Abstract
Background:
Bridge therapy is associated with an increased risk of major bleeding in patients with atrial fibrillation and venous thromboembolism (TE) without a corresponding reduction in TE. The benefits of bridge therapy in patients with mechanical heart valve (MHV) prostheses interrupting warfarin for invasive procedures are not well described.
Methods and Results:
A retrospective cohort study was conducted at an integrated health-care delivery system. Anticoagulated patients with MHV interrupting warfarin for invasive diagnostic or surgical procedures between January 1, 2006, and March 31, 2012, were identified. Patients were categorized according to exposure to bridge therapy during the periprocedural period and TE risk (low, medium, and high). Outcomes validated via manual chart review included clinically relevant bleeding, TE, and all-cause mortality in the 30 days following the procedure. There were 547 procedures in 355 patients meeting inclusion criteria. Mean cohort age was 65.2 years, and 38% were female. Bridge therapy was utilized in 466 (85.2%) procedures (95.2%, 77.3%, and 65.8% of high, medium, and low TE risk category procedures, respectively). The 30-day rate of clinically relevant bleeding was numerically higher in bridged (5.8%; 95% confidence interval [CI], 3.9%-8.3%) versus not bridged procedures (1.2%; 95% CI, <0.1%-6.7%; P = .102). No TEs or deaths were identified.
Conclusion:
The use of bridge therapy is common among patients with MHV and may be associated with increased bleeding risk. Further research is needed to determine whether bridge therapy reduces TE in patients with MHV interrupting warfarin for invasive procedures.
Introduction
Anticoagulation with warfarin is the gold standard for the prevention of thromboembolism (TE) in patients with mechanical heart valve (MHV) prostheses. 1 Approximately 10% of patients receiving anticoagulation undergo invasive diagnostic or surgical procedures each year. 2 Clinicians caring for these patients are faced with a common clinical predicament: balancing the risk of bleeding if anticoagulation continues throughout the procedure against the risk of TE if anticoagulation is interrupted.
Given the gradual offset of warfarin’s effect following discontinuation and the delayed time to therapeutic anticoagulation following resumption, the risk of TE is likely increased for patients requiring warfarin interruption in the perioperative period. The short-acting anticoagulant (eg, low-molecular-weight heparin) has been used during periprocedural warfarin withdrawal and reinitiation in an attempt to mitigate this risk. This strategy, referred to as “bridge therapy,” is commonly recommended for patients deemed at high TE risk during warfarin interruption. 2
The subset of patients who benefit from bridge therapy is unknown. The American College of Chest Physicians Evidence-Based Clinical Practice Guidelines classifies periprocedural risk as high (>10% per year), moderate (5%-10% per year), or low (<5% per year) depending on the annual risk of TE without anticoagulation therapy. 2 Historically, bridge therapy has been suggested for most patients receiving warfarin for MHV at moderate or high risk of TE. 2
Recent evidence has confirmed bridge therapy is associated with an increased risk of major bleeding in patients with atrial fibrillation (AF) and venous thromboembolism (VTE) without the corresponding reduction in TE. 3,4 This has prompted widespread reevaluation of bridge therapy protocols and calls to reduce patient exposure to bridge therapy, including those with MHV. 5 As bleeding and TE outcome-associated bridge therapy in patients with MHV are not well described, 6 –11 the aim of this study was to compare the 30-day rates of clinically relevant bleeding, TE, and all-cause mortality among patients with MHV prosthesis that interrupted warfarin for an invasive surgical or diagnostic procedure who did or did not receive bridge therapy.
Methods
Study Design and Setting
This retrospective cohort study was conducted at Kaiser Permanente Colorado (KPCO), an integrated health-care delivery system that provides care to >635 000 members. Each year there are approximately 2400 procedures requiring coordination of periprocedural warfarin therapy at KPCO. The centralized, telephone-based Clinical Pharmacy Anticoagulation and Anemia Service (CPAAS) provides anticoagulation services within KPCO. 12 Periprocedural warfarin therapy plans are developed by CPAAS pharmacists using a collaborative drug therapy management guideline and are approved by the referring physician and proceduralist. Detailed information regarding each periprocedural plan is recorded in an electronic patient tracking tool (DAWN AC; 4S Information Systems, Ltd, Cumbria, United Kingdom) and the electronic medical record (EMR). The institutional review board of KPCO approved all study activities. Informed consent was waived as this was a retrospective study.
