Abstract
Background:
In a randomized trial (ie, Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer [CLOT]) that evaluated secondary prophylaxis of recurrent venous thromboembolism (VTE) in patients with cancer, dalteparin reduced the relative risk by 52% compared to oral vitamin K antagonists (VKAs; hazard ratio = 0.48, P = .002). A recent subgroup analysis in patients with moderate to severe renal impairment also revealed lower absolute VTE rates with dalteparin (3% vs 17%; P = .011). To measure the economic value of dalteparin in these populations, a pharmacoeconomic analysis was conducted from the Dutch health-care system perspective.
Methods:
Resource utilization data contained within the CLOT trial database were extracted and converted into direct cost estimates. Univariate analysis was then conducted to compare the total cost of therapy between patients randomized to dalteparin or VKA therapy. Health state utilities were then measured in 24 members of the general public using the time trade-off technique.
Results:
When all of the cost components were combined for the entire population (n = 676), the dalteparin group had significantly higher overall costs than the VKA control group (dalteparin = €2375 vs VKA = €1724; P < .001). However, dalteparin was associated with a gain of 0.14 (95% confidence interval [CI]: 0.10-0.18) quality-adjusted life years (QALYs) over VKA. When the incremental cost was combined with the utility gain, dalteparin had a cost of €4,697 (95% CI: €3824-€4951) per QALY gained.
Conclusion:
Secondary prophylaxis with dalteparin is a cost-effective alternative to VKA for the prevention of recurrent VTE in patients with cancer.
Keywords
Introduction
Venous thromboembolism (VTE), characterized by deep vein thrombosis (DVT) and/or pulmonary embolism (PE), is a common complication in patients diagnosed with cancer. 1 Keeping in mind the caveats with making comparisons across tumor types, it has been estimated that approximately 15% of all patients with cancer will develop VTE during the course of their disease. 2 Once a patient has been admitted into an oncology service, the type of anticancer therapy can also elevate the risk of VTE. Chemotherapy is a risk factor, with the relative risk (RR) reaching 6-fold compared to patients who are not receiving chemotherapy. 3,4 In addition, targeted agents such as sunitinib, sorafenib, lenalidomide, and bevacizumab are also associated with an elevated risk of VTE. 5,6
In addition to the clinical impact in terms of increased risk of mortality. 1,4 In 1 observational study from France, which assessed 62 365 and 45 551 patients with breast and prostate cancer, there were 1271 (2.0%) and 997 (2.2%) hospital admissions for VTEs during the first year. 7 At the 2-year follow-up, 346 (15%) of the 2268 patients presented with a recurrent VTE event requiring a second hospital admission. Over the course of management, these admissions translated into added costs totaling €3.41 million over 2 years. 7 The high cost of VTE, especially in patients with cancer having recurrent events, has also been reported in the United States. 8,9
Given the clinical and economic impact of VTE in patients with cancer, both health-care providers and payers have a desire to eliminate or reduce the number of events developing. However, the effect size associated with primary prevention in the general cancer population is modest. In 1 randomized trial, 1166 patients with solid tumors receiving chemotherapy were randomized 2 to 1 to subcutaneous (SC) injections of nadroparin (3800 IU once a day, n = 769) or placebo (n = 381). Patients were treated for up to 4 months. The primary end point was a composite of symptomatic venous or arterial thromboembolic events, as assessed by an independent adjudication committee. Overall, 15 (2.0%) of the 769 patients in the nadroparin group and 15 (3.9%) of the 381 patients treated with placebo had a VTE (1-sided P = .02). 10
Despite this being reported as a positive trial, the number needed to treat (NNT) to prevent 1 primary VTE from occurring would be approximately 53 patients. Therefore, the American College of Chest Physicians does not recommend primary prophylaxis in outpatients with cancer that do not have additional risk factors for VTE. 11 Additional risk factors included immobilization, hormonal therapy, angiogenesis inhibitors, thalidomide, lenalidomide, or a prior thromboembolic event. 11
An international standard of care for patients with cancer and symptomatic VTE (acute treatment followed by extended secondary VTE prophylaxis) is 6-month treatment with agents such as dalteparin. 12,13 The advantages of dalteparin over oral vitamin K antagonists (VKAs) such as warfarin include a comparable safety profile with reduced variability in the anticoagulation response, which eliminates the need for continuous laboratory monitoring. Dalteparin is also more effective than oral anticoagulation therapy for secondary prophylaxis with an overall relative risk reduction of approximately 50% and an NNT of 13 (absolute difference = 7.8%), which was demonstrated in the multinational randomized Comparison of Low-Molecular-Weight Heparin versus Oral Anticoagulant Therapy for the Prevention of Recurrent Venous Thromboembolism in Patients with Cancer (CLOT) trial. 14 In contrast, it is interesting to note that in a recent study that also evaluated secondary prophylaxis of VTE in patients with cancer, the absolute difference in the occurrence of VTE was not significantly different between VKA and tinzaparin, a different low-molecular-weight heparin (LMWH) with different chemical characteristics and pharmacodynamic actions (7.2% vs 10.5%; P = .07). 15
Dalteparin also provides benefits across important patient subgroups. In a recent post hoc analysis of the CLOT trial conducted by Woodruff et al, dalteparin patients with moderate (creatinine clearance [CrCl]: 30-59 mL/min) to severe (CrCl: 15-29 mL/min) renal impairment at randomization had lower absolute rates of VTE than those in the VKA group (3% vs 17%; P = .011; NNT = 7), whereas the frequency of major bleeding was similar between subgroups (2.0% vs 2.4%; P = .46). 16 In contrast, the difference in absolute rates of VTE between tinzaparin and VKA within the renal impairment subgroup was not statistically significant (RR = 0.93; 95% confidence interval [CI]: 0.39-2.18). 17
One of the advantages of the multinational CLOT trial was that health-care resource use data were collected prospectively. Therefore, the 2 main objectives of the current study were to conduct an economic analysis of secondary VTE prophylaxis in all patients with cancer and then extend the analysis to patients with moderate to severe renal impairment. Both analyses were conducted from the perspective of the publicly funded health-care system of the Netherlands.
Methods
Clinical Trial
The main source for the clinical data supporting the economic evaluation was the multinational CLOT trial, which randomized patients to receive dalteparin (200 IU/kg SC once daily in the first month, then 150 IU/kg SC once daily from months 2 to 6) or 5 to 7 days of dalteparin (200 IU/kg SC once daily) overlapped with and followed by an oral VKA for a planned 6 months. 14 The main inclusion criterion was prior acute symptomatic VTE, and the primary end point was the first objectively documented, symptomatic recurrent DVT or PE during the 6-month study period. Over the course of the study, 676 patients having cancer with a newly diagnosed VTE were enrolled. By the end of the study, 27 (8.0%) of the 336 patients in the dalteparin group developed a recurrent VTE compared to 53 (15.8%) of the 336 in the VKA control group (hazard ratio = 0.48; P = .002). Safety was comparable between groups with no significant differences in major bleeding events (dalteparin = 6% vs VKA = 4%; P = .27). 14
Data Extraction From the CLOT Trial
In addition to the clinical efficacy and safety data, the CLOT trial also collected health-care resource use information. Even though the original CLOT trial was not designed to formally collect health-care resource data for an economic evaluation, sufficient data were available to allow a patient-level economic analysis. This consisted of information related to dose and duration of therapy of study drugs, routine laboratory tests, patient international normalization ratio (INR) in the case of VKA, patient telephone contact, unscheduled clinical visits, diagnostic tests associated with VTE recurrence, hospital length of stay, blood transfusions, and the occurrence of major bleeding events that were possibly or probably due to the study drugs.
