Abstract
Despite the identification of a wide range of inherited and acquired risk factors for arterial ischemic stroke (AIS) in children, genetic risk factors are incompletely characterized and may vary among different populations. We investigated the role of individual and combined inherited prothrombotic and intermediate-risk factors in 73 children with perinatal (n = 35) and childhood AIS (n = 38) and 100 age- and sex-matched controls. Ten polymorphisms in 8 candidate genes encoding coagulation and fibrinolytic proteins (factor V [FV] Leiden, FV HR2, factor II [FII] G20210A, β-fibrinogen [β-FBG]-455G>A, factor XIII [FXIII]-A p.Val34Leu, plasminogen activator inhibitor 1 4G/5G), homocysteine metabolism (methylenetetrahydrofolate reductase [MTHFR] C677T, MTHFR A1298C), and intermediate-risk factors (angiotensin-converting enzyme I/D, apoE ∊2-4) were detected using a multilocus genotyping assay. Allele-specific polymerase chain reaction was used for the determination of human platelet alloantigens (HPA-1, HPA-2, HPA-3, and HPA-5). Factor V Leiden was associated with an increased risk of AIS (odds ratio [OR]: 4.72, 95% confidence interval [CI]: 1.22-18.27) and perinatal AIS (OR: 8.29, 95% CI: 1.95-35.24). Human platelet antigen-3b allele carriers had a 2-fold lower risk of AIS (OR: 0.51, 95% CI: 0.26-0.98) and perinatal AIS (OR: 0.40, 95% CI: 0.18-0.92). A 2.21-fold increased risk of childhood AIS (95% CI: 1.03-4.73) was identified in FXIII-A Leu34 allele carriers. Combined FV Leiden/FV HR2, FV Leiden/MTHFR A1298C, FV Leiden/MTHFR C677T/MTHFR A1298C, and FV Leiden/FV HR2/MTHFR A1298C heterozygosity was identified in children with AIS but not in controls, which revealed a statistically significant difference. This case–control study shows that besides already documented association between FV Leiden and AIS, other previously unreported polymorphisms (FXIII-A p.Val34Leu, HPA-3) and several genotype combinations that always include heterozygous FV Leiden can be related to AIS in Croatian population.
Keywords
Introduction
Stroke is predominantly a disease occurring in adults. Although being relatively a rare disease in children, it is increasingly recognized as an important cause of childhood disability and lifelong morbidity. 1 The most frequently diagnosed type of stroke in children is arterial ischemic stroke (AIS) that is subdivided according to the time of onset into perinatal and childhood AIS. Perinatal and childhood AIS represent 2 distinct categories characterized by different etiologies, presentation signs and symptoms, and estimated incidence rates. With the estimated incidence ranging from 17 to 63 per 100 000 live births, perinatal AIS is diagnosed even 17 times more often compared to childhood AIS (annual incidence rates ranging from 0.2-7.9 per 100 000 live births). 2 –5
Extensive investigation of AIS in children during the last 20 years has led to the identification of a wide range of inherited and acquired risk factors, such as congenital heart malformations, hemolytic anemias, vasculopathies, and collagen vascular diseases, as well as some rare inborn metabolic disorders, trauma, and infection. 6 –8 The importance of thrombophilia in the etiology of AIS has been emphasized in recent years, based on the reported associations between polymorphisms in genes encoding proteins of the coagulation system and homocysteine metabolism found in numerous population studies. The best-studied genetic polymorphisms associated with an increased risk of AIS are factor V (FV) Leiden (F5 1691 G>A, rs6025), factor II (FII) G20210A (F2 20210 G>A, rs1799963), and the common polymorphism in the methylenetetrahydrofolate reductase [MTHFR] gene C677T (MTHFR 677 C>T, rs1801131). 9 –13 Apart from them, only a few case–control studies have investigated the impact of other polymorphisms in coagulation and fibrinolysis factor genes in the etiology of AIS, such as FV HR2 haplotype (F5 4070 A>G, rs1800595), β-fibrinogen (FBG)-455G>A (FBG-455G>A, rs1800790), factor XIII-A (FXIII-A) p.Val34Leu (F13A1, rs5985), and plasminogen activator inhibitor 1 (PAI-1) 4G/5G (SERPINE1, rs1799889). 14 –19 The role of platelets is still relatively unknown as only the human platelet alloantigen 1 (HPA-1; ITGB3, 176 T>C, rs5918) has been investigated among platelet glycoprotein receptor polymorphisms and conflicting results were reported. 14 –16 Moreover, the second common polymorphism involved in homocysteine metabolism, MTHFR A1298C (MTHFR 1298 A>C, rs1801131), has also been associated with AIS. 14,20
Although intermediate-risk polymorphisms are routinely studied as part of the workup in adults with AIS, this is not the case in children. A possible influence of apoE ∊2-4 (APOE rs429358 and rs7412) has only recently been evaluated, 21,22 whereas to date, there are no available data regarding the angiotensin-converting enzyme (ACE I/D; ACE rs1799752) polymorphism.
Taking into consideration that genetic risk factors are incompletely characterized at present and that the frequency and distribution of genetic factors may vary among different populations, the aim of this study was to investigate the possible role of individual and combined inherited prothrombotic and intermediate-risk factors in children with a confirmed diagnosis of AIS and to check the possible association of investigated risk factors with the disease.
Patients and Methods
Study Population
Cases
A total of 73 unrelated Caucasian children (48 boys and 25 girls) aged ≤18 years with a confirmed diagnosis of AIS, who were admitted to the Department of Neuropediatrics, Children’s Hospital Zagreb, and the Department of Pediatric Neurology, University Hospital Centre Zagreb, between February 1998 and September 2010 were included in the present study. Data were collected from medical records of children with AIS. Arterial ischemic stroke was defined as any neurological event including a seizure occurring in association with corresponding ischemic lesions in arterial distribution, shown by brain imaging. 23,24 The diagnosis was made after a careful analysis of clinical history and physical and neurological examination based on clinical symptoms and signs and confirmed in all cases by magnetic resonance imaging or/and computed tomography. Magnetic resonance angiography, transfontanellar ultrasonography (US), and transcranial color Doppler US were performed in selected cases as ancillary techniques. Furthermore, electroencephalographic examinations, electrocardiography, and echocardiography were performed in all patients. Children with AIS were further classified according to the time of onset into perinatal AIS, occurring between 20 weeks of fetal life through the 28th postnatal day, and childhood AIS, occurring from 29 days of life to 18 years. The perinatal AIS group consisted of newborns with neonatal AIS (neurological presentations within 28 postnatal days) and infants in whom the diagnosis of presumed perinatal AIS was made retrospectively with neurologic presentations after 1 month of age and imaging indicating old arterial ischemic infarction. 25
Controls
The control group comprised 100 age- and sex-matched children (63 boys and 37 girls) aged ≤18 years (mean age 6.5 years) from the same geographical region with no history of neurologic or thromboembolic diseases who were recruited from children awaiting minor surgery and children with respiratory diseases at routine visits.