Study Patients
This study included patients who had invasive diagnostic or surgical procedures between January 1, 2006, and March 31, 2012. Included patients (1) were at least 18 years, (2) were monitored by CPAAS, (3) received warfarin therapy for MHV prosthesis, (4) had an international normalized ratio (INR) ≤1.5 on the day of or on the day before the procedure, (5) had at least 180 consecutive days of KPCO membership prior to the procedure, (6) resumed warfarin therapy within 30 days postprocedure, and (7) did not have a subsequent procedure-related warfarin interruption within 90 days.
Study Outcomes
The primary efficacy outcome was objectively confirmed TE (including stroke, arterial TE, and heart valve thrombosis). The primary safety outcome was clinically relevant bleeding. Clinically relevant bleeding was defined as any clinically overt bleeding, regardless of severity, resulting in hospitalization or emergency department (ED) visit, or bleeding that complicated the procedure. The secondary outcome was all-cause mortality. Outcomes were identified from the day of procedure through the following 30 days. Thirty-day follow-up was chosen as events occurring during this period are considered to most likely represent procedure-related events. 13
Data Collection
Potential study patients were identified using KPCO electronic administrative data sets. Information pertaining to the type of procedure was gathered from DAWN AC and/or the EMR. Procedures were categorized as gastrointestinal endoscopy; spinal/intracranial; orthopedic; dermatologic; abdominal/thoracic (major and nonmajor); urologic; dental; vascular; ears, eyes, nose, throat; pacemaker/implantable cardiac defibrillator; or other procedures. Bleeding and TE events were identified administratively using predefined International Classification of Diseases, Ninth Revision (ICD-9) diagnosis codes and confirmed via manual chart review by 2 study team members (S.M.M., N.P.C.) using a standardized abstraction form. Disagreements were resolved by a third reviewer (D.M.W.). Comorbidities, including history of stroke/transient ischemic attack (TIA), diabetes, heart failure, hypertension, and AF, present in the 180 days prior to the procedure were identified administratively using predefined ICD-9 codes. The use of bridge therapy was determined by identifying dispensings of parenteral anticoagulants recorded in the KPCO pharmacy database and the manual review of periprocedural plans recorded in DAWN AC.
Data Analysis
Patients could contribute multiple procedures to the analysis provided that each met inclusion criteria and was separated from the previous procedure by at least 90 days. No formal power calculation was performed as all procedures meeting inclusion criteria were analyzed. Patient characteristics were summarized using descriptive statistics. Patients were categorized according to the TE risk definitions outlined in the ninth edition of the American College of Chest Physician Guidelines on antithrombotic therapy (high, medium, and low—see Table 1 for definitions) 2 and bridging status (bridged vs not bridged). Thirty-day bleeding and TE rates were calculated by dividing the counts of each event by the total number of included procedures and multiplying by 100. Categorical and interval-level data were reported as percentages and means (±standard deviations), respectively. Chi-square tests of association and t tests were used to compare categorical and interval-level variables, respectively. Subanalysis was performed on the bleeding outcome using only a patient’s first procedure during the study period. Adjustment for potential confounding factors was not performed due to the low rates of outcomes in the not bridged group. Statistical analyses were performed using SAS 9.4 (SAS, Cary, North Carolina) and Intercooled STATA 9.0 (StataCorp, College Station, Texas).
Thromboembolism Risk Stratum for Patients With Mechanical Valve Prosthesis.

Abbreviation: AT9, Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. 2
Results
There were 547 procedures in 355 patients meeting inclusion criteria (Figure 1). The mean age of the cohort was 65.2 ± 10.9 years, and 38% were female (Table 2). Warfarin therapy was most commonly interrupted for gastrointestinal endoscopic, orthopedic, and nonmajor abdominal/thoracic procedures (Table 3).
