Abstract
Contrary to well-recognized bleeding diathesis in chronic liver disease, thrombotic events can occur in these patients due to reduction or loss of synthesis of anticoagulant proteins. Forty-seven consecutive patients with end-stage liver disease (ESLD) were investigated for activity of protein C, protein S, antithrombin, and factor V Leiden mutation. Forty-two (89.4%) patients had low levels of at least 1 while 33 (70.2%) patients were deficient for all anticoagulant proteins studied. Forty-six (97.9%) patients were negative for factor V Leiden mutation. The deficiencies were more marked in hepatitis C virus–positive patients and patients with model for end-stage liver disease (MELD) score >15. Six (12.8%) patients had portal vein thrombosis (PVT), and all had diminished protein S activity. In conclusions, deficiency of anticoagulant proteins occur in early phase of chronic liver disease. The severity of deficiency is proportional to the severity of liver disease. Despite the high prevalence of hypercoagulability, the incidence of PVT is low. Further studies with larger cohort of patients are needed to support these conclusions and to study other associated factors.
Introduction
The regulation of blood coagulation is dependent on a complex interplay between procoagulant, anticoagulant, and fibrinolytic proteins.1 –3 The liver plays a central role by being involved in the synthesis of both procoagulant and anticoagulant proteins. Hence, hepatic insufficiency is not only accompanied by numerous changes in the hemostatic system secondary to reduction in protein synthesis but also due to dysfunction in clearance of activated hemostatic proteins and protein inhibitor complexes from the circulation. The resulting clinical state (bleeding or thrombosis) will be determined by type of complex hemostatic mechanisms which predominates.1 –3
While thrombocytopenia and prolongation of prothrombin time (PT) are the well-recognized abnormalities in chronic liver disease,1 –7 the abnormalities related to anticoagulant proteins were not well elucidated. Reductions in the synthetic capacity of anticoagulant proteins (protein C, protein S, and antithrombin) have been demonstrated to be responsible for a hypercoagulable state.1 –7 This might result in thrombotic complications, which may be clinically evident as portal vein thrombosis (PVT) or peripheral thrombosis in the extremities with associated pulmonary emboli.8 –11
The aims of this study were (1) to examine the prevalence and degree of deficiency of anticoagulant proteins in patients with end-stage liver disease (ESLD), (2) to determine the correlation between the degree of deficiency and severity of liver disease, and (3) to examine the impact of anticoagulant protein deficiency on the prevalence of PVT.
Materials and Methods
In this study, we retrospectively examined 47 patients with ESLD that underwent hypercoagulable workup at our institution between January 2010 and December 2010. The institutional review board approved the study.
Clinical data points coded for analysis included baseline demographics, etiology of liver disease, presence of hepatocellular carcinoma (HCC), and model for end-stage liver disease (MELD) score. Only 1 patient had a history of deep vein thrombosis and none had pulmonary embolism.
Routine comprehensive metabolic and hematological parameters included PT/international normalized ratio (INR) and activated partial thromboplastin time. The hypercoagulation workup consisted of activity of protein C, protein S, antithrombin, and factor V Leiden mutation. Routine imaging included color Doppler ultrasound and contrast-enhanced computed tomography/magnetic resonance imaging of the abdomen to exclude HCC and PVT.
Statistical Analyses
Results were expressed as mean ± standard deviation unless otherwise specified. Comparisons were made using chi-square tests for categorical variables. Unpaired t test was used to compare the means. SPSS version 18.0 (SPSS, Inc, Chicago, Illinois) statistical software was used for analysis and differences were considered significant when P < .05.
Results
The patient’s baseline characteristics are given in Table 1 . The mean age was 56 ± 6.9 years and 33 (70.2%) were men. Chronic hepatitis C virus (HCV) infection was the commonest cause of ESLD involving 32 (68.1%) patients. Eight (17%) patients had HCC. The mean MELD score was 13 ± 4.7 (range: 6-23). Thirty-five (74.5%) patients had an MELD score ≤15 at the time of evaluation. Overall, the mean platelet count was 92.5 ± 59.2 (range: 21-274) × 10 3 /mm 3 and the INR was 1.3 ± 0.2. Seventeen (36.1%) patients had platelet count ≥100 × 10 3 /mm 3 and 33 (70.2%) patients had INR <1.5 including 6 (12.7%) patients with INR <1.