Written informed consent was obtained from parents of all children included in the study. The present study was performed in accordance with ethical standards established in the updated version of the 1964 Declaration of Helsinki, and the University Hospital Centre Zagreb ethics committee approved the study.
Blood Sampling and DNA Analysis
Genomic DNA was extracted from peripheral blood leukocytes by the standard salting-out method. 26 Ten polymorphisms in 8 candidate genes encoding proteins of the coagulation and fibrinolytic systems (FV Leiden, FV HR2, FII G20210A, β-FBG-455G>A, FXIII-A p.Val34Leu, PAI-1 4G/5G), homocysteine metabolism (MTHFR C677T, MTHFR A1298C), and intermediate-risk factors (ACE I/D, apoE ∊2-4) were detected using a commercial multilocus genotyping assay (CVD StripAssays; ViennaLab Diagnostics, Vienna, Austria). Briefly, each DNA sample was amplified in 2 parallel multiplex polymerase chain reactions (PCRs) using biotin-labeled primers. Amplification products were selectively hybridized to a test strip containing allele-specific oligonucleotide (ASO) probes immobilized as an array of parallel lines. Bound PCR fragments were detected using a streptavidin–alkaline phosphatase conjugate and color substrates. Purple staining, visible after 15 minutes and representing positive reaction, enabled assignment of genotypes for each polymorphism. For the apoE ∊2-4 polymorphism, 6 possible homozygous or heterozygous genotypes could be obtained (∊2∊2, ∊2∊3, ∊2∊4, ∊3∊3, ∊3∊4, and ∊4∊4).
Human platelet alloantigen 1, HPA-2, HPA-3, and HPA-5 were detected using ASO-PCR with sequence-specific primers according to Klüter et al. 27 In addition, genotypes for HPA-1 determined by ASO-PCR were compared by those obtained with the CVD StripAssays, and a 100% concordance rate was obtained.
Statistical Analysis
Statistical analyses were performed using the MedCalc software package version 9.3.2.0 (Frank Schoonjans, the Netherlands). Distributions of continuous variables were expressed as mean ± standard deviation (SD), and qualitative data are presented as frequencies. Each investigated polymorphism was tested for the Hardy-Weinberg equilibrium and the χ2 analysis was applied.
Association between each polymorphism with the disease risk was assessed using the logistic regression model for the calculation of odds ratios (ORs) with corresponding 95% confidence intervals (CIs). The “dominant” model (homozygous or heterozygous variant compared with homozygous wild type) was applied for all investigated polymorphisms, except for the analysis of the apoE ∊2-4 polymorphism that was performed by grouping genotypes with at least 1 ∊4 allele, that is, ∊2∊4, ∊3∊4, and ∊4∊4, versus those without ∊4 alleles, that is, ∊2∊2, ∊2∊3, and ∊3∊3. Univariate logistic regression analysis was initially used to explore the effect of each polymorphism separately. Subsequent multivariate logistic regression analysis was performed for those variables that were statistically significant (P < .100 in univariate analysis).
Obtained genotypes of FV Leiden and FV HR2, as well as of MTHFR C677T and MTHFR A1298C, were combined and expressed as frequencies. The wild types of combined genotypes were used as reference, and the prevalence of combined genotype frequencies was calculated using Pearson χ2 analysis or Fisher exact test, depending on sample size. A 2-tailed P value <.05 was considered statistically significant.
Results
The analysis of data from medical records for 73 children with AIS revealed that perinatal AIS was established in 35 children and childhood AIS in 38 children. Perinatal AIS was detected in 4 newborns after convulsions in the first days after birth, whereas the diagnosis of presumed perinatal AIS was made retrospectively after the clinical presentation of hemiparesis or seizures in 31 children. Childhood AIS is manifested as hemiplegia, aphasia, visual or balance disturbance, or seizures. The characteristics of children with AIS, including gender, age at testing, age at diagnosis, brain imaging data, and identified risk factors are summarized in Table 1.
Characteristics of Children With Arterial Ischemic Stroke.
