Abstract
Introduction:
Antiphospholipid antibodies (APLAs) have been variably reported in 14% to 75% of patients with immune thrombocytopenia (ITP). There is lack of Indian data on incidence of APLA in ITP.
Objective:
We studied the incidence of APLA in patients with pediatric and adult Indian ITP.
Materials and Methods:
We prospectively studied 100 patients including acute (n = 37), persistent (n = 13), and chronic (n = 50) ITP. Male to female ratio was 1.22:1. Median age was 18 years (1.5-56). All patients underwent investigations for lupus anticoagulant (LA), anticardiolipin (aCL) immunoglobulin G (IgG) and IgM antibodies, and anti-β2 glycoprotein 1 (β2GP1) IgG and IgM antibodies. Patients with secondary ITP were excluded. Bleeding manifestations were recorded. Patients with acute and persistent ITP were assessed for steroid response. Response rates were compared between APLA-positive and APLA-negative patients.
Results:
Antiphospholipid antibodies were detected in ∼12% of patients with ITP: 8.1% (3 of 37) in acute, 0% (0 of 13) in persistent, and 18% (9 of 50) in chronic ITP. Anti-β2GP1 antibodies were most frequent (9%). Only 2 patients each were positive for anti-aCL antibodies and LA. Although platelet counts were significantly higher in APLA-positive patients, there was no significant difference in bleeding between the APLA-positive versus APLA-negative patients with ITP. There was also no significant difference in steroid response between APLA-positive and APLA-negative patients with acute/persistent ITP. In the short follow-up (median 8 months), none of the APLA-positive patients developed thrombosis.
Conclusions:
Incidence of APLA in Indian population was lower than reported in the West, which indicates that not all patients of ITP need to be subjected to these manifestations upfront at diagnosis.
Introduction
Immune thrombocytopenia (ITP) is an acquired immune-mediated disorder characterized by isolated thrombocytopenia (defined as a platelet count less than 100 × 109/L) in the absence of any obvious initiating and/or underlying cause. 1 It is also classified into primary ITP where no obvious cause can be demonstrated and secondary ITP that may be due to autoimmune disorders, lymphoproliferative diseases, drug related, or secondary to various infections. Antiphospholipid syndrome (APS) is recognized as an important cause of secondary ITP. Thrombocytopenia is found in 20% to 40% of patients with APS 2 but is not included in the diagnostic criteria for APS. 3
Antiphospholipid antibodies (APLAs) have been demonstrated in some cases of ITP. These patients may not fulfill the clinical criteria for APS at the time of detection of APLA. Harris et al first demonstrated the presence of APLA in 30% of patients with ITP from United Kingdom. 4 The incidence of antibodies, however, is reported from ∼14% to 75%. 5 –9 The reported incidence from France is ∼26%, with most patients positive for anti-β2 glycoprotein 1 (β2GP1) antibodies. 2 Stasi et al reported an incidence of 46.3% in 149 newly diagnosed Italian patients, with ∼36.2% of patients positive for lupus anticoagulant (LA). 5 A very high incidence of APLA in ITP has been reported from the United States. 7 Indian data regarding the presence of APLA are minimal with a single article by Dash et al 10 who found 27.5% of pediatric patients with ITP had presence of LA. Data on anticardiolipin (aCL) antibodies and anti-β2GP1 antibodies have not been reported from India.
The present study aimed at studying the prevalence of APLA in ITP and correlating it with bleeding symptoms, response to steroid therapy, and development of thrombosis.
Materials and Methods
Study Population
This prospective study was performed at a tertiary care center in North India from September 2011 to June 2013. Ethical clearance was obtained from the institutional ethics committee, and patients were enrolled after taking written informed consent. A total of 100 cases of ITP fulfilling the following inclusion and exclusion criteria were enrolled.
Inclusion Criteria
All confirmed cases of ITP, regardless of age, sex, and duration of symptoms were enrolled. Immune thrombocytopenia was defined as platelet count <100 × 109/L in the absence of other causes or disorders that may be associated with thrombocytopenia. 1 Only those patients were included who consented to undergo tests for APLA-LA, aCL (IgG) and aCL (IgM) antibodies, and anti-β2GP1(IgG) and β2GP1 (IgM)antibodies.
