Abstract
Objectives:
To investigate whether LEP G2548A and LEPR Q223R polymorphisms influence serum lipid levels and whether the 2 polymorphisms affect the efficacy of simvastatin treatment in Chinese patients with primary hyperlipidemia.
Methods:
We used an extreme sampling approach by selecting 212 individuals from the top and bottom 15% of adjusted lipid-lowering response residuals to simvastatin (n = 106 in each group of good or bad response) from a total of 734 samples with primary hyperlipidemia. They were treated with simvastatin orally 20 mg/d. Fasting serum lipids were measured at baseline and after 4 and 8 weeks of treatment. Genotyping was carried out using polymerase chain reaction-restriction fragment length polymorphism.
Results:
More patients in the good response group (27%) had LEPR Q223R than in the bad response group (16%, P = .046). Secondary stratified analyses showed that patients carrying the RR genotype of the LEPR Q223R gene had significantly higher high-density lipoprotein cholesterol levels than those with the QR genotype at baseline (P = .034) among good responders. After 29 consecutive days of treatment with simvastatin, patients carrying the RR genotype had a significantly larger decrease in triglycerides (change: −0.74 ± 0.92, P = .036) and total cholesterol levels (change: −1.77 ± 0.68, P = .023) compared with those carrying QR genotype among bad responders. After Bonferroni correction, the results were not statistically significant.
Conclusion:
LEPR Q223R polymorphism, but not LEP G2548A, could modulate the efficacy of simvastatin in Chinese patients with primary hyperlipidemia.
Introduction
Cardiovascular disease (CVD) is a leading cause of death worldwide. Hyperlipidemia is one of the traditional risk factors for CVD. 1 High cholesterol levels have been associated with CVD. 2 Statins, as the first-line lipid-lowering drug, have been widely used in clinical practice.
3-Hydroxy-3-methylglutaryl-coenzyme A (HMG-CoA) reductase inhibitors, known as statins, are potent inhibitors of cholesterol biosynthesis that are the most widely used cholesterol-lowering drugs to treat patients with hypercholesterolemia. 3,4 Statins interrupt cholesterol biosynthesis by inhibiting activity of enzyme HMG-CoA reductase, the rate-limiting step in cholesterol synthesis. And the inhibition of cholesterol synthesis leads to increased levels of low-density lipoprotein (LDL) receptors, 5 which results in increased uptake and degradation of low-density lipoprotein cholesterol (LDL-C), a reduction in cholesterol accumulation and esterification, and decreased lipoprotein secretion and cholesterol synthesis. 6
Although a large body of studies confirmed that statins can significantly decrease total cholesterol (TC) and LDL-C as well as weakly increase high-density lipoprotein cholesterol (HDL-C), the lipid-lowering effect of statins among individuals exhibited considerable variation. In addition, statins have side effects, including muscle pain, 7 increased risk of diabetes mellitus, and abnormalities in liver enzyme tests. 8 Previously, numerous studies demonstrated that genetic polymorphisms of drug-metabolizing enzymes, transport proteins, and receptors could contribute to the interindividual variability in the response to statins. 9
Leptin may have a regulatory role in cholesterol metabolism. 10 Leptin has been shown to downregulate hepatic activity of HMG-CoA reductase and upregulate activities of both sterol 27-hydroxylase and cholesterol 7α-hydroxylase, which cause a decrease in plasma very low-density lipoprotein cholesterol (VLDL-C) concentrations. 11 Moreover, leptin has also been shown to phosphorylate and activate adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK) via central and peripheral mechanisms. 12 AMP-activated protein kinase is a protein kinase that phosphorylates and inactivates HMG-CoA, therefore, it helps inhibit cholesterol synthesis, triglyceride syntheses, adipocytes lipolysis, and lipogenesis. 13
The LEP gene, located at chromosome 7q31.3 and consisting of 3 exons separated by 2 introns, encodes a 16-kDa protein that has been consistently shown to be associated with endocrinologic metabolism. 14 Leptin exerts its physiological action by binding and activating its receptor (LEPR, a single transmembrane protein distributed in many types of tissues with gene located at chromosome 1p31) in the hypothalamus, 15 with pathways involving janus kinase (JAK)/signal transducer and activator of transcription (STAT), phosphatidylinositol 3-kinase (PI3K), and mitogen-activated protein kinase (MAPK)/extracellular signal-regulated kinase (ERK) systems and protein kinase C (PKC). 16 –18
LEP and LEPR genes are highly polymorphic, and a number of single-nucleotide polymorphisms (SNPs) have been identified in these 2 genes. 19,20 For example, there are at least 2627 reported SNPs in the LEP gene region and 20 010 reported SNPs in the LEPR gene region (http://www.ncbi.nlm.nih.gov/snp, update September/08/2015). However, only a few of these reported SNPs are potentially functional. G2548A is one of the SNPs in the LEP gene, consisting of G to A substitution at nucleotide −2548 upstream from the ATG start site, which has been reported to be associated with lipid levels. 21 Q223R is another SNP of the LEPR gene. It is located within the region encoding the extracellular domain of the leptin receptor and results in the change of glutamine to arginine at position 223 of the LEPR protein. Q223R polymorphism is associated with a decrease in leptin-binding activity resulting in leptin resistance. 22 The Q223R polymorphism was also found to be associated with lipid profiles. 15
Our present study aimed to investigate the effect of LEP G2548A and LEPR Q223R polymorphisms on lipid levels and further investigated the influence of the polymorphisms on lipid-lowering response to simvastatin in Chinese patients with primary hyperlipidemia.
