Abstract
Cancer-associated venous thromboembolism (VTE) is one of the leading causes of mortality and morbidity among patients with malignancy. The Khorana risk score (KRS) is currently the best validated risk assessment model to stratify risks of VTE development in ambulatory patients with cancer. In the current study, we assessed the performance of KRS in patients with hepatocellular carcinoma (HCC). We retrospectively analyzed patients with diagnosis of HCC (screened by International Classification of Diseases [ICD-9] and ICD-10 code, confirmed with radiographic examination and/or histopathology) at a large public hospital over 15 years (January 2000 through July 2015). Cases with VTE were identified through radiographic examination and blindly adjudicated. Khorana risk score was calculated for each patient, and its association with VTE development and mortality was assessed. Among 270 patients with HCC, 16 (5.9%) cases of VTE were identified, including 7 (43.8%) pulmonary embolism, 4 (25%) peripheral deep vein thrombosis, and 6 (37.5%) intra-abdominal thrombosis. One hundred eighty-four (68.1%) patients had a KRS of 0 and 86 (31.9%) patients had a KRS >0. Most of the thrombotic (n = 9, 56%) events occurred in the low-risk group. In univariate analysis, only prechemotherapy leukocyte count equal to or greater than 11 000/μL was statistically significant in the prediction of VTE incidence. After adjusting for confounding factors in multivariate analysis, KRS >0 was not predictive of VTE (hazard ratio [HR] = 1.83, 95% confidence interval [CI] = 0.81-4.15, P = .15) or mortality (HR = 1.61, 95% CI = 0.92-2.81, P = .09). Khorana risk score did not predict VTE development or mortality in patients with HCC. Design of HCC-specific risk assessment model for VTE development is necessary.
Introduction
Venous thromboembolism (VTE) is a common complication in patients with cancer and represents a leading cause of morbidity and mortality in this population. 1,2 The reported cumulative incidence of VTE in overall patients with cancer varies (1%-8%) among studies, 3 depending predominantly on the type of primary cancer. Overall, the risk of VTE is increased by 4- to 7-folds in patients with cancer as compared to the general population. 4 –6 In an attempt to risk stratify which patients with cancer may benefit from primary prevention strategies against VTE, independent risk factors for cancer-associated VTE have been described in prior studies and included primary site of cancer, presence of metastatic disease, chemotherapy, and surgery.4,7,8
To date, Khorana risk score (KRS) is the best-validated risk assessment model to predict VTE in cancer population. 9 This score used 5 clinical and laboratory values to classify the patients as low to high risk for VTE—site of primary cancer, body mass index (BMI), prechemotherapy leukocyte count, prechemotherapy platelet count, and hemoglobin (or use of erythropoiesis-stimulating agents). 10 A modification of the KRS was externally validated in Vienna Cancer and Thrombosis Study 11 ; however, in subsequent cohorts of specific cancer types, the score has been challenged and deserves retesting.9,12,13 In addition, it has been suggested that the KRS may add information on the prediction of mortality, 14 and this has not been tested in patients with hepatocellular carcinoma (HCC).
Hepatocellular carcinoma is the fifth most frequently diagnosed cancer in adult men and seventh in women globally, leading to 25 000 to 1 million death per annum. 15 With often concurrent chronic liver disease, 16 HCC poses high risk for VTE development. The present study aims to evaluate the performance of KRS in the prediction of VTE or mortality in patients with HCC.
Materials and Methods
Study Participants
We retrospectively reviewed the electronic medical records of patients who presented to Cook County Health and Hospital System, Chicago between January 1, 2000, and July 31, 2015. Initial screening was conducted with International Classification of Diseases (ICD)-9 code (=155) and/or ICD-10 code (=C22) for malignant neoplasm of liver and intrahepatic biliary duct. We reviewed and selected the patients if they had histopathology-proven or radiographically proved HCC by triple-phase enhanced computed tomography and/or magnetic resonance imaging of the abdomen (intense enhancement during arterial phase followed by “washout pattern” during delayed phase17,18). We excluded the patients if they had incomplete data, were less than 18 years of age, had VTE prior to the diagnosis of HCC, or had incomplete follow-up of less than 1 month in our institution.