Baseline Patient Characteristics

Abbreviations: n, number; SD, standard deviation; MELD, model for end-stage liver disease; HCV, hepatitis C virus; EtOH, ethanol; NASH, nonalcoholic steatohepatitis; HBV, hepatic B virus; PBC, primary biliary cirrhosis; PSC, primary sclerosing cholangitis.
Anticoagulant Proteins
Overall, 42 (89.4%) patients were deficient for at least 1 of the 3 anticoagulant proteins. Thirty-three (70.2%) patients were deficient for all three anticoagulant proteins studied (Table 2 ). Activity of protein C, protein S, and antithrombin III was deficient in 39 (83%), 37 (78.7%), and 38 (80.9%) patients, respectively. The activity of protein C in 24 (51.1%) patients, protein S in 5 (13.5%) patients, and antithrombin in 14 (37.8%) patients was less than 50% of the lower limit of their normal ranges. Forty-six (97.9%) patients were negative for factor V Leiden mutation.
Correlation of Anticoagulant Proteins Activity With MELD Score
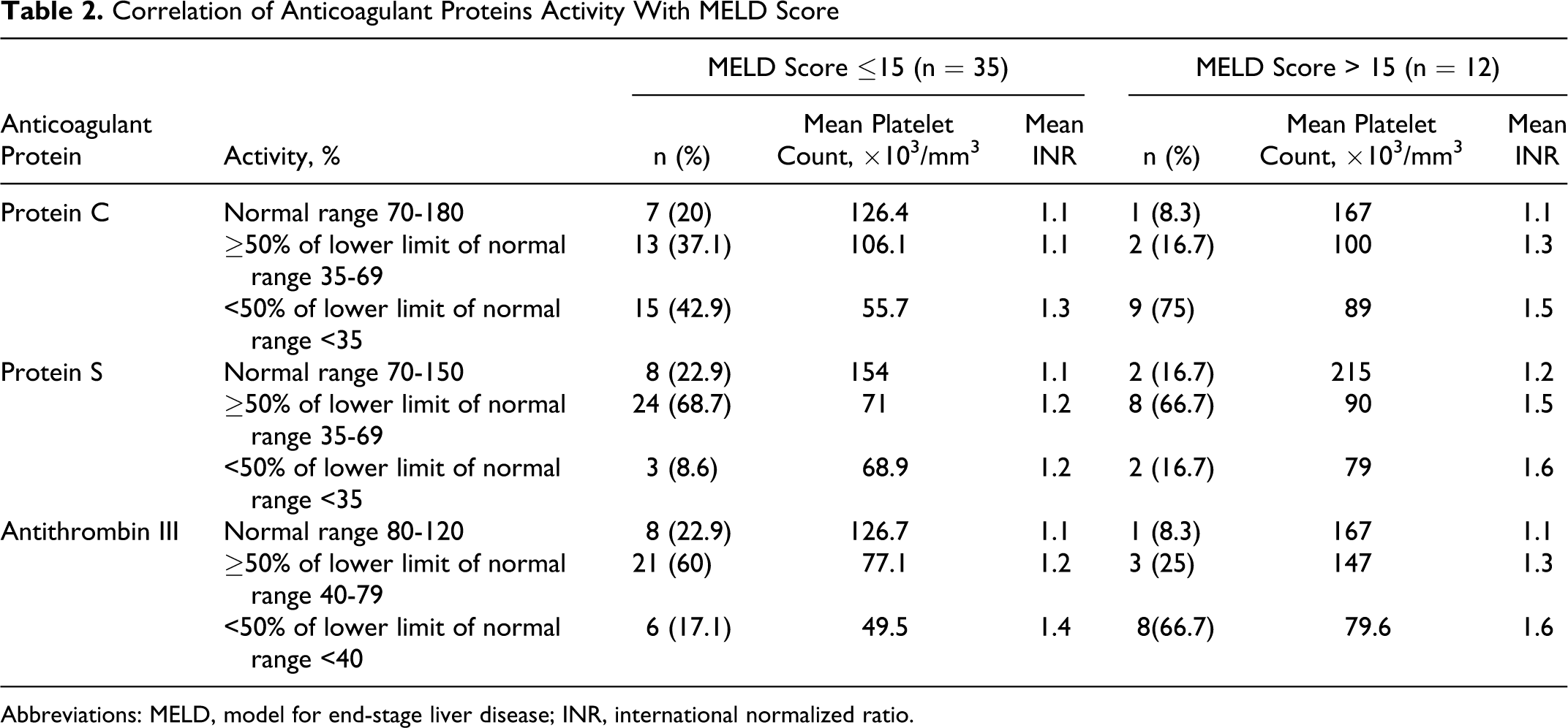
Abbreviations: MELD, model for end-stage liver disease; INR, international normalized ratio.
