various thromboelastometry variables linked to fibrinolysis, for example, maximum lysis or lysis onset time. In the present study, we were unable to detect any effect on fibrinolysis, simply because none of the study participants had any thromboelastometric signs of fibrinolysis in postoperative baseline samples (data not shown).
The effect of FXIII supplementation in the present study was limited. With the highest dose of FXIII, EXTEM clotting time was shortened by approximately 10% (in both groups) and FIBTEM clot firmness increased by 25% in the cardiac surgery group, compared to baseline. Furthermore, the effects of FXIII alone were markedly less than when fibrinogen or platelets were added simultaneously to the samples (Figures 1 and 2 and Table 2). This corroborates current guidelines suggesting that FXIII supplementation should only be considered in patients with diffuse bleeding and adequate fibrinogen levels or when fibrinogen supplementation is not successful. 10,11 In fact, the present results in combination with previous observations on the dose-dependent improvement in clot firmness after the addition of increasing doses of fibrinogen and/or platelets 32 suggest that higher doses of fibrinogen or platelet concentrates would be more effective in restoring clot firmness than adding FXIII concentrate. This is also supported by the only large-scale randomized trial with FXIII supplementation in cardiac surgery patients, which was unable to show any effect on bleeding and transfusions with recombinant FXIII. 33
Interestingly, the effect of FXIII—both when given as monotherapy and in combination with fibrinogen or platelets—was comparable in both cardiac and scoliosis surgery patients despite the large differences between the groups. The groups in fact differed in most patient characteristics and laboratory tests, including, for example, age, gender, body mass index, and fibrinogen concentration (Table 1). This strengthens the validity of our results and indicates that they may also be relevant in other surgical populations.
The study had important limitations. The one that is inherent in any ex vivo study of clot formation is whether the results can be related to the clinical setting. The ex vivo model did not include the influence of the vascular wall and the endothelium, and of blood flow, on clot formation. Another limitation was that the samples were collected from normal postoperative cardiac and scoliosis surgery patients without excessive bleeding. The absolute thromboelastometry values cannot be directly compared to those in other studies performed with undiluted samples. The strengths of the study were that the samples were collected from patients (and not healthy volunteers) and that we measured changes in clot formation before and after FXIII, fibrinogen, and platelet supplementation in the same blood samples and that the same degree of hemodilution was maintained in all samples.
In conclusion, the results of this ex vivo study show that supplementation with clinically relevant doses of FXIII improves clot formation moderately in samples from cardiac and scoliosis surgery patients in a dose-dependent manner. The effect of FXIII alone was markedly less than the effect that was achieved with FXIII in combination with fibrinogen or platelets.
Footnotes
Authors’ Note
An earlier version of this manuscript was included in a doctoral thesis from Sahlgrenska Academy, University of Gothenburg, Sweden (![]() ). The study sponsors had no influence on the analysis and interpretation of the data, in the writing of the report, or in the decision to submit the report for publication. The authors have access to the data.
). The study sponsors had no influence on the analysis and interpretation of the data, in the writing of the report, or in the decision to submit the report for publication. The authors have access to the data.
Acknowledgments
The authors thank the staff of the Regional Blood Bank for the preparation of the platelet concentrates.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: AJ has received honoraria for presentations and advisory boards from CSL Behring.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by the Swedish Heart and Lung Foundation (grant number 20140281 to AJ), Sahlgrenska University Hospital (ALF/LUA grant number 146281 to AJ and number 428161 to HB), and CSL Behring, Marburg, Germany.
