Abstract
Objectives:
To test for the presence of inflammatory biomarkers in blood taken from varicose veins versus antecubital blood of the same patient and compare this to levels in healthy controls.
Methods:
Using a multiplex biochip array method (Randox, United Kingdom), the interleukins (ILs) IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10; vascular endothelial growth factor; interferon γ, tumor necrosis factor α ; monocyte chemotactic protein 1 (MCP-1); and epidermal growth factor were measured in citrated plasma samples drawn from the arms and legs of 24 patients with varicose veins and 24 controls.
Results:
Expressed as median (interquartile range) in pg/mL, leg samples from patients with varicose veins had significantly higher levels of IL-8 and MCP-1 compared to their own arm samples (IL-8: local 2.3 [1.71-3.3] vs systemic 2.3 [1.62-2.98], P = .023; MCP-1: local 114.42 [84.29-139.05] vs systemic 103.56 [79.75-126.42], P < .0005). This was not observed in the control group. Leg samples from both patients with varicose vein and controls had higher levels of IL-6 compared to their own arm samples (patients: local 1.67 [0.82-4.48] vs systemic 1.24 [0.58-3.26], P = .002; controls: local 1.23 [0.83-1.7] vs systemic 1.03 [1.7-1.52], P = .005). No significant differences were detected with the other biomarkers.
Conclusions:
Blood drawn from the site of varicose veins appears to have significantly increased concentrations of IL-6, IL-8, and MCP-1 when compared to the same patient’s arm blood. This supports the hypothesis that inflammation is activated from the tissues drained by the varicose veins.
Keywords
Introduction
Primary varicose veins are an objective clinical sign that may indicate the presence of significantly impaired venous drainage from the leg. The condition of impaired venous drainage is termed chronic venous insufficiency (CVI). Invariably, the cause in primary CVI is reflux or reverse flow against the direction of the venous valves. In this situation, the normal process of antegrade flow toward the heart is hampered by incompetent perforating veins and saphenous trunks that allow gravitational forces to direct venous flow back toward the foot. This creates an environment where the venous pressure within the veins of the lower leg is elevated and sustained during dependency because of a continuous hydrostatic column of blood. In health, the muscle pumps of the leg interrupt the hydrostatic column and offset the raised pressure by pumping blood upward. However, in disease, the relief from venous pressure is short lived from the rapid cascade of retrograde flow after calf muscle pump activity. In patients with CVI, the antigravitational mechanisms of the leg have failed, and the consequence is a pathologically sustained increase in the venous pressure. 1
Most patients with pathological CVI, or healthy persons subjected to physiological CVI from prolonged standing, may experience leg pain on dependency, 2 edema, and later on may develop skin changes in the gaiter region with hyperpigmentation, fibrosis, and even venous ulceration. This indicates the presence of inflammatory processes at work, mediating tissue destruction. While the causes of a raised venous pressure are known, the inflammatory cytokines that provoke the final pathways of tissue damage and that are responsible for the clinical features of CVI are awaiting discovery. Inflammation is an essential process in tissue destruction, but it is likewise vital in tissue recovery and healing.
The aim of this study was to measure the concentrations of inflammatory cytokines in the arm, where the venous pressures are normal, and blood is taken usually and compared to samples taken from the ankle region of the leg. This was performed in patients with known CVI and compared to healthy controls without clinical evidence of venous disease.
To identify which cytokines are associated with CVI, this project used a batch analysis of an array of 12 known inflammatory cytokines that have been identified as possible components of the molecular process involved in CVI. 3 –9
Methods
Study Design
This was a dual center study on 24 patients with varicose veins and 24 controls. Patient recruitment and blood drawing took place at Ealing Hospital with subsequent analysis at Loyola University Medical Center. All the patients were awaiting endothermal laser ablation to their great saphenous vein (GSV). They were referred initially from the family doctor with leg symptoms suggestive of venous origin.
A duplex ultrasound examination, performed standing, was used to confirm the presence of reflux in the GSV to fulfill the inclusion criteria. Reverse flow provoked by a manual calf compression and release maneuver with a duration >1 second was used to define significant reflux for this study. 10 The diameter of the GSV was measured in the upper thigh while standing as a rough estimate of disease severity. 11 The presence of deep venous abnormalities such as wall irregularities/fibrosis, intraluminal echogenicity, incomplete collapse on compression, or a reduction in luminal diameter implied a previous deep vein thrombosis (DVT). These patients were excluded. Exclusion criteria were also a history of DVT, previous venous interventions, on medications that affect coagulation, known disorders of coagulation, inability to comprehend study demands, active malignancy, and comorbidity.
