Abstract
Background:
Increased coagulation activity has been established in paroxysmal atrial fibrillation (PAF), but data on the anticoagulant system are scarce.
Purpose:
To examine the protein C anticoagulant pathway in the early hours of the disease.
Materials and Methods:
Fifty-one patients (26 men and 25 women; mean age 59.84 ± 1.60 years) and 52 controls (26 men and 26 women; mean age 59.50 ± 1.46 years) were selected for the study. Protein C antigen and its activity, total protein S, free protein S and its activity, soluble forms of endothelial protein C receptor (sEPCR), and thrombomodulin (sTM) were examined in the plasma.
Results:
The indicators were studied in patients between the 2nd and the 24th hour after the onset of arrhythmia. Levels of protein C were significantly elevated in patients compared to controls (111.40% ± 6.66% vs 94.83% ± 4.47%; P = .039). Protein C activity showed significant reduction in PAF (73.13% ± 5.80% vs 103.3% ± 3.80%; P < .001). Total protein S levels did not differ significantly (108.20% ± 4.07% vs 102.40% ± 3.65%; P = .30). Free protein S (76.81% ± 6.01% vs 122.10% ± 3.97%; P < .001) and its activity (71.39% ± 6.27% vs 119.50% ± 6.54%; P < .001) were reduced in patients. Higher levels of sEPCR (203.10 ± 10.33 vs 133.10 ± 7.37 ng/mL; P < .001) and sTM (6.50±0.40 vs 4.48±0.28 ng/mL; P < .001) were measured in PAF.
Conclusion:
Protein C activity is reduced still in the first hours (until the 24th hour) of PAF clinical manifestation, determining reduced activity of the anticoagulant pathway as a whole. The established low levels of free protein S and its activity as well as low sEPCR and sTM levels are a possible explanation of the changes in protein C activity.
Keywords
Introduction
Atrial fibrillation (AF) is the most common arrhythmia in clinical practice, affecting >1% of the general population. 1 Paroxysmal atrial fibrillation (PAF; duration of episodes of arrhythmia <7 days) represents between 25% and 60% of all cases of the disease. 2 It has attracted the interest of more researchers, as epidemiological studies found a high incidence of thromboembolic events similar to that of chronic AF. 3 –5
The reasons for the significant embologenic potential of the disease are most probably diverse and complex. It is well known that blood stasis in a poorly contractile left atrium is directly related to thrombogenesis. 6 Data have been accumulated in the recent years, some of it contradictory in nature, but giving sufficient grounds to assume that a tendency for hypercoagulability is developed in PAF. In this sense, high plasma levels of the thrombin–antithrombin III complex, fibrinogen, and fibrinopeptide A were established. 7 –10
Blood coagulation in the human body is controlled by several natural anticoagulant systems, which normally dominate the procoagulant systems. 11,12 One of the most important anticoagulant systems is the protein C pathway. 13,14 Its main components are protein C, thrombomodulin (TM), endothelial cell receptor for protein C (EPCR), and protein S.
Protein C is a vitamin K-dependent anticoagulant synthesized primarily in the liver. 14 It circulates in plasma as a proenzyme and is activated by thrombin connected to TM. Thrombomodulin is a transmembrane protein that is expressed on the surface of endothelial cells and acts as a thrombin receptor. 15 The formation of the thrombin–TM complex is important for initiating the activation of the protein C anticoagulant pathway, wherein thrombin converts into a protein whose procoagulant activity is blocked. 16,17 At the same time, it acquires a number of important qualities such as the ability to activate protein C, to inhibit protein C inhibitor, and others. 18 The soluble form of TM (sTM), resulting from the enzyme cleavage of the membrane form, circulates freely in the plasma. Its plasma levels reflect the degree of TM loss from the endothelial surface. 19
The EPCR is also a transmembrane protein that is located on the surface of endothelial cells in the immediate vicinity of TM. 20 The activation of protein C by the thrombin–TM complex is further amplified after its binding to EPCR. 21 This bond is reversible; therefore, the activated protein C (APC) is released into the plasma. The soluble form of EPCR (sEPCR) in plasma is also a normal finding, exhibiting affinity for protein C and APC, similar to the membrane form. 13,22 In contrast to it, however, sEPCR inhibits both protein C activation and APC anticoagulant activity. 23
Protein S is a vitamin K-dependent plasma protein whose biosynthesis takes place in the liver. In human plasma, 60% to 70% of protein S circulates in complex with the complement regulator C4b-binding protein. 24 The rest circulates as free protein S, which acts as APC cofactor, increasing its potency approximately 20 times. 25,26 This makes APC a major inhibitor of coagulation factors VIIIa and Va. 11 Furthermore, APC indirectly affects fibrinolysis by inhibiting the activation of both thrombin activiatable fibrinolytic inhibitor and plasminogen activator inhibitor 1. 27,28
The high incidence of thromboembolic complications in patients with PAF has aroused a clinical interest in the anticoagulation system that still remains not well studied. The purpose of this research was to study in patients with PAF the protein C anticoagulant pathway still in the early hours of the clinical manifestation of the disease.
Materials and Methods
Study Design
Correct assessment of the onset and duration of AF episodes was essential for the study. Therefore, we screened only patients who could clearly determine the onset of the rhythm disorder. They reported sudden manifestations of “palpitations” continuing up to hospitalization. The patients were diagnosed with “AF” after electrocardiographic examination performed immediately after admittance to the ward.
Object of research interest were only patients with PAF episode duration <48 hours. Long-lasting (>48 hours) AF episodes were excluded from the study after thorough medical history in which patients categorically denied previous similar episodes as well as accidental, even asymptomatic detection of irregular heartbeat in medical examinations or by themselves.
The control group was formed to match the gender, age, accompanying diseases, and medical treatment of the patient group. The participants were selected among volunteers who visited their doctor for prophylactic annual examinations. By the time of the survey, they had no history or electrocardiographic evidence of AF.