Exclusion Criteria
All patients who had an identifiable primary cause of thrombocytopenia were excluded. Cases positive for any of the viral markers (HIV, hepatitis B surface antigen [HBsAg], and hepatitis C virus [HCV]), antinuclear antibodies (ANAs), direct Coomb test (DCT), history of thrombosis, or APS were excluded. Patients who had received corticosteroids in the past 1 month at an adequate dose were also excluded. The patients were not tested for Helicobacter pylori unless they had suggestive clinical manifestations such as dyspepsia. Since none of the patients reported any clinical manifestations, they were not tested for the same.
Based on these exclusion criteria, 31 patients of a total of 131 patients were excluded. Since our hospital is a tertiary care center, we had to exclude 27 patients due to corticosteroid use. These were patients with acute ITP who had been diagnosed in the previous 1 month of reporting to our hospital and received corticosteroid therapy in any dosage for some or most part of this 1 month. Two patients were excluded due to a positive ANA, one each due to a positive DCT (Evan syndrome) and HCV positivity.
Baseline Evaluation
A detailed history addressing presence of bleeding symptoms, severity of bleeding, and requirement of any transfusions due to increased bleeding was taken. The bleeding symptoms recorded were—skin bleeds including purpura, petechiae, ecchymosis, epistaxis, oral bleeds, menorrhagia, gastrointestinal bleeds, hematuria, and hemoptysis. The severity of bleeding manifestations was recorded as per the World Health Organization (WHO) bleeding scale. 11 Routine tests for ITP, such as hemogram and biochemistry (urea, creatinine, sodium, potassium, and liver function test), viral markers (HBsAg, anti-HCV, and HIV), ANA, and anti-double-stranded DNA, were done in all cases. All patients underwent a bone marrow aspiration and trephine biopsy before starting therapy. Even though the guidelines do not recommend bone marrow examination for the diagnosis of ITP, it is part of the departmental protocol in order to rule out acute leukemia or aplastic anemia. This policy has been adopted by our department as some patients with partially treated acute leukemia may have a similar blood picture as ITP. Immune thrombocytopenia was subclassified into newly diagnosed, persistent, and chronic ITP as per the International Working Group definitions. 1
A total of 100 patients were enrolled in the present study including 37 newly diagnosed, 13 persistent, and 50 patients with chronic ITP. The median age of these 100 patients was 18 years (1.5-56 years). The male to female ratio was 1.22:1 (55 males and 45 females).
Lupus anticoagulant was tested using 2 techniques: Kaolin clotting time (KCT). It was a manual test, and index of circulating anticoagulant was calculated. 11 A value of >16% was taken as positive for LA. Dilute Russell Viper venom time (DRVVT) using the Diagnostica Stago commercial kit (STA Staclot DRVV LA assays, Parsippany, New Jersey), and results were interpreted according to the kit insert. A patient was considered LA positive when either KCT or DRVVT or both were positive.
Anticardiolipin antibodies (aCL [IgG] and aCL [IgM]) were tested using the enzyme-linked immunosorbent assay (ELISA) manufactured by Akesulia Cardiolipin-GM test kit (Aesku. Lab Diagnostika, Germany). A cutoff value more than 15 GPL/MPL units for IgG and IgM antibodies, respectively, was used to determine positivity.
Anti-β2GP1 IgG and IgM antibodies were tested using an ELISA capable of detecting IgG and/or IgM antibodies against β2GP1 manufactured by Generic Assays (GA Generic Assays GmbH, Dahlewitz, Germany). A cutoff value ≥10 U/mL was taken as a positive result.
A group of 150 healthy controls was analyzed using the same kits and tests. None (0%) of the 150 patients were positive for LA. Two (1.33%) of the 150 controls were positive anti-β2GP1 antibodies. Two (1.33%) of the 150 controls were positive for aCL antibodies, both were IgG subtypes. None of the patients tested positive for aCL IgM. Using the same criteria, a cohort of 32 patients with systemic lupus erythematosus (SLE) who underwent all these tests in 2010 showed a 12.5% of incidence of APLA.
