Abstract
Interleukins play a central role in the immune system and are involved in a variety of immunological, inflammatory, and infectious disease states including sepsis syndrome. Levels of interleukins may correlate with overall survival and may directly or indirectly affect some of the regulators of coagulation and fibrinolysis, thereby disrupting hemostasis and thrombosis. Our hypothesis is that in sepsis-associated coagulopathies (SACs), interleukins may be upregulated, leading to hemostatic imbalance by generating thrombogenic mediators. We profiled the levels of interleukins IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10 in addition to
Introduction
Interleukins are a group of more than 36 cytokines involved in intercellular communication that have various origins, target receptors, and multiple functions. These signaling proteins have a central role in the immune system and are involved in a variety of immunological, inflammatory, and infectious disease states including sepsis syndrome. 1
Interleukins, including the proinflammatory IL-6 and the anti-inflammatory IL-10, have been shown to be involved in the immune response in sepsis, and the levels of these cytokines may correlate with overall survival. The immune response in sepsis can be characterized into 2 stages: a cytokine-mediated hyperinflammatory phase with a high survival rate, followed by a secondary phase of immune suppression. 2 Serum levels of IL-6 have been shown to be related to disease severity and correlate with the outcome of patients with sepsis. 3 In addition, the levels of IL-10 are elevated in patients with sepsis production of interferon γ, and tumor necrosis factor α (TNF-α) is impaired. 4 Previous studies demonstrated that a lower production of IL-6 was associated with a better prognosis, whereas an increase in IL-10 was predictive of severity and a fatal outcome. 5
Interleukins may affect survival in sepsis through their involvement in sepsis-associated coagulopathies (SACs).
6
The SAC is a complex syndrome that is characterized by widespread coagulation followed by possible induction of severe bleeding with multiorgan failure.
7
Interleukins may directly or indirectly affect regulators of coagulation and thereby disrupt hemostasis. In vitro, IL-1β and other inflammatory cytokines can elicit prothrombotic changes in treated cells.
8
Additionally, IL-6 has been shown to stimulate the formation of new platelets with increased prothrombotic activity due to increased sensitivity to thrombin activation.
8
van der Poll et al demonstrated that in models of sepsis in chimpanzees, elimination of IL-6 attenuates coagulation.
9
Cytokines such as IL-6 and TNF-α mediate tissue factor-dependent hypercoagulable reactions triggered by endotoxin in gram-negative sepsis.
10
In a recent study, comparing inflammatory mediators and hemostatic markers in patients with sepsis and in patients with overt disseminated intravascular coagulation (DIC), the levels of IL-6 and IL-10 were shown to be higher in patients with overt DIC than in patients with sepsis and non-overt DIC.
11
Another analyzed
Interleukins have been considered a possible therapeutic target for increasing patient survival in sepsis. Historically, a recombinant human IL-1 receptor antagonist failed to show an increase in survival time in patients with sepsis syndrome. 13 More recently, IL-7 has been investigated as possible therapeutic agent in sepsis with some promising results. Kasten et al showed that treatment with IL-7, an antiapoptotic cytokine-accelerated neutrophil recruitment, improved bacterial clearance and enhanced survival in sepsis. 14 Others have shown that in addition to its already known efficacy in bacterial and viral infections, IL-7 improves survival in fungal sepsis. 15 To better understand the role of interleukins in the pathogenesis of SAC, we undertook a systematic assessment of interleukins in patients with SAC. We hypothesized that in sepsis, interleukins may be upregulated, leading to hemostatic imbalance through the generation of thrombogenic mediators.
Materials
Plasma samples (N = 130) from patients with presumed sepsis-associated clotting disorders were retrospectively collected from Loyola University under an approved institutional review board. Patients older than 18 years with sepsis as defined by the American College of Chest Physicians/Society of Critical Care Medicine consensus definition and patients with DIC as defined by the International Society on Thrombosis and Hemostasis scoring system were included. This scoring system is based on platelet count and prothrombin time–international normalized ratio. Citrated platelet poor plasma was aliquoted and stored at −70ºC and thawed immediately before analysis. The DD levels were determined in patients with SAC to determine the extent of coagulation. Coagulopathy was evident in all baseline plasma samples from patients with SAC as the DD levels are markedly increased in comparison to the normal plasma samples (Table 1).