Exclusion criteria from the study were multiple diseases, conditions, and medications related to hemostasis. They were identical for the patient and the control groups: Cardiovascular diseases: known coronary artery disease (stable angina pectoris; previously symptomatic obstructive or nonobstructive coronary artery disease, which has become asymptomatic with treatment; conditions after percutaneous coronary intervention or coronary artery bypass graft; myocardial infarction regardless of duration; and acute coronary syndromes) or suspected coronary artery disease (note 1), acute or chronic heart failure (note 2), uncontrolled hypertension (note 3), implantation of cardiac rhythm devices, myocarditis, pericarditis and infectious endocarditis, congenital heart diseases, moderate or severe acquired valve diseases, and cardiomyopathies. Other diseases: renal failure, liver cirrhosis, respiratory failure, inflammatory, and/or infectious diseases for the previous 3 months; neoplastic or autoimmune diseases; and diseases of the endocrine nervous system (except for diabetes mellitus type 2, noninsulin dependent, well controlled). Intake of hormone-replacement therapy or contraceptives, pregnancy, systematic intake of analgesics (incl. nonsteroidal anti-inflammatory drugs), anticoagulants and antiplatelet agents, body mass index (> 35), history of thromboembolic events, namely, pulmonary embolism, deep vein thrombosis, cerebrovascular events, extremity thrombosis, and surgery in the last 6 months.
Seven indicators of the protein C anticoagulant pathway were studied once in each participant: protein C antigen protein C activity, total protein S, free protein S, activity of protein S, sEPCR, and sTM. Blood samples were taken in patients immediately after hospitalization and prior to administration of medications. Blood was collected in controls during preventive outpatient examination.
Study Population
From a total of 338 screened patients, 51 were consecutively selected (26 men, 25 women), with mean age of 59.84 ± 1.60 years. The remaining 287 patients were excluded due to the exclusion criteria. While forming the control group, from a total of 169 screened, 52 were selected (26 men and 26 women; mean age 59.50 ± 1.46 years).
The study was conducted in the Intensive Coronary Care Units of First Cardiology Clinic at the University Hospital Saint Marina—Varna during the period October 2010 to May 2012 after approval by the Ethics Commission of Research at the same hospital and in accordance with the Declaration of Helsinki. 29 Participants were included in the study after previously signing an informed consent to participate.
Venous Blood Sampling
Venous blood, obtained from the antecubital vein, was collected into 0.109 mol/L (3.2%) sodium citrate coagulation tubes (VACUETTE coagulation tube, Greiner Bio-One, Kremsmünster, Austria). Immediately after that the blood samples were centrifuged at 2000 g for 15 minutes and the resulting plasma was separated. It was stored in plastic Eppendorf tubes at –20°C for up to 1 month.
Collection and storage of blood samples were carried out in full compliance with the manufacturer’s requirements for the tests used in the study (Diagnostica Stago, Asnieres, France).
Laboratory Procedures
Quantitative determination of protein C antigen (%) was performed using a commercially available enzyme immunoassay Asserachrom Protein C (Diagnostica Stago, Asnieres, France). Protein C activity (%) was measured with a Clotting Assay STA-Staclot (Diagnostica Stago). Total protein S (%) was measured with a microlatex particle-mediated immunoassay Liatest (Diagnostica Stago). An enzyme-linked immunosorbent assay Imulise Monoclonal (Diagnostica Stago) was used to determine free protein S (%). A clotting assay (Bioclot; Diagnostica Stago) was used to study the activity of protein S (%). Quantitative determination of sEPCR (ng/mL) was performed by an enzyme immunoassay (Asserachrom; Diagnostica Stago, Asnieres, France). Quantitation of sTM (ng/mL) was determined by an enzyme-linked immunosorbent assay (Imubind; American Diagnostica Inc, Stamford, Connecticut). The coefficients of variation (both interassay and intra-assay reproducibility) were within 6.1% except for protein S activity where they were less than 11.8%.
All laboratory procedures were carried out in full compliance with the tests used. The indicators were determined twice in each sample, taking the average value. Refreezing of samples was not allowed.
Statistical Analysis
Descriptive statistics was used to calculate the mean values, standard error of the mean (SEM), relative shares, and central tendency (Mo = mode). The analysis of the hypotheses for differences in 2 means and relative share equality was done by the Student’s 2-tailed t test for unpaired data for normal distributions.
Statistical data processing was carried out using the special purpose statistical package GraphPad PRISM, Version 5.00. The results were presented as mean ± standard error of the mean or n (%). Values of P < .05 were considered statistically significant.
Results
Patient and Control Characteristics
Table 1 summarizes the patient and control group characteristics. There was no statistically significant difference between the 2 groups regarding demographic indicators, clinical features, bad habits, and BMI (Table 1; P > .05).
Clinical Features of Patient and Control Groups.
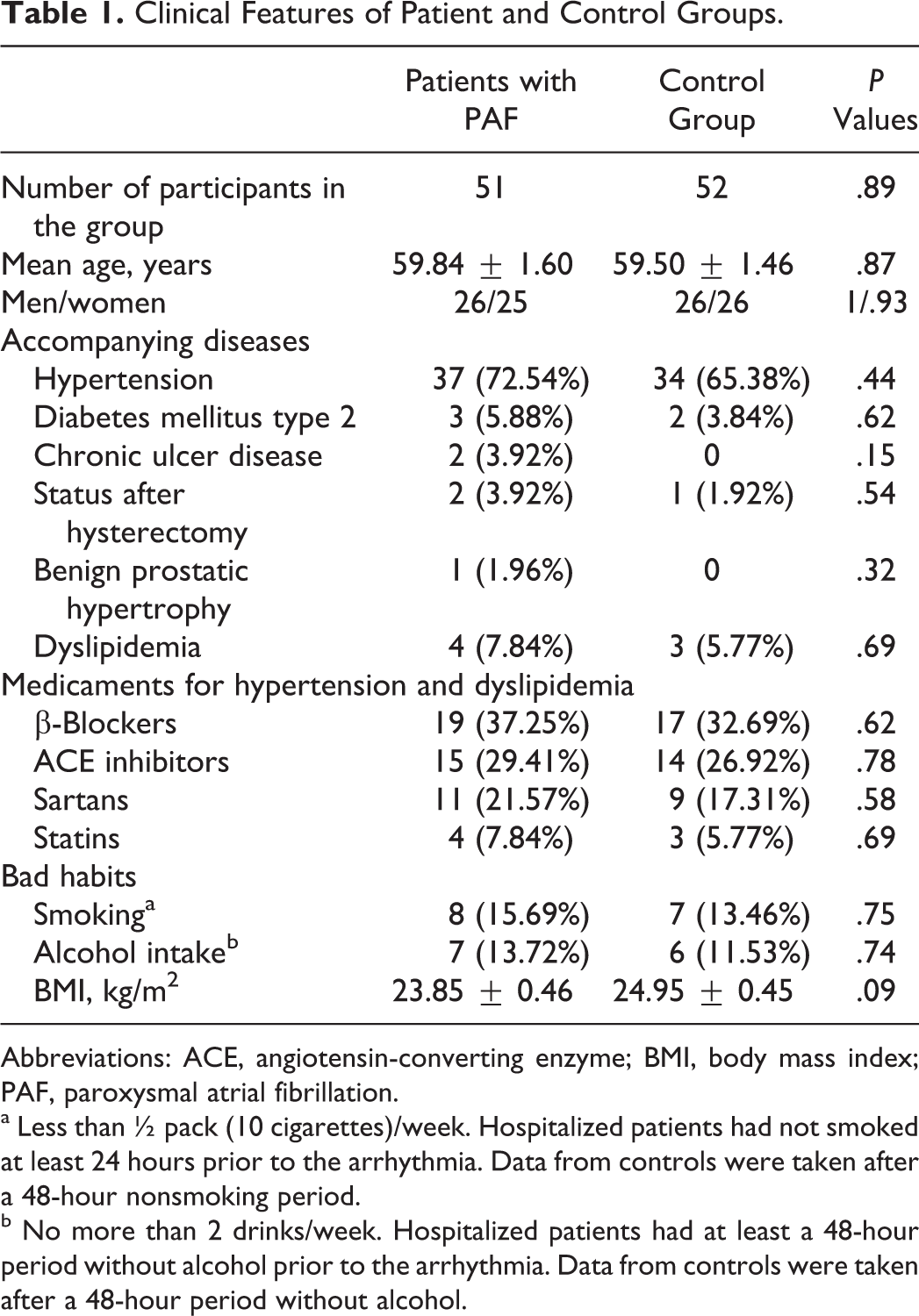
Abbreviations: ACE, angiotensin-converting enzyme; BMI, body mass index; PAF, paroxysmal atrial fibrillation.