Specific treatment data for the management of patients who developed recurrent VTE or for the management of treatment-related side effects were not collected. Therefore, the Dutch, European, and international literature was reviewed in order to obtain cost estimates for treating a DVT, PE, fatal PE, as well as the following adverse events: heparin-induced thrombocytopenia, hematochezia, hematoma, hematuria, hematemesis, malena, retroperitoneal bleeding, intracranial bleeding, and intraperitoneal bleeding. 18 –23 Only events of grade ≥III (National Cancer Institute’s Common Terminology Criteria for Adverse Events, NCI CTCAE V4.0) were included in the economic analysis. Unless specified within the CLOT trial database, it was assumed that patients developing DVTs were treated in the outpatient setting.
Unit cost data were obtained from several Dutch sources such as the Dutch Health Care Authority, The Netherlands Healthcare Institute, and The Drug Costs website of the Care Institute, the Netherlands. All direct costs were reported in 2015 Euros, and cost estimates from previous years were converted into 2015 Euros using the consumer price index as reported by the European Central Bank. The daily cost of supplies for dalteparin administration and the monthly pharmacy dispensing fee (ie, €6.60) were also included in the analysis. Care was taken to ensure patient demographic and associated clinical outcome data had been properly linked with the appropriate costs in patients randomized to the dalteparin and VKA groups. The final outcome of this process allowed for a comparison of overall resource use and cost between groups.
Cost Comparison Within Full CLOT Population
Following extraction of available health-care resources from the full CLOT intention-to-treat (ITT) population, the unit costs were linked with each resource item. This was followed by a patient-level univariate economic analysis between patients randomized to receive dalteparin or VKA within the CLOT trial. The difference in cost between groups was then used as the numerator in the subsequent cost-effectiveness analysis, which estimated the incremental cost per VTE avoided and quality-adjusted life year (QALY) gained with dalteparin. The estimation of the incremental cost per QALY gained with dalteparin would allow for comparison with other supportive care cancer drugs used in patients with cancer in the Netherlands and across Europe.
Cost Comparison in Patients With Moderate to Severe Renal Impairment
One of the main objectives of the current study was to estimate the economic value of extended treatment with dalteparin for secondary prophylaxis of VTE in patients with cancer having moderate to severe renal impairment at the time of randomization. Within the CLOT trial, 162 (24%) of the 676 patients had renal impairment at baseline (74 dalteparin and 88 VKA). A univariate economic analysis of the renally impaired subgroup was then conducted to determine the magnitude of the cost difference between patients treated with dalteparin relative to those who received oral anticoagulation therapy. This cost difference was then used in the subsequent cost–utility analysis to estimate both the incremental cost per VTE avoided and the QALY gained with dalteparin in renally impaired subgroup.
Statistical Analysis
The unpaired t test and the nonparametric Wilcoxon rank sum test were used to evaluate the significance of the difference between groups in the overall cost of therapy per patient in the ITT population and in the renally impaired subgroup. A multivariable quantile regression analysis was then employed to compare the total cost for patients treated with dalteparin relative to the control group. All of the statistical analyses were performed using Stata, release 11.0 (Stata Corp, College Station, Texas).
Treatment Preferences and Health State Utilities
The health-related quality-of-life values measured in the analysis were patient preferences for alternative health state, with the associated number of QALYs. The 4 health states were secondary prophylaxis with a VKA or dalteparin for up to 6 months to prevent a recurrent VTE within the general CLOT trial population or within the patient subgroup with renal impairment. In the current study, QALYs were measured as “healthy month equivalence” for the time spent in each health state using the time trade-off technique. 24,25 Healthy month equivalence scores measure patient utilities for a given health state. A lower healthy month equivalence for the total time spent in a given health state suggests poorer quality of life during that time period (eg, the time period during which a VTE recurs). The scores in months were then converted to utility measures between 0 and 1, where 0 represented death and 1 was a state of perfect health or optimal quality of life. Gains in healthy month equivalence were also converted into QALYs by dividing by 12 months. As an illustration, a 12-month health state may have a healthy month equivalence of 6 months. Therefore, the utility would be 0.5, on a scale between 0 and 1 (6 months of healthy month equivalence/12 months in the given health state).
As recommended by the globally accepted guidelines such as those developed by the Canadian Agency for Drugs and Technology in Health and by the Task Force from the International Society of Pharmacoeconomics and Outcomes Research, health state utilities were measured from a randomly selected sample from the general public. 26,27 Therefore, for convenience, a sample of 24 respondents from the Canadian tax paying public were selected using a multistage, random cluster sampling technique. With a sample of 24 respondents, healthy month equivalence was measured with a precision of ±1.0 month, with a 95% probability. The Canadian utility data were then applied to the Dutch population. Such extrapolations from one country to another are commonly done in cost–utility studies.
To be eligible for the utility assessment, individuals had to be 18 years of age or older, have permanent resident status in Canada, be indirectly supporting the health-care system through tax contributions, and give informed consent to participate in the interview. Permission and approval to conduct the study was received by the Quorum ethics review board. After informed consent was obtained, respondents were presented with information about the natural history of VTE in cancer, followed by a description of the VKA and dalteparin administration protocol. This included information on the method of administration, monitoring requirements, and the associated risks and benefits. During the final part of the interview, the clinical outcomes from the CLOT trial were presented. This consisted of the risk for recurrent DVTs, fatal and nonfatal PEs, and major bleeding events associated with each therapy. Respondents were then asked how many months of “optimal health” they considered being equivalent to the time spent in each of the less than optimal health states described. These measures were used to weigh the duration within each health state by the quality of life experienced by a patient living through that time period. The interview process was then repeated with the presentation of data for patients with moderate to severe renal impairment. Printed interview tools were used to facilitate the participant’s understanding of the time trade-off technique.
Pharmacoeconomic Evaluation
Taking a Dutch health-care system perspective, the pharmacoeconomic analyses for both the full ITT CLOT population and the renally impaired subgroup estimated the incremental cost per VTE avoided and the QALY gained when dalteparin is used as an alternative to VKA over a 6-month time horizon. The 2 economic outcomes were calculated by dividing the difference in cost between dalteparin and VKA therapy (numerator) by the difference in VTE avoided or QALYs gained (denominator). Future costs and benefits were not discounted because of the short 6-month time period.