Patients were stratified to define the study population using validated questionnaires; the clinical part of the revised clinical, etiological, anatomical, pathophysiological (CEAP) classification 12 and the venous clinical severity score (VCSS). 13 The former stages the venous severity between C0 representing the absence of venous disease through to C6 that represents a venous ulcer. The remaining categories are, in order: C1 (telangiectasia or reticular veins), C2 (varicose veins), C3 (edema), C4a (eczema or pigmentation), C4b (lipodermatosclerosis or atrophie blanche), and C5 (healed venous ulcer). The categories C4 and C6 are associated with active inflammation. The latter questionnaire includes patient symptoms and is acknowledged to be more responsive to treatment. The VCSS ranges between 0, which is normal, and a potential maximum score of 30.
The control group consisted of healthy volunteers without any symptoms or clinical signs of venous disease. They were matched in terms of age and sex so that there would be no statistical difference between the groups regarding these parameters. This was an essential part of the study design in order to determine whether regional and systemic differences in inflammatory cytokines were exclusive to the patients.
The study protocol was approved by the Regional Ethical Committee (REC number 12/LO/0024) and the Research and Development Department of Ealing Hospital. All participants received a study information leaflet and signed a consent form.
Venous Blood Samples
All blood samples were drawn manually by direct puncture of a vein using a blue, 23-gauge, 25-mm-long needle. This was done first from the ankle region and then from the antecubital vein after at least 5 minutes of rest in the sitting position. In patients, the ankle sample was from the lowest varicose vein, and in the controls, this was from a foot vein near the ankle or the GSV near the medial malleolus. The ankle samples were uncuffed from a dependent leg, but the arm samples were extracted with the use of a proximal tourniquet. Therefore, both samples were taken from distended pressurized veins. 14
After 5 mL were extracted, these were transferred into citrated tubes and centrifuged at 3500 rpm for 10 minutes. The supernatant plasma was separated manually and stored at −20°C for batch analysis. On completion of the study, the plasma samples were all transferred under dry ice protection by air courier to the Thrombosis and Hemostasis Research Laboratories at Loyola University Medical Center, Maywood, Illinois, United States.
The concentrations of the following 12 inflammatory cytokines were assayed: 7 interleukins (ILs; IL-1α, IL-1β, IL-2, IL-4, IL-6, IL-8, and IL-10), vascular endothelial growth factor, interferon γ, tumor necrosis factor α, monocyte chemotactic protein 1 (MCP-1), and epidermal growth factor. These were performed on all the samples in a batch by following the manufacturer’s instructions from a commercially available kit (Randox Laboratories Ltd, London, United Kingdom). This was possible because of a recent technological advance termed biochip array technology, which allows simultaneous multianalyte detection.
Statistical Analysis
A normal distribution of plasma concentrations of inflammatory biomarkers was not assumed. Therefore, all the statistics were performed using nonparametric principals with medians, interquartile ranges (IQR), and ranges. The statistical software IBM SPSS version 22 was used throughout and data imported following their tabulation in Excel spreadsheets. Statistical differences within individual patients (arm vs leg) were evaluated using the Wilcoxon signed rank test with correlations using the Spearman ρ test. Differences between the groups (patients vs controls) were evaluated using the Mann-Whitney U test. Significance was achieved when P < .05.
Results
Demographics
The median (range) ages of the patients and controls were 45 (25-91) years and 45 (24-89) years, respectively (P = .483: Mann-Whitney U test). In both groups, 17 of the 24 were male. The right leg was tested in 11 of the 24 patients and in 12 of the 24 controls.
In the patients, the clinical CEAP classification was C2 (6), C3 (4), C4a (1), C4b (6), C5 (5), and C6 (2). The median (range) VCSS was 6 (4-10), and the diameter of the refluxing GSV measured with duplex ultrasound was 8.2 (6-12) mm.