The institutional review board of Cook County Health and Hospitals System, Chicago approved the present study. The database of HCC was set up and maintained by faculty of Department of Internal Medicine, Cook County Health and Hospitals System, Chicago.
Data Collection
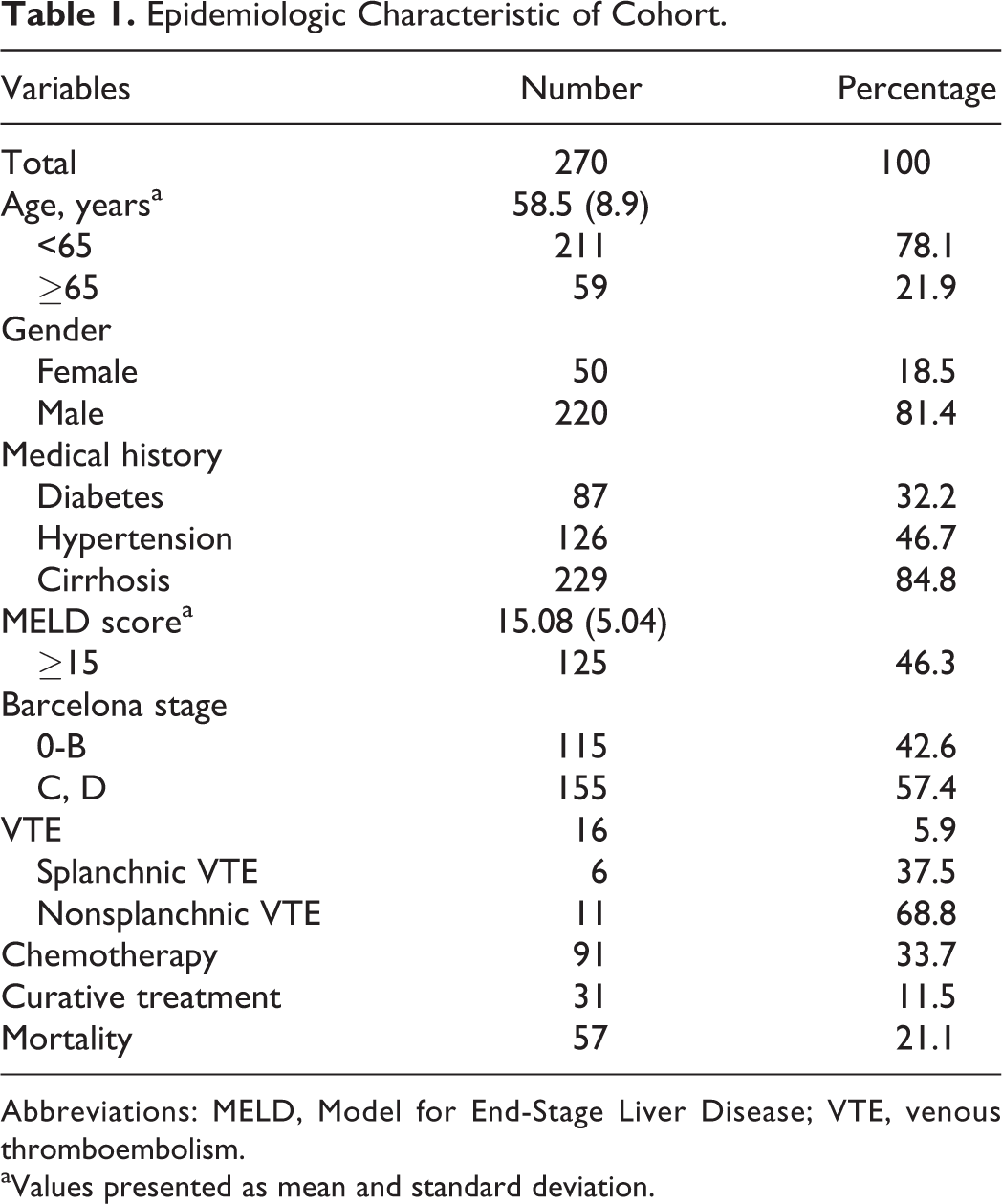
Eligible patients were censored if there was discontinuation of follow-up. We collected demographic variables including age, gender, medical history (hypertension, diabetes), BMI at diagnosis; tumor-related risk factors including etiology of liver disease, Child-Pugh score, number of intrahepatic lesions, extrahepatic metastasis, and Barcelona staging of HCC 19 ; treatment variables including chemotherapy with sorafenib, curative treatment (liver transplant, resection, radiofrequency ablation); and baseline laboratory at diagnosis (white blood cell count, neutrophil count, lymphocyte count, platelet, hemoglobin, total bilirubin, international normalized ratio [INR]). Information of outcome during follow-up period (outpatient, hospice care, death) was also collected (Table 1).