To test for uncertainly in both the ITT and renally impaired populations, a deterministic 1-way sensitivity analysis was conducted on the base case results. This consisted of a reanalysis of the primary results using the upper and lower 95% CIs of the differences in total treatment costs and utility estimates. In addition, a reassessment was done using alternative estimates of costs for recurrent DVT and nonfatal PE. 28 All cost–effectiveness ratios were rounded to the nearest hundred.
Results
Comparison of Health-Care Resource Use
For the full ITT CLOT trial population, a univariate comparison of baseline patient and clinical variables suggested that patients randomized into the dalteparin and VKA groups were well balanced (Table 1). However, there were slightly more patients in the VKA group who had moderate to severe renal impairment at baseline (26.0% vs 21.9%), but the difference failed to reach statistical significance (P = .21). Since balance was present between groups, an unbiased comparison of resource use and total cost could be conducted.
Demographic and Clinical Characteristics for all Patients at Randomization.
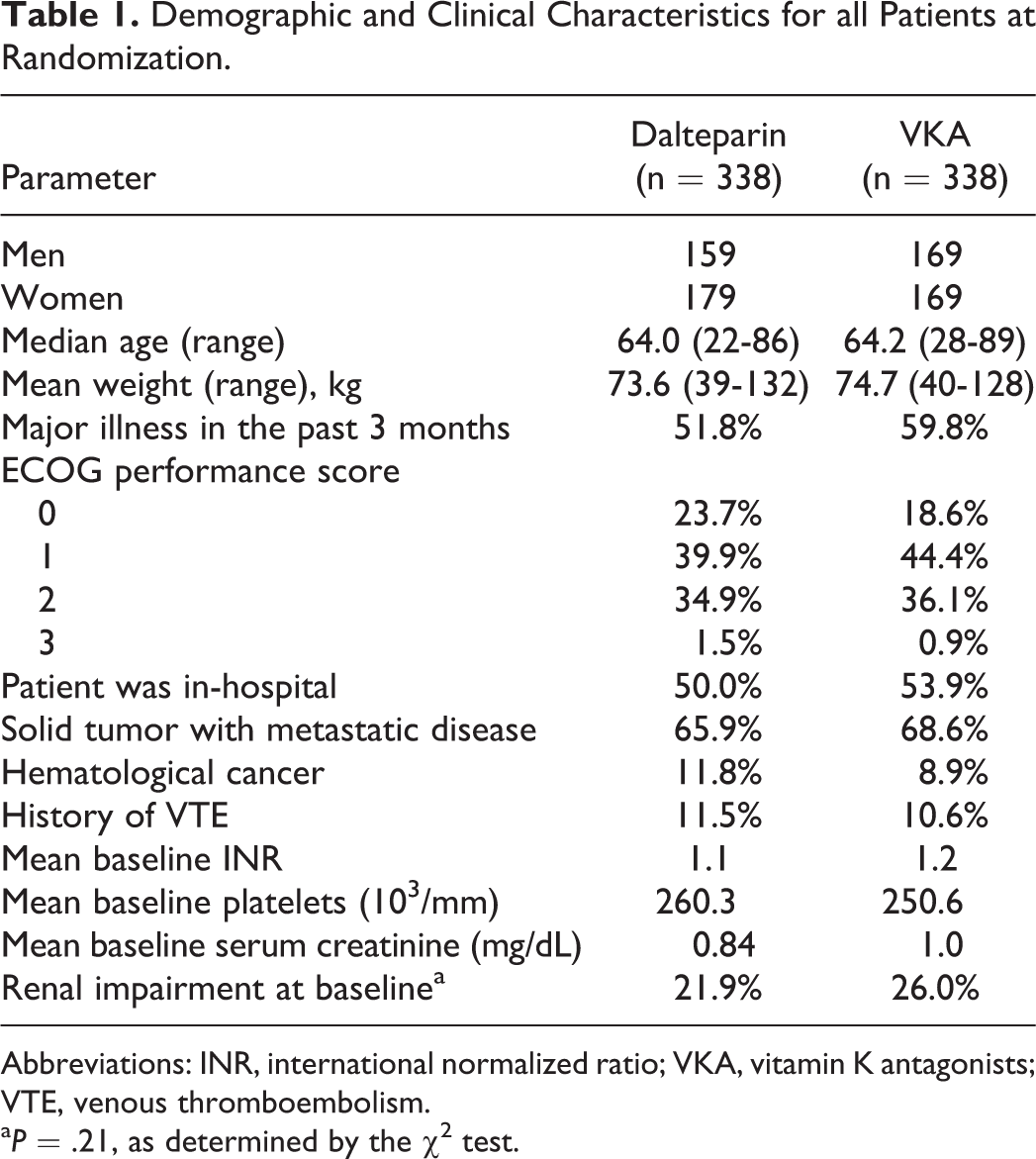
Abbreviations: INR, international normalized ratio; VKA, vitamin K antagonists; VTE, venous thromboembolism.
a P = .21, as determined by the χ2 test.
Health-care resource utilization data collected during the CLOT trial for all patients were extracted to undertake a comparison between groups. Over the 6-month trial horizon, patients in the experimental group received dalteparin for a mean of 126.3 days compared to 8.0 days in the VKA group (Table 2). Following the initial 8 days of therapy with dalteparin, which was overlapped with VKA, patients in the control group received oral VKA therapy only, which was given for a mean of 116.9 days. The findings also revealed that treatment compliance was significantly different in favor of the dalteparin group by approximately 10% (P < .001).
Univariate Statistical Comparison of Health-Care Resource Use for all Patients at Randomization.