a Less than ½ pack (10 cigarettes)/week. Hospitalized patients had not smoked at least 24 hours prior to the arrhythmia. Data from controls were taken after a 48-hour nonsmoking period.
b No more than 2 drinks/week. Hospitalized patients had at least a 48-hour period without alcohol prior to the arrhythmia. Data from controls were taken after a 48-hour period without alcohol.
Transthoracic echocardiography found no significant differences between the patient and the control groups (Table 2; P > .05). Statistical analysis showed that all 51 patients were hospitalized between the 2nd and the 24th hour after the onset of the arrhythmia, most commonly during the 5th hour (Mo = 5; 10 of all 51 patients). The mean duration of AF episodes until hospitalization was 8.14 ± 0.76 hours.
Echocardiographic Evaluation of Patient and Control Group.
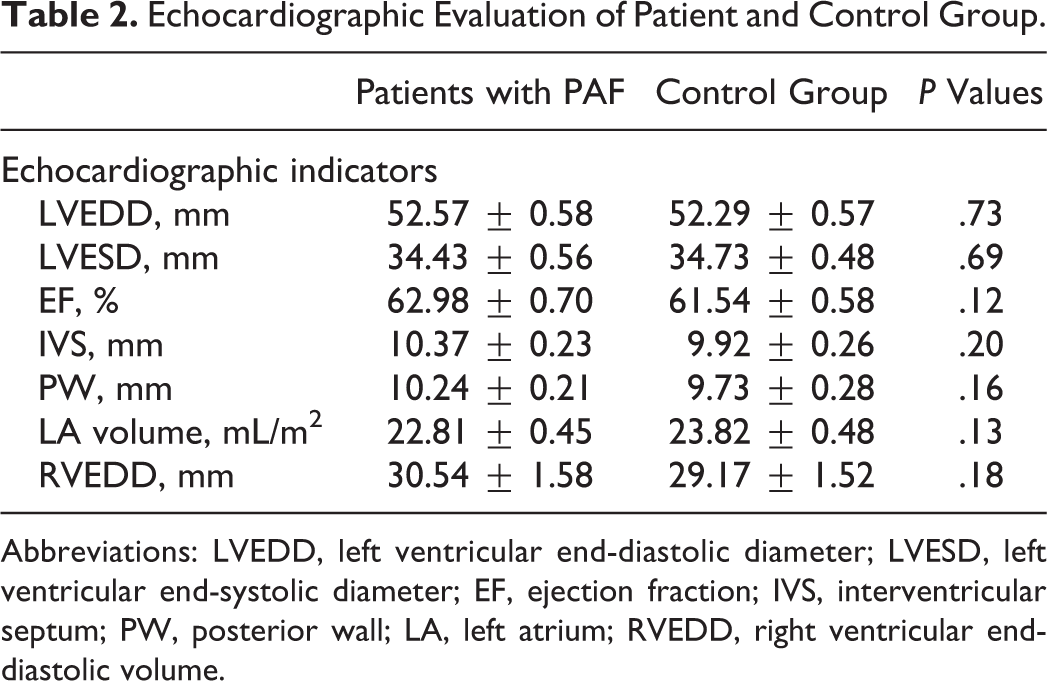
Abbreviations: LVEDD, left ventricular end-diastolic diameter; LVESD, left ventricular end-systolic diameter; EF, ejection fraction; IVS, interventricular septum; PW, posterior wall; LA, left atrium; RVEDD, right ventricular end-diastolic volume.
Hemostatic Markers (Indicators)
Plasma levels of protein C were significantly higher in patients with PAF than controls (111.40% ± 6.66% vs 94.83 ± 4.47%; P = .039; Figure 1). Anticoagulant activity of protein C in the patient group was significantly reduced (73.13% ± 5.80% vs 103.3% ± 3.80%; P < .001; Figure 2). There was no significant difference between the 2 groups in the levels of total protein S (108.20% ± 4.07% vs 102.40% ± 3.65%; P = .30; Figure 3). Free plasma protein S levels were reduced in patients (76.81% ± 6.01% vs 122.10% ± 3.97%; P < .001; Figure 4). Activity of protein S was also reduced in the patient group (71.39% ± 6.27% vs 119.50% ± 6.54%; P < .001; Figure 5). Levels sEPCR were higher in the patient group (203.10 ± 10.33 vs 133.10 ± 7.37 ng/mL; P < .001; Figure 6). Levels of sTM were also elevated (6.50 ± 0.40 vs 4.48 ± 0.28 ng/mL; P < .001; Figure 7).