Epidemiologic Characteristic of Cohort.
Abbreviations: MELD, Model for End-Stage Liver Disease; VTE, venous thromboembolism.
aValues presented as mean and standard deviation.
Venous thromboembolism was diagnosed based on radiographic examinations including compression ultrasonography, contrast-enhanced computed tomography, and pulmonary angiogram. There was no systematic VTE screening. We categorized VTE according to the anatomical site into pulmonary embolism (PE), deep vein thrombosis (DVT), and intra-abdominal venous thrombosis (superior mesenteric vein, inferior vena cava [IVC], hepatic vein). Portal vein thrombosis was not included given limitations on objective differentiation between tumor invasion and true thrombosis. Blinded event adjudication was conducted for all outcome data by 3 physicians; in case of discrepancy, there was a majority vote to adjudicate.
Khorana risk score was calculated based on the above information collected at the time of diagnosis of HCC, according to the following formula—1 point for BMI ≥35 kg/m2, prechemotherapy platelet count ≥350 000/μL, hemoglobin level <10g/dL, or on treatment with a red-cell growth factor, prechemotherapy leukocyte count ≥11 000/μL. No point was given for cancer type as HCC is not regarded as high-risk or very high-risk type of primary cancer.
Statistical Analysis
Descriptive data were summarized to characterize the distribution of variables. Continuous laboratory values were dichotomized into categorical variables according to the KRS calculation as outlined above, and Khorana score was calculated. As in the current cohort, few cases fell into high-risk group for VTE development according to KRS, intermediate-risk and high-risk groups were unified as elevated risk (KRS >0), as compared to low risk whereas KRS equals to 0. The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), positive likelihood ratio, and negative likelihood ratio were calculated for elevated risk group in regard to overall VTE development, splanchnic VTE development, nonsplanchnic VTE development, and overall survival. We assessed each variable included in KRS calculation and the final score in univariate analysis. Survival analysis was then conducted with cox proportional hazards model, in which the development of VTE was treated as a time-dependent variable to eliminate the effect of misclassification from immortal time bias. Univariate analysis was performed for each variable and KRS group (elevated risk as compared to low risk). Variables with statistical significance in univariate analysis (P < .05) were included in multivariate analysis to identify independent risk factors for mortality. Statistical analysis was performed with STATA (version 13.0). P value of less than .05 indicated statistical significance.
Results
Patient Characteristics
Among the 308 patients identified by initial screening, 34 patients were excluded as they had biopsy-proven cholangiocarcinoma or metastatic adenocarcinoma, 4 patients were excluded with a follow-up period less than 1 month. For analysis, we included 270 patients who fulfilled the selection criteria (Table 1). The mean age at diagnosis was 58.5 years with range from 26 to 80 years. There were 115 (42.6%) patients who had HCC with Barcelona stage of 0-A and 155 (57.4%) patients who had advanced HCC with Barcelona stage of C or D. A total of 31 (11.5%) patients received curative treatment, 91 (35.9%) patients received chemotherapy with sorafenib, and 57 (21.1%) patients died during follow-up (Table 1)
Venous Thromboembolism Incidence and Khorana Score Calculation
There were 16 (5.93%) cases of VTE documented during a mean follow-up period of 11.9 months, including 6 cases of PE, 5 cases of intra-abdominal VTE, 4 cases of peripheral DVT, and 1 case of concurrent PE and intra-abdominal VTE. The 2-year cumulative incidence of VTE after diagnosis of HCC was 5.93%, with 75% (12 cases) of VTE occurred within 3 months of diagnosis of HCC. Among the 16 patients, 9 (56.3%) endorsed symptoms corresponding to VTE, the remaining 7 cases (43.8%) were identified incidentally. The treatment strategies were not uniform, 7 (43.8%) patients were treated with enoxaparin subcutaneous injection as outpatient, 3 (18.8%) had IVC filter placement, and 6 (37.5%) remained untreated because of concurrent variceal bleeding, high bleeding risk with thrombocytopenia and coagulopathy, or receiving palliative care.