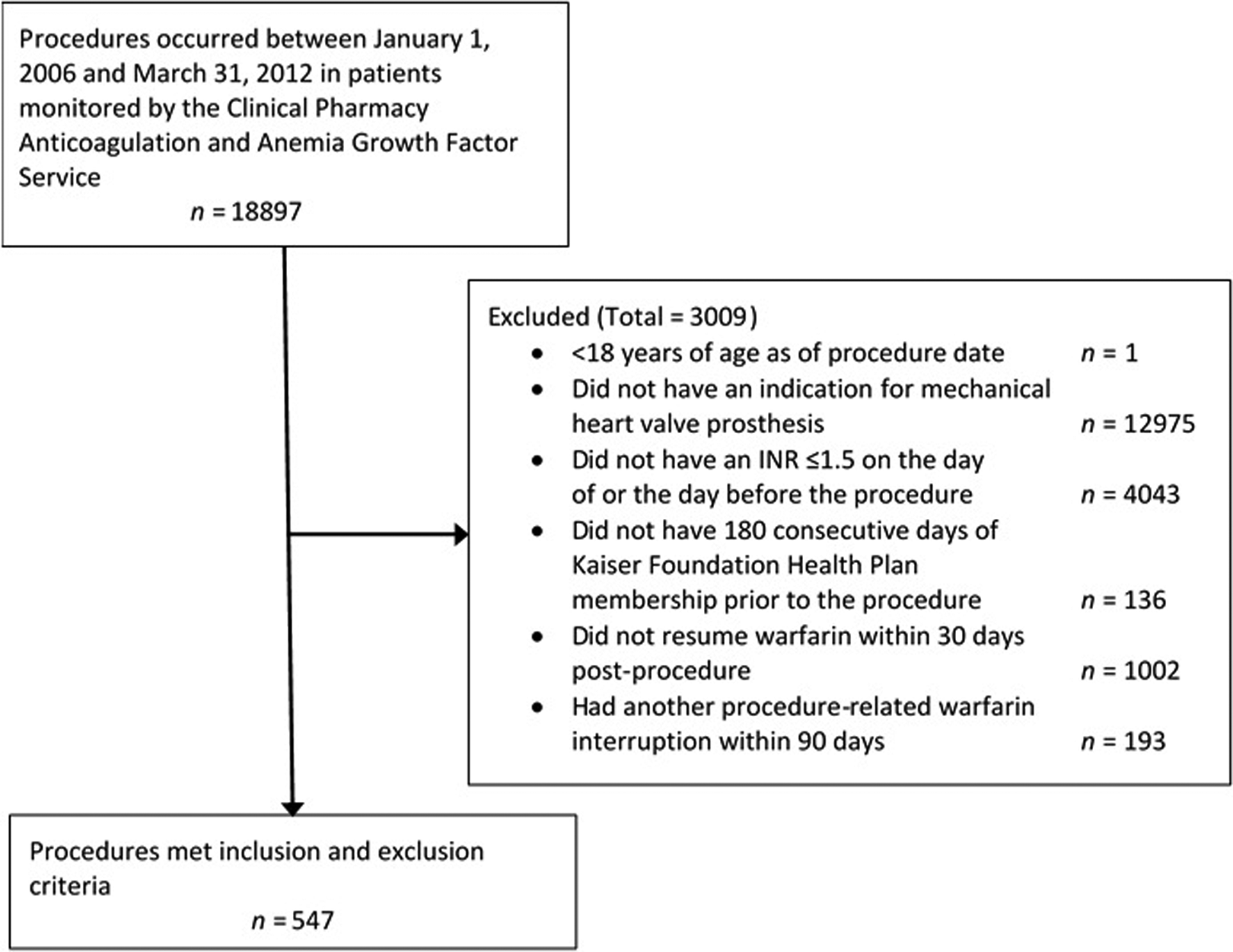
Patient dispositions.