General
The differences between local (leg) and systemic (arm) inflammatory cytokine levels in patients with CVI and healthy volunteers are summarized in Table 1. Statistically significant differences, where local concentrations were greater than systemic concentrations, occurred only with IL-6, IL-8, and MCP-1. Concentrations of IL-6 were higher in 19 of the 24 patients’ legs and 17 of the 24 control legs, whereas those of IL-8 and MCP-1 were increased only in the patients’ legs at 17 of the 24 and 21 of the 24, respectively. This indicates that the most relevant inflammatory biomarkers in CVI appear to be IL-6, IL-8, and MCP-1. These are assessed in more detail subsequently.
Plasma Levels of Inflammatory Cytokines Taken From Ankle Veins (Local) and Antecubital Veins (Systemic) in Patients With Varicose Veins and Healthy Volunteers (Controls).a
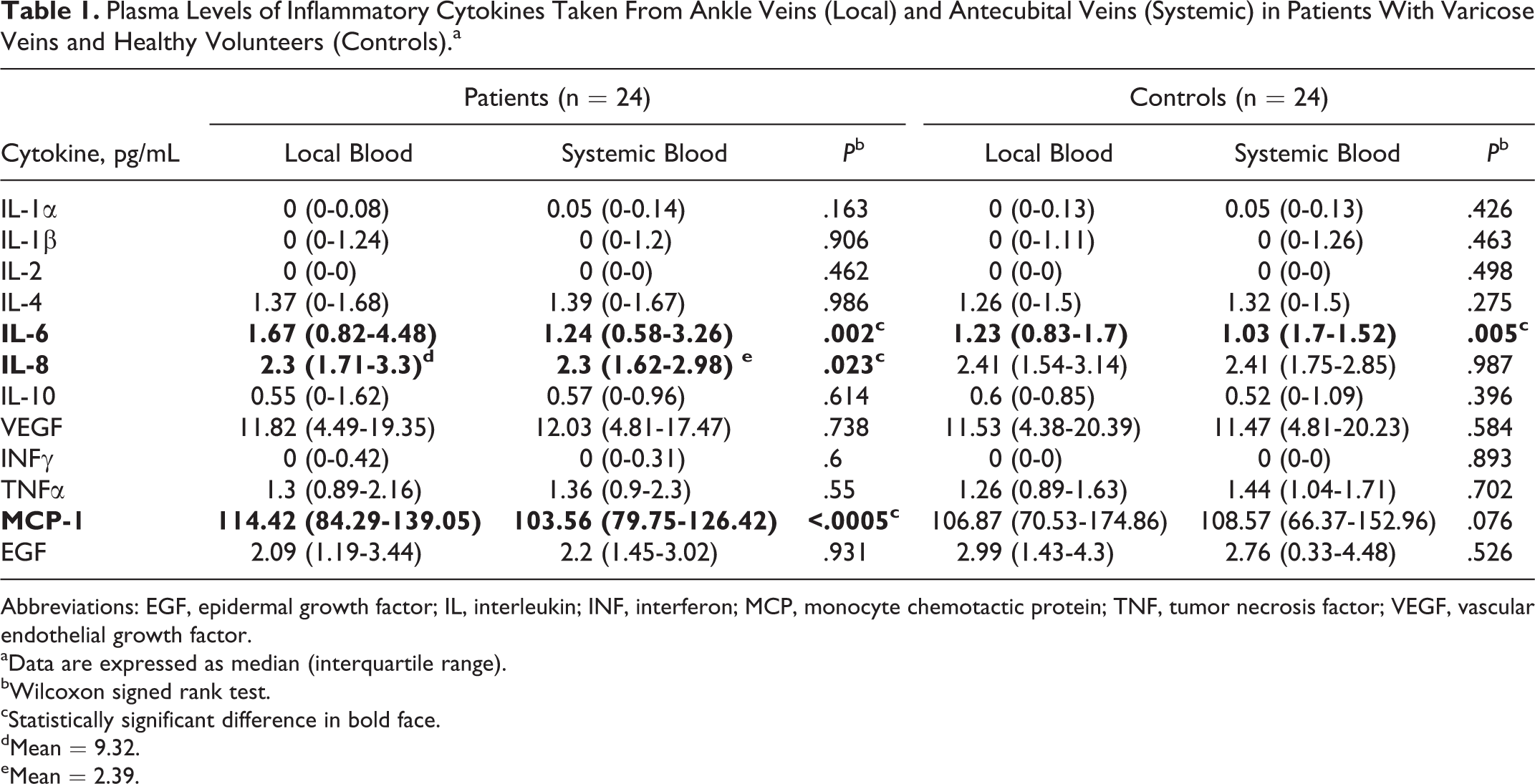
Abbreviations: EGF, epidermal growth factor; IL, interleukin; INF, interferon; MCP, monocyte chemotactic protein; TNF, tumor necrosis factor; VEGF, vascular endothelial growth factor.