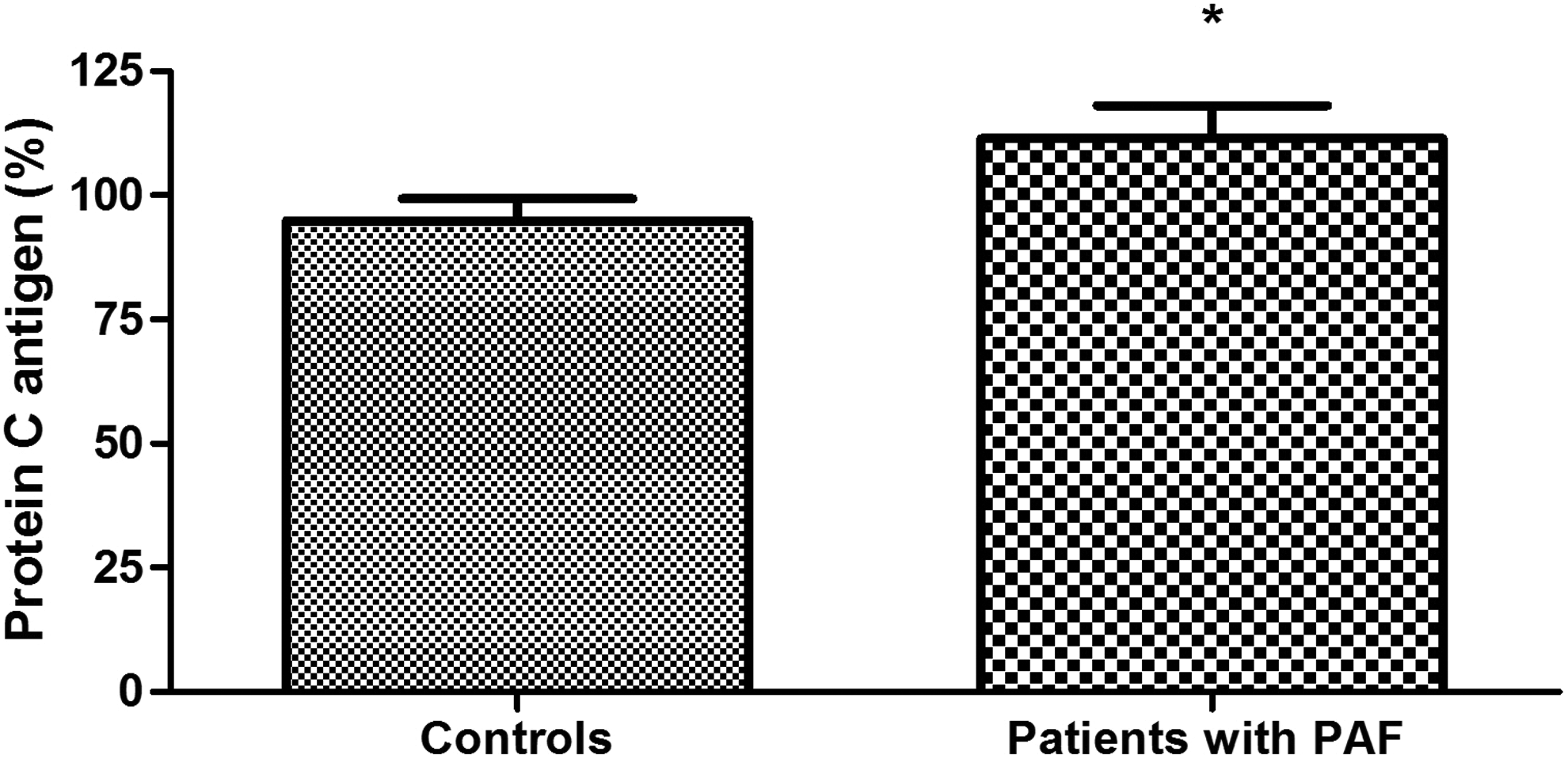
Plasma levels of protein C antigen in patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (*P < .05).
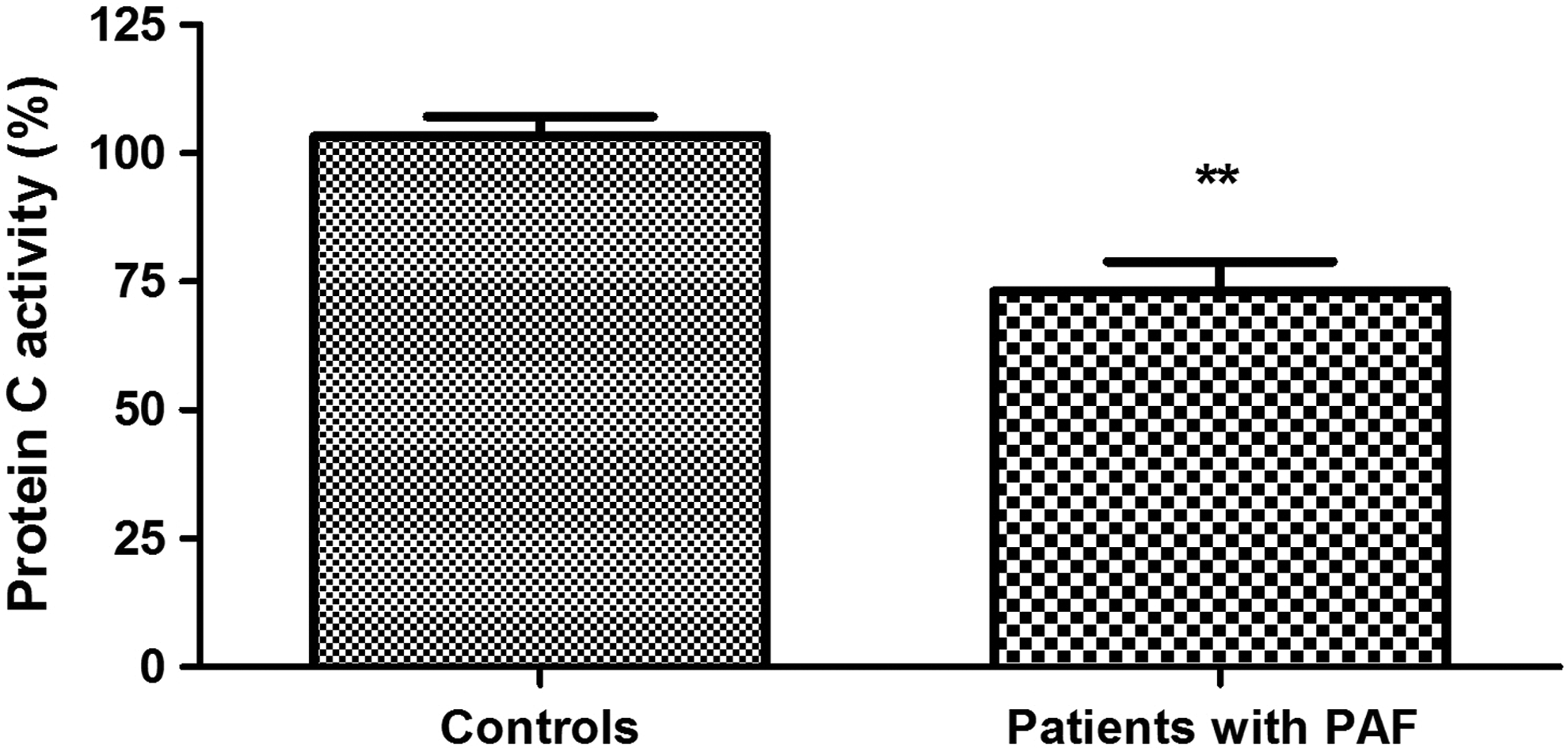
Protein C anticoagulant activity in patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (**P < .001).