Patient Characteristics Overall and by Bridging Status.a
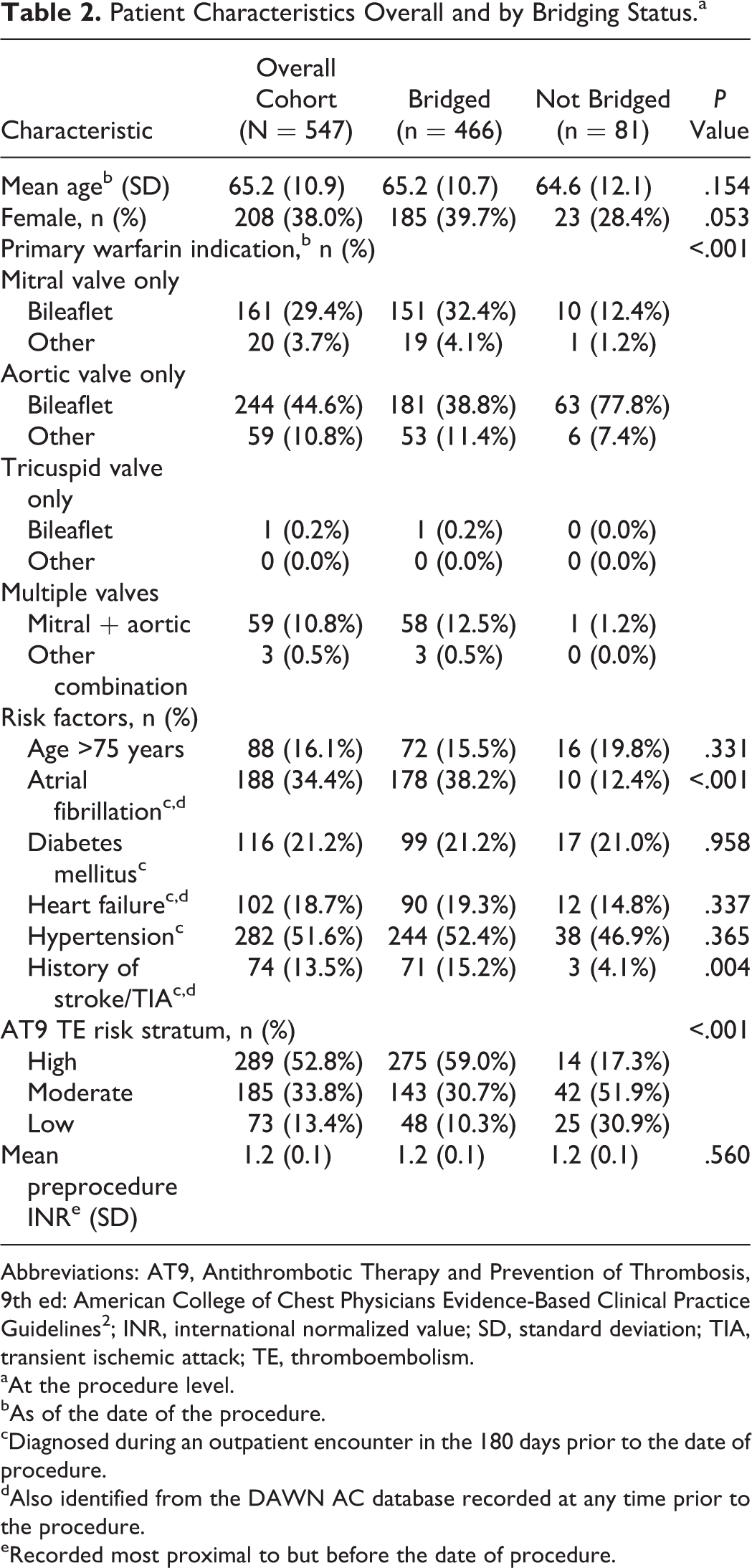
Abbreviations: AT9, Antithrombotic Therapy and Prevention of Thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines 2 ; INR, international normalized value; SD, standard deviation; TIA, transient ischemic attack; TE, thromboembolism.
aAt the procedure level.
bAs of the date of the procedure.
cDiagnosed during an outpatient encounter in the 180 days prior to the date of procedure.
dAlso identified from the DAWN AC database recorded at any time prior to the procedure.
eRecorded most proximal to but before the date of procedure.
Procedures Characteristics Overall and by Bridging Status.