aData are expressed as median (interquartile range).
bWilcoxon signed rank test.
cStatistically significant difference in bold face.
dMean = 9.32.
eMean = 2.39.
Age and Severity of Disease
The age of the patient and the severity of disease were the variables that appeared to impact most on the results of IL-6, IL-8, and MCP-1 as shown in Figures 1 and 2, respectively. Increasing age correlated significantly with increasing IL-6 concentrations. Below the age of 64 years, the majority of IL-6 levels were <2 pg/mL, whereas in patients older than 64 years, all the IL-6 values were >1.5 pg/mL. In the patients, there were no significant correlations between IL-8 and MCP-1 versus age. This is in direct contrast to the control group where IL-8 and MCP-1 concentrations appeared to increase with increasing age.
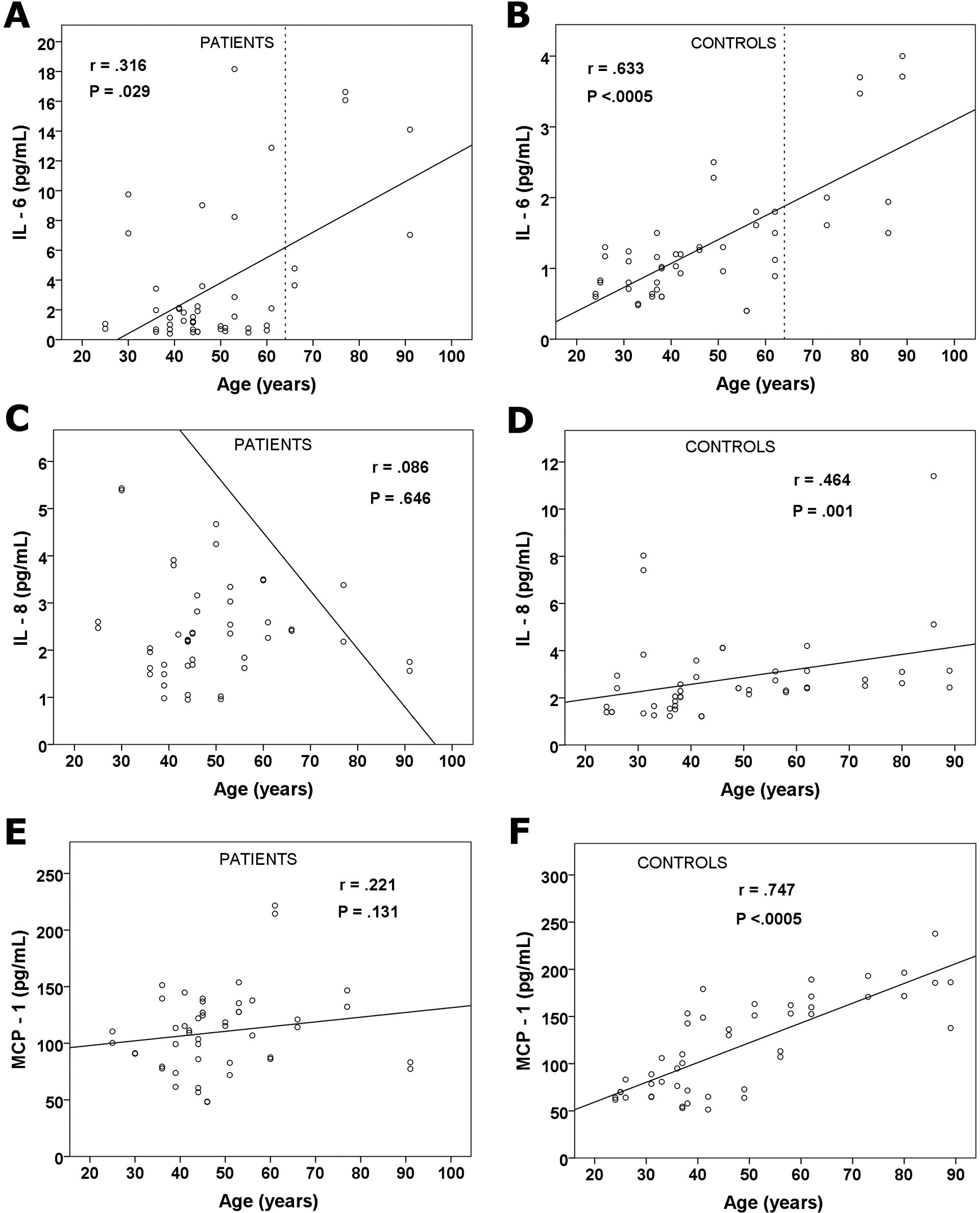
Relationship between interleukin (IL) 6 (A and B), IL-8 (C and D), and monocyte chemotactic protein 1 (E and F) concentration versus age on all the arm and leg samples from patients (n = 48) and controls (n = 48). Correlations (r; Spearman) are given with their significance (P). Dashed line represents the age cutoff of 64 years.