Therapy
Patients with newly diagnosed and persistent ITP were given 1 mg/kg dose of prednisolone for 2 weeks followed by tapering of dosage over next 2 to 4 weeks. Patients with chronic ITP were given dapsone 50 to 100 mg/d after ascertaining glucose-6-phosphate dehydrogenase status.
Response Assessment
Response to steroid therapy was recorded as per the International Working Group definition. 1 The patients were classified into categories of complete response, response, and no response.
Only patients with newly diagnosed and persistent ITP who received only steroids were included for assessment of response status. Patients who received intravenous Ig or anti-D were excluded. The median follow-up was 5 months (2-12 months) for acute ITP, 8 months (3-11 months) for persistent ITP, and 9 months (3-21 months) for chronic ITP.
Statistical Analysis
For statistical analysis, newly diagnosed and persistent ITP were clubbed together. For the analysis of categorical/quantitative variables, Fisher exact test or continuity correction was used. For the analysis of continuous variables, Mann-Whitney test was used. A P value of <.05 was taken as significant.
Results
The median platelet count was 13.5 × 109/L (2-77 × 109/L). Most patients presented with various bleeding manifestations. The patients presented with skin bleeds, oral bleeds, epistaxis, gastrointestinal bleeding, hemoptysis, and hematuria. Females, in addition, had menorrhagia as a common presenting complaint. The most common presenting symptom was skin bleeds seen in 64 patients followed by epistaxis in 33 patients and oral bleeds in 22 patients. Hematuria and hemoptysis were uncommon and were seen in 2 and 1 patients, respectively. Of the 45 female patients evaluated, menorrhagia was seen in 20 (44.44%) patients and was the second most common symptom after skin bleeds. Only 11 patients did not have any bleeds at presentation. The WHO bleeding scale was used to assess the severity of bleeding. 12 The median bleeding scale was 2. Fifty-seven patients had a bleeding scale 2, whereas only 13 patients had a bleeding scale of 1. Severe bleeds, that is, WHO scales 3 and 4 were seen in 18 and 1 patients, respectively.