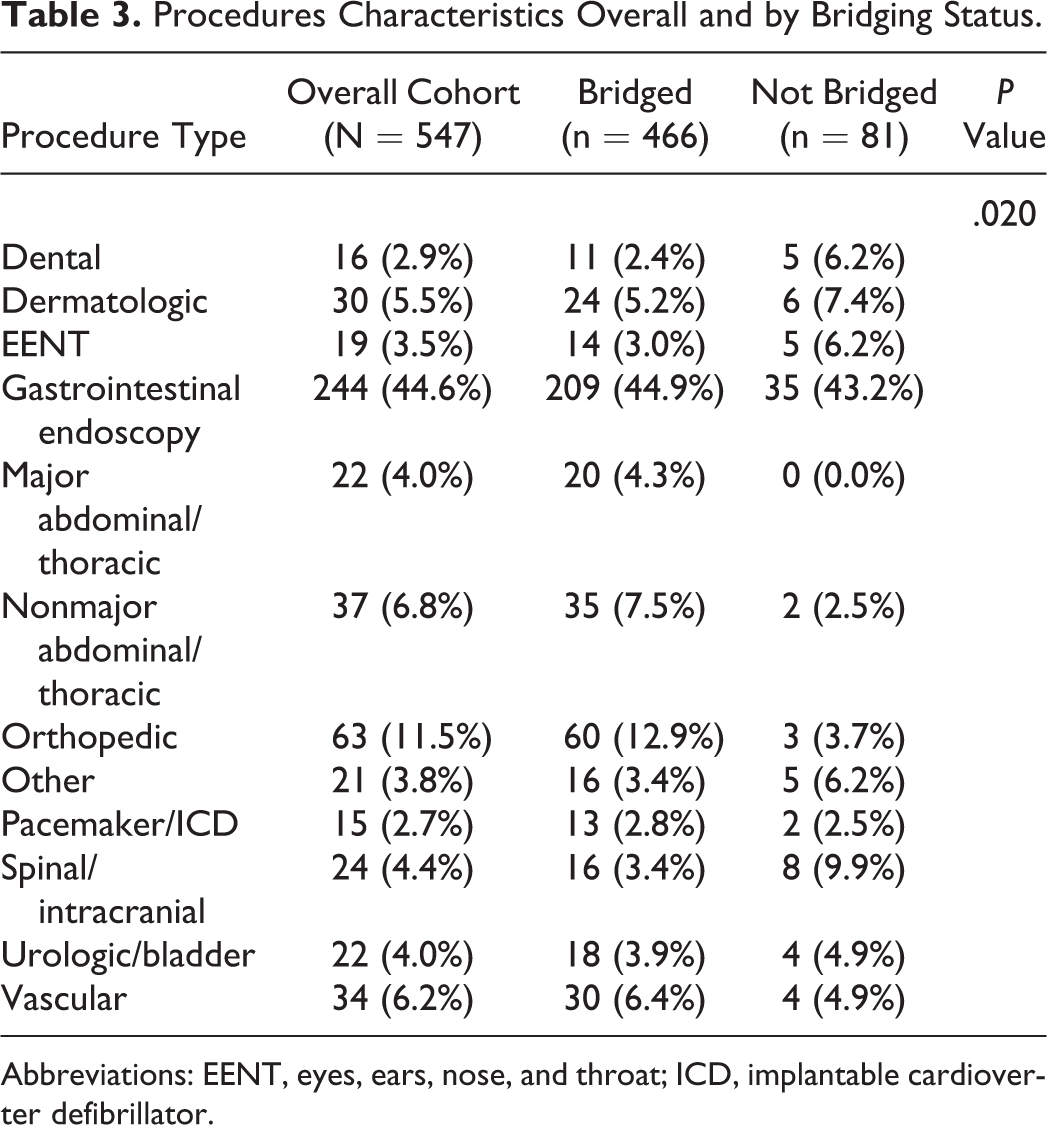
Abbreviations: EENT, eyes, ears, nose, and throat; ICD, implantable cardioverter defibrillator.
Bridge therapy was utilized in 466 (85.2%) procedures (95.1%, 77.5%, and 65.8% of high, medium, and low TE risk category procedures, respectively). Bridge therapy was used more often for patients with mitral bileaflet valves, whereas patients with aortic bileaflet valves without other risk factors were less likely to have been bridged (P < .001). In addition, bridged procedures were more common among patients with AF or history of stroke/transient ischemic events (both P < .01). Bridged procedures were classified as low, moderate, and high TE risk in 10.3%, 31.1%, and 58.6% of procedures, respectively. Procedures performed in patients classified as high TE risk were more likely to have received bridge therapy than those with a low or moderate TE risk (P < .001).