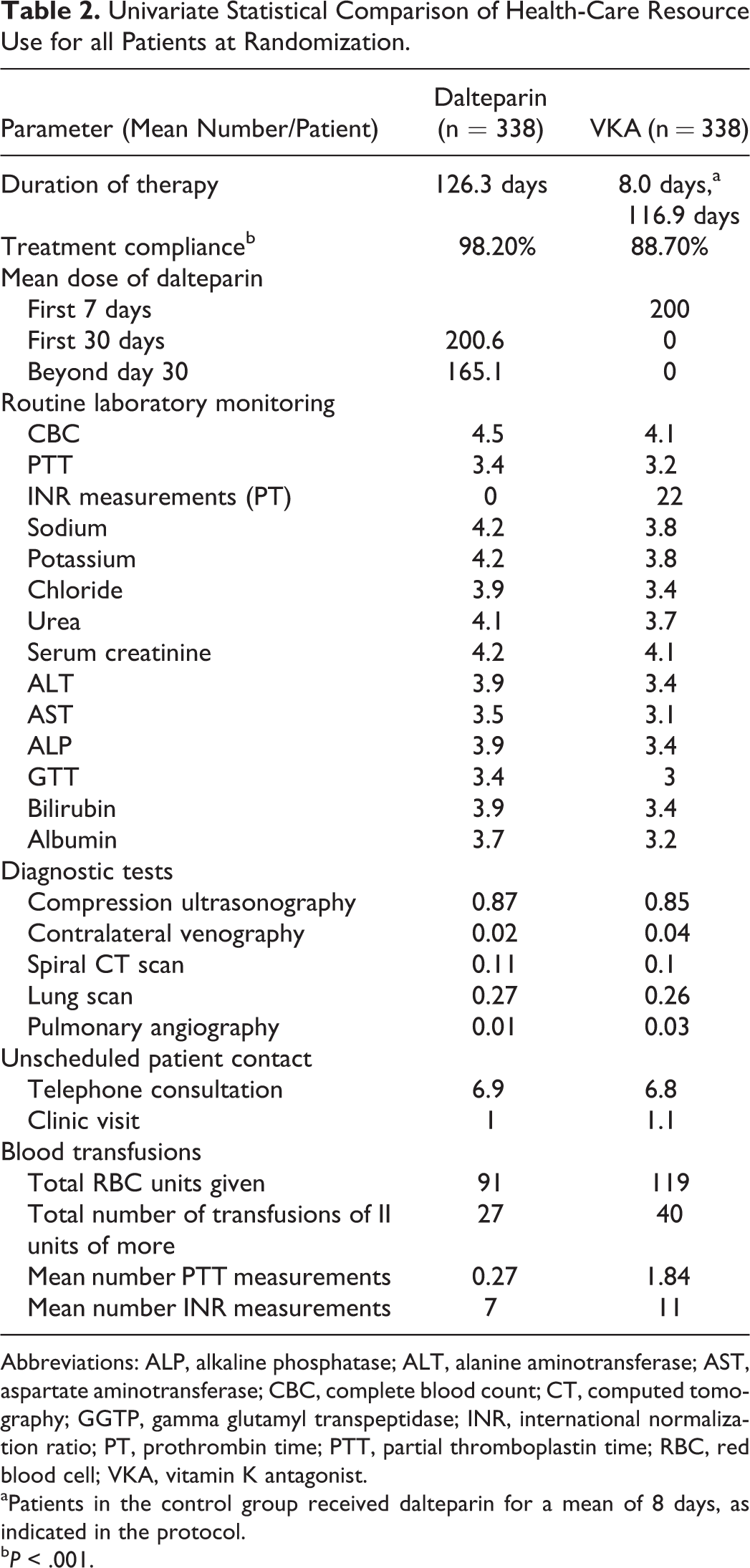
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CBC, complete blood count; CT, computed tomography; GGTP, gamma glutamyl transpeptidase; INR, international normalization ratio; PT, prothrombin time; PTT, partial thromboplastin time; RBC, red blood cell; VKA, vitamin K antagonist.
aPatients in the control group received dalteparin for a mean of 8 days, as indicated in the protocol.
b P < .001.
An evaluation of laboratory test use indicated comparable utilization, with the exception of INR measurements, which are required for monitoring oral anticoagulation therapy with VKAs. An average of 22 INR tests were ordered per patient randomized to oral anticoagulation therapy. Furthermore, there were more unscheduled clinic visits (absolute number = 352 vs 387), blood transfusions, and partial thromboplastin time/INR measurements in the VKA group (Table 2).
Duration of drug therapy and health-care resource use was also compared between groups in patients who had renal impairment at baseline. The mean duration of dalteparin therapy was 121.8 and 7.8 days in the experimental and control groups, respectively (Table 3). Patients in the control group then received a mean of 113.8 days of a VKA following the initial 7-day induction with dalteparin. As seen in the evaluation of the ITT population, health-care resource use in terms of routine laboratory monitoring (with the exception of INR), diagnostic tests, and unscheduled patient contact was comparable between groups. However, total red blood cell units given, the frequency of blood transfusions, and the associated mean number of INR measurements were elevated in renally impaired patients who received VKA (Table 3).
Univariate Statistical Comparison of Health-Care Resource Utilization in Patients With Moderate to Severe Renal Impairment.
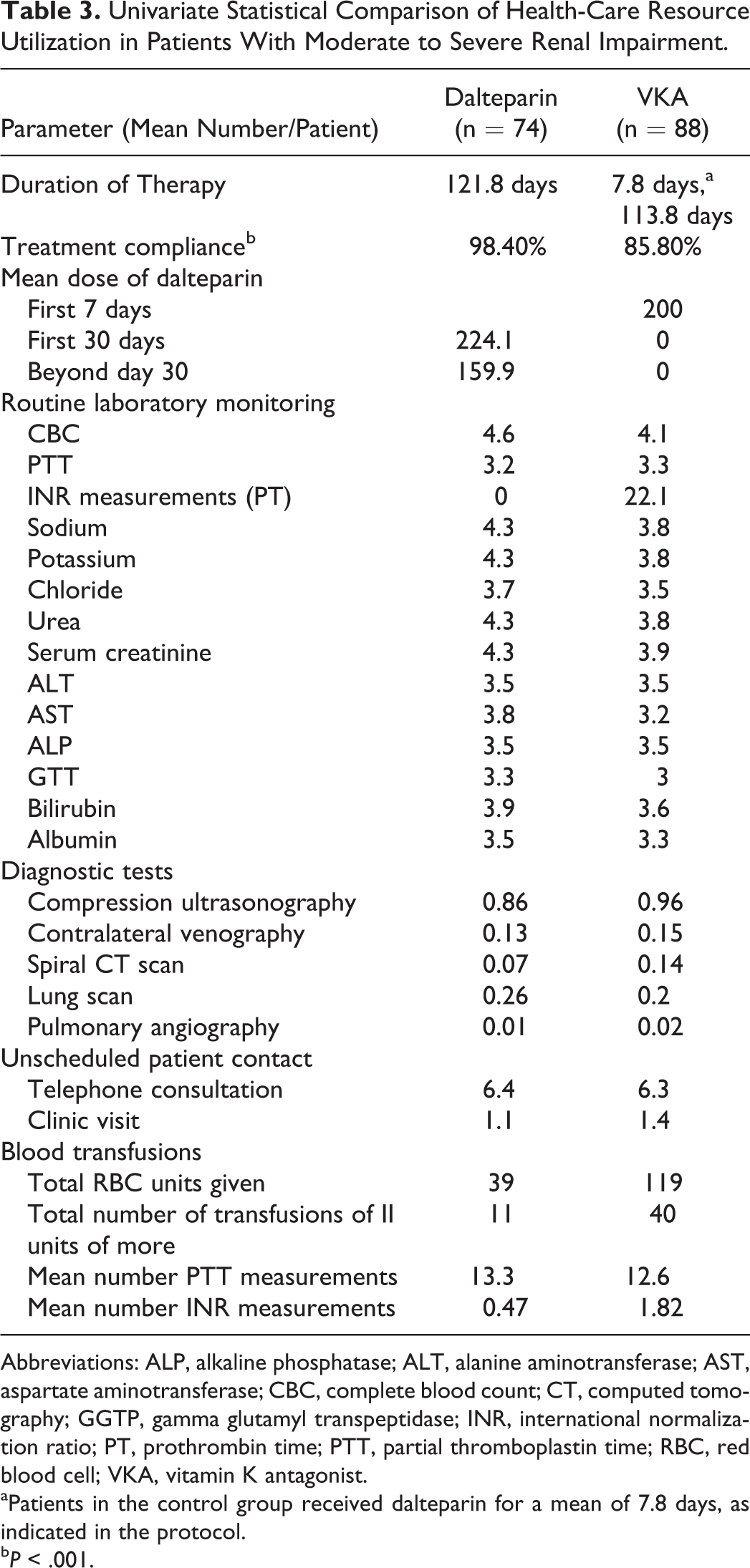
Abbreviations: ALP, alkaline phosphatase; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CBC, complete blood count; CT, computed tomography; GGTP, gamma glutamyl transpeptidase; INR, international normalization ratio; PT, prothrombin time; PTT, partial thromboplastin time; RBC, red blood cell; VKA, vitamin K antagonist.
aPatients in the control group received dalteparin for a mean of 7.8 days, as indicated in the protocol.
b P < .001.