There was no statistical difference in mean platelet counts between patients who bled or had no bleeding in patients with newly diagnosed/persistent ITP (15.5 × 109/L vs 16.1 × 109/L, P = .99) and in chronic ITP (24.71 × 109/L vs 19.71 × 109/L, P = 1.00).
The patient characteristics and difference between chronic ITP and newly diagnosed and persistent ITP are shown in Table 1. There was no significant association between ITP subtype and sex, skin bleeds, epistaxis, gastrointestinal bleeding, oral bleeds, menorrhagia, and bleeding scale. However, the platelet count in patients with chronic ITP was significantly higher than platelet count in patients with newly diagnosed and persistent ITP. There was also no significant difference between age of patients and ITP subtypes.
The Demographic Characteristics and Difference Between ITP Subtypes.
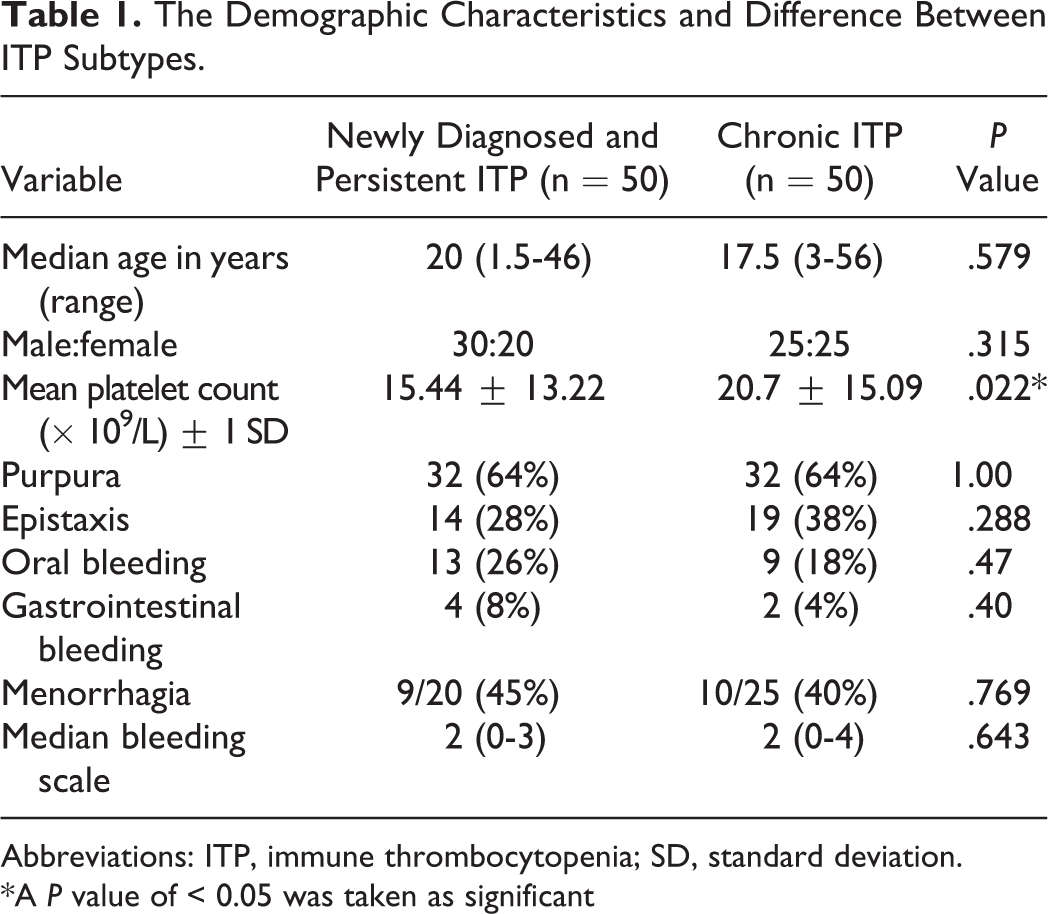
Abbreviations: ITP, immune thrombocytopenia; SD, standard deviation.
*A P value of < 0.05 was taken as significant
The APLAs evaluated were LA, aCL (IgG and IgM), and β2GP1 (IgG and IgM). Overall, of the 100 patients, 12 (12%) were positive for any APLA. The subtypes of APLA and their distribution are presented in Table 2. All patients with persistent ITP were negative for APLA. Only 1 patient with newly diagnosed ITP was positive for both β2GP1 and aCL (IgM). When the incidence of APLA was compared between healthy controls and patients with ITP, it was seen that only the incidence of β2GP1 antibodies was higher in patients with ITP (9 of 100 vs 2 of 150, P = .008). There was no difference between incidence of LA and aCL antibodies between healthy controls and patients with ITP studied. A comparison of APLA-positive and APLA-negative patients is presented in Table 3. There was no correlation between age, sex, bleeding symptoms, and bleeding score between APLA-positive and APLA-negative patients. It was observed that initial platelet counts in APLA-positive patients were significantly higher than APLA-negative patients. Since β2GP1 antibodies were the most commonly detected, a comparison between positive and negative patients was performed and is presented in Table 3. Only the platelet count was higher in β2GP1-positive patients with ITP.
The Various Subtypes of APLA Detected in 100 Patients With ITP.