The 30-day rate of clinically relevant bleeding was numerically higher in bridged procedures (5.8%; 95% confidence interval [CI], 3.9%-8.3%) compared to not bridged procedures (1.2%; 95% CI, <0.1%-6.7%), but this did not reach statistical significance (P = .102; Table 4). No TEs or deaths were identified. Subanalysis using only the first procedure for each patient (n = 355) provided similar results (30-day rates of clinically relevant bleeding for the bridged and not bridged groups were 5.7% and 1.9%, respectively; P = .242). Of the 27 bleeding events in bridged procedures, 12 (44.4%) were procedure complications and 7 (25.9%) were gastrointestinal bleeding. Of the bleeding events in the bridged group, 17 (63.0%), 6 (22.2%), and 4 (8.3%) occurred in patients with high, moderate, and low TE risk, respectively. The lone clinically relevant bleeding event complicating a procedure in the absence of bridge therapy was gynecological bleeding, which occurred in a low TE risk patient.
Thirty-Day Outcome Rates and 95% Confidence Intervals (CI) by Bridging Status.a,b
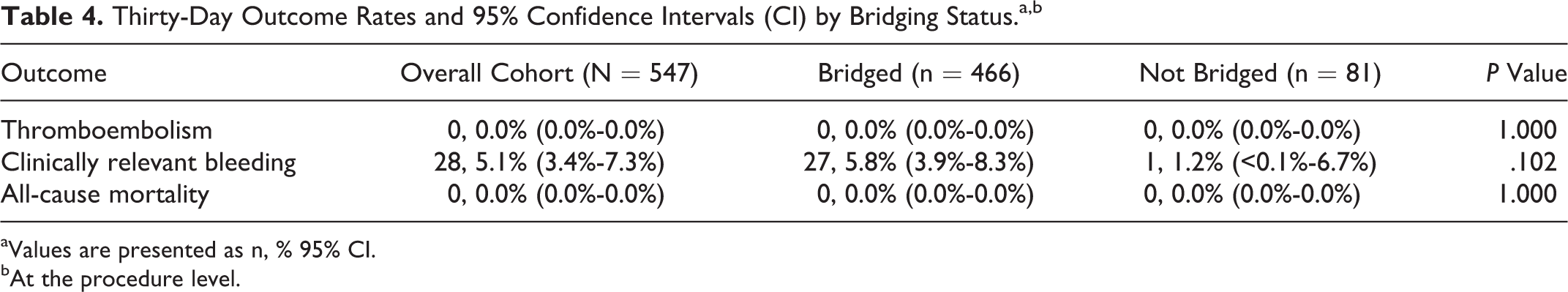
aValues are presented as n, % 95% CI.
bAt the procedure level.
Discussion
Our retrospective cohort study identified that the use of bridge therapy was common among patients with MHV prostheses requiring elective invasive diagnostic or surgical procedures. Patients at higher risk of TE according to the American College of Chest Physicians definitions were more likely to have received bridge therapy. We identified a nearly 4-fold increase in clinically relevant bleeding for procedures that received bridge therapy. Although this result did not achieve statistical significance, it is consistent with the growing body of evidence that suggests bridge therapy is associated with a higher risk of periprocedural bleeding without concomitant reduction in TE risk. 3,4
Existing research examining the risks and benefits of bridge therapy largely has focused on patients with AF or VTE. A recent randomized controlled trial by Douketis and colleagues found placebo injections were noninferior to therapeutic dalteparin bridge therapy for the prevention of stroke and systemic embolism in patients with AF. 3 In addition, dalteparin bridge therapy leads to an almost 3-fold increase in the risk of bleeding. 3 In patients receiving warfarin for VTE, we previously identified a 17-fold increased risk of clinically relevant bleeding in patients who received bridge therapy without a significant difference in the rate of recurrent VTE compared to nonbridged patients. 4 McBane and colleagues did not report statistically significant differences in bleeding outcomes among patients with VTE receiving or not receiving bridge therapy. 14 However, major bleeding occurred in 14 (2.8%) of 499 bridged procedures versus 2 (0.8%) of 256 unbridged procedures in that study. 14
The outcomes of patients with MHV receiving or not receiving bridge therapy at the time of an invasive procedure are not well described. This is a critical gap in clinical knowledge as patients with MHV may be at greater risk for life-threatening TE compared to those with AF or VTE. 15 A prospective cohort of patients with MHV who had a dental procedure in the setting of bridge therapy identified a 30-day bleeding rate of 5.4% with 1 TE complication (0.6%). 16 Unfortunately, outcomes in patients not receiving bridge therapy were not assessed. 16 Daniels and colleagues performed a cohort study of patients with MHV who had an invasive procedure with or without bridge therapy. 17 Similar to our study, the rates of TE were very low and equivalent in the bridged and unbridged groups, while major bleeding was numerically greater in the setting of bridge therapy. 17 Likewise, low INR excursions identified during the course of chronic warfarin therapy likely result in a low risk for subsequent TE complications. 18 To our knowledge, no study has shown low-molecular-weight heparin to be effective in reducing TE complications in patients with MHV during warfarin interruption for invasive procedures.