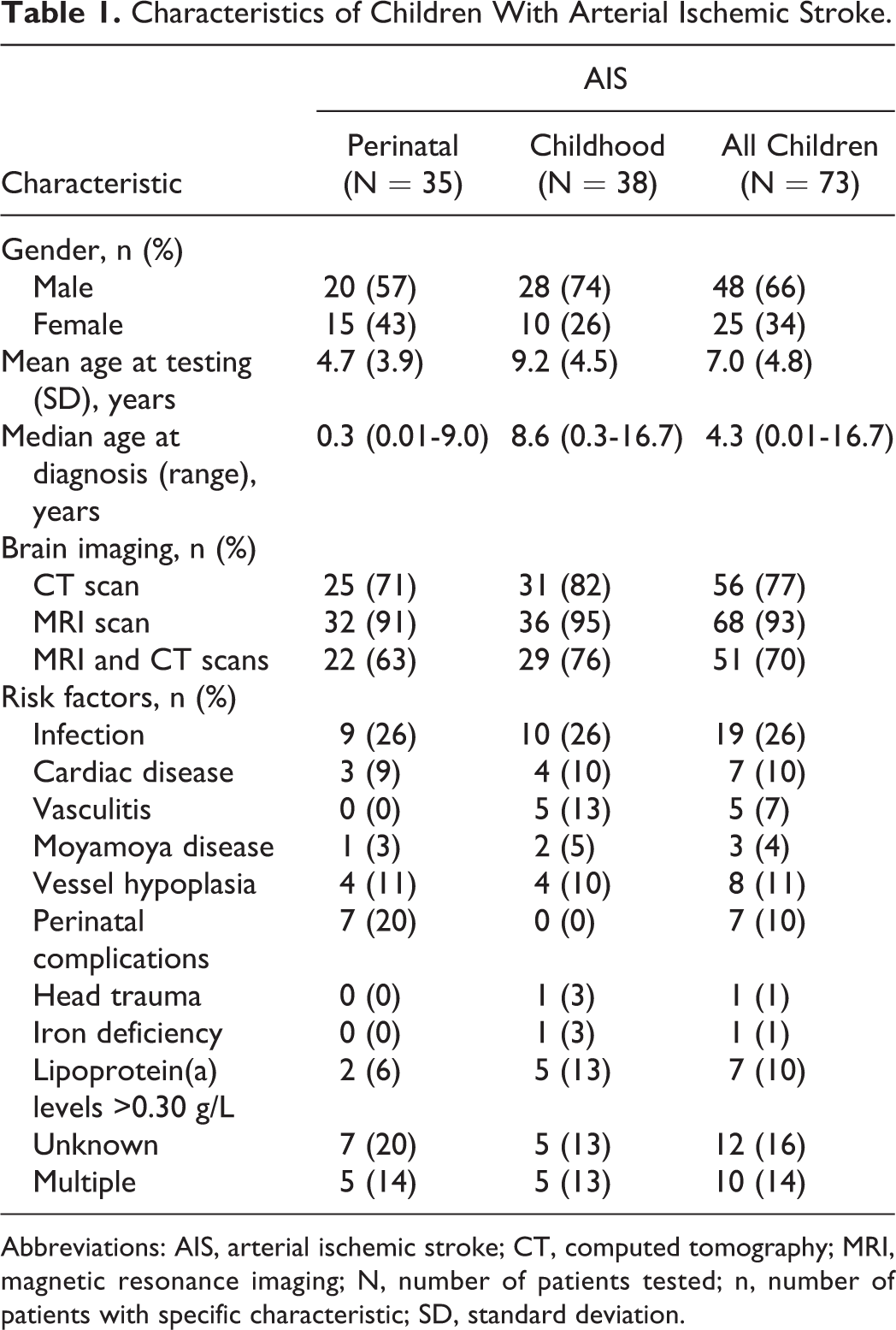
Abbreviations: AIS, arterial ischemic stroke; CT, computed tomography; MRI, magnetic resonance imaging; N, number of patients tested; n, number of patients with specific characteristic; SD, standard deviation.
A preponderance of boys was found among children with AIS, as well as among children with either perinatal or childhood AIS. The occurrence of perinatal AIS was 1.33 times more frequent in boys compared to girls, whereas childhood AIS occurred in boys even 2.80 times more frequently than in girls.
At least 1 risk factor was identified in the majority of children with AIS (84%), with infections as the leading risk factor in both AIS subgroups. Among 10% of patients with cardiac disease as the underlying condition, 4 patients with childhood AIS had congenital heart disease, whereas 3 patients with perinatal AIS had isolated patent foramen ovale. Elevated lipoprotein (a) [Lp(a)] concentration was not identified in any patient with cardiac disease. Not a single case of AIS was detected during or after surgery or cardiac catheterization. Multiple risk factors were present in 10 (14%) of 73 children with AIS and were equally distributed in perinatal and childhood AIS.
Distribution of genotype frequencies of investigated polymorphisms in children with perinatal, childhood AIS, all children with AIS, and controls is presented in Table 2. All genotypic distributions of investigated polymorphisms, both in cases and controls, were in Hardy-Weinberg equilibrium, except for HPA-1 (P = .018) in children with AIS. The results of the univariate and multivariate logistic regression analyses between children with AIS and controls for genotypic distributions of all investigated polymorphisms with calculated ORs and the corresponding CIs are presented in Table 3.
Distribution of Genotype Frequencies of Investigated Polymorphisms in Children With Perinatal, Childhood Arterial Ischemic Stroke, All Children With Arterial Ischemic Stroke, and Controls.
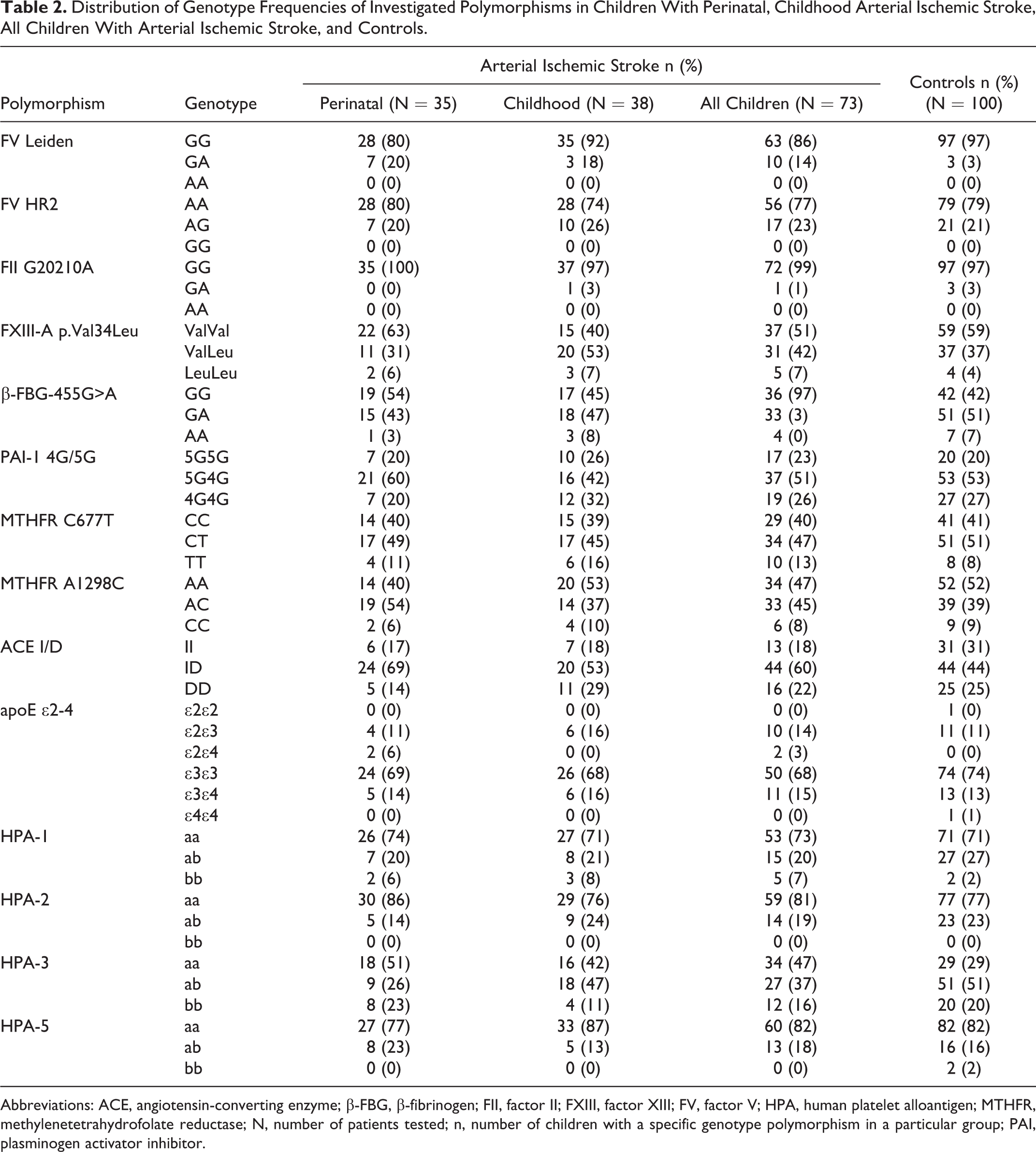
Abbreviations: ACE, angiotensin-converting enzyme; β-FBG, β-fibrinogen; FII, factor II; FXIII, factor XIII; FV, factor V; HPA, human platelet alloantigen; MTHFR, methylenetetrahydrofolate reductase; N, number of patients tested; n, number of children with a specific genotype polymorphism in a particular group; PAI, plasminogen activator inhibitor.