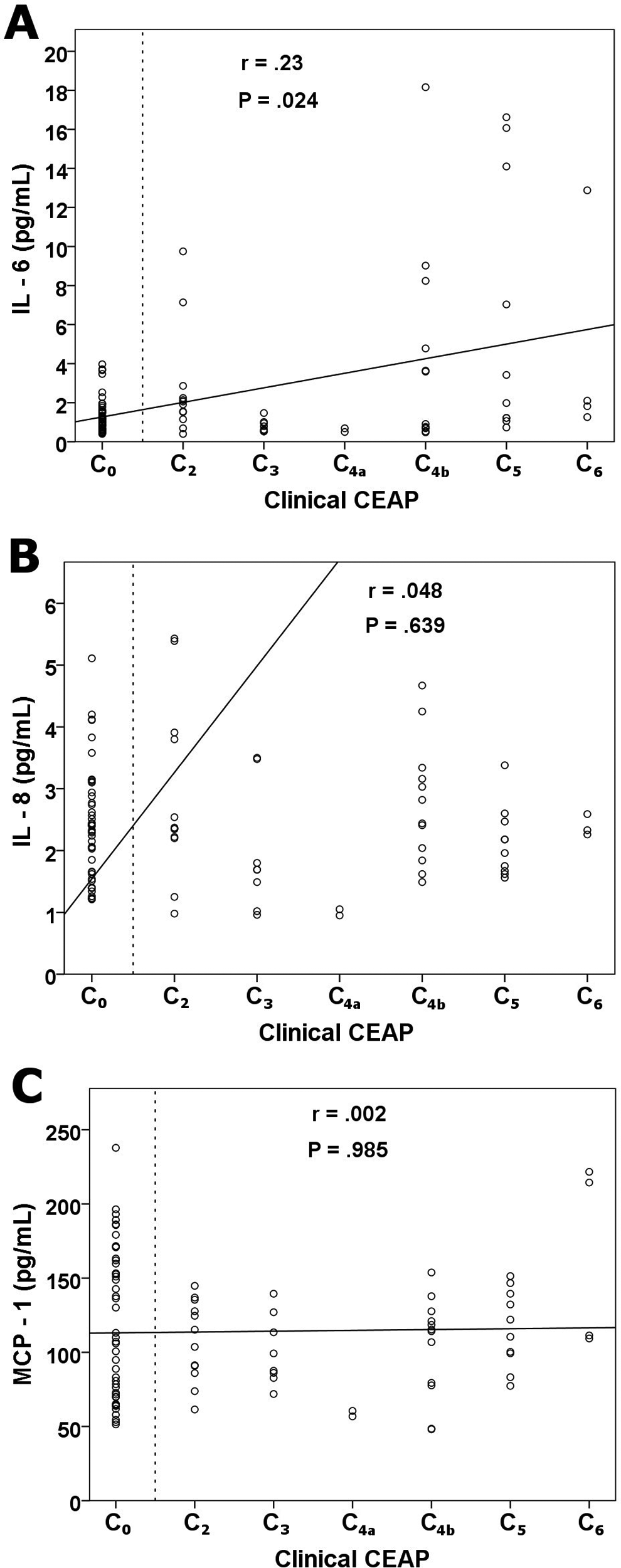
Relationship between interleukin (IL) 6 (A), IL-8 (B), and monocyte chemotactic protein 1 (C) concentration versus clinical severity of venous disease on all arm and leg samples from patients and controls (n = 96). Correlations (r; Spearman) are given with their significance (P). Dashed line separates the patients from the controls.