The primary clinical end point of the full ITT population was the occurrence of recurrent VTE over the 6-month trial horizon. These consisted of DVTs as well as fatal and nonfatal PEs. As reported in the trial for the full CLOT ITT population, there was a statistically significant reduction in the occurrence of new VTE in patients randomized to the dalteparin group (Table 4). Overall, there were 23 fewer DVT events and 3 fewer PE events in the dalteparin group. In patients from the full CLOT ITT population who were receiving therapy in the outpatient setting, there was a hospital admission rate (regardless of cause) of 25.1% in the dalteparin group compared to 28.5% in the control (P = .33; not statistically significant). When the cause for admission was limited to a recurrent VTE, bleeding, or heparin-induced thrombocytopenia, the rates of hospital admission were 3.2% for dalteparin and 3.8% in the oral therapy group (P = .68). Overall, these rates translated to 32 additional hospital days for dalteparin patients and 40 days in the control group (Table 4).
Treatment-Associated Clinical Outcomes and Resource Use in all Patients and in Those With Moderate to Severe Renal Impairment.
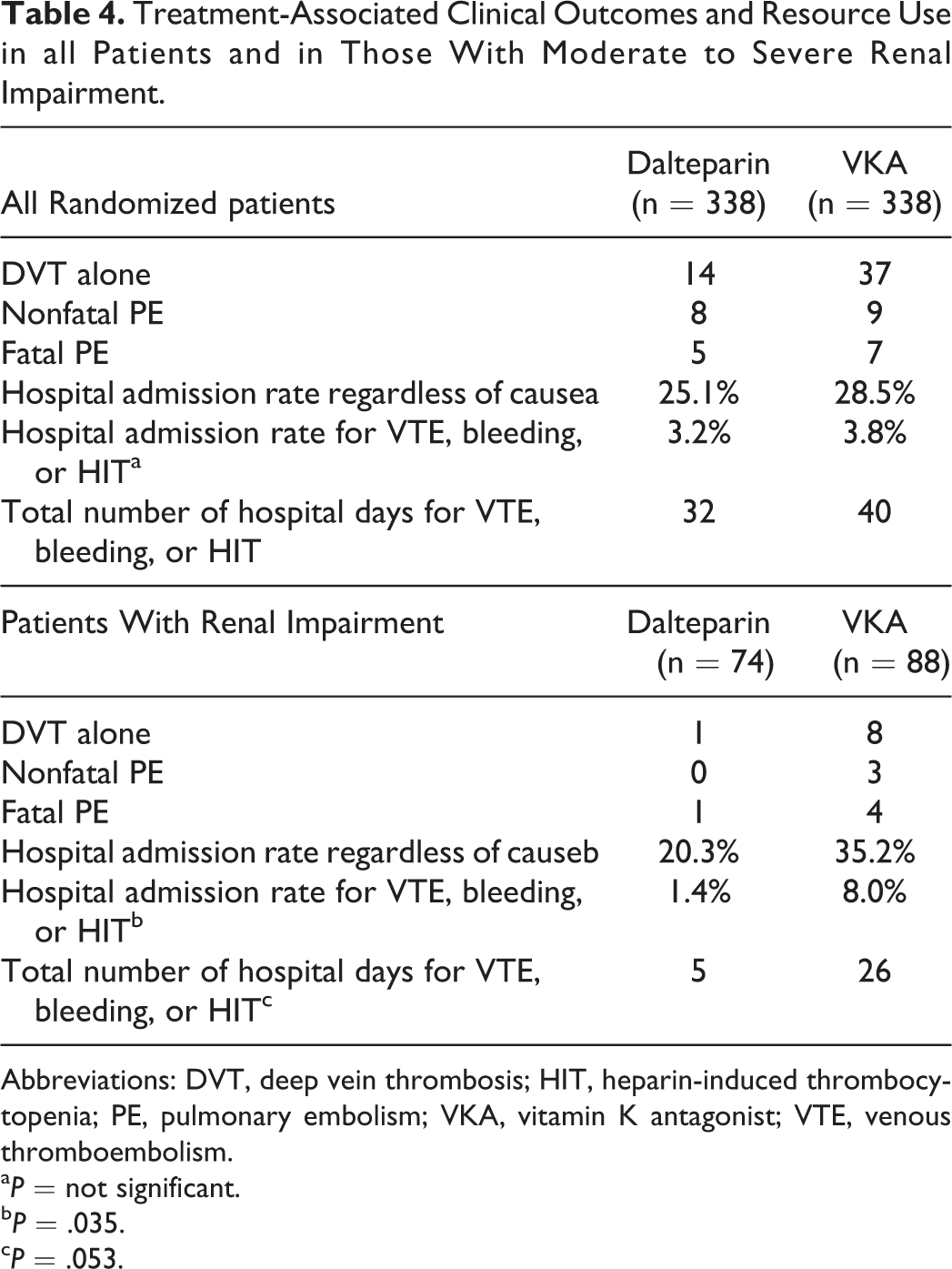
Abbreviations: DVT, deep vein thrombosis; HIT, heparin-induced thrombocytopenia; PE, pulmonary embolism; VKA, vitamin K antagonist; VTE, venous thromboembolism.
a P = not significant.
b P = .035.
c P = .053.
A statistically significant reduction in recurrent VTE was also observed in the renally impaired subgroup treated with dalteparin. There were a total of 15 thrombotic events in the 88 patients who received a VKA compared to 2 events in the 74 dalteparin patients (P = .01). Furthermore, there were significant reductions in hospitalizations and in the total number of hospital days for managing VTE, bleeds, and treatment-related complications in this important patient subgroup (Table 4).
Cost Comparison
The next phase of the analysis was to apply the Dutch unit costs to all health-care resource consumption in all patients randomized in the CLOT trial. The intent was to compare the total cost of care between the dalteparin and VKA groups, as well as in the subgroup of patients with moderate to severe renal impairment at baseline. Dalteparin patients had higher costs for drug therapy and for the treatment of major bleeding events relative to the VKA control group (Table 5). In contrast, the VKA group had increased costs for laboratory monitoring, for blood transfusions, and for the management of VTE recurrences. When all of the costs were combined, the total mean cost for all CLOT patients randomized into the dalteparin group was €2375 (95% CI: €2184-€2567) compared to €1724 (95% CI: €1574-€1874) in the VKA group (P < .001). However, in the subgroup analysis consisting of renally impaired patients, the difference in total cost between the dalteparin and VKA group was no longer statistically significant (€2111 [95% CI: €1769-€2311] vs €1810 [95% CI: €1418-€12 202)]; P = .50). The largest cost contributor in the dalteparin group was drug acquisition at 49% of the total (vs 5% in the control), while at 40%, VTE management was the largest cost component in the VKA group.