The
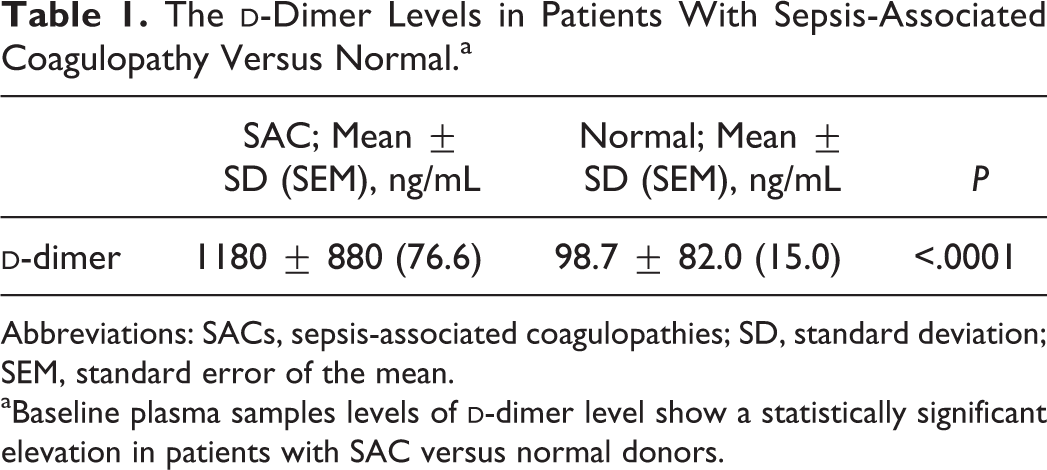
Abbreviations: SACs, sepsis-associated coagulopathies; SD, standard deviation; SEM, standard error of the mean.
aBaseline plasma samples levels of
Plasma samples (n = 50) from normal donors (CORE Set; George King Bio-Medical, Inc, Overland Park, Kansas) were utilized as controls. Donors came from a geographically diverse population, are nonsmoking, are nonmedicated, and are confirmed to be nonreactive for HIV, Hepatitis C Virus (HCV), and Hepatitis B surface antigen (HBsAg).
Methods
A Randox Technology Cytokine Array I and High-Sensitivity Biochip Array (Antrim, Northern Ireland) was used for the quantitation of IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10 in SAC and normal samples.
The DD levels in SAC and normal samples were quantified using an enzyme-linked immunosorbent assay method (Hyphen BioMed, Andresy, France). Absorbance measurements were obtained using an ELISA plate reader SPECTRAmax PLUS (Molecular Devices, Sunnyvale, California).
Statistical Analysis
Graphpad Prism Software (Version 7.0) was utilized to statistically analyze and graphically visualize interleukin and DD levels in plasma samples from patients with SAC versus normal donors.
Descriptive analysis was used to obtain mean, standard deviation, standard error of the mean, and quartiles for interleukin levels in plasma samples from patients with SAC versus normal donors. Additionally, Student