Results of Univariate and Multivariate Logistic Regression Analysis of Investigated Polymorphisms Between Children With Arterial Ischemic Stroke and Controls for Genotypic Distributions and Calculated Odds Ratios With Corresponding 95% Confidence Intervals.
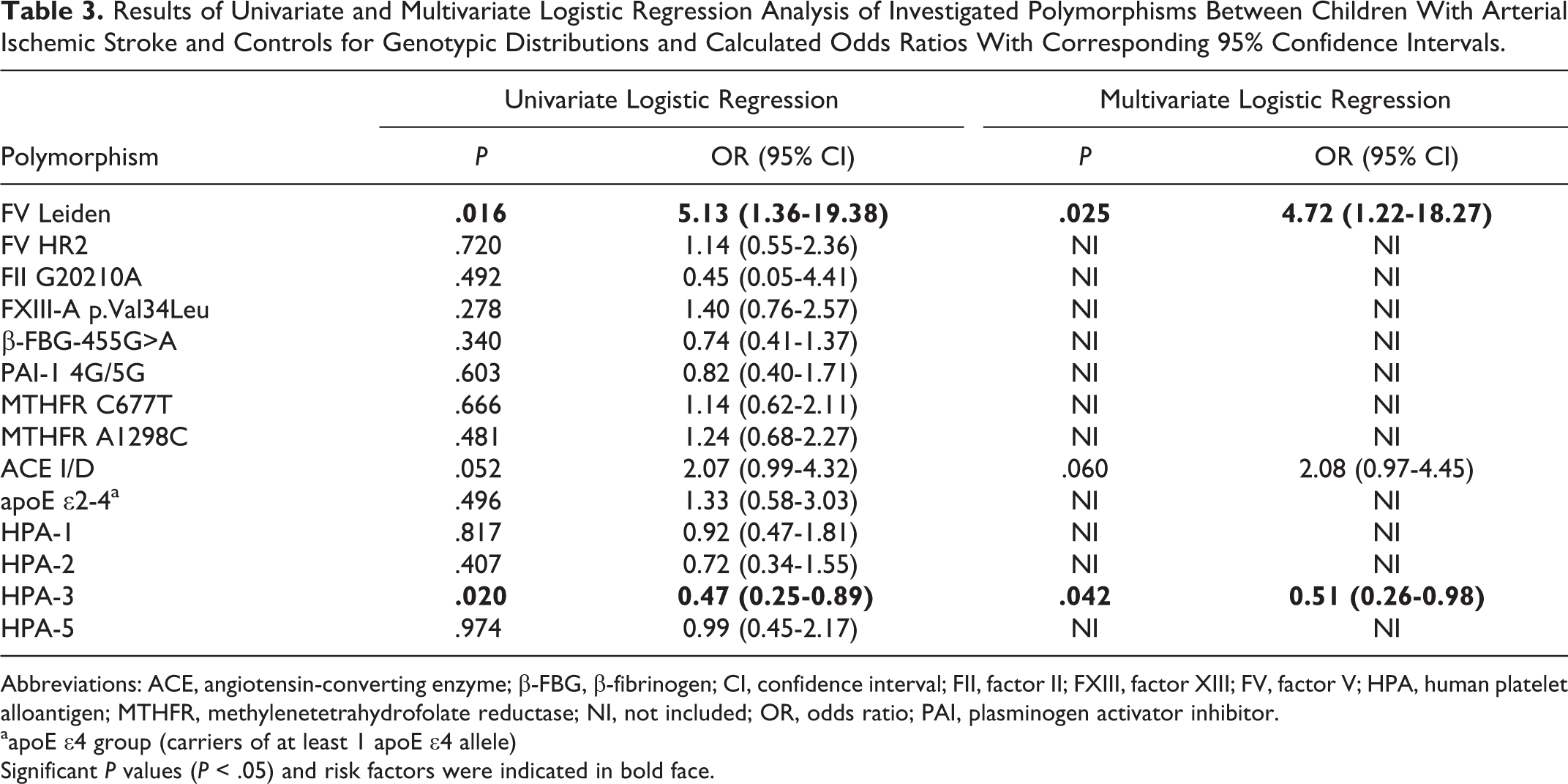
Abbreviations: ACE, angiotensin-converting enzyme; β-FBG, β-fibrinogen; CI, confidence interval; FII, factor II; FXIII, factor XIII; FV, factor V; HPA, human platelet alloantigen; MTHFR, methylenetetrahydrofolate reductase; NI, not included; OR, odds ratio; PAI, plasminogen activator inhibitor.
aapoE ∊4 group (carriers of at least 1 apoE ∊4 allele)
Significant P values (P < .05) and risk factors were indicated in bold face.