Severity of Liver Disease (MELD score, Child-Pugh class)
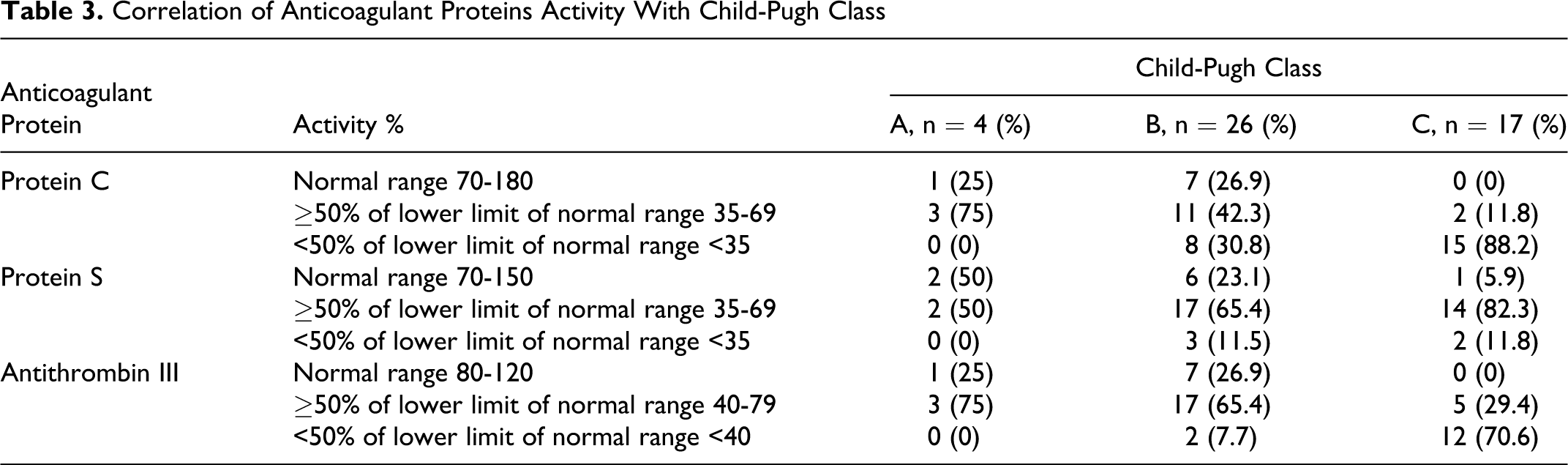
Twenty-four (73%) of 33 patients with decreased activity of anticoagulant proteins had MELD score ≤15. Thirty-one (88.5%) of 35 patients with MELD score ≤15 and 11 (91.6%) of 12 patients MELD score >15 had deficiency of at least 1 anticoagulant protein. In patients with MELD score ≤15, the activity of protein C, protein S, and antithrombin was deficient in 28 (71.7%), 27 (73%), and 27 (71%) patients, respectively. A higher percentage of patients with MELD score >15 had activity <50% of the lower limit of the normal range of respective anticoagulant proteins compared with patients with MELD score ≤15 (P >0.05; Table 2). The severity of their deficiency was proportional to the prolongation of INR and decrease in platelet count. The severity of decreased activity of anticoagulant proteins increases with the severity of liver disease manifested by Child-Pugh class (Table 3 ). A higher percentage of patients with child’s class C had activity <50% of the lower limit of the normal range of respective anticoagulant proteins compared with patients with class A or B (P < .05; Table 3).