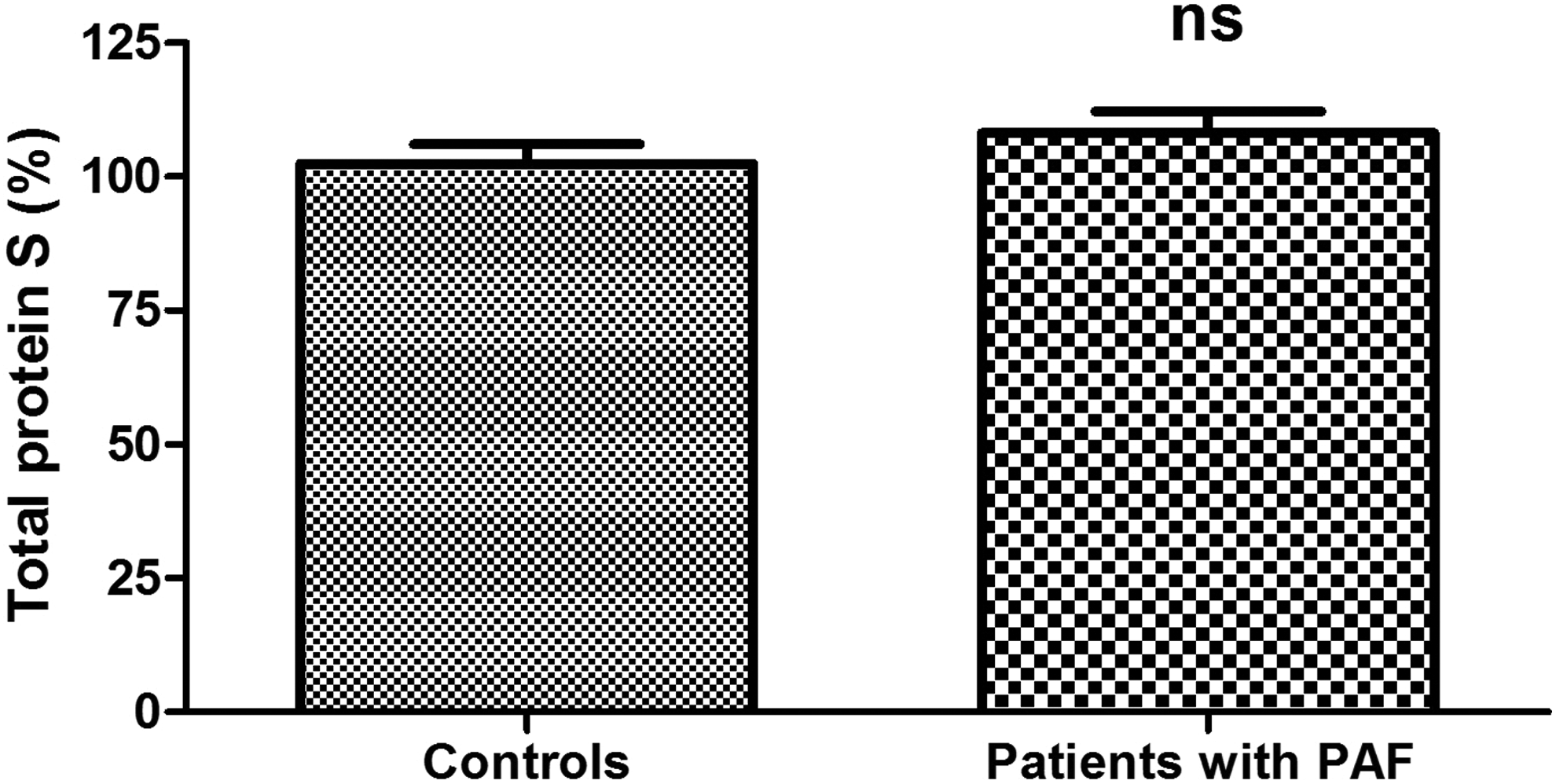
Total protein S levels in plasma in patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (ns, not statistically significant).