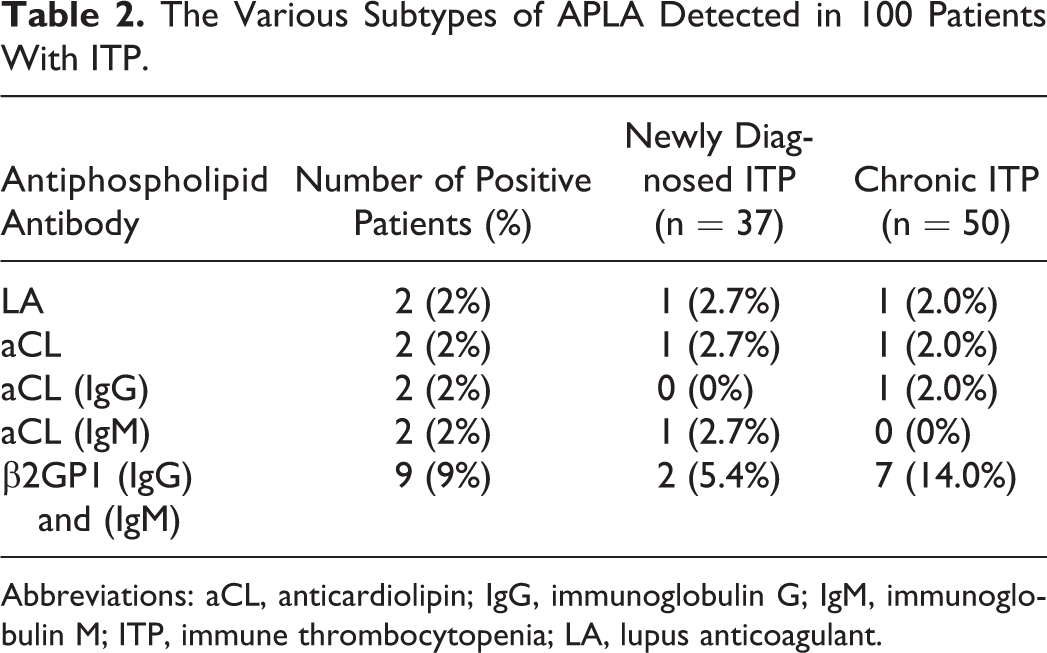
Abbreviations: aCL, anticardiolipin; IgG, immunoglobulin G; IgM, immunoglobulin M; ITP, immune thrombocytopenia; LA, lupus anticoagulant.
A Comparison of Clinical Characteristics of Antiphospholipid Positive and Negative Patients.
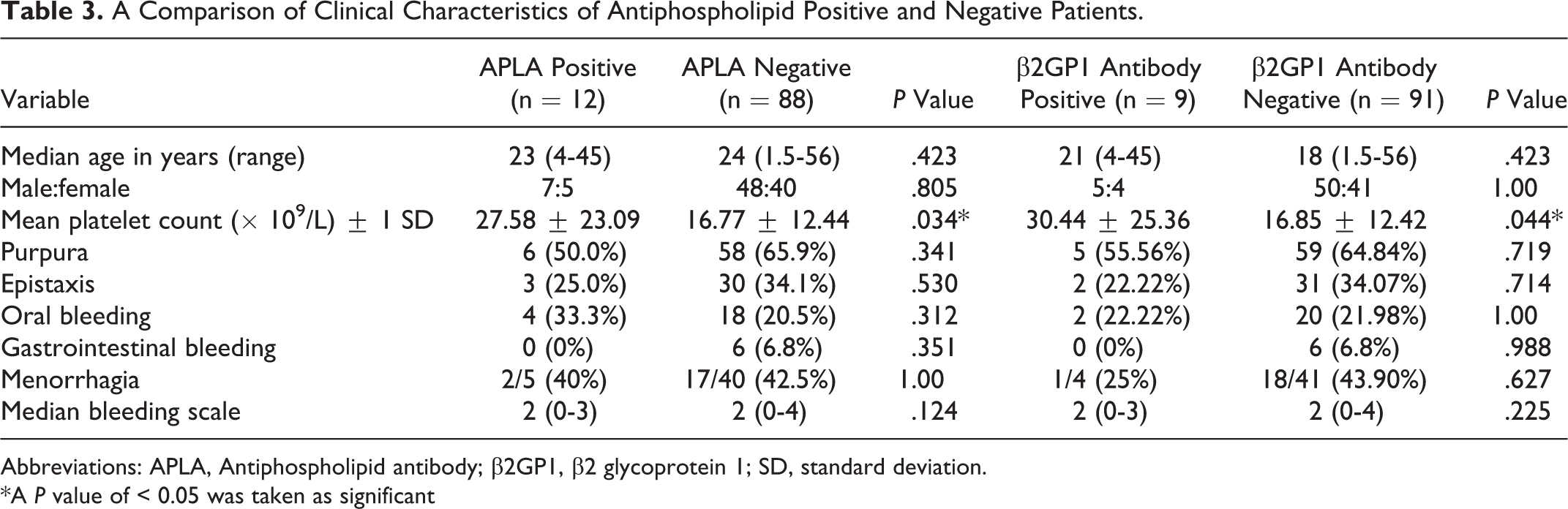
Abbreviations: APLA, Antiphospholipid antibody; β2GP1, β2 glycoprotein 1; SD, standard deviation.
