Abstract
The purpose of the study was to analyze a systemic activation of hemostasis and concentration of matrix metalloproteinase 10 (MMP-10) in patients with primary varicose veins (PVVs). A study group consisted of 41 patients with noncomplicated PVVs. A control group consisted of 30 age- and sex-matched healthy individuals without varicose veins. The concentration of
Introduction
It has not been unequivocally established whether primary varicose veins (PVVs) are or not a risk factor for deep venous thrombosis (DVT). Varicose veins of lower limbs are cited as a risk factor for DVT in the guidelines of the 7th conference of the American College of Chest Physicians (ACCP) published in 2004 1 but were not mentioned in the guidelines of 8th ACCP conference in 2008. 2
There are few data on hemostatic activation in patients with varicose veins of the lower limbs without venous thrombosis.
3
Ikeda and colleagues found elevated levels of
A possible role of extracellular matrix metalloproteinases (MMP) in the pathogenesis of PVVs has been suggested. The presence of larger amounts of MMP-1 and smaller amounts of MMP-9 as well as their different distribution in the wall of the varicose great saphenous veins (GSVs) in comparison with healthy veins has been demonstrated. 6,7 Metalloproteinase-10 is a protease that degrades various components of basement membrane such as collagen type IV, laminin, and proteoglycans and activates other metalloproteinases (MMP-1, -8, and -13) that degrade the extracellular matrix. 8,9 We have shown that prothrombotic/proinflammatory stimuli increase the expression of MMP-10 in vitro and in vivo, 10 –12 thus linking hemostasis and inflammation. However, no studies have examined its role in patients with PVVs.
Patients and Methods
After approval by the local ethical committee, 2 groups of patients were included: (1) a group of 41 patients with PVVs and GSV incompetency, qualified to an invasive treatment of varicose veins; (2) a control group of 30 age- and sex-matched healthy individuals without varicose veins.
In all study patients data on demographic factors, medical history, comorbidities, and risk factors for DVT were collected. Prior to the inclusion to the study, a duplex Doppler of lower limb venous system was performed. Patients with ultrasound signs of acute or chronic DVT and superficial thrombophlebitis were excluded from the study. The remaining exclusion criteria were as follows: patients under 18 years of age, pregnancy and breastfeeding, previous ipsilateral GSV surgery, chronic and acute lower limb ischemia, known thrombophilia or other coagulation disorders, lymphedema, any acute or chronic inflammatory disease, active cancer or current chemotherapy or radiotherapy, symptomatic coronary artery disease, any surgery or trauma within the last 4 weeks, aortic aneurysms, patients with cirrhosis, patients receiving oral anticoagulants or heparin, and patients with any ongoing inflammatory disease including thrombotic inflammation of the superficial veins, at the time of the study or during the preceding 4 weeks.
In all study patients thorough physical examination was performed. A clinical severity of chronic venous insufficiency was scored to according to Clinical-Etiology-Anatomy-Pathophysiology (CEAP) classification. 13 A degree of reflux in the GSV trunk was determined during duplex Doppler examination according to Hach classification. 14 No signs of systemic inflammation or renal insufficiency were observed in the group of patients since the leukocyte count, serum creatinine, and estimated glomerular filtration rate were within normal ranges.
Sample Collection
Venous samples were collected from superficial elbow vein into closed Vacutainer tubes (BD, Plymouth, United Kingdom) containing sodium citrate at a concentration of 0.129 mol/L for plasma preparation and into Vacutainer tubes (BD) containing SST II Advance gel for serum preparation. Anticoagulated blood was centrifuged at 2500 rpm for 15 minutes and platelet-poor plasma prepared. Serum was prepared from the pellet during centrifugation. Plasma and serum samples were frozen and stored at −80°C until assayed. The blood samples were always collected and the same time of a day at 8.00 in the morning.
Analytical Measurements
The concentrations of DD, F1+2, vWF, and activity of PAI-1 were measured in the plasma. The DD was measured by an immunoturbidimetric (INNOVANCE DD in a BCS analyzer, Siemens, Munich, Germany) and results expressed in ng/mL. The F1+2 was measured by an enzyme immunoassay (Enzygnost F1+2, Dade Behring GmbH, Marburg, Germany) and results expressed in pmol/L. The PAI-1 activity was determined with a chromogenic assay (Stachrom PAI-1, Diagnostica Stago, Asnières sur Seine, France) and expressed in U/mL; vWF was determined by immunoturbidimetric assay (STA LIATEST vWF, Diagnostica Stago, Asnières sur Seine, France) and expressed as percentage.
The concentration of MMP-10 was determined in serum by enzyme-linked immunosorbent assay (R&D Systems, Minneapolis, MN, USA) and expressed in pg/mL. Coefficients of variations for all immunological assays were <8%.