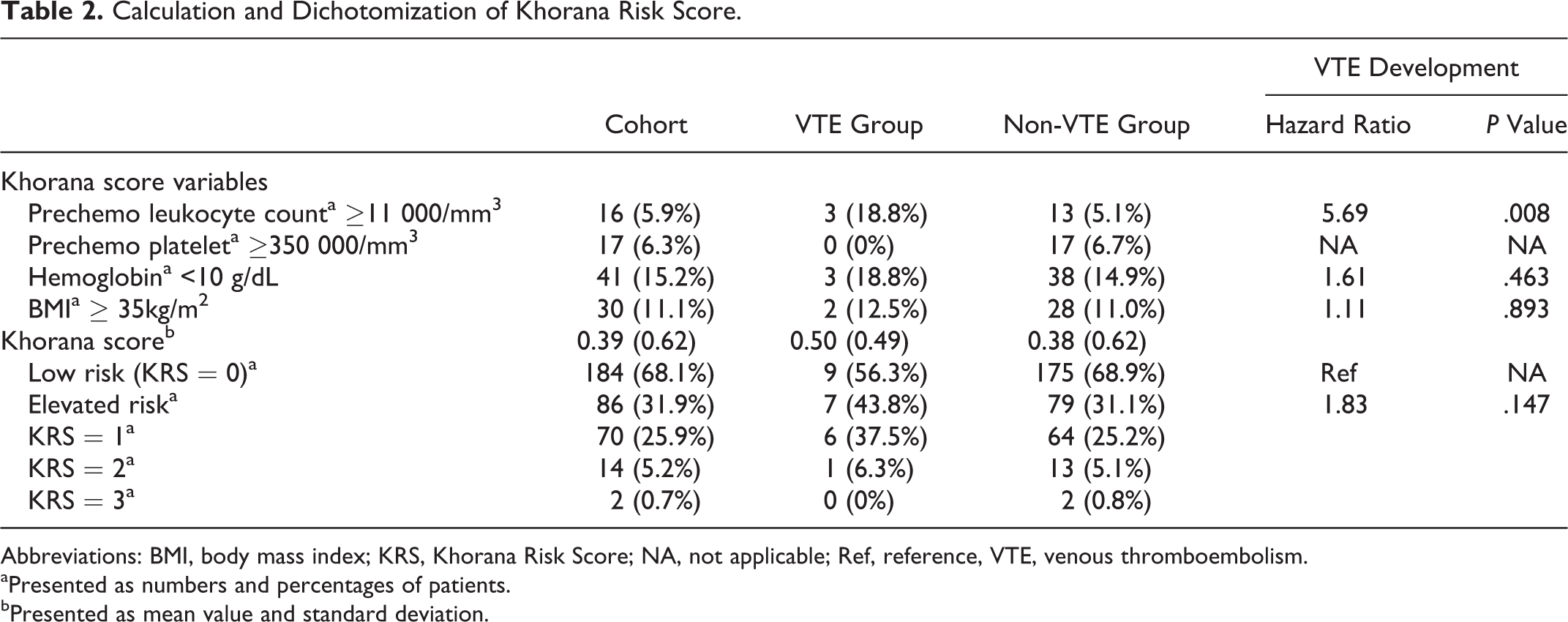
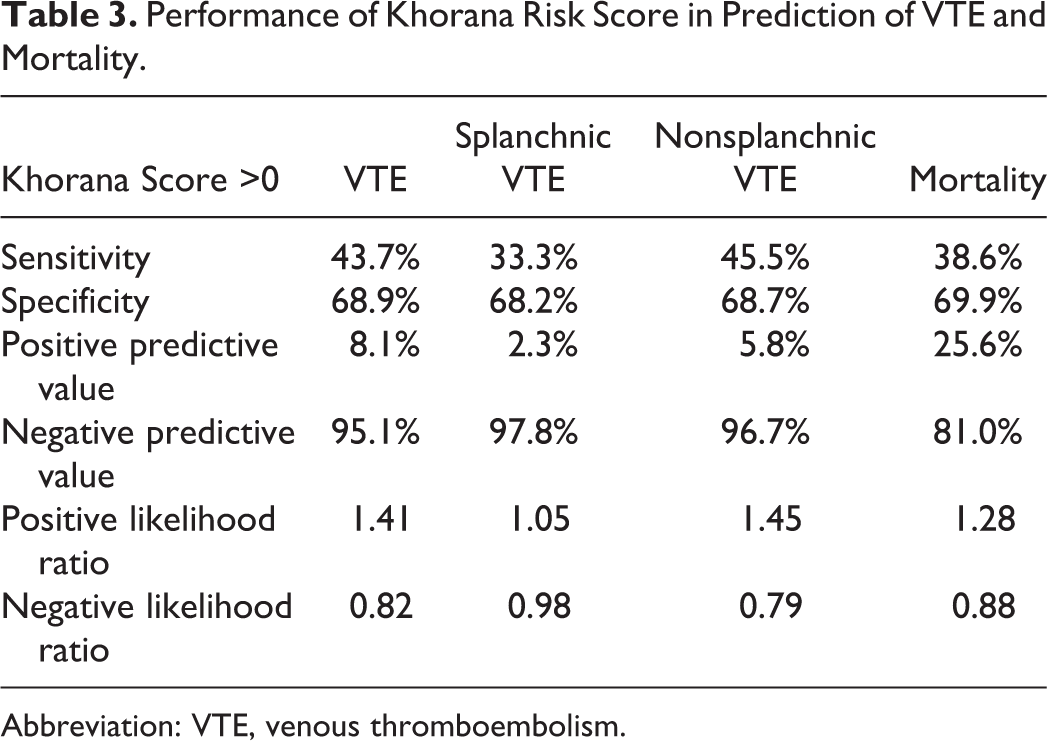
In the present cohort, 184 (68.1%) patients had KRS of 0, 70 (25.9%) had KRS of 1, 14 (5.2%) had KRS of 2, and only 2 (0.7%) patients had KRS of 3 thus regarded as high risk (Table 2). Most of the thrombotic (n = 9, 56%) events occurred in the low-risk group. On univariate analysis, only prechemotherapy leukocyte count ≥11 000/μL predicted VTE development with statistical significance. After dichotomization into low-risk group (KRS = 0) and elevated risk group (KRS >0) for reasons depicted in Section “Statistical Analysis,” the incidence of VTE in elevated risk group was not different than that of low-risk group (hazard ratio [HR] = 1.83, 95% confidence interval [CI] = 0.81-4.15: P = .15; Table 2). For overall VTE, the sensitivity and specificity of KRS >0 were 43.7% and 68.9%, respectively. Of note, KRS >0 had high NPVs for VTE development (95.1%) and mortality (81.0%); however, the PPVs were low (8.1% and 25.6%, respectively). The sensitivity, specificity, PPV, NPV, positive likelihood ratio, and negative likelihood ratio for overall VTE, splanchnic VTE, nonsplanchnic VTE, and mortality were presented in Table 3.
Calculation and Dichotomization of Khorana Risk Score.
Abbreviations: BMI, body mass index; KRS, Khorana Risk Score; NA, not applicable; Ref, reference, VTE, venous thromboembolism.
aPresented as numbers and percentages of patients.
bPresented as mean value and standard deviation.
Performance of Khorana Risk Score in Prediction of VTE and Mortality.
Abbreviation: VTE, venous thromboembolism.