There was a weak correlation between the severity of venous disease as assessed by the C of the CEAP classification and IL-6 concentrations, as shown in Figure 2A. All volunteers had IL-6 values <4 pg/mL. Of the patients’ samples, 2 were >4 pg/mL in CEAP class C2, compared to 9 samples >4 pg/mL in the advanced CEAP class C4b to C6. Relationships between disease severity versus IL-8 and MCP-1 were not observed (Figure 2B and C).
Local–Systemic Relationship
Highly significant correlations (P < .0005) were observed between local (leg) blood and systemic (arm) blood in regard to IL-6, IL-8, and MCP-1 (Figure 3). Although IL-6 concentrations were statistically greater in the legs (vs arm) of patients and controls (P = .002 and P = .005, respectively), there was an excellent correlation between the increased leg values and the corresponding arm samples (Figure 3). This suggests that IL-6 is released from the leg and may get diluted once it appears in arm blood. There was a trend also toward higher IL-6 levels in patients legs versus control legs (P = .09, Mann-Whitney U test). Significant local/systemic correlations were also detected in the patients IL-8 and MCP-1 values at r = .863 (P < .0005) and r = .939 (P < .0005), respectively (Figure 3).
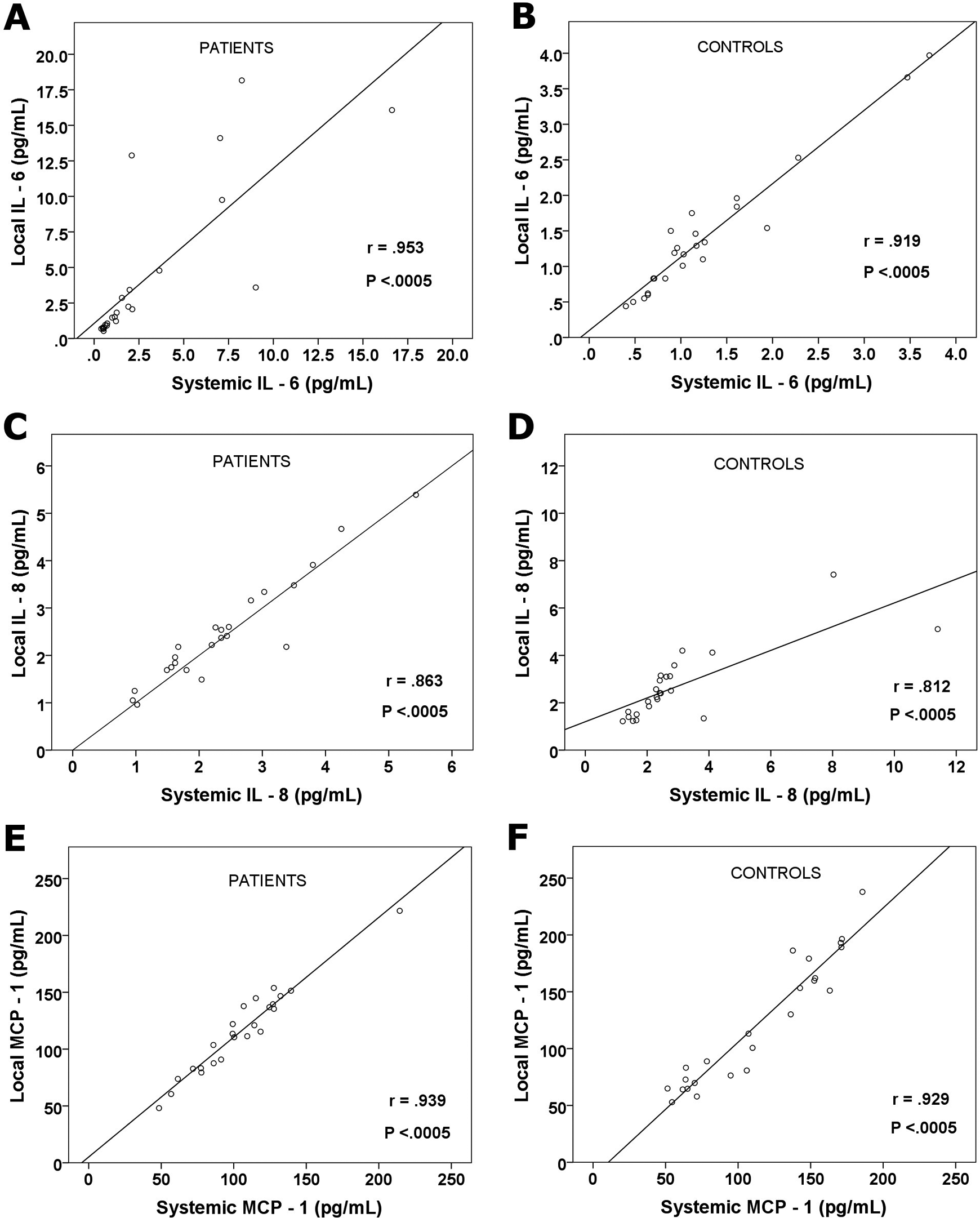
Correlation (Spearman) between local versus systemic interleukin (IL) 6 (A and B), IL-8 (C and D), and monocyte chemotactic protein 1 (E and F) concentrations in patients samples (n = 48) and control samples (n = 48). Correlations (r; Spearman) are given with their significance (P).
Discussion
Although the clinical features of CVI are caused by a hemodynamic imbalance in terms of sustained elevations in venous pressure from the forces of gravity, 15 the molecular pathways involved in cellular damage are unclear. The main factors that are recognized to exert an important role include hypoxia, 16 hemosiderin deposition, 17 fibrinogen/fibrinolysis, 18 and endothelial/glycocalyx dysfunction. 19 Central to the process of damage is inflammation, and the cell from which most cytokines are released is the leukocyte. 20, 21
This study investigated the levels of 12 inflammatory cytokines to discover which ones were most important in CVI. Venous blood samples were taken locally from the ankle region, where venous pressures are the greatest and compared to systemic blood samples taken from the arm. This was undertaken in patients with CVI because they had evidence of venous disorder as well as from healthy controls who are more protected from sustained rises in the venous pressure.