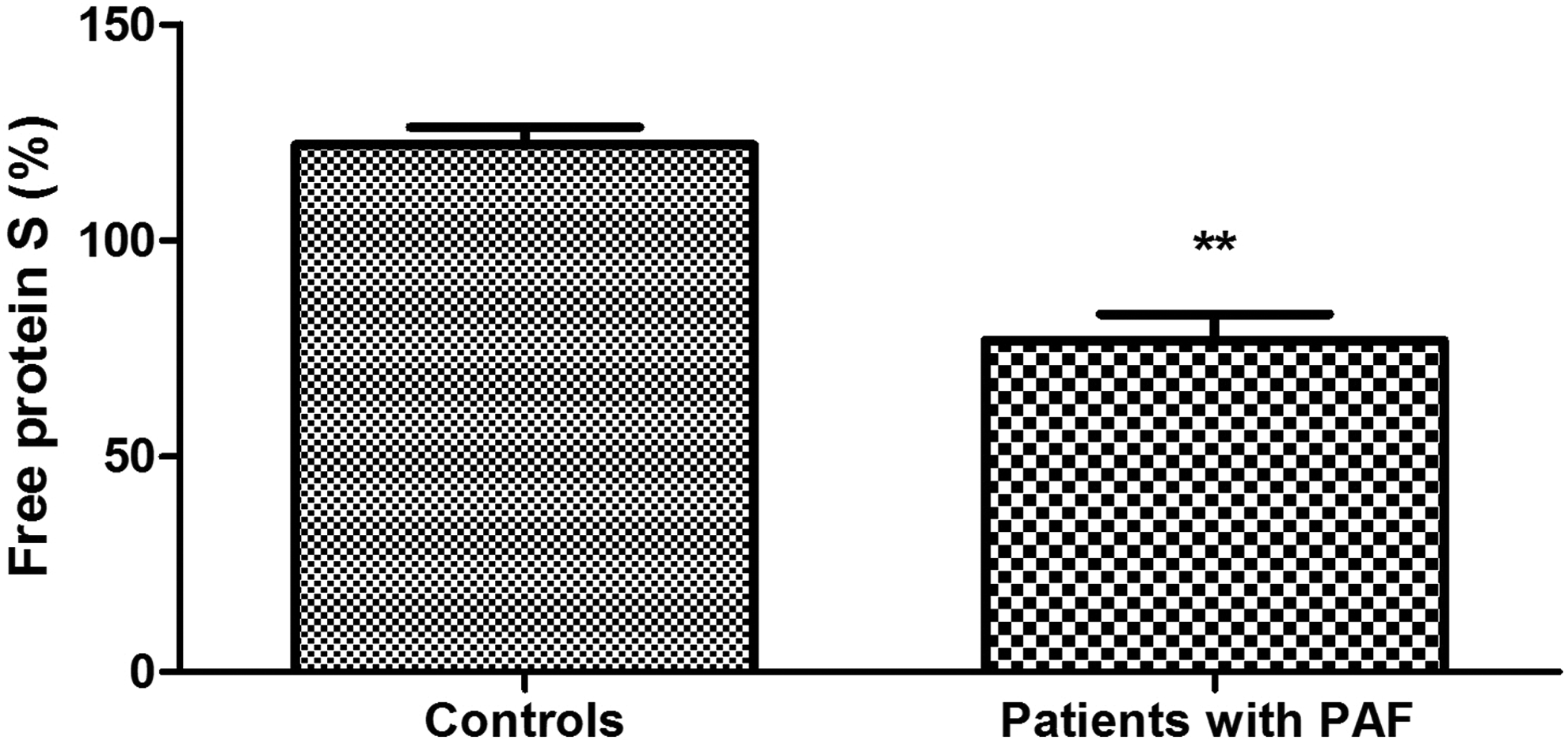
Free protein S levels in plasma of patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (**P < .001).
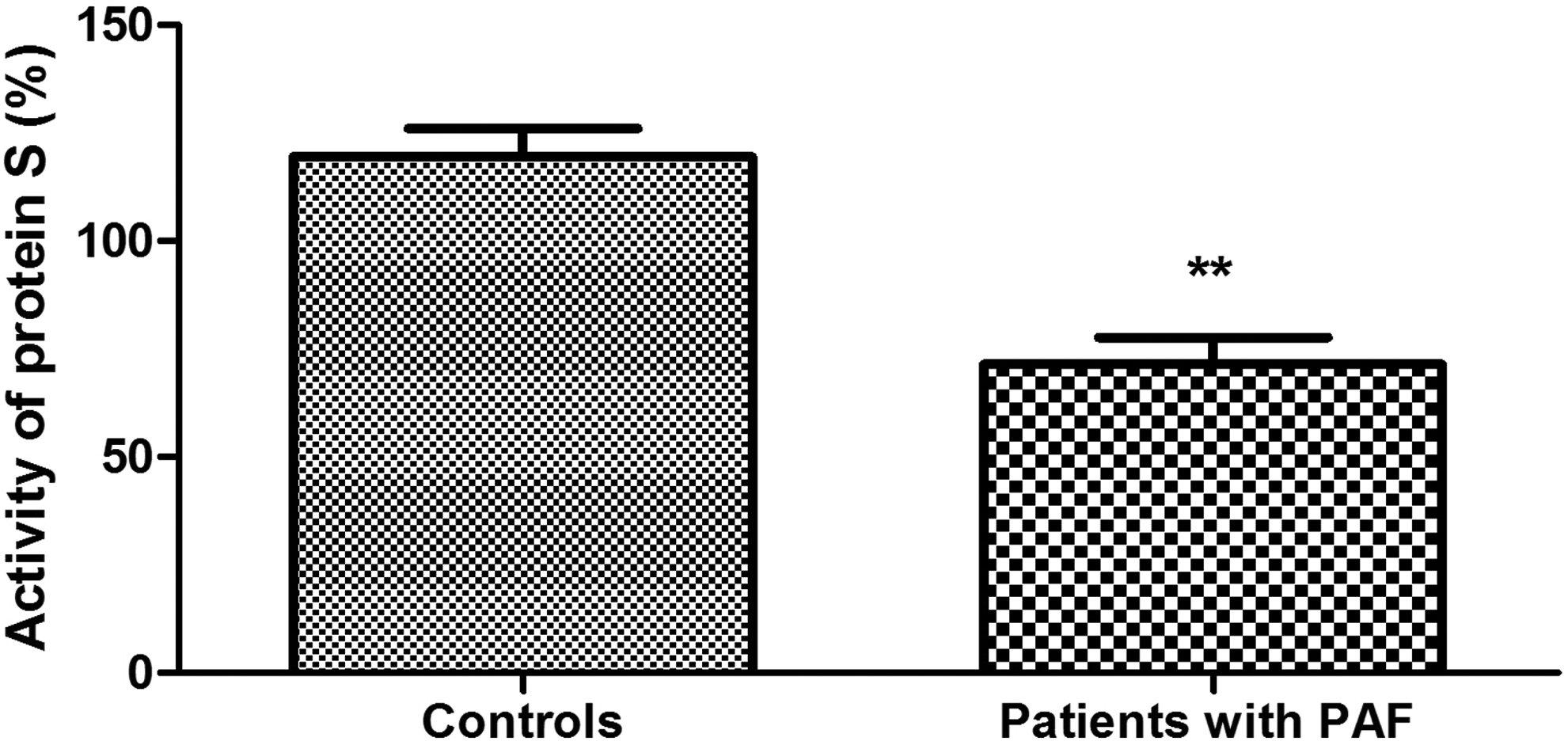
Anticoagulant activity of free protein S in plasma of patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (**P < .001).