Survival Analysis
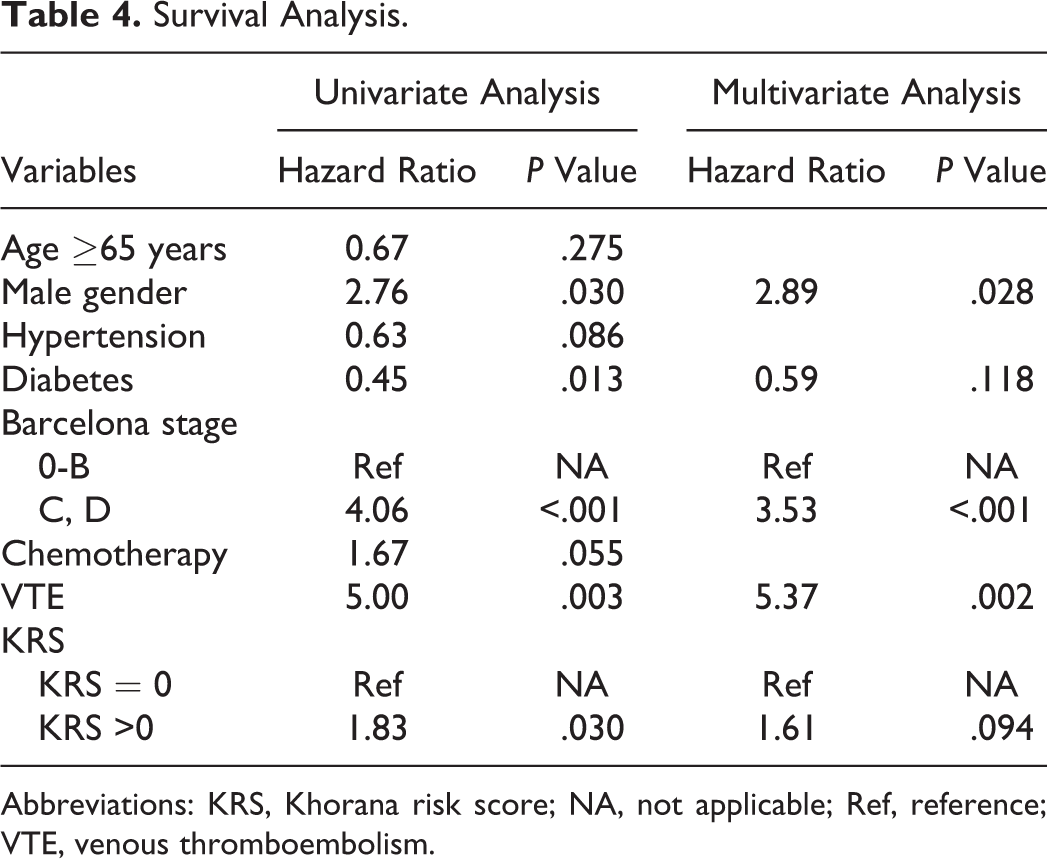
In univariate analysis, statistically significant risk factor for mortality included male gender, absence of diabetes, advanced HCC (Barcelona stage of C and D as compared to Barcelona stage of 0, A, B), VTE development, and elevated risk group by KRS (KRS >0; Table 4). After controlling of confounding factors in the multivariate analysis, the independent risk factors for mortality that remained in the model were male gender (HR = 2.89, 95% CI = 1.12-7.43, P = .03), advanced HCC (HR = 3.53, 95% CI = 1.82-6.87, P < .01), and development of VTE (HR = 5.37, 95% CI = 1.84-15.6, P < .01). Elevated risk group (KRS >0) did not remain as a significant predictor of overall survival (HR = 1.61, 95% CI = 0.92-2.81, P = .09; Table 4).
Survival Analysis.
Abbreviations: KRS, Khorana risk score; NA, not applicable; Ref, reference; VTE, venous thromboembolism.