The acquisition of local blood samples draining the site of potential inflammation was an important part of the study design. Our group has reported previously, on the same set of patients, that prothrombotic biomarkers from systemic blood samples do not necessarily represent the biochemical activities occurring in the leg. For example, thrombin–antithrombin concentrations were significantly higher in leg blood with significantly reduced antithrombin activity.
22
Furthermore,
The observation that IL-6 was elevated in the legs of patients and controls may suggest that this IL is not related to CVI per se and may represent instead a normal physiological response to the higher venous pressures typical of leg veins in the gaiter region. This may reflect the higher sensitivity of IL-6 to venous pressure increases in contrast to IL-8 and MCP-1. However, IL-8 and MCP-1 may be more specific for inflammation in CVI. Since IL-6 was identified as the biomarker with the most interest, other factors were examined which may influence IL-6 concentrations. Age and disease severity were shown to correlate significantly with increases in IL-6 concentration.
Inflammation is an intrinsic part of the process of cellular damage as well as healing and recovery. While concentrations of IL-6, IL-8, and MCP-1 were found to be higher in the legs of patients, the assumption that this is causing injury may not be correct. These cytokines could equally be essential for tissue repair. For example, IL-6 has been discussed as both a proinflammatory and an antiinflammatory molecule. 25 Furthermore, in experimental studies, IL-6 may promote hepatocyte 26 and intestinal cell 27 regeneration. Matrix metalloproteinases (MMPs), not tested in this study, likewise, have important roles in venous damage as well as healing. 15,28
The identification of elevated blood concentrations of IL-8 and MCP-1 in the legs of patients may indicate that they have a specific role in the development of varicose veins. Prolonged ambulatory venous hypertension results in a pressurized venous system causing vein wall damage and the adhesion of leukocytes to the endothelium. 29 It is known that IL-8 and MCP-1 are cytokines that attract neutrophils and monocytes, respectively, and that these are expressed by smooth muscle cells and fibroblasts as well as endothelial cells. 30 Since normal veins have few inflammatory cells in their wall, this may indicate the pivotal role of IL-8 and MCP-1 in varicose vein development. 31 However, circulating IL-8 and MCP-1 are also increased in obesity 32 and many other inflammatory situations. 33 Minciullo et al have demonstrated that there is a relationship between many other cytokines with increasing age. 34 However, to our knowledge, here is the first time that a relationship between IL-8 and MCP-1 versus age has been demonstrated.