Statistical Analysis
Statistical analysis was performed with SPSS for Windows version 15.0. Quantitative variables with normal distribution were described as mean and standard deviation and compared with unpaired t test. Quantitative variables not normally distributed were described with median and interquartile range and compared with Mann-Whitney test. Nominal variables were compared with Fisher exact test or chi-square test. The correlations between variables were analyzed with Spearmann test. The differences were considered statistically significant with double tailed P < .05.
Results
The patient’s group included 17 men and 24 women (mean age 55.6 ± 13.4 years). The control group consisted of 13 men and 17 women (mean age 55.7 ± 2.1 years). There were no statistically significant differences between the study group and the control group with regard to age and sex, P = .96 and P = .84, respectively.
The median values of the analyzed parameters are shown in Table 1. The group of patients showed a significant increase in DD (P < .001), F1 +2 (P < .001), PAI-1 (P < .001), vWF (P = .027), and MMP-10 (P < .001) as compared with controls.
Markers of Hemostatic Activation and MMP-10 in Patients and Controls.
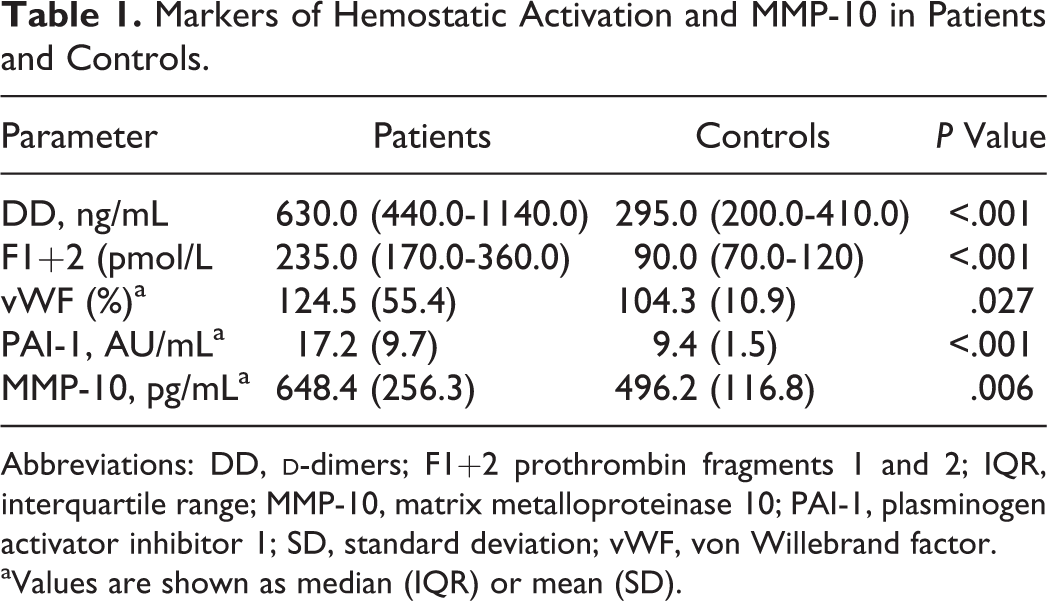
Abbreviations: DD,
aValues are shown as median (IQR) or mean (SD).
Univariate analysis showed that there was a positive, moderate correlation between concentrations of DD and F1+2 (r = .33, P = .04) and also a significant positive correlation was found between serum levels of DD and vWF (r = .34, P = .03; Figure 1). There was no correlation between circulating levels of MMP-10 and DD, F1 +2, PAI-1, and vWF. In patients with PVVs, there was a positive correlation between age and DD (r = .33, P = .035) and age and vWF (r = .32, P = .04).