Comparison of Costs Between Groups in all Patients and in Those With Moderate to Severe Renal Impairment.
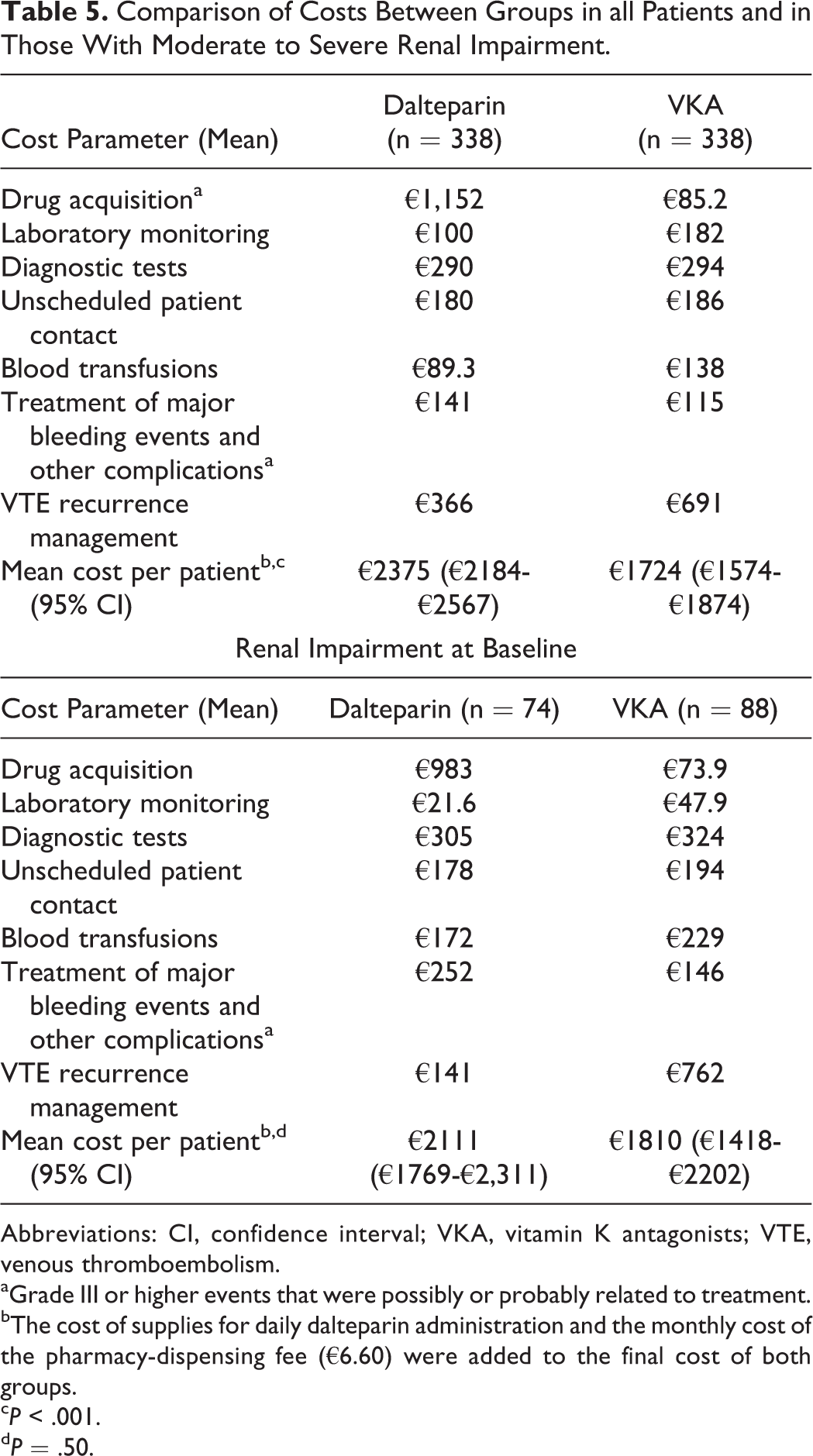
Abbreviations: CI, confidence interval; VKA, vitamin K antagonists; VTE, venous thromboembolism.
aGrade III or higher events that were possibly or probably related to treatment.
bThe cost of supplies for daily dalteparin administration and the monthly cost of the pharmacy-dispensing fee (€6.60) were added to the final cost of both groups.
c P < .001.
d P = .50.
The findings of the multivariable analysis confirmed the results of the univariate analysis, which showed that treatment with dalteparin was associated with a significantly higher total cost. An examination of the variable “dalteparin group” for the ITT CLOT population indicated that while controlling for other factors such as performance status, gender, and renal impairment, patients in the dalteparin group had a median incremental cost of €1115 per patient compared to oral anticoagulation therapy (P < .001). However, what was of particular interest and unexpected was the finding in the renal impairment subgroup analysis via the interaction between group and renal impairment (data not shown). Namely, dalteparin patients with renal impairment at baseline were less costly to manage than those treated with VKA (median difference of €266; P = .027). The cost savings was due to the avoidance of costly recurrent thromboembolic events.
Treatment Preferences and Health State Utilities
Treatment preferences and health state utilities for each of the 4 outcomes were measured from a sample of 24 members of the general public who were selected via a random multistage sampling strategy. The mean age of respondents was 52 years (range: 32-73 years), with 11 (45.8%) of the 24 being female. One (4.2%) of the 24 participants had previously experienced a VTE, which was treated with warfarin.
Once all of the information had been presented on both treatments, respondents were asked to select their preferred secondary prophylactic intervention from an overall CLOT trial perspective. Overall, 21 (87.5%) of the 24 respondents selected dalteparin over VKA as their drug of choice. Healthy month equivalence scores and health state utilities for each alternative were then estimated from the sample. The higher preferences for dalteparin translated into higher health state utility scores (measured as healthy month equivalence). The utility of the dalteparin health state was almost 2-fold higher than treatment with a VKA, suggesting improved quality of life (Table 6). The preferences and utility scores for dalteparin were due to a combination of factors such as improved efficacy, a demonstrated safety profile with extended use, and the ability to eliminate continuous INR monitoring. The main positive attribute that respondents indicated about VKA was the oral route of administration. Overall, the gain in healthy month equivalence was approximately 1.67 months with dalteparin for the full CLOT population, which corresponded to an additional gain of approximately 0.14 QALYs.
Cost-Effectiveness and Cost–Utility Analysis.