Graphs were generated to compare mean and standard deviation of each interleukin in patients with SAC versus normal donors. In addition, a boxplot analysis was generated for each interleukin.
The fold increase for each interleukin in patients with SAC with respect to normal donors was calculated and graphed with a logarithmic y-axis. Finally, Spearman
Results
Descriptive statistics show that the levels of all interleukins are increased in plasma from patients with SAC compared to normal donors (Table 2). Additionally, this increase is statistically significant (
Profile of Interleukins in Patients With Sepsis-Associated Coagulopathy Versus Normal Donors.a
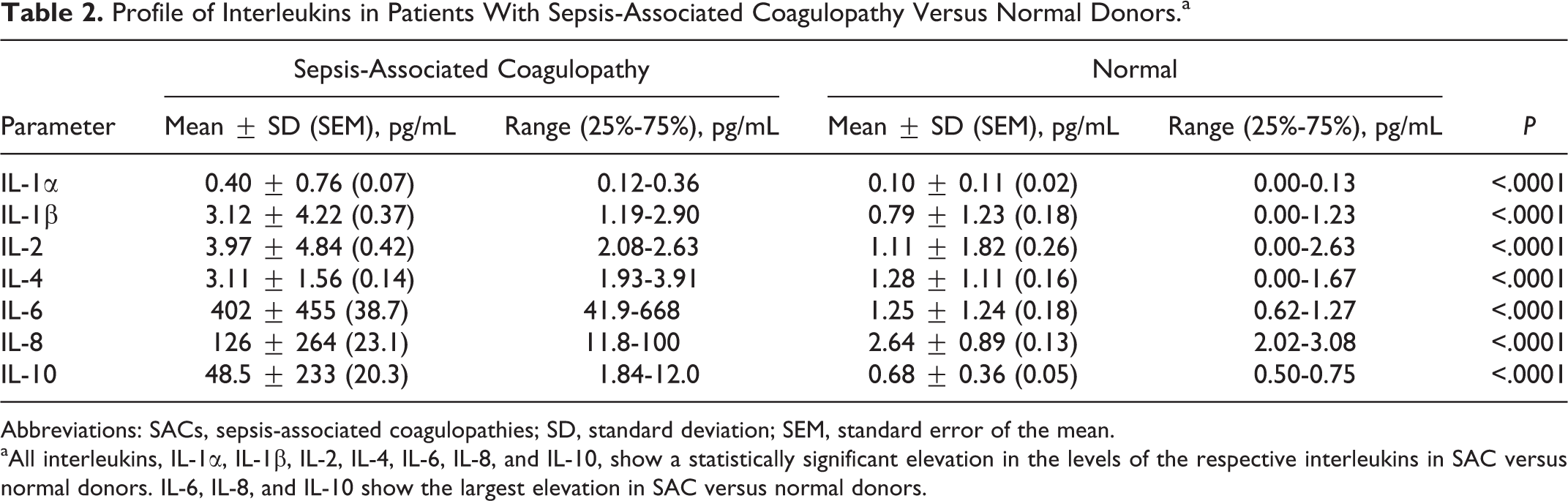
Abbreviations: SACs, sepsis-associated coagulopathies; SD, standard deviation; SEM, standard error of the mean.
aAll interleukins, IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10, show a statistically significant elevation in the levels of the respective interleukins in SAC versus normal donors. IL-6, IL-8, and IL-10 show the largest elevation in SAC versus normal donors.
Increase in Interleukin Levels in Patients With SAC Versus Normal Donors
Figure 1 tabulates the mean and standard deviation of interleukins in patients with SAC compared to healthy donors. There was a statistically significant elevation (
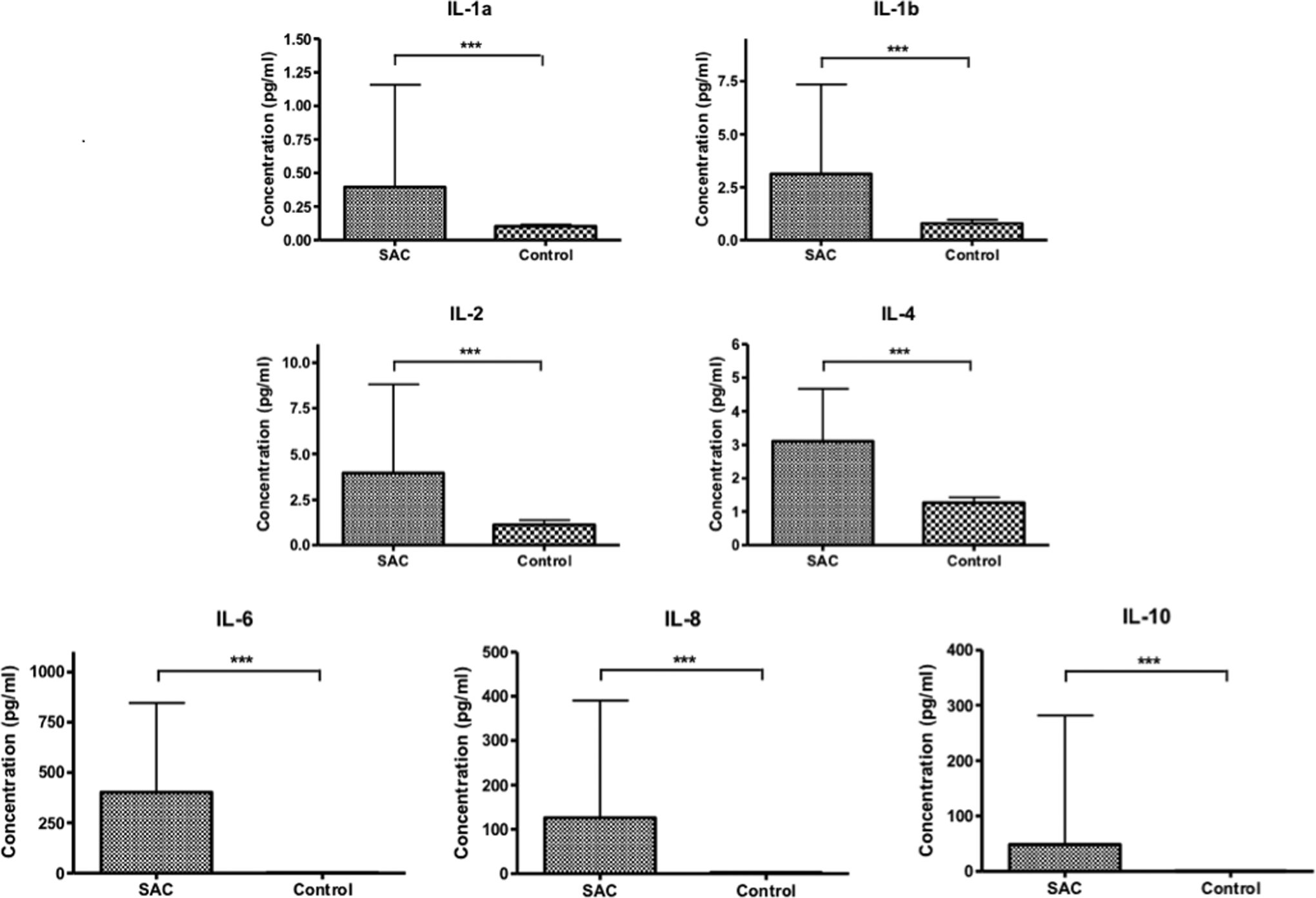