Correlation of Anticoagulant Proteins Activity With Child-Pugh Class
Etiology of Liver Disease
Twenty-four (73%) of 33 patients with deficient anticoagulant proteins had HCV-related ESLD. Activity of protein C, protein S, and antithrombin was deficient in 27 (69.2%), 25 (67.5%), and 27 (71%) patients, respectively. Interestingly, a higher percentage of patients with HCV-related ESLD had a greater deficiency (<50% of the lower limit) compared with non-HCV patients (P > .05; Table 4 ).
Correlation of Anticoagulant Proteins Activity With and Without HCV-Related Liver Disease

Abbreviation: HCV, hepatitis-C virus.
Portal Vein Thrombosis
Overall, 6 (12.8%) patients had PVT (Table 5 ). All patients with PVT were deficient for protein S activity, while 4 (66.6%) were deficient for all 3 anticoagulant proteins. Two patients with PVT (# 2 and 3) had normal protein C and antithrombin activity and had low MELD scores. Five (83.3%) patients had PVT involving whole portal vein. Three patients had nonocclusive PVT. Four (67%) of 6 patients with PVT also had superior mesenteric vein thrombosis. None of the patient with PVT had HCC.
Activity of Anticoagulant Proteins in Patients With PVT
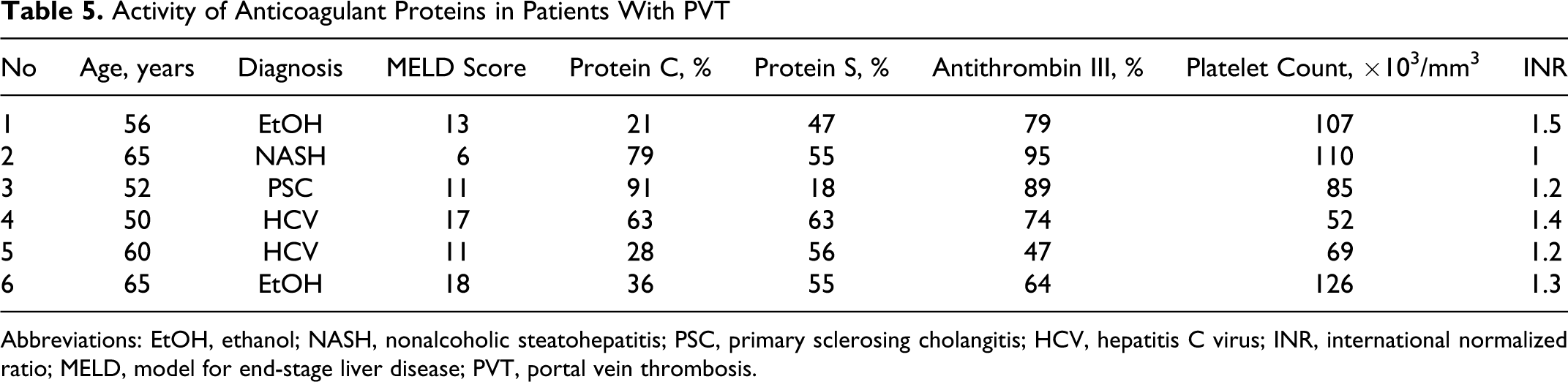
Abbreviations: EtOH, ethanol; NASH, nonalcoholic steatohepatitis; PSC, primary sclerosing cholangitis; HCV, hepatitis C virus; INR, international normalized ratio; MELD, model for end-stage liver disease; PVT, portal vein thrombosis.