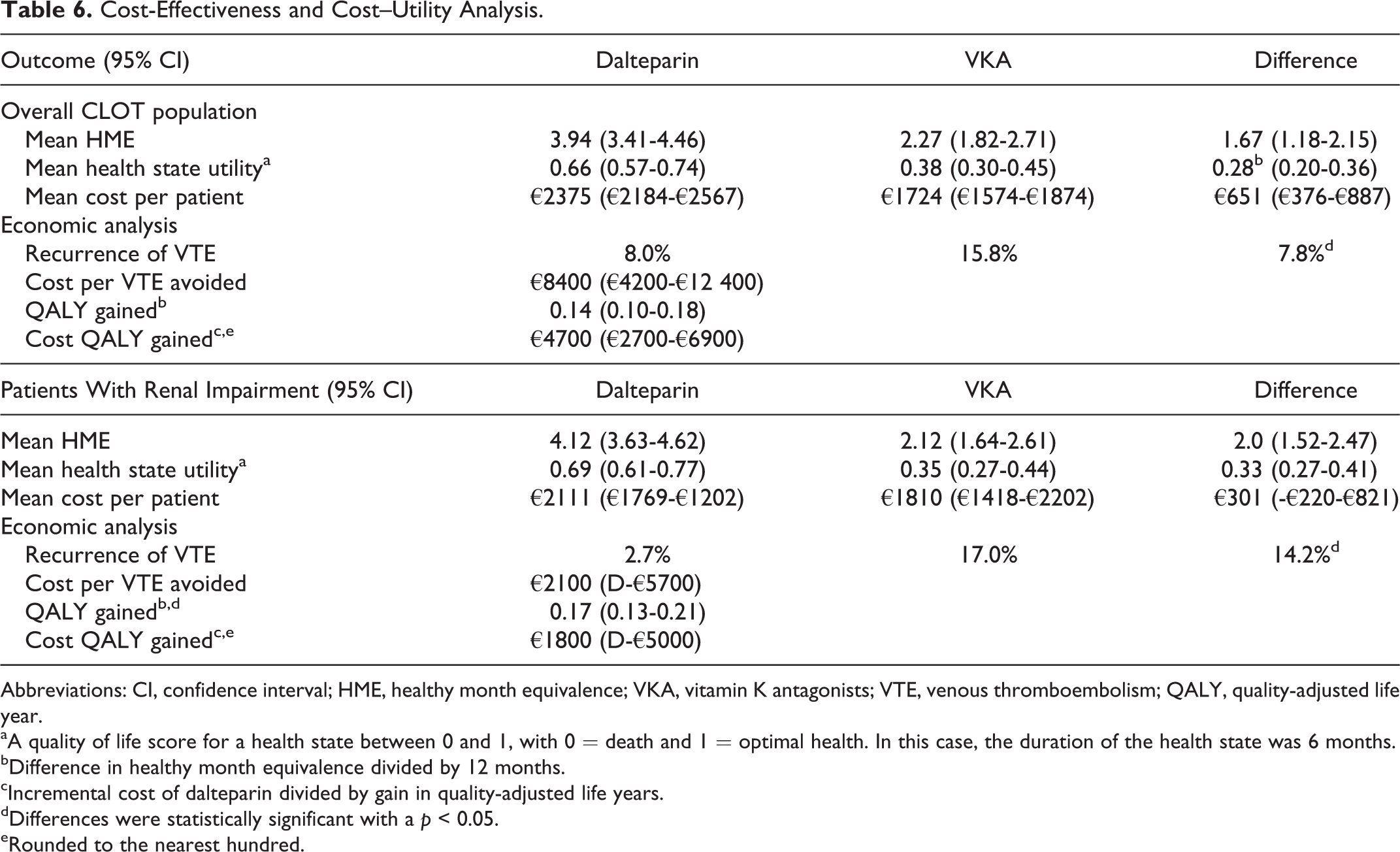
Abbreviations: CI, confidence interval; HME, healthy month equivalence; VKA, vitamin K antagonists; VTE, venous thromboembolism; QALY, quality-adjusted life year.
aA quality of life score for a health state between 0 and 1, with 0 = death and 1 = optimal health. In this case, the duration of the health state was 6 months.
bDifference in healthy month equivalence divided by 12 months.
cIncremental cost of dalteparin divided by gain in quality-adjusted life years.
dDifferences were statistically significant with a p < 0.05.
eRounded to the nearest hundred.
The utility assessment was then extended to patients with moderate to severe renal impairment. Under this treatment setting where dalteparin demonstrated an even greater effect size (ie, NNT = 7 vs 13 for the overall population), 23 (96%) of the 24 respondents selected dalteparin as their preferred treatment. This higher preference translated into a healthy month equivalence gain of 2.0 months, which corresponded to an additional gain of approximately 0.17 QALYs (Table 6).
Pharmacoeconomic Analysis
The differences in cost and effectiveness between the dalteparin- and VKA-treated patients were then used in a pharmacoeconomic analysis from a Dutch health-care system perspective. The 2 end points used to assess overall product value were the incremental cost per VTE avoided and the incremental cost per QALY gained. The latter parameter would allow secondary dalteparin prophylaxis to be compared to other supportive care drugs used in oncology.
The use of dalteparin in the full ITT CLOT population was associated with an incremental cost of approximately €8400 (95% CI: €4200-€12 400) per VTE avoided (Table 6). However, this cost–effectiveness ratio fails to incorporate the patient quality of life and the utility gain associated with the avoidance of VTE. Such benefits are quantified with the QALY. To provide such information, the incremental cost of dalteparin (ie, €651) was combined with the 0.14 QALYs gained (a gain of 1.67 health month equivalence corresponds to a gain of 0.14 QALY). The findings generated an incremental cost of approximately €4700 per QALY gained (95% CI: €2700-€6400), consistent with the hypothesis that dalteparin is a cost-effective intervention in patients with cancer at risk for recurrent VTE (Table 6).
The analysis was repeated for the renally impaired patient subgroup. Since the VTE risk reduction from dalteparin in this patient subgroup is even greater than in the general CLOT trial population (ie, absolute risk reduction of 14% vs 7.8%), the value of dalteparin for patients was improved, as indicated by the lower incremental cost–effectiveness ratios. When used in patients with moderate to severe renal impairment, dalteparin was associated with an incremental cost per VTE avoided and a QALY gained of €2100 and €1800, respectively (Table 6). It was also interesting to see that the lower boundary of the 95% CI interval was less than 0 for both outcomes, suggesting potential economic dominance where one of the alternatives (ie, dalteparin) is both more effective and cost saving.