Comparison of mean and standard deviation of interleukin levels in patients with sepsis-associated coagulopathies (SACs) versus normal donors. The
Distribution of Various Interleukins in Plasma Samples Collected From Patients With SAC Compared to Healthy Individuals
Figure 2 shows a boxplot analysis generated for each interleukin that visually depicts a wide variation in distribution of the levels of interleukins in patients with SAC. Additionally, for most interleukins, the median and interquartile range (IQR) of interleukin levels in patients with SAC are higher than that of normal donors. The median (IQR) values of IL-1α were 0.17 (0.12-0.36) for patients with SAC and 0.10 (0.00-0.13) for normal donors. The median (IQR) values of IL-1β were 1.72 (1.19-2.90) for patients with SAC and 0.00 (0.00-1.23) for normal donors. The median (IQR) values of IL-2 were 2.51 (2.08-2.63) for patients with SAC and 0.00 (0.00-2.63) for normal donors. The median (IQR) values of IL-4 were 2.65 (1.93-3.91) for patients with SAC and 1.41 (0.00-1.67) for normal donors. The median (IQR) values of IL-6 were 189 (41.9-668) for patients with SAC and 0.94 (0.62-1.27) for normal donors. The median (IQR) values of IL-8were 34.6 (11.8-100) for patients with SAC and 2.59 (2.02-3.08) for normal donors. The median (IQR) values of IL-10were 4.39 (1.84-12.0) for patients with SAC and 0.60 (0.50-0.75) for normal donors.