*A P value of < 0.05 was taken as significant
The incidence of APLA in newly diagnosed ITP, persistent ITP, and chronic ITP was 8.1% (3 of 37), 0% (0 of 13), and 18% (9 of 50), respectively. When cases with newly diagnosed ITP and cases with persistent ITP were combined, the incidence of APLA was 6% (3 of 50). On comparison between newly diagnosed and persistent ITP with patients having chronic ITP, a trend was observed toward a higher occurrence of APLA with chronic ITP (P = .065). A comparison between APLA-positive and APLA-negative patients with chronic ITP did not reveal any significant differences with respect to demographic variables, platelet count, and bleeding symptoms.
Response to Steroid Therapy in Newly Diagnosed and Persistent ITP
Of the 50 cases, response to steroid therapy could be assessed in 49 patients. One case with newly diagnosed ITP had received intravenous Ig therapy and was excluded. The response rates in newly diagnosed and persistent ITP are given in Table 4. It was observed that the rate of complete response with steroid therapy was higher in newly diagnosed ITP in comparison to patients with persistent ITP (P = .008). However, there was no difference between the rate of response to steroid therapy when APLA-positive patients with newly diagnosed and persistent ITP were compared to those who were APLA negative (P = .667).
Responses in Patients With Newly Diagnosed and Persistent ITP.
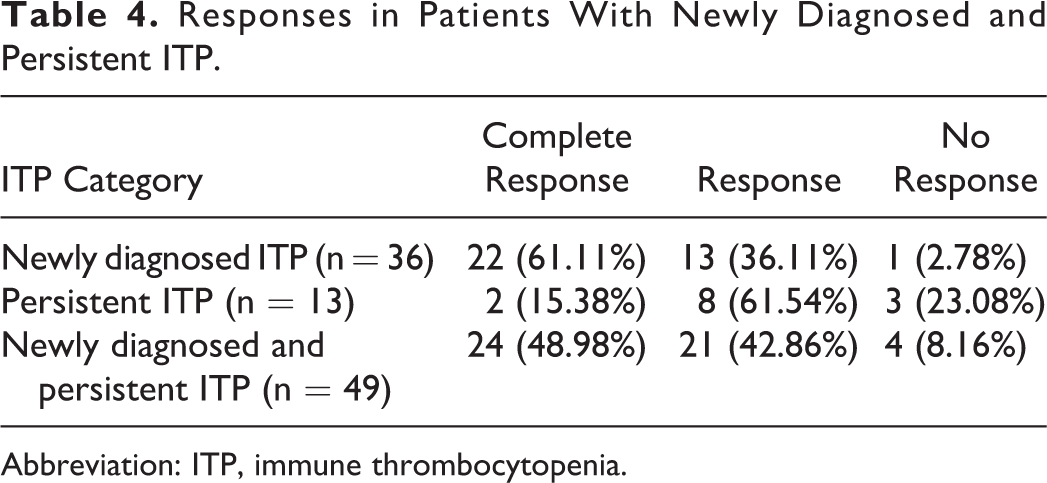
Abbreviation: ITP, immune thrombocytopenia.