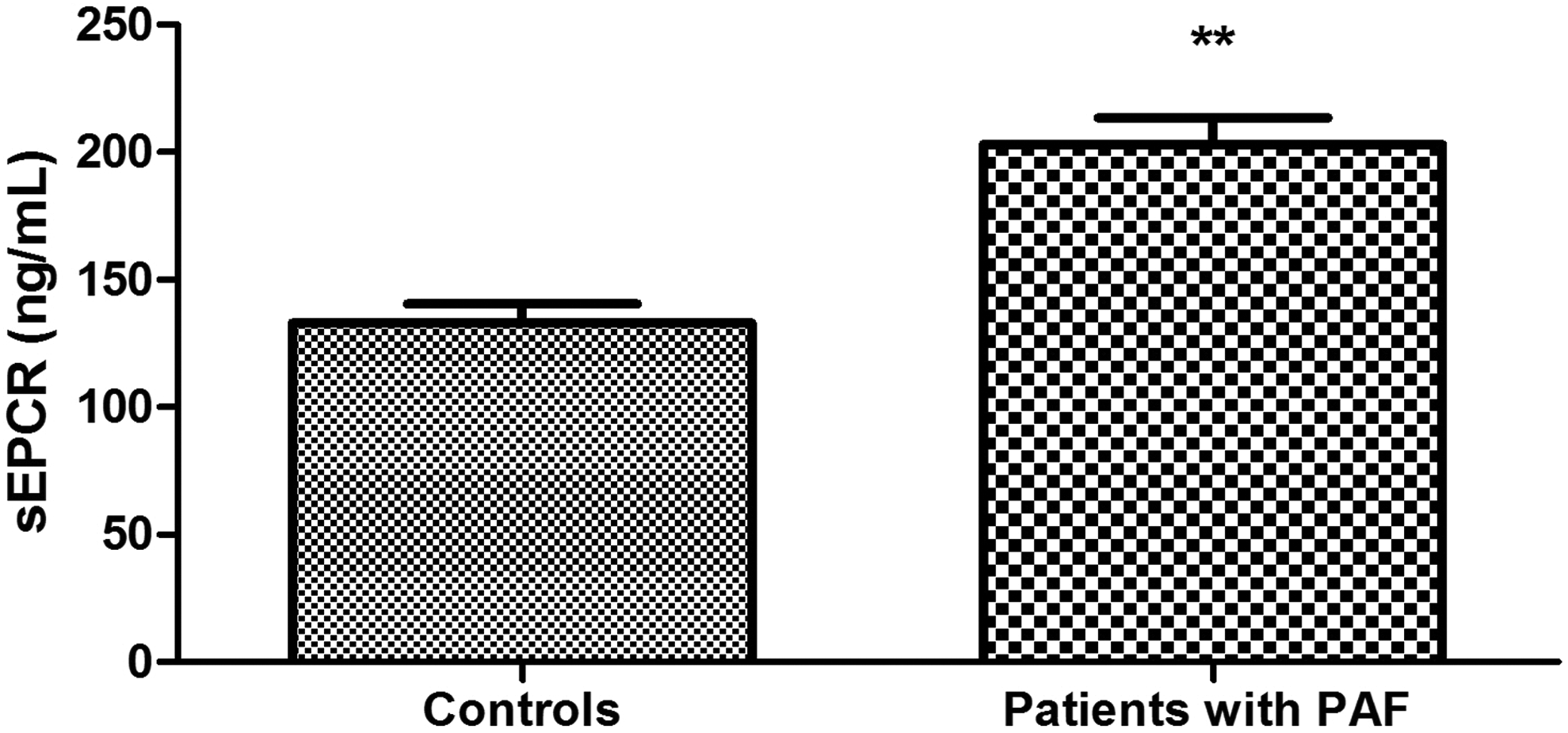
Plasma levels of soluble forms of endothelial protein C receptor (sEPCR) in patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (**P < .001).
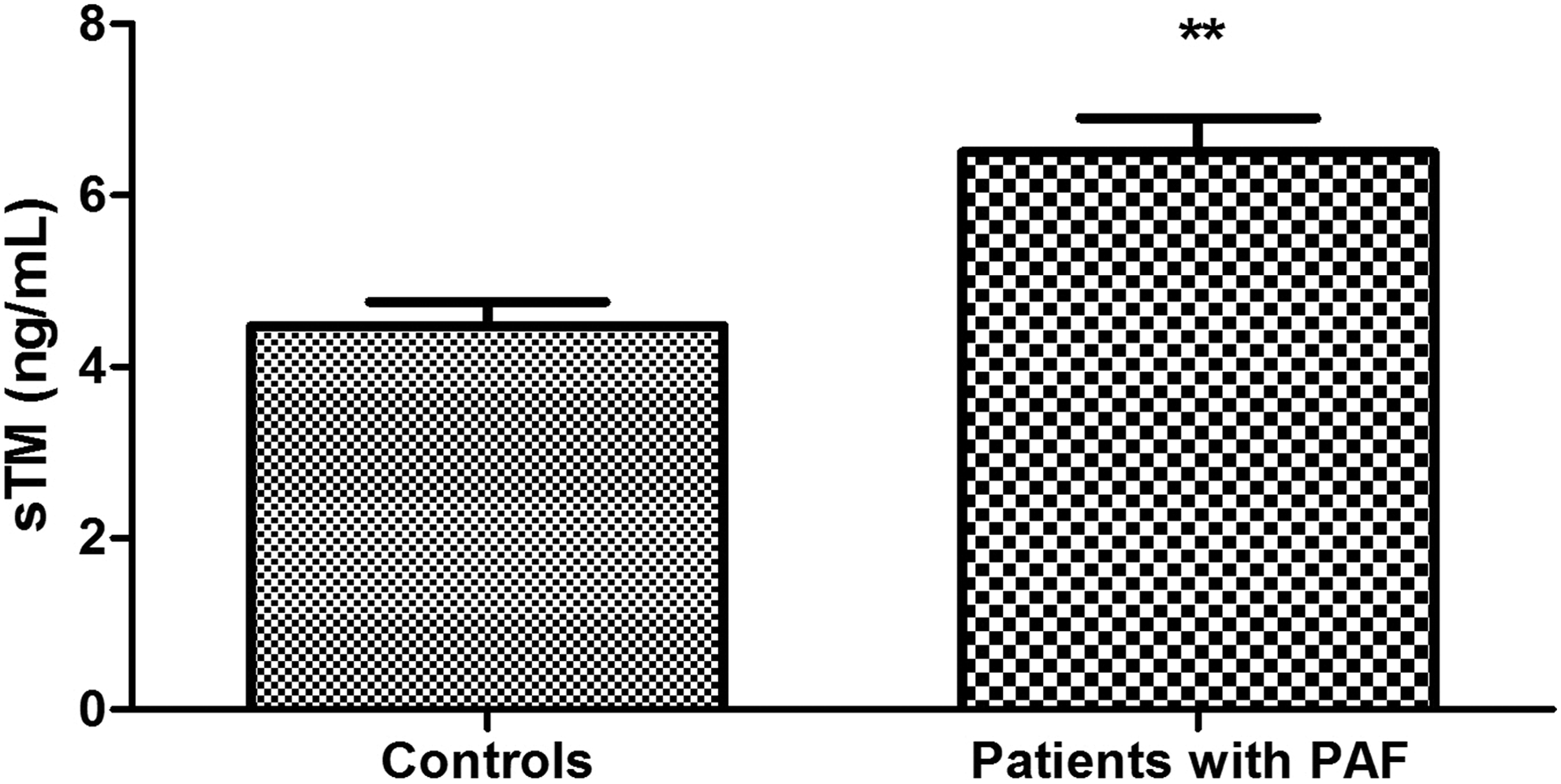
Plasma levels of soluble forms of thrombomodulin (sTM) in patients with paroxysmal atrial fibrillation (PAF) and controls in sinus rhythm (**P < .001).