Of the cytokines IL-4 and IL-10, which both have an antiinflammatory action, IL-10 is the most potent. It is an inhibitor of cell proliferation, dilutes the immunological effects of antigen presentation, and hampers the manufacture of proinflammatory cytokines. 35 In the current study, neither cytokine appeared to have an association with venous insufficiency or sampling site.
Future studies are required to focus on the dual roles of inflammation in mediating tissue destruction as well as tissue repair. This could be achieved by taking local blood samples after prolonged standing and by comparing them in the same patient after prolonged elevation recovery. Local blood samples taken from patients with varicose vein after standing for 30 minutes have revealed significantly increased intracellular adhesion molecules 29 and MMP-9, suggesting that these are influential in pressure-mediated damage. 36
Molecular targets extracted from venous blood may be interpreted in 2 different ways: As evidence to support the role of inflammation in causing venous wall damage and valve incompetence. This has been discussed in relation to the etiology of varicose veins themselves, but usually from tissue samples, regarding hypoxia,
37
iron overload,
38
and inflammation from cytokines IL-8 and MCP-1.
39
However, venous wall concentrations may not represent the concentrations in streaming blood. As evidence that there is tissue damage from sustained venous pressure. This is reflected by blood samples from the veins that drain these regions. The current study is in support of the latter interpretation because of the universal belief that the destruction from CVI is caused by sustained venous pressure.
40
The rationale is that the gaiter region, the area of maximal venous pressure, has a greater volume of potential tissue damage than the venous wall itself. This is also the region that develops ulceration.
41
Furthermore, one of the commonest valves to become incompetent is at the sapheno–femoral junction that is remote from the region of elevated ambulatory venous pressure.
41
However, the situation may be different in a patient with the postthrombotic syndrome where the vein wall has developed extensive fibrosis. It would be interesting to compare the biomarkers of inflammation in this group to patients with reflux alone.
Currently, this work has identified 3 inflammatory cytokines, whose levels are increased in the legs of patients with CVI: IL-6, IL-8, and MCP-1. This may be a small step in the future discovery of a biomarker specific for CVI. Once available, such a biomarker would have potential utility for monitoring the effects of treatment and for increased understanding of the mechanisms of pressure-mediated cellular damage and repair.
Limitations
Potential limitations include small sample sizes and overlapping IQRs which may lessen the impact of any conclusions.
Conclusion
This study has confirmed that some inflammatory processes are activated in patients with varicose veins. Furthermore, blood samples taken from the ankle region may be a way forward in identifying which inflammatory cytokines are most influential in the pathophysiology of CVI. The ones of most relevance in this study appear to be IL-6, IL-8, and MCP-1. However, increased local concentrations do not establish a cause and effect relationship in the development of the skin problems of CVI, and this is a subject of future research.
Footnotes
Authors’ Note
This work was presented as an oral abstract in the European Venous Forum, June 25-28, 2014, Paris, France, and in the American Venous Forum, February 25-27, 2015, Palm Springs, CA, USA. The demographic details of the study participants are held at the Josef Pflug Vascular Laboratory, Ealing Hospital, Middlesex, United Kingdom. The inflammatory biomarker data and samples from which they arise are held at Loyola University Medical Center, Department of Pathology and Pharmacology, Maywood, IL, USA.
Acknowledgments
The authors wish to thank Daneyal Syed, BS, from the Thrombosis & Hemostasis Research Laboratories at Loyola University Medical Center for the analysis of the plasma samples. We also thank the pathology department of Ealing Hospital, United Kingdom, for their kind permission to use the centrifugation and freezing facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The primary author received a research grant from the Research & Development Department, Ealing Hospital, Middlesex, UB1 3HW, United Kingdom.