The association with an increased risk of AIS was not found for the majority of investigated single gene polymorphisms. Univariate logistic regression analysis has demonstrated statistically significant association with AIS for 2 polymorphisms: FV Leiden (P = .016) and HPA-3 (P = .020), whereas a borderline statistical significance was found for the ACE I/D polymorphism (P = .052). The multivariate analysis revealed that the presence of FV Leiden was associated with a 4.72-fold increased risk of AIS (95% CI = 1.22-18.27; P = .025), whereas the carriers of at least 1 HPA-3b allele had a 2-fold lower risk for the development of AIS (OR = 0.51; 95% CI = 0.26-0.98; P = .042).
As shown in Table 4, different polymorphisms were found to be statistically significantly associated with perinatal and childhood AIS. The strongest association was identified between the presence of FV Leiden and perinatal AIS, indicating an 8-fold increased risk of perinatal AIS (OR = 8.29; 95% CI = 1.95-35.24; P = .004). Furthermore, similar to the results obtained for children with AIS (Table 3), carriers of at least 1 HPA-3b allele had a more than 2-fold lower risk of perinatal AIS (OR = 0.40; 95% CI = 0.18-0.92; P = .032). The same associations were not found in children with childhood AIS, in whom the presence of at least 1 FXIII-A Leu34 allele revealed a modestly increased risk (OR = 2.21; 95% CI = 1.03-4.73; P = .042).
Results of Univariate and Multivariate Logistic Regression Analysis for Children With Perinatal and Childhood Arterial Ischemic Stroke Compared to Controls for Genotypic Distributions and Calculated Odds Ratios With Corresponding 95% Confidence Intervals.
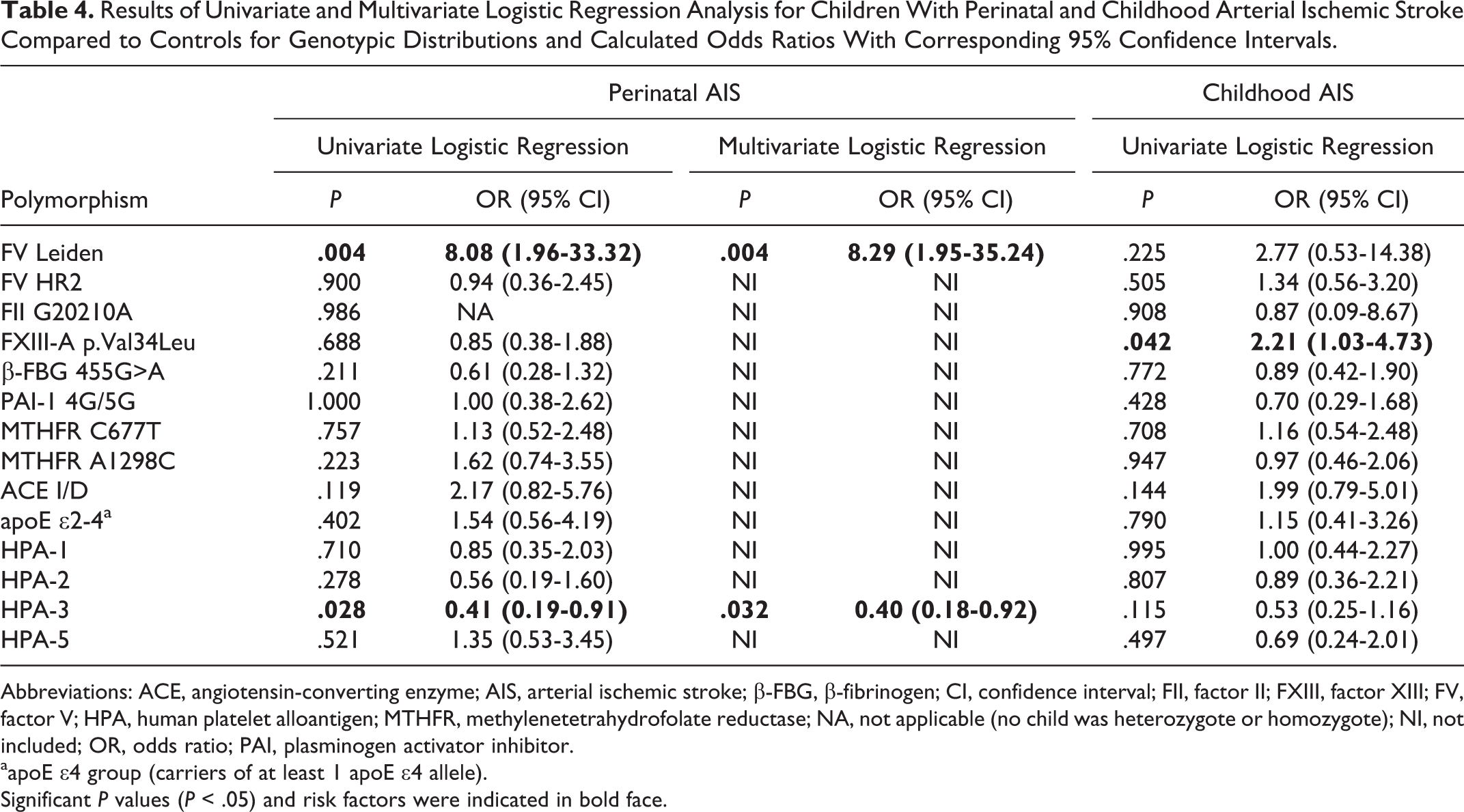
Abbreviations: ACE, angiotensin-converting enzyme; AIS, arterial ischemic stroke; β-FBG, β-fibrinogen; CI, confidence interval; FII, factor II; FXIII, factor XIII; FV, factor V; HPA, human platelet alloantigen; MTHFR, methylenetetrahydrofolate reductase; NA, not applicable (no child was heterozygote or homozygote); NI, not included; OR, odds ratio; PAI, plasminogen activator inhibitor.
aapoE ∊4 group (carriers of at least 1 apoE ∊4 allele).
Significant P values (P < .05) and risk factors were indicated in bold face.