A study by Talwar and colleagues described a reduction in valve thrombosis with the use of enoxaparin bridge therapy after MHV implantation; however, the onset of thrombosis was 3 to 6 months after surgery. 19 More recently, a study has challenged the use of bridge therapy for patients having mechanical aortic valve replacement surgery. 20 Bridge therapy in that study was associated with a greater risk of bleeding and pericardial effusion compared to no bridging without a reduction in thrombosis during hospitalization. 20
Bridge therapy increases the risk of bleeding in patients with AF or VTE and it is reasonable to expect that similar risk exists for patients with MHV receiving bridge therapy. 3,4 The role of bridge therapy continues to evolve and there may be opportunities to improve the safety of anticoagulant therapy for patients with MHV without incurring avoidable TE in the perioperative setting. Direct evidence demonstrating bridge therapy benefits is absent for any warfarin indication. However, studies have not adequately assessed the safety of a “no bridging” option for high-risk MHV populations, such as those with active or recent thrombosis in the previous 3 months, MHV in the mitral position, caged-ball or tilting disc valves, or patients with a history of stroke or TIA during warfarin therapy or a previous warfarin interruption. 5 Until more data comparing bridging and no bridging outcomes in patients with MHV become available, continuing to offer bridge therapy to these high-risk MHV populations is sensible. On the other hand, low-molecular-weight heparin bridge therapy can be avoided in patients with an uncomplicated bileaflet MHV in the aortic position. 2
Our study had several limitations. Although no TE events were observed in either the bridged or the not bridged groups, this supports the premise that the potential to reduce the risk of TE during invasive procedures using bridge therapy is small, particularly in comparison with the magnitude of increased bleeding risk. We defined bleeding as clinically relevant without further categorization of bleeding severity (eg, “major” bleeding). Our definition of bleeding included only events that necessitated ED or hospital readmission or those complicating the procedure. We believe these events are important from the patient’s perspective and both the clinical and resource utilization viewpoint. The low bleeding event rate in our not bridged group precluded us from performing adjusted analysis to control for any potential confounding factors.
Both our study and the study by Daniels and colleagues 17 were likely underpowered to detect statistically significant differences in bleeding rates between the bridged and not bridged groups. However, the findings from these 2 analyses should be used as pilot data to justify a randomized, placebo-controlled trial of low-molecular-weight heparin bridge therapy for patients with MHV requiring warfarin interruption for elective invasive procedures.
Conclusion
The use of bridge therapy is common among patients with MHV interrupting warfarin for elective invasive procedures and may be associated with an increased bleeding risk. Further research with adequate power to identify important differences in TE risk among patients with MHV receiving and not receiving bridge therapy is needed.
Footnotes
Authors’ Note
This study was conducted at Kaiser Permanente of Colorado. At the time of the study, S.M.M. was a Post-Graduate Year 2 Pharmacy Resident at Kaiser Permanente Colorado. Portions of this study were presented by S.M.M. at the Mountain States Conference in Salt Lake City, Utah, on May 13, 2015. T.D. and N.P.C. had full access to all of the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis, including and especially any adverse effects. T.D., S.M.M., D.J., D.M.W. J.D., and N.P.C. contributed substantially to the study design, data analysis and interpretation, and the writing and revision of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for the conduct of the research and/or preparation of the article was provided by Kaiser Permanente Colorado. The sponsor had no role in study design, collection, analysis and interpretation of data, writing of the report, or the decision to submit the article for publication.