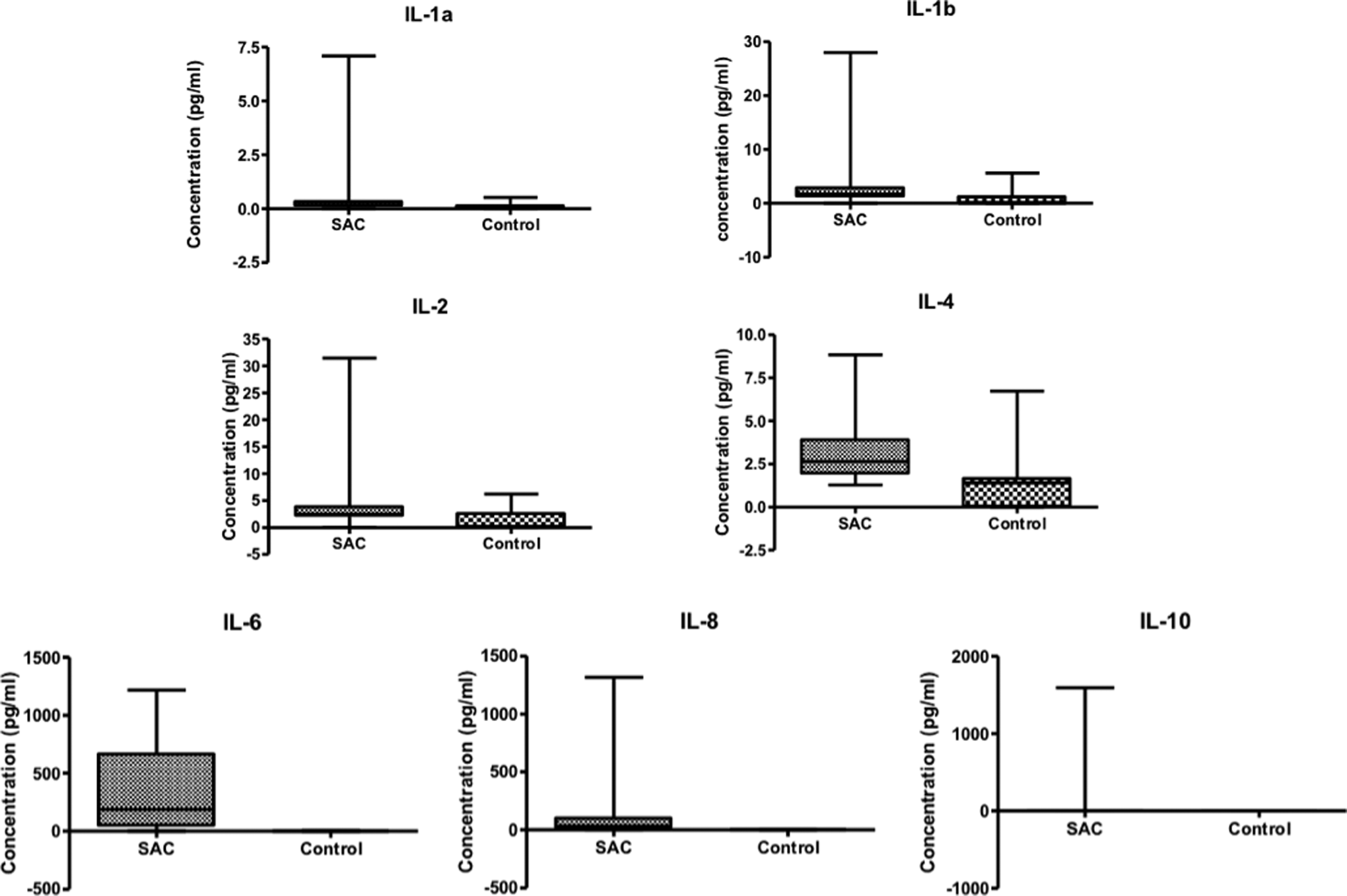
Boxplot analysis to show distribution of various interleukin in plasma samples collected from patients with sepsis-associated coagulopathies (SACs) versus normal donors. Median and interquartile range (IQR) are reported. The interleukins IL-1α (SAC: 0.17 [IQR: 0.12-0.36]; normal: 0.10 [IQR: 0.00-0.13]), IL-1β (SAC: 1.72 [IQR: 1.19-2.90]; normal: 0.00 [IQR: 0.00-1.23]), IL-2 (SAC: 2.51 [IQR: 2.08-2.63]; normal: 0.00 [IQR: 0.00-2.63]), IL-4 (SAC: 2.65 [IQR: 1.93-3.91]; normal: 1.41 [IQR: 0.00-1.67]), IL-6 (SAC: 189 [IQR: 41.9-668]; normal: 0.94 [IQR: 0.62-1.27]), IL-8 (SAC: 34.6 [IQR: 11.8-100]; normal: 2.59 [IQR: 2.02-3.08]), and IL-10 (SAC: 4.39 [IQR: 1.84-12.0]; normal: 0.60 [IQR: 0.50-0.75]).
Relative Increase in Various Interleukin Levels in Patients With SAC Compared to Healthy Individuals
As shown in Figure 3, the most pronounced increases in interleukins in patients with SAC compared to healthy individuals were observed in IL-6 with a fold increase of 322, IL-8 with a fold increase of 48, and IL-10 with a fold increase of 72. Less pronounced fold increases were also observed in IL-1α with a fold increase of 3.88, IL-1β with a fold increase of 3.95, IL-2 with a fold increase of 3.58, and IL 4 with a fold increase of 2.44.