Distribution of genotype frequencies of investigated polymorphisms in children with and without identified etiology of AIS is presented in Table 5. Genotype distribution of all investigated polymorphisms was not statistically significantly different between the cases with and without identified etiology. In contrast to perceived overall preponderance of boys among children with AIS, in children without identified etiology, a male to female ratio of 0.5 was in favor of girls.
Distribution of Genotype Frequencies of Investigated Polymorphisms in Children With and Without Identified Etiology of Arterial Ischemic Stroke.
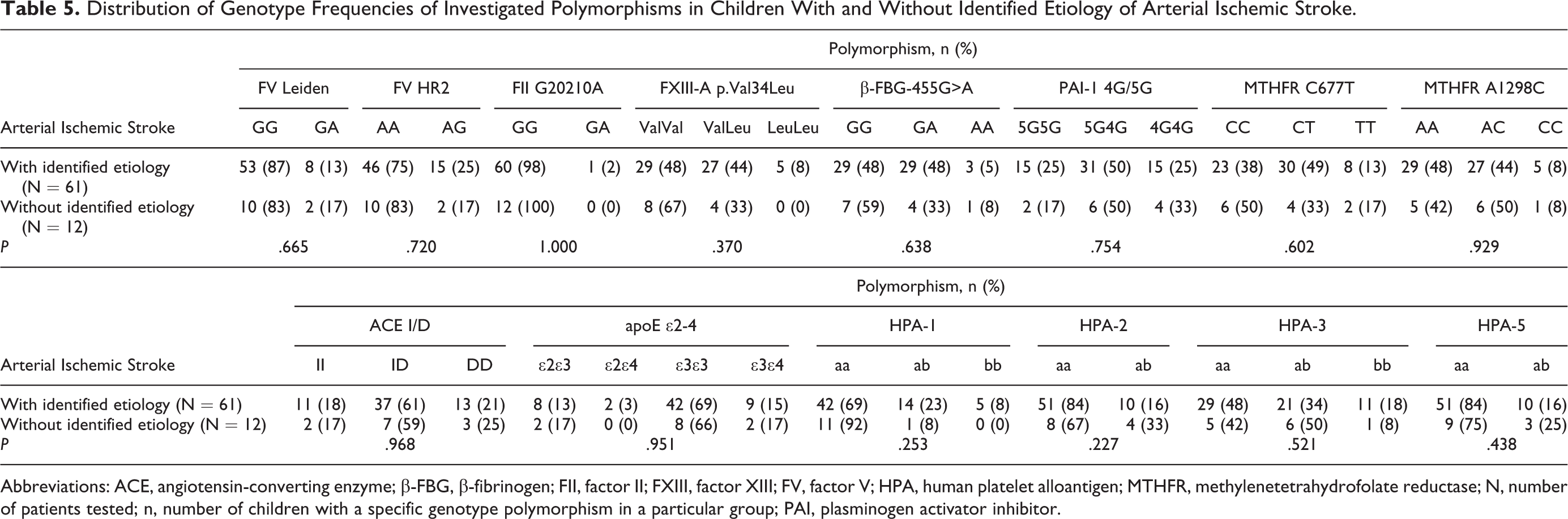
Abbreviations: ACE, angiotensin-converting enzyme; β-FBG, β-fibrinogen; FII, factor II; FXIII, factor XIII; FV, factor V; HPA, human platelet alloantigen; MTHFR, methylenetetrahydrofolate reductase; N, number of patients tested; n, number of children with a specific genotype polymorphism in a particular group; PAI, plasminogen activator inhibitor.
Frequencies of combined genotypes for FV Leiden, FV HR2, MTHFR C677T, and MTHFR A1298C in children with AIS, AIS subgroups, and controls are presented in Table 6.
Distribution of Frequencies of Combined Genotypes for FV Leiden, FV HR2, MTHFR C677T, and MTHFR A1298C Polymorphisms in Children With Perinatal, Childhood Arterial Ischemic Stroke, All Children With Arterial Ischemic Stroke, and Controls.
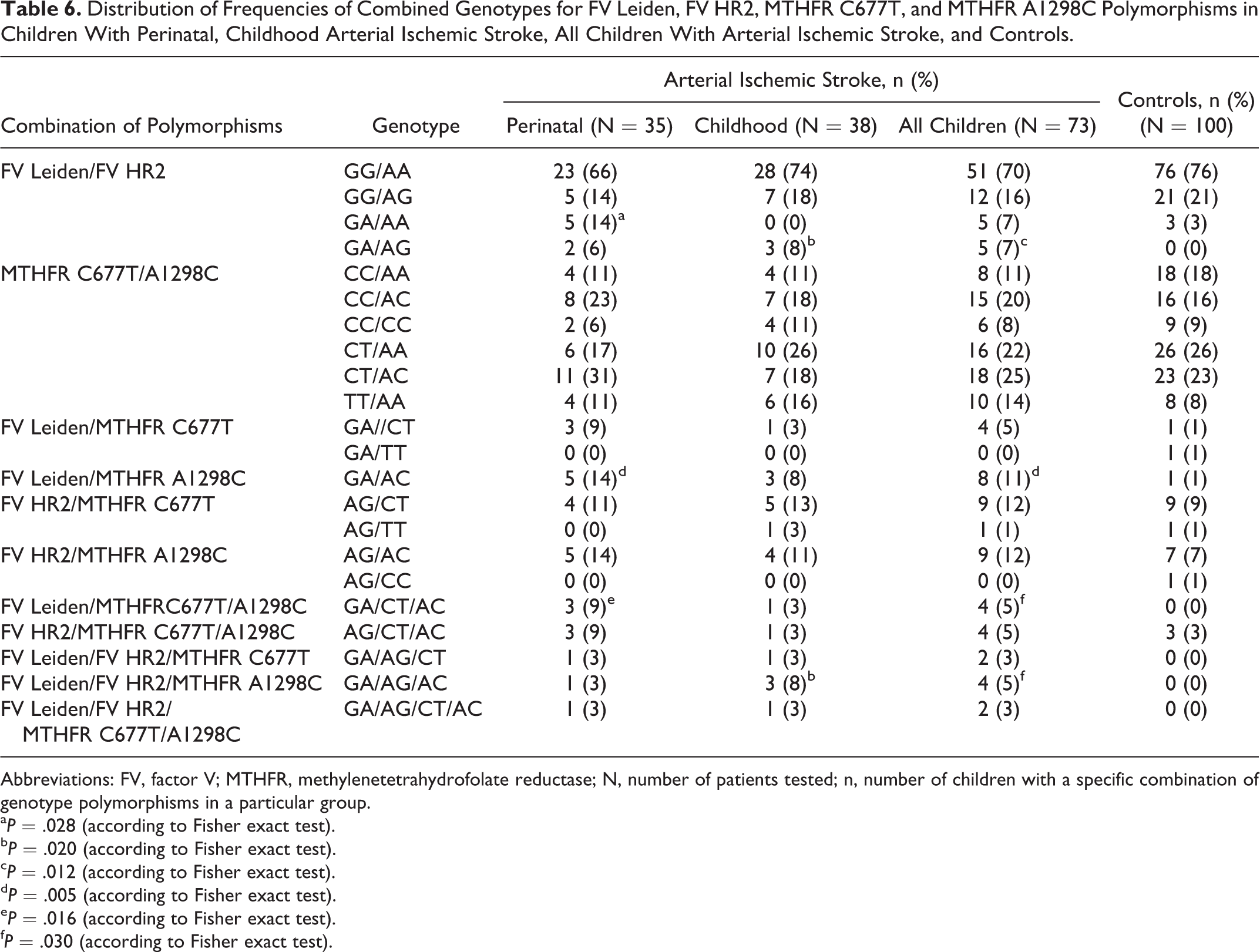
Abbreviations: FV, factor V; MTHFR, methylenetetrahydrofolate reductase; N, number of patients tested; n, number of children with a specific combination of genotype polymorphisms in a particular group.