Discussion
In this cohort of 270 patients with HCC, 16 (5.9%) cases developed VTE, which is within the expected incidence of cancer-associated thrombosis. 20 Among patients with HCC, an elevated KRS (KRS >0) was not predictive of VTE nor morality. Consistent with the original validation cohort, KRS had a high NPV for overall VTE (95.1%), splanchnic VTE (97.8%), and nonsplanchnic VTE (96.7%), but the PPV was only 8.1% for overall VTE. Only 43.8% patients who developed VTE had an elevated KRS. Therefore, KRS could be used to identify patients with HCC having low risk of VTE, however, not sufficient for prediction of VTE development overall.
Our results are in line with prior cancer-specific studies that suggested inadequate performance of the score in patients with specific cancer types. In their study, Mansfield et al 9 retrospectively analyzed 719 cases of lung cancer among which 83 VTEs were diagnosed during a median follow-up of 15.2 months and reported that KRS did not accurately identify patients with high risk of VTE. In the large FRAGMATIC trial, which included 2202 patients with lung cancer, the KRS was also not predictive VTE events.21,22 Similarly, Ramos et al reviewed 943 patients with urothelial carcinoma and variant histology with 93 (9.9%) VTEs and found no significant difference in overall VTE rate between different KRS risk groups, 12 which was also in line with the conclusion by Martin et al in their cohort of 571 patients with ambulatory pancreatic cancer. 23 Taken together, the score has failed to perform in large cancer-specific cohorts, and the limitation appears to be systematic and not limited by sample size. Given that the KRS is heavily dependent on cancer type, it does not adequately discriminate specific groups of patients. In contrast, among 1717 patients with lymphoma in a meta-analysis of 12 studies, 12% was classified as high risk and had a 6.6% risk of VTE at 6 months, higher than the incidence among patients with low risk (2.2%). 24 Thus, although the score appears to perform well in some subsets, before wide implementation, it needs to be stressed that the predictive value varies depending on the cancer type. Moreover, we estimate cancer-specific predictive tools need to be created for prevalent cancers.
Among Khorana variables, only prechemotherapy leukocyte count ≥11 000/mm3 was associated with VTE incidence in our analysis. This finding was also described in previous study 12 and deemed to be probably related with release of neutrophil extracellular traps. Neutrophil extracellular traps promote coagulation and increased fibrin deposition by providing nidus for platelet adhesion, activation, and aggregation, therefore theoretically leading to increased VTE development.25,26 Conversely, in present cohort, 6.3% patients had prechemotherapy platelet ≥350 000/mm3, and 15.2% patients had hemoglobin <10g/dL, both significantly different from the original study (22.4% and 6.6%, respectively). The above findings were anticipated as thrombocytopenia and anemia secondary to hypersplenism are commonly associated with cirrhosis, 27 which is prevalent in 84.8% patients in the current cohort. The unique clinical features of HCC severely limited the efficiency of the variables included in KRS to predict VTE and suggested the need for a HCC-specific risk assessment model for VTE.
Originally designed to predict the occurrence of VTE, KRS was also demonstrated to predict mortality. Kuderer et al retrospectively analyzed the 4405 patients involved in the original Khorana study and concluded KRS could predict early mortality (<120 days) and cancer progression. 14 In their cohort of resectable pancreatic adenocarcinoma, Sohal et al 13 also reported KRS was associated with early mortality (within 30 days), which is in line with the findings in lung cancer. 9 In the present cohort, however, KRS >0 was not associated with higher mortality in multivariate analysis.
Several limitations existed for the present study: First, our study was retrospective in design and limited by the inherent confounders as patients may have been systematically excluded. However, our VTE cases were objectively confirmed with blinded event adjudication, and database was complete in all 270 patients to calculate Khorana score. Second, although the incidence detected was consistent with that reported in other studies, 11 our analysis might be underpowered to detect true association between KRS and VTE development or mortality. Of note, the present data are based on a relatively large cohort of patients with HCC and have allowed us to demonstrate that the implied clinical characteristics expected in this population limit the applicability of platelet count and anemia as predictive variables.
In conclusion, KRS did not accurately stratify cumulative VTE risks in patients with HCC. Venous thromboembolism, but not the KRS, was an independent predictor of death among patients with HCC. In patients with HCC, variables included in KRS might be confounded by concurrent cirrhosis. Therefore, design of HCC-specific risk assessment model for VTE development is needed.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