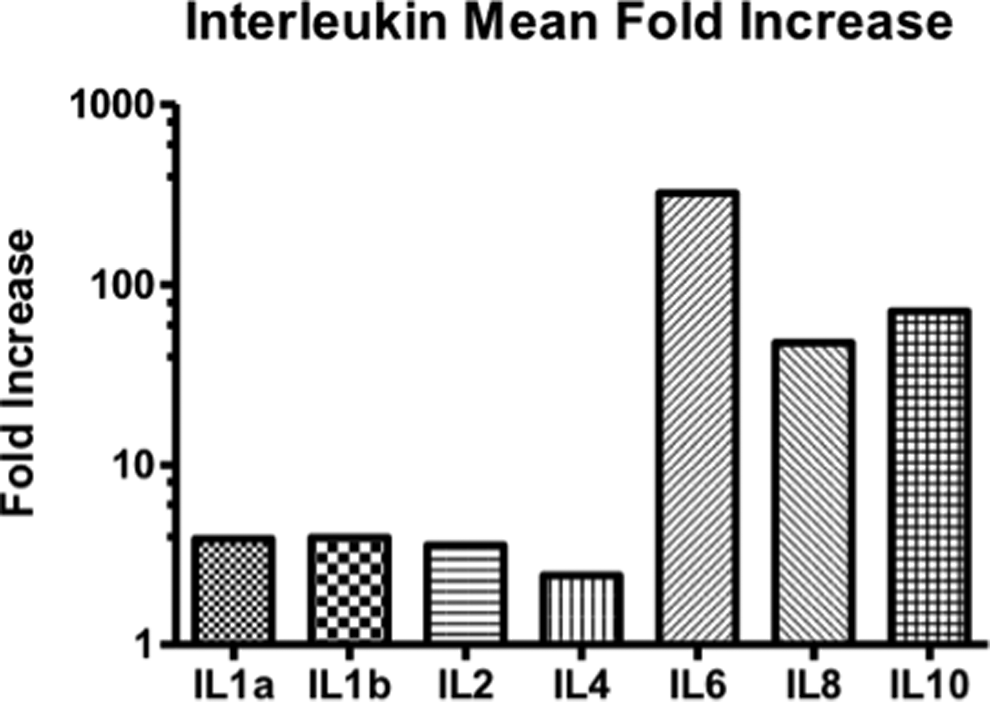
Relative increase in various interleukin levels in patients with sepsis-associated coagulopathies (SACs) as referenced against normal donors. The most pronounced increase in interleukins in patients with SAC versus normal is observed in IL-6 (fold increase: 322), IL-8 (fold increase: 48), and IL-10 (fold increase: 72). Less pronounced fold increase was observed in IL-1a (fold increase: 3.88), IL-1b (fold increase: 3.95), IL-2 (fold increase: 3.58), and IL 4 (fold increase: 2.44).
The Spearman
Spearman
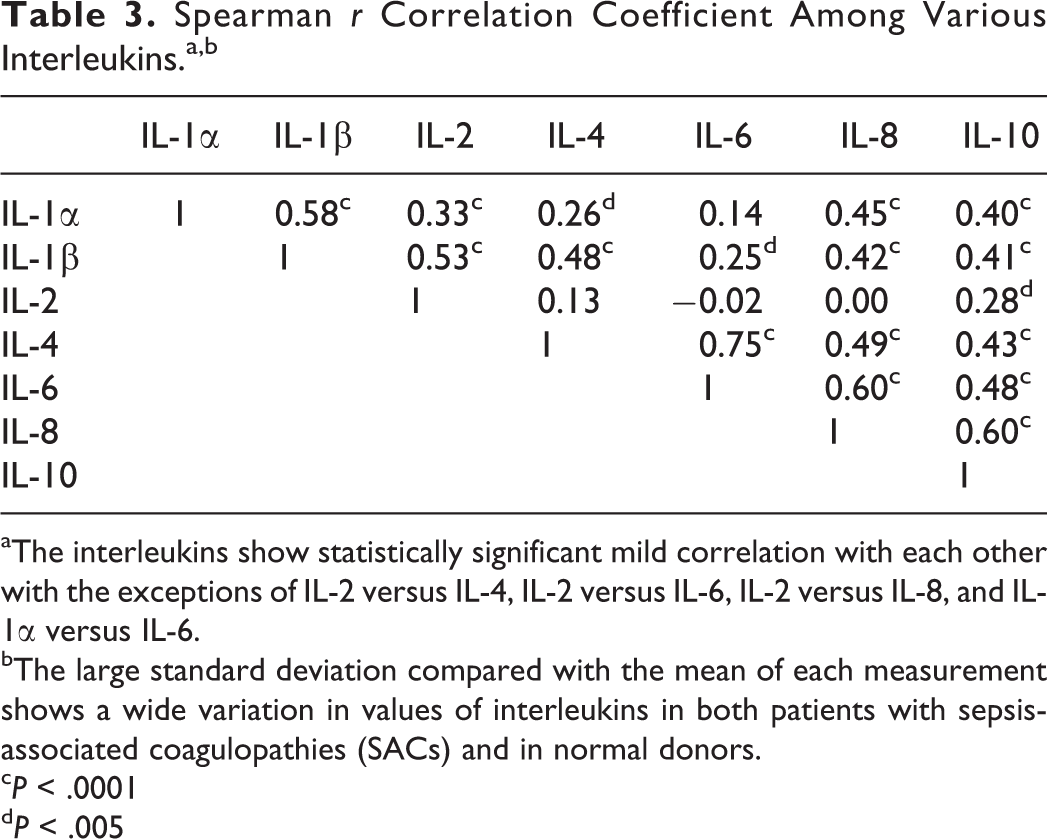
aThe interleukins show statistically significant mild correlation with each other with the exceptions of IL-2 versus IL-4, IL-2 versus IL-6, IL-2 versus IL-8, and IL-1α versus IL-6.
bThe large standard deviation compared with the mean of each measurement shows a wide variation in values of interleukins in both patients with sepsis-associated coagulopathies (SACs) and in normal donors.
c
d
Discussion
Previous studies have demonstrated the involvement of specific interleukins in the regulation of coagulation. However, these studies were performed in tissue culture, 8 animal models, 9 and gram-negative sepsis. 10 This study utilized available plasma samples (N = 130) from a well-defined population of patients with SAC to assess the role of interleukins in this population.
All interleukins showed an increase in patients with SAC compared to healthy individuals. Most notably, IL-6, IL-8, and IL-10 increased dramatically, with increases of 322-fold, 48-fold, and 72-fold, respectively. The DD levels were also significantly increased 18-fold.
The increase in both proinflammatory and anti-inflammatory interleukins in sepsis may represent the body’s feedback mechanism to prevent an out-of-control inflammatory and immune response. The simultaneous activation of thrombotic markers such as DD is highly suggestive of an interplay between interleukins and thrombogenesis in SAC. Previous studies demonstrated that lower production of IL-6 was associated with a better prognosis, whereas an increase in IL-10 was predictive of severity and fatal outcome. 