Discussion
As pointed out earlier, the protein C anticoagulant pathway plays a crucial role in the hemostatic balance of the human body. 13,30 It has a direct contribution to the anticoagulant system response while exercising indirect control on coagulation and fibrinolytic systems. 30 Therefore, thromboembolic complications in PAF require protein C pathway study. A number of indicators should be examined with protein C itself taking central place.
Plasma levels of protein C are significantly higher in the PAF group than those in the control group (P < .05; Figure 1). Elevated values are present very early in the course of the disease, considering the fact that patients were hospitalized between the 2nd and 24th hour from the onset of the arrhythmia. It is well known that protein C is synthesized in the liver. Its levels are extremely sensitive to changes in liver function. 31 Therefore, they are a good indicator of its evaluation. 32,33 In this sense, the high levels of protein C in PAF could be interpreted as indicative of enhanced liver function and, respectively, enhanced protein C biosynthesis and secretion in the early hours of the rhythm disorder. The formation of “pure” groups in terms of comorbidities and medications affecting hemostasis is an important prerequisite for this claim (see Table 1).
Protein C levels do not always correlate proportionally to its activity. 34 In some cases of both congenital and acquired protein C deficiency, normal immunological levels are associated with a decreased functional activity of the protein. This implies simultaneous determination of both indicators. In our study, high plasma concentrations of protein C in PAF are characterized by a significantly lower activity compared to controls (P < .001; Figure 2). Thus, the presented changes are indicative of the presence of protein C deficiency in the early hours of the clinical presentation of the disease. We consider the high plasma levels of the indicator as a compensatory reaction to the low activity.
Activated protein C plays a leading role in the regulation of thrombin formation. It cleaves critical sites in the activated procoagulant factors V and VIII, thus inactivating these enzymes. 35 In this sense, it is acceptable to say that the established low protein C activity in PAF suggests a downregulation of thrombin generation by FVa and FVIIIa in the early hours of the arrhythmia.
It is well known that precisely the functional adequacy of protein C plays a critical role for the efficiency of the protein C pathway. 36 Therefore, our results give serious grounds to believe that the anticoagulant potential of protein C pathway is reduced in the early hours of PAF. The decreased activity is a prerequisite for breaking the delicate balance between procoagulant and anticoagulant forces and creating a prothrombotic state. 37
The molecular mechanisms governing the anticoagulant activity of protein C pathway are inherently complex. Protein S is their important component. Its free form is one of the APC cofactors and, in this sense, it is directly responsible for the maximum catalytic efficiency of the anticoagulant. 38 Total plasma levels of protein S in patients with PAF do not differ significantly from controls (P > .05; Figure 3), while the levels of free protein S and its functional activity are significantly reduced (P < .001; Figures 4 and 5). Given the aforementioned data, the results are reason to assume with high probability that the established variations in protein S are accountable for the low activity of protein C. The distinction in examining total and free protein S is extremely important. It is a reason to say that the reduced activity of the cofactor is associated with deficiency in its free form (Figure 4), most likely due to its transition into the connected form.
It is known that protein S also possesses an APC-independent anticoagulant activity. 39 Protein S inhibits the tissue factor activity by enhancing the interaction between the tissue factor pathway inhibitor (TFPI) and factor Xa and thereby reduces coagulation. Thus, the estimated protein S deficiency in the patient group increases the risk of thrombosis not only by violating the protein C pathway activity but also by reducing the capacity of TFPI to inhibit the extrinsic coagulation pathway.
Thrombomodulin and EPCR cell receptors are an integral part of the protein C pathway by promoting protein C activation. Their soluble forms, sTM and sEPCR, respectively, also affect the anticoagulant potential of the pathway and present research interest. Soluble EPCR, unlike the membrane-associated form, inhibits protein C activation and APC anticoagulant activity through connecting, respectively, with protein C and APC. 40 Levels of sEPCR were significantly higher in patients than in controls (P < .001; Figure 6). Given the effect of sEPCR on the protein C pathway, the established deviations might be a possible explanation for the decreased activity of protein C. It is well known that sEPCR is released into plasma during thrombin formation and therefore it is considered as a potential marker of prothrombotic state. 41 Its high levels in plasma in PAF could be regarded as indicative of an increased thrombogenic risk.
In this respect, we also observed sTM. It is well known that its plasma levels are indicative of the degree of proteolytic cleavage of the TM membrane form. 42 Therefore, we interpret the high values of the index as an increased TM loss on the endothelial cell surface in patients with PAF (Figure 7), which in turn implies reduced activation of protein C.
Our literature search showed that there is no data on changes at such an early state in the activity of the protein C anticoagulant pathway. It is appropriate to note that the selected by us patient and control groups are pure in regard to a number of diseases that lead to changes in coagulation, and none of the participants conducted anticoagulant therapy during the study. All these facts gave us reasons to assume that the identified changes are closely associated with the clinical manifestation of PAF.
The studies to date on the activity of the protein C anticoagulant pathway in PAF are few, and the resulting data are contradictory. Marin et al established significantly elevated sTM plasma levels in acute-onset AF. 43 Unlike them, however, Olivot et al and Freestone et al did not register significant changes in the values of the indicator. 19,44 Ambiguous are also data on the state of the protein C system in persistent and permanent AF. The results presented by Mondillo et al do not establish changes in the plasma levels of protein C in isolated nonrheumatic permanent AF, while Alonso et al showed an association between low levels of protein C and the clinical manifestation of the rhythm disorder. 45,46
In conclusion, we could say that the early hours of the PAF clinical manifestation (up to the 24th hour) are associated with significant changes in the protein C anticoagulant pathway. The activity of its main element protein C is reduced, determining reduced activity of the anticoagulant pathway as a whole. The established low free protein S and its activity as well as low levels of sEPCR and sTM are a possible explanation of the changes in protein C activity.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