aP = .028 (according to Fisher exact test).
bP = .020 (according to Fisher exact test).
cP = .012 (according to Fisher exact test).
dP = .005 (according to Fisher exact test).
eP = .016 (according to Fisher exact test).
fP = .030 (according to Fisher exact test).
The highest frequency was found for the wild-type combined genotypes for FV Leiden/FV HR2 (genotype combination GG/AA). A total of 4 combined genotypes for FV Leiden and FV HR2 were identified in children with AIS and 3 in controls. Combined FV Leiden and FV HR2 heterozygosity (genotype combination GA/AG) was identified in children with AIS (P = .012) and in both AIS subgroups (perinatal AIS P = .071; childhood AIS P = .020), but not in controls. A genotype combination GA/AA was observed in the perinatal AIS subgroup only and in controls. The statistically significant difference (P = .028) indicated a 5.39-fold increased risk of perinatal AIS (95% CI = 1.22-23.88).
Six combined genotypes MTHFR C677T/MTHFR A1298C were observed in children with AIS, AIS subgroups, and in controls. No cases of double homozygosity or a combination of heterozygosity and homozygosity were found. Although the highest frequency for double heterozygosity was identified in children with AIS and in the perinatal AIS subgroup, the statistical significance was not reached (P = .942 and P = .446, respectively). Additional analysis was performed for combined genotypes for FV Leiden, FV HR2, MTHFR C677T, and MTHFR A1298C.
Factor V Leiden/FV HR2/MTHFR C677T/MTHFR A1298C heterozygosity was detected in children with AIS and in the AIS subgroups, but not in controls. However, this association did not reach statistical significance (P = .177). Among other analyzed genotype combinations, 8 were detected in AIS, whereas 5 of them were detected in controls. A statistically significant difference in frequencies was found for the following combinations: FV Leiden/MTHFR A1298C (P = .005) and FV Leiden/MTHFR C677T/MTHFR A1298C (P = .016) in the perinatal AIS subgroup and FV Leiden/FV HR2/MTHFR A1298C (P = .020) in the childhood AIS subgroup. The calculated OR for FV Leiden/MTHFR A1298C combination revealed 16.5-fold increased risk of perinatal (95% CI = 1.85-146.78) and 12.18-fold risk of AIS (95% CI = 1.49-99.73). Regarding other genotype combinations, the only case with FII G20210A mutation (heterozygote) was the carrier of 6 additional heterozygous mutations (FV HR2, β-FBG-455G>A, PAI-1 4G/5G, MTHFR C677T, HPA-2, and HPA-3).
None of children with moyamoya disease had FV Leiden, FV HR2, or FII G20210A identified, whereas all 3 children were carriers of apoE ∊3∊3 and were at the same time heterozygotes for PAI-1 4G/5G. Among other identified polymorphisms, 2 of 3 children with moyamoya were also homozygotes for the rare HPA-1b allele (HPA-1 b/b genotype) and heterozygotes for the ACE I/D polymorphism. One child with moyamoya having HPA-1b/b genotype was also homozygote for MTHFR C677T, heterozygote for FXIII-A p.Val34Leu, β-FBG 455G>A, HPA-3, and HPA-5, and had elevated Lp(a) concentration (0.72 g/L).
Among the investigated cases, 4 children had a recurrent AIS, corresponding to an overall recurrence rate of 5.5%, and all were the boys. As none of the children with perinatal AIS had a recurrent stroke, the recurrence rate for childhood AIS corresponded to 10.5%. Among patients with recurrent stroke, none of the patients had FV Leiden, FV HR2, or FII G20210A, 1 was homozygous for MTHFR C677T, whereas 1 had elevated Lp(a) concentration. Furthermore, HPA 2a/b genotype was identified in half of the cases with recurrent childhood AIS as compared to 7 of 34 cases with nonrecurrent childhood AIS (50% vs 20%). Three of 4 patients with recurrent childhood AIS had combined apoE ∊2∊3 and ACE I/D genotype, compared to 2 of 34 cases with nonrecurrent childhood AIS (75% vs 6%).
Discussion
Stroke in children is a heterogeneous multifactorial disorder, with a wide range of inherited and acquired risk factors identified to date. Despite extensive investigation of potential additional risk factors, especially in the last 10 years, the etiology of AIS is still undetermined in up to 30% of children. 1,10,12,28,29
The association of FV Leiden, FII G20210A, and MTHFR C677T with AIS in children has been demonstrated in numerous population studies including a recently published meta-analysis by Kenet et al. 30 However, obtained results have shown great variation in frequencies and distribution among different populations. When considering these 3 common polymorphisms, results of our case–control study confirmed only the association between FV Leiden and AIS. On the contrary, no association with FII G20210A and MTHFR C677T was identified, corroborating the findings already obtained in our previous 31 and other population studies. 32 –39 Taking into consideration that genetic risk factors are still incompletely characterized, we extended the genetic profile to other polymorphisms for proteins involved in thrombosis and hemostasis in order to investigate their possible role in the occurrence of AIS. Further genetic analysis focused on the genes encoding proteins of the coagulation (FV HR2, β-FBG-455G>A, FXIII-A p.Val34Leu) and fibrinolytic (PAI-1 4G/5G) systems, platelet function (HPA-1, HPA-2, HPA-3, and HPA-5), homocysteine metabolism (MTHFR A1298C), and intermediate-risk factors (ACE I/D, apoE ∊2-4), which have been stated to predispose for thrombotic events.