5 The effects of interleukins in sepsis can be both positive by containing an infection and negative by mediating tissue and organ damage. Further questions to be addressed in this include the specific role of IL-10 in the regulation of thrombotic and hemostatic processes and the potential for IL-10 as a therapeutic target in SAC.
Conclusion
Profiling interleukins and thrombotic markers not only provides an understanding of mechanisms involved in SAC-associated pathogenesis but may help in the risk stratification of these patients. Furthermore, the understanding of the potential link between inflammation and thrombogenesis may help in the development of combined anti-inflammatory and anticoagulant pharmacotherapy. The increase in cytokines such as IL-6, Il-8, IL-10, IL-18, and TNF-α may have implications in diagnosis and treatment of sepsis. 5 The IL-7 has been investigated as a possible therapeutic agent in sepsis with some promising results. 14,15 This study suggests that IL-6 and IL-10 may be candidates for therapeutic targeting due to their association with SAC and decreased survival.
Footnotes
Authors’ Note
This research was conducted as a part of the Loyola University Chicago Stritch School of Medicine 2013 Student Training in Approaches to Research (STAR) program.
Acknowledgments
The authors are grateful to Dr Linda Brubaker, Dean and Chief Diversity Officer, Loyola University Chicago Stritch School of Medicine, and to Dr Gail Hecht, MD, MS, Assistant Dean for Medical Student Research, Loyola University Chicago Stritch School of Medicine. The authors are also grateful to Dr Eva Wojick, Chairperson, Department of Pathology, Loyola University Medical Center, for support and encouragement in facilitating this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