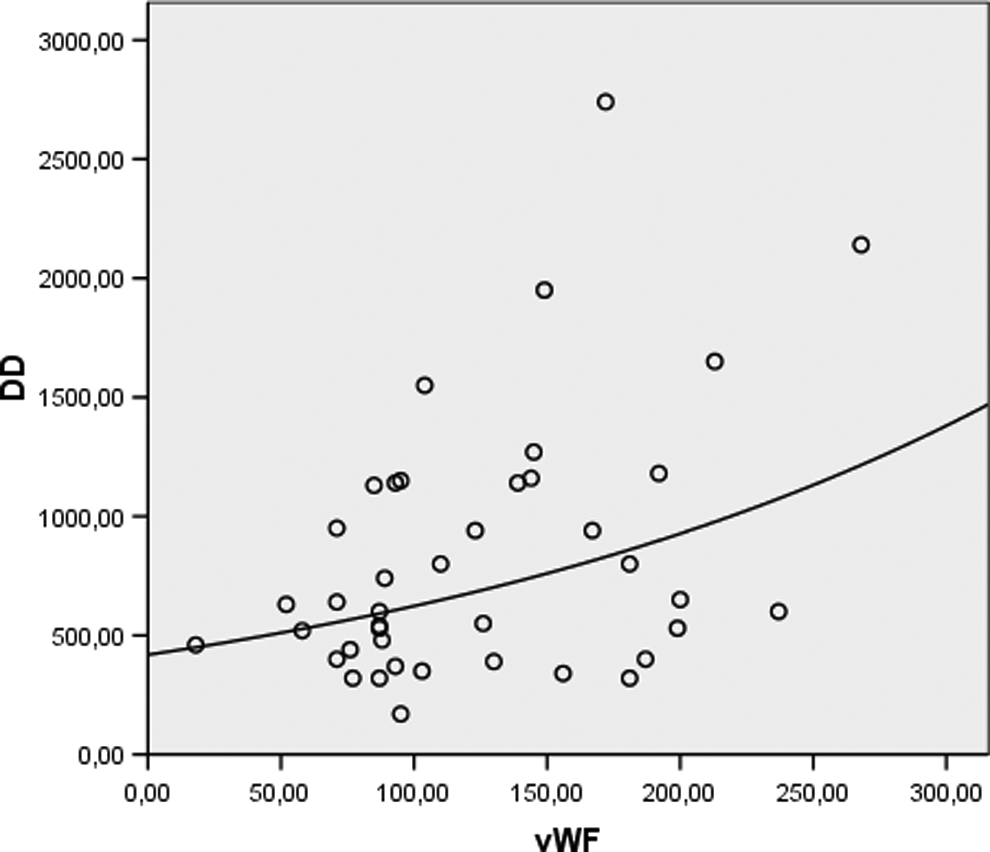
Correlation between plasma levels of
There were no statistical significant differences in the levels of analyzed parameters according to the CEAP clinical stage.
Discussion
This study has objectively demonstrated a prothrombotic activation of hemostasis and a proinflammatory stage in patients with PVVs, without any ongoing thrombotic or inflammatory process. An important advantage of this study is the fact that it has analyzed a homogenous group of patients with PVVs due to saphenous vein insufficiency. This is of vital importance in view of existence of different definitions of varicose veins. In addition, patients enrolled did not have any ongoing inflammatory or significant comorbid conditions that could affect the concentration of analyzed markers.
Elevated values of DD and F1+2 concentrations are associated with an increased risk of developing DVT. That association was observed not only in patients undergoing total hip replacement, chemotherapy, insertion of central venous catheter, or after discontinuation of anticoagulant therapy after DVT 15 –18 and also in a general population. 19 In our study, patients with PVVs showed increased PAI-1 activity. It has been shown that elevated PAI-1 levels may be associated with an increased risk of deep vein thrombosis and metabolic syndrome. 20 –22 Taken together, elevated DD and F1 +2 concentrations as well as elevated PAI-1 activity in patients with PVVs would reflect a hypercoagulable state associated with an increased risk of venous thromboembolism.
We show for the first time that the level of MMP-10, a metalloproteinases that is significantly expressed after proinflammatory/prothrombotic conditions, 10 –12 was significantly expressed in patients with PVVs. A role of this and other MMPs in the pathogenesis of PVVs cannot be excluded. 7
A positive correlation between DD and F1+2 and vWF seems to suggest the association of hemostatic activation and endothelial damage in PVVs. The vWF is a marker of endothelial damage that correlated with the severity of venous insufficiency in another study. 23 The lack of correlation between hemostatic parameters and MMP-10 would suggest additional mechanisms involved in the inflammatory response in PVVs.
The observed prothrombotic activation of hemostasis in patients with PVVs may have clinical implications. The reported incidence of DVT after varicose veins surgery is 5.3%. 24 Our results suggest that it would be possible to identify subgroups of patients with PVVs requiring thromboprophylaxis on the basis of their prothrombotic/proinflammatory profile.
The cause of that hypercoagulable state in patients with PVVs is another interesting issue. A correlation between serum DD and vWF may suggest the relationship between systemic activation of coagulation and endothelial damage due to venous hypertension. Positive correlations between age and DD and vWF may further support this hypothesis since it has been shown that the severity of chronic venous insufficiency increases with the age of the patients. 25 A slow blood flow in the varicose veins may also contribute to the formation of mural microthrombi. The immunohistochemical studies showed signs of prothrombotic hemostatic activation in the endothelium and deeper layers of the varicose veins. 26
Due to a wide range of age of the patients, they might have had some degree of arterial damage that could be responsible for observed abnormalities. However, all the patients with symptomatic coronary or peripheral arterial disease had been excluded from the study, so we believe that the observed prothrombotic and proinflammatory state can be attributed to the presence of varicose veins.
We conclude that noncomplicated PVVs are associated with markers of hemostatic activation and elevated concentration of MMP-10, which may have pathophysiological and therapeutic implications.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