Sensitivity Analysis
Deterministic 1-way sensitivity analyses were then conducted using the 95% CI of the differences in healthy month equivalence scores (1.18-2.15) and cost (€376-€887) between the 2 treatments in the full ITT CLOT population. Under the worst-case scenario for dalteparin where the highest cost difference between the 2 treatments was used (ie, €887) along with the lowest gain in healthy month equivalence (ie, 1.18), the cost per QALY gained with dalteparin increased to approximately €9000. Under the best-case scenario for dalteparin where the lowest cost difference (ie, €376) was used along with the highest gain in healthy month equivalence (ie, 2.15), the cost per QALY gained with dalteparin was reduced to €2,100 in the ITT CLOT population
One of the key offsets associated with the overall economic value of dalteparin is the cost of managing a recurrent VTE. In a recent paper, Fernandez and colleagues reviewed the international literature that estimated the cost of treating a VTE. 28 A total of 18 studies were identified, 13 from the United States, 1 from Canada, and 4 from Europe. Among the European countries, there were no VTE costing studies from the Netherlands. The investigators identified variability with the cost of treating DVT and PE in Europe, which was due to differences in hospital length of stay and inconsistencies in the use of outpatient VTE management strategies across the continent. For instance, the cost of treating a DVT in France varied from €523 if the patient was managed outside hospital to €3181 in cases where the patient was hospitalized. 28
To address this uncertainty, the data from the current study were reanalyzed under the assumption that all recurrent DVTs observed within the CLOT trial were treated in the outpatient setting using the French reported cost of €523 per event. As expected, the incremental cost per VTE avoided and QALY gained increased to €11 800 and €6500, respectively.
In patients with renal impairment, the cost per VTE avoided and QALY gained increased to €4800 and €2200, respectively, under this extreme scenario. These findings imply that even with extreme variations, such as assuming that all patients with a DVT are treated in the less costly outpatient setting, estimates of cost per QALY gained remained relatively stable, which supports the hypothesis that dalteparin is a cost-effective therapy in this treatment setting.
Discussion
One of the greatest challenges faced by health-care authorities globally is the high cost of new medicines, especially drugs used in cancer care. 29,30 High costs for cancer drugs are not only straining pharmacy budgets but also impacting the patient, a term that has been coined, “financial toxicity.” 30 In the United States, for instance, a national survey of insured patients with cancer determined that approximately 25% of respondents reported that they had consumed all or most of their personal savings to treat their disease. 31 Cancer was also reported to be the most common diagnosis when medical reasons were cited as the main cause for declaring personal bankruptcy. 32 In one population-based study, patients diagnosed with cancer were also 2.7 times more likely to file for personal bankruptcy in the 5-year period following their diagnosis compared to a matched sample of people without a cancer diagnosis. 33 Given the challenges faced by both payers and patients, there is a need to identify therapies that are both cost-effective and cost saving. Secondary VTE prophylaxis with dalteparin, as an alternative to low-cost daily VKA, seems to be such a therapy for patients with cancer.
The results from this patient-level economic analysis suggest that from the perspective of the Dutch health-care system, secondary dalteparin prophylaxis in patients with cancer having prior symptomatic VTE is a cost-effective alternative to oral daily VKA therapy, with an incremental cost of €4700 (95% CI: €3800-€5000) per QALY gained. Furthermore, the economic value of dalteparin was especially high in patients with moderate to severe renal impairment, with the cost per QALY gained being less than €2000. Such low incremental cost–effectiveness ratios are rare in oncology today.
One of the advantages of using the cost per QALY as an outcome measure in economic evaluations is that it allows a comparison to other interventions across therapeutic areas. Supportive care drugs commonly used in patients with cancer include pegfilgrastim, recombinant erythropoietin alpha, and palonosetron. A review of the contemporary economic literature from the Netherlands did not identify any cost–utility studies for such agents. Therefore, the search was extended to Europe to find relevant studies for comparison. Liu et al conducted a cost–utility study to measure the value of primary prophylaxis using pegfilgrastim compared with filgrastim in patients with breast cancer receiving chemotherapy in the United Kingdom. Using an economic modeling approach from the health-care system perspective of England, it was estimated that pegfilgrastim was associated with a higher cost of £4161 or €5700 per QALY gained. 34
In another study evaluating the cost-effectiveness of recombinant erythropoietin in France, Fagnoni et al determined that the use of recombinant erythropoietin alpha during breast cancer chemotherapy was associated with a cost of €310 577 per QALY gained from the perspective of the French health-care system. 35 In a study from the United States, Avritscher et al concluded that palonosetron-based regimens when compared to ondansetron-based 2-drug therapy had a cost per QALY of US$115 490 when used for the prevention of emesis in patients with breast cancer receiving chemotherapy. 36 Therefore, dalteparin provides much better value than all of these other drugs that are commonly used in cancer supportive care.
One of the main strengths of the current pharmacoeconomic study is that health-care resource use data were used from patients who were part of a multinational randomized trial, which included patients from the Netherlands. Despite this advantage, there are several limitations that have to be addressed. Some of the hospital resources such as INR tests and clinic visits were protocol driven and may not completely reflect standard practice. In clinical practice, there may be fewer INR tests ordered, so this would have lowered the total cost in the VKA control group. These factors may reduce the generalizability (external validity) of the results to patients outside the optimal situation created by a randomized trial. Detailed health-care utilization data for the management of all recurrent VTE were not available. Therefore, we had to rely on literature-reported cost estimates for managing such events in both the hospitalized and outpatient settings. Another limitation in the cost–utility analysis was that a health-care system perspective was taken. In the Netherlands, the Dutch health-care authority prefers a societal perspective, where indirect costs such as lost productivity, travel costs, and informal care giver are also included in the analysis. The inclusion of these indirect costs would have improved the economic profile of dalteparin secondary to fewer recurrent VTE.
The Dutch health-care authority also prefers a life-time horizon for the overall analysis. However, there is no evidence that LMWH alter the natural history of cancer. Lastly, members of the Canadian tax paying public instead of a sample from the Dutch population were used to measure health state utilities. Since a formal comparison to utilities from a similar sample of respondents has not been done, the utility data from the former group may not be fully generalizable to the latter. Furthermore, using general public for assessing utilities always presents with challenges in terms of optimizing their understanding of the health states under investigation. As a final point, a major economic driver in the current analysis was the cost of treating a DVT or a PE. The recent review by Fernandez et al revealed considerable variability across Europe in the cost of DVT and PE treatment. 28 Therefore, the results from the perspective of the Dutch health-care authority may not be fully generalizable to other European countries. Country-specific analyses are therefore recommended.
Conclusion
This patient-level cost–utility analysis revealed that the use of dalteparin to reduce the risk of VTE recurrence in patients with cancer with a prior VTE is cost-effective from the Dutch health-care system perspective. In patients with cancer having moderate to severe renal impairment, the economic value of dalteparin is further improved. Given the long history of this drug in terms of safety, improved efficacy, and convenience, extended duration dalteparin is a cost-effective alternative to VKA for secondary VTE prophylaxis and should be offered to clinically eligible patients with cancer at risk of recurrent VTE.
Footnotes
Authors’ Note
All authors confirm they were involved in study design, data analysis and interpretation, drafting of the paper, and final approval of the current version to be published. The corresponding author had full access to the data, designed the study, conducted the analysis, interpreted the results, and had the final responsibility for the decision to submit the paper for peer-reviewed publication.
Acknowledgments
The study investigators will like to express their gratitude to Dr Job F. M. van Boven, Unit of Pharmacoepidemiology and PharmacoEconomics, Department of Pharmacy, University of Groningen, Groningen, the Netherlands, for his contributions to the study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Some of the authors (L.S., S.W., and L.B.) are employees of the sponsor.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by Pfizer Inc.