Data from the literature regarding the role of these polymorphisms in the etiology of AIS in children are scarce and rely on studies of single polymorphisms in selected populations, whereas a broader genetic profile has been investigated to date in few studies only. 14 –16 Moreover, no positive associations were found, except the association of HPA-1 with AIS in Indian children that was reported in the study of Biswas et al. 14 Logistic regression analysis in the present study revealed moderate protective effect of the presence of at least 1 HPA-3b allele in AIS and confirmed our previously reported association with AIS. 31 As no other study has investigated this polymorphism in children with AIS, it is not clear whether this association is limited to our population or represents a protective factor against the development of the disease.
Polymorphisms of intermediate-risk factors, such as ACE I/D and apoE ∊2-4, are traditionally investigated in adult patients with ischemic stroke as part of the routine genetic profile. In contrast, no study to date has explored the possible association of the ACE I/D polymorphism with AIS in children, whereas the effect of the apoE ∊2-4 polymorphism has been recently explored in 2 studies only. 21,22 The results of our study revealed a modestly increased risk of AIS onset in carriers of at least 1 ACE D allele (OR: 2.07, 95% CI: 0.99-4.32), but this association did not reach statistical significance (P = .052) in neither univariate nor in multivariate logistic regression analysis. As a borderline association was observed, we think that this is worthy to be tested in a higher number of children both in our and other populations.
Elevated Lp(a) levels were found in 7 (10%) of 73 children with AIS, of whom 5 (13%) were from the group of children with childhood AIS. This frequency of children with elevated Lp(a) levels are slightly lower than that reported by Nowak-Göttl et al 40 (26.4%) and Teber et al 41 (26.9%). In 6 of 7 children, of whom 1 had a recurrent stroke, elevated Lp(a) was the only identified risk factor. The remaining child, besides the elevated Lp(a) level, had moyamoya as the underlying condition with several combinations of polymorphisms. These results are similar to the data reported in the study by Teber et al. 41 These authors confirmed elevated Lp(a) as a risk factor for AIS in children as well as relationship of solely elevated Lp(a) for recurrent stroke.
Our study and other studies so far showed that identified risk factors for perinatal and childhood AIS are rather variable. 34 Considering this fact, we also analyzed the genotypic distributions of all investigated polymorphisms separately in both AIS subgroups. Indeed, the present study revealed that distinct polymorphisms were associated with perinatal (FV Leiden and HPA-3) and childhood AIS (FXIII-A p.Val34Leu). Moreover, in perinatal AIS, moderate protective effect was identified in carriers of at least 1 HPA-3b allele, similar to that observed for AIS in all children. Factor V Leiden was significantly associated with perinatal AIS and AIS in all children, but the association was almost 2-fold stronger for perinatal AIS. The associations of these polymorphisms (FV Leiden and HPA-3) were not observed in childhood AIS, where a moderately increased risk in carriers of at least 1 FXIII-A Leu34 allele was obtained. It has been demonstrated that the presence of the FXIII-A Leu34 allele is associated with a 2.5-fold faster release of the activation peptide, resulting in increased activation rate of FXIII with thrombin that could lead to ineffective cross-linking. 42 According to available data, this is the first study reporting this association, in contrast to results obtained in Turkish 18 , Greek, 15 and Polish 43 children with AIS.
Altogether, it is quite evident that single thrombophilia cannot fully explain AIS in children as it represents only a mild risk factor, as seen in numerous population studies, including this study and a meta-analysis of Kenet et al. 30 Based on this knowledge, it is reasonable to expect that the presence of combined polymorphisms could have a stronger effect compared to single polymorphisms. In line with this, it has been reported that combined FV Leiden/FV HR2 heterozygosity is associated with 3- to 6-fold increased relative risk of venous thromboembolism compared to FV Leiden alone. 44,45 In the present study, we identified double heterozygosity for the combination of polymorphisms FV Leiden and FV HR2 (genotypes GA/AG) and triple heterozygosity for the combinations FV Leiden/FV HR2/MTHFR A1298C and FV Leiden/MTHFR C677T/MTHFR A1298C in children with AIS, but we could not estimate the exact risk, as these genotypic combinations were not found in controls. Similar to the finding of distinct single polymorphisms associated with the occurrence of perinatal and childhood AIS, the extended analysis of combined polymorphisms revealed different combinations that were statistically significantly related to perinatal and childhood AIS. Although FV Leiden alone was not found to be associated with childhood AIS in our study, another 2 mutations (FV HR2 and MTHFR A1298C) in all 3 heterozygous FV Leiden carriers had a significant additive effect (P = .016). In contrast to childhood AIS, synergistic effect of HR2 heterozygosity was not noticed in FV Leiden heterozygous carriers but, on the contrary, HR2 wild-type homozygosity reduced the risk of perinatal AIS (OR from 8.29 to 5.39). Moreover, in perinatal AIS, a strong additional effect of MTHFR polymorphism was noticed where MTHFR A1298C heterozygosity doubled the risk of perinatal AIS onset in FV Leiden heterozygous carriers (OR, 16.5 in comparison with 8.29). This is in line with reported OR = 18.75 for ≥2 genetic traits as risk factors for AIS onset calculated in a meta-analysis by Kenet et al. 30
In conclusion, this case–control study confirmed the already documented association between FV Leiden and AIS in children. It also showed that other previously unreported single polymorphisms (FXIII-A p.Val34Leu, HPA-3) and several genotype combinations that always include heterozygous FV Leiden can be related to AIS in Croatian population. Our study clearly shows that perinatal and childhood AIS do not share the same inherited prothrombotic risk factors and corroborates the opinion of Trenor and Michelson about different underlying mechanisms and possibly different role of thrombophilia in these AIS subgroups. 46
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Croatian Science Foundation (Grant IP-2014-09-2047).