Discussion
In our study of 100 patients with ITP, the median platelet count was significantly higher in patients with chronic ITP as compared to newly diagnosed and persistent ITP. This finding corroborates with a prior study in ITP, which also showed that patients with chronic ITP present with a higher platelet count than cases with newly diagnosed ITP. 13
We studied the incidence of APLA including LA, aCL (IgG and IgM), and anti-β2GP1 (IgG and IgM) antibodies in patients with ITP. Various studies in the existing literature have reported an incidence of 14% to 76% in patients with ITP. 2,5,14 –17 We obtained an incidence of 12% for occurrence of any APLA in patients with ITP. This incidence was lower than that seen in the studies by Harris et al (31.3%), 4 Lipp et al (73%),14 Bidot et al (66.7%), 15 Despujol et al (26%), 2 Yang et al (28.5%), 17 and El-Bostany et al (76.1%). 18 However, it was almost similar to the incidence documented in the study by Dasanu and Codreanu (14%). 16 Lower positivity rate seen in the present study may be attributed to ethnicity and variable assessment techniques. Another reason for a lower incidence of APLA is due to the only inclusion of ANA-negative cases and exclusion of all patients with criteria proven APS or any coexisting autoimmune disorders. A few prior studies have included ANA-positive patients 9,18 and even patients with a positive DCT.19 It is worth noting that in the study that reported the highest incidence of APLA, 7 of 42 patients positive for APLA subsequently were diagnosed as SLE. 18 Also, a few authors have studied APLA against other phospholipid antigens such as phosphatidyl serine, phospatidyl choline, phosphatidyl ethanolamine, and factor VII/VIIa, 15 ,19 which have not been tested in the present study and may also account for a lower incidence. However, these antibodies also do not form a part of the diagnostic criteria laid down for the diagnosis of APS and hence are not widely tested in routine practice. 3
The most common APLA detected was anti-β2GP1 in 9% of patients with ITP, and this was significantly higher than controls. Detection of anti-β2GP1 antibodies was made mandatory in the testing for APS only in 2006, and hence, only a few studies have reported the incidence of these antibodies. 2,15,20 The incidence seen in the present study is lower than the 17.5% to 26% reported in the literature. 2,15,20 The incidence of aCL antibodies in the present study (2%) is comparable to that detected by Sakai et al (0%) 19 but is lower than the 25% to 47.6% reported in other studies. 2,15 Lupus anticoagulant was also detected in only 2% of patients. Only a few studies have studied LA in ITP and have found variable rates between 0% to 27.5%. 2,10,15 It is also interesting to note that for both aCL antibodies and LA, the incidence observed in patients with ITP was not different from the healthy controls.
In the short study duration, we did not observe any thrombotic events in these cases with ITP, and hence, a comment on the association of APLA with thrombosis in ITP cannot be made in the present study. The fact that none of the patients developed thrombosis may be due to the short follow-up (median: 8 months), a lower incidence of APLA than previously reported, or may be due to the lack of many dual- or triple-positive patients as only 1 case showed a dual positivity for anti-β2GP1 and aCL while none of the cases were triple positive. Another reason may be the very low incidence of LA in the present study as LA has been associated with a higher risk of thrombosis in antiphospholipid positive patients with ITP.20
A trend was observed toward a higher occurrence of APLA positivity in chronic ITP, but statistical significance was not reached (P = .065). None of the other published cases have compared the incidence of APLA in newly diagnosed and chronic ITP. In patients with newly diagnosed and persistent ITP, APLA positivity was seen in 6%. Only in a Japanese study, it was seen that 14% of patients with newly diagnosed ITP were positive for an APLA. 19 Other studies have reported a much higher incidence of APLA in newly diagnosed ITP ranging from 27% to 46%. 5,8,10,18 The only Indian study to report data on APLA was done on patients with newly diagnosed pediatric ITP 10 and reported an incidence of 27.5% LA positivity while other antibodies were not tested. The incidence of APLA in chronic ITP was 18%. This is similar to the 18.8% observed by Sakai et al. 19 Both lower and higher incidence have been observed. Kaburaki et al 21 have reported the lowest incidence of these antibodies in ITP at 7.2%. Other authors have reported rates between 25% and 100%. 6,9,18 A 100% rate was reported by El-Bostany et al in pediatric patients with chronic ITP. 18 Cases with newly diagnosed ITP showed a significantly higher complete response rate than cases with persistent ITP. When these 2 subtypes of ITP were combined, it was seen that there was no statistically significant association seen between APLA-positive and APLA-negative patients with a degree of response.
Conclusion
Antiphospholipid-positive patients with ITP do not differ significantly from APLA-negative patients with respect to demographics and bleeding characteristics. Only platelet counts tend to be higher in APLA-positive versus APLA-negative patients. The incidence obtained in the present study is lower than that reported in the Western literature. There was no impact of APLA positivity on thrombosis during the short follow-up in the present study. However, a larger study with a longer follow-up is required to ascertain the impact of APLA in thrombosis in ITP in the Indian population and not all patients with ITP need to undergo testing for APLA at diagnosis if other autoimmune causes have been excluded.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