Discussion
Hypercoagulability plays an important role in many aspects of acute and chronic liver disease. An average patient with liver failure may remain in hemostatic balance despite prolonged routine coagulation tests. Since both procoagulant and anticoagulant are affected in liver disease, the abnormalities of latter are not well reflected in the current routine coagulation testing, and their potential role remains underrecognized.1 –3
Proteins C and S are vitamin K-dependent proteins, while antithrombin is a nonvitamin K-dependent glycoprotein synthesized mainly by hepatocytes.12,13 During acute or chronic liver disease, their concentrations decrease concomitantly with the other coagulation factors, mainly due to reduced synthesis. In the present cohort, 70.2% patients were deficient for all anticoagulant proteins. Protein C was the most common and severely affected anticoagulant protein. Tripodi et al reported the similar conclusions that the hypercoagulability in patients with cirrhosis is due to the increased levels of factor VIII and decreased levels of protein C. 14 Surprisingly, more than 70% of our patients with deficient anticoagulant proteins had MELD score ≤15 and INR <1.5. This is in contrast to earlier reports suggesting the deficiency of anticoagulant proteins as an indicator of advanced liver disease.1 –3 In addition, it is certain that the remaining 27% patients with MELD >15 also have had these deficiencies during early stages of liver disease. We agree that the levels of both procoagulant and anticoagulant proteins decline as liver disease progresses. But the preliminary results of our study indicate that the decline in plasma activity of anticoagulant proteins actually occurs much earlier and constitute an early disturbance of the coagulation system in patients with chronic liver disease.
Consistent with the current prevalence of HCV infection in the United States, 73% patients with deficient anticoagulant proteins in our study had HCV-related ESLD. Interestingly, in patients with chronic hepatitis C, concomitant reduction in plasma levels of antithrombin, protein C, and/or protein S has been consistently associated with more severe fibrosis stage.9,11 It may be prudent to comment that ongoing necroinflammatory activity inherent to HCV may further contribute to more severe deficiency of anticoagulant proteins in the patients with HCV compared with non-HCV patients.
It is known that the incidence of thrombotic events increases with the increase in severity of cirrhosis, being less than 1% in patients with compensated cirrhosis, 15 but ranges from 8% to 25% in candidates for liver transplantation. 16 In the present study, despite a deficiency of natural anticoagulant proteins in 70.2% patients, the overall prevalence of PVT was 12.8%. This verified the fact that thrombosis in liver disease is multifactorial, which include poor flow, vasculopathy associated with a chronic inflammatory state, and the reduced synthesis of the natural anticoagulant proteins. Tissue factor pathway inhibitor concentration decreases in advanced liver disease, and this may also contribute to PVT. 17 Unexplainably, all patients with PVT had a deficiency of protein S, while 66.6% patients were deficient for all 3 anticoagulant proteins. In addition, 66.6% of the patients with PVT had MELD score ≤15. Hereditary or acquired deficiencies of protein S activity have been associated with venous and less commonly arterial thromboses.18,19
This initial study is retrospective and entails certain limitations including small sample size and lack of randomization, necessitating validation of findings with a larger patient population using prospective methodology. The next step would be to follow anticoagulant protein levels in patients after liver transplantation. The findings of the present study also led us to consider whether we should routinely investigate the activity of anticoagulant proteins in patients with chronic liver disease as an early sign of decompensation. However, until more data are available, PT represents the best answer.
Conclusions
Based on the findings of present study, we consider that deficiency of anticoagulant proteins occur in early phase of chronic liver disease, even before prolongation of PT. The severity of deficiency was proportional to the severity of liver disease. Patients with HCV-related ESLD had greater deficiency of anticoagulant proteins, probably due to ongoing necroinflammatory process. Despite a high prevalence of deficiency of anticoagulant proteins, PVT was found only in 12.8% patients. Activity of protein S was deficient in all patients with PVT. Further studies with larger cohort of patients are needed to support these conclusions.
Footnotes
Authors’ Note
These data have been presented at 17th Annual International Congress of International Liver Transplantation Society, June 22-25, 2011, Valencia, Spain.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