Patients and Methods
Study Population
Totally, 734 patients with primary hyperlipidemia were recruited for the study from Beijing and Anhui, China. In the present study, we used an extreme sampling approach by selecting 220 individuals (212 remained after excluding 8 with unreliable genotyping information) from the top and bottom 15% of lipid-lowering response residuals to simvastatin (n = 106 in each group of good or bad response) from all samples and adjusted for age and gender. We screened eligible patients with hyperlipidemia through medical history, physical examination, and clinical laboratory evaluation, including the lipid profile. No participant had any of symptomatic ischemic heart disease or any other vascular diseases. Any lipid-lowering drugs had to have been discontinued for at least 4 weeks. All participants gave informed consent, and the study protocol was approved by the ethics committee of the Institute of Biomedicine, Anhui Medical University.
Participants who met the following criteria of fasting serum lipid levels were recruited as having primary hyperlipidemia: TC: 5.72 to 8.32 mmol/L and/or LDL: 3.64 to 6.50 mmol/L and TG ≥ 1.70 mmol/L. 23,24 To avoid potential side effects, patients with any of these characteristics were excluded: (1) impaired hepatic function (aminotransferase levels > 2 × normal and history of chronic liver disease, such as cirrhosis or alcohol abuse), (2) impaired renal function (serum creatinine levels >1.8 mg/dL and/or a history of chronic renal disease, such as glomerulonephritis, chronic pyelonephritis, obstructive renal disease, or proteinuria), (3) diabetes mellitus (fasting blood glucose >126 mg/dL), (4) raised thyroid-stimulating hormone levels (>5.0 μU/L), and (5) any medical conditions that might preclude successful completion.
Simvastatin Treatment
After a washout period of 7 to 10 days, all participants were treated orally with a fixed dosage of simvastatin 20 mg/d for 8 consecutive weeks. Participants were required to take drugs between 8
Laboratory Determinations
Venous blood samples were drawn between 8
Genotyping of LEP G2548A and LEPR Q223R
DNA was extracted from EDTA-treated whole blood and stored at −20°C. Genotyping of LEP G2548A (rs7799039) and LEPR Q223R (rs1137101) was carried out using the polymerase chain reaction-restriction fragment length polymorphism (PCR-RFLP) assay. Their primer pairs for PCR amplification were as follows: forward: 5′-TTT CCT GTA ATT TTC CCG TGA G-3′, reverse: 5′-AAA GCA AAG ACA GGC ATA AAA A-3′ for LEP G2548A gene; forward: 5′-ACC TCT GGT TCC CCA AAA AG-3′, reverse: 5′-TCA TCA TTT TAG TGC ATA ACT TAC CC-3′ for LEPR Q223R gene. Polymerase chain reaction amplification was carried out in 25 μL PCR mixture in a PCR amplifier (long gene) as follows: an initial denaturation at 95°C for 60 seconds, followed by 35 cycles of denaturation at 95°C for 30 seconds, annealing at 56°C for 45 seconds, and extension at 72°C for 30 seconds, then a final extension at 72°C for 7 minutes for LEP G2548A gene; an initial denaturation at 94°C for 2 minutes, followed by 35 cycles of denaturation at 94°C for 30 seconds, annealing at 56°C for 45 seconds, and extension at 72°C for 45 seconds, then a final extension at 72°C for 6 minutes for LEPR Q223R gene. After PCR amplification, 15 μL was digested with restriction enzymes, HhaI 25 for LEP, 37°C for 4 hours, and MspI 25 for LEPR, 37°C for 5 hours. Then, the digestion products were separated on 3% agarose gels stained with ethidium bromide. The 242 base pair (bp) fragment was divided into 242, 181, and 61 bp; the 212-bp fragment was divided into 212, 151, and 61 bp. Ten percent of all the samples were selected for replication with 100% concordance.
Statistical Analysis
Mean ± standard deviation (SD) was calculated for continuous variables. The χ2 test was used for categorical variables. Paired Student t test was used to compare lipid levels before and after simvastatin treatment. Multiple linear regression analysis was used to estimate the associations between LEP and LEPR polymorphisms and serum lipid levels at baseline and lipid response at 8 weeks after simvastatin treatment. Lipid response was defined as serum lipid levels after treatment − serum lipid levels at baseline. Bonferroni correction was used for multiple testing corrections. We adjusted for age and gender in regression models to get the residuals that we used for selecting top and bottom 15% of response to treatment. All statistical analyses were performed with IBM SPSS software package (version 19.0 for windows; IBM Inc, Armonk, New York). P < .05 was considered to be significant.
Results
Baseline Characteristics of Patients
A total of 212 patients with complete genotyping information and primary hyperlipidemia at baseline and prescribed simvastatin were selected based on good or bad response to treatment. The clinical and epidemiologic characteristics of the population at baseline grouped by simvastatin treatment response are shown in Table 1. The bad response group had markedly higher HDL-C levels (1.84 ± 0.66 mmol/L) than the good group (1.48 ± 0.36 mmol/L; P < .001). In contrast, significantly higher levels of body mass index (BMI), TC, and LDL-C were observed in the good response group than in the bad group. The distribution of occupation was significantly different between the 2 groups (P = .001). There were no significant differences in age, waist circumference (WC), systolic blood pressure, diastolic blood pressure, or TG between the 2 groups. The distributions of sex, smoking, and drinking within the 2 groups also had no statistically significant differences.
Baseline Clinical and Epidemiologic Characteristics of Samples Grouped by Treatment Response.a,b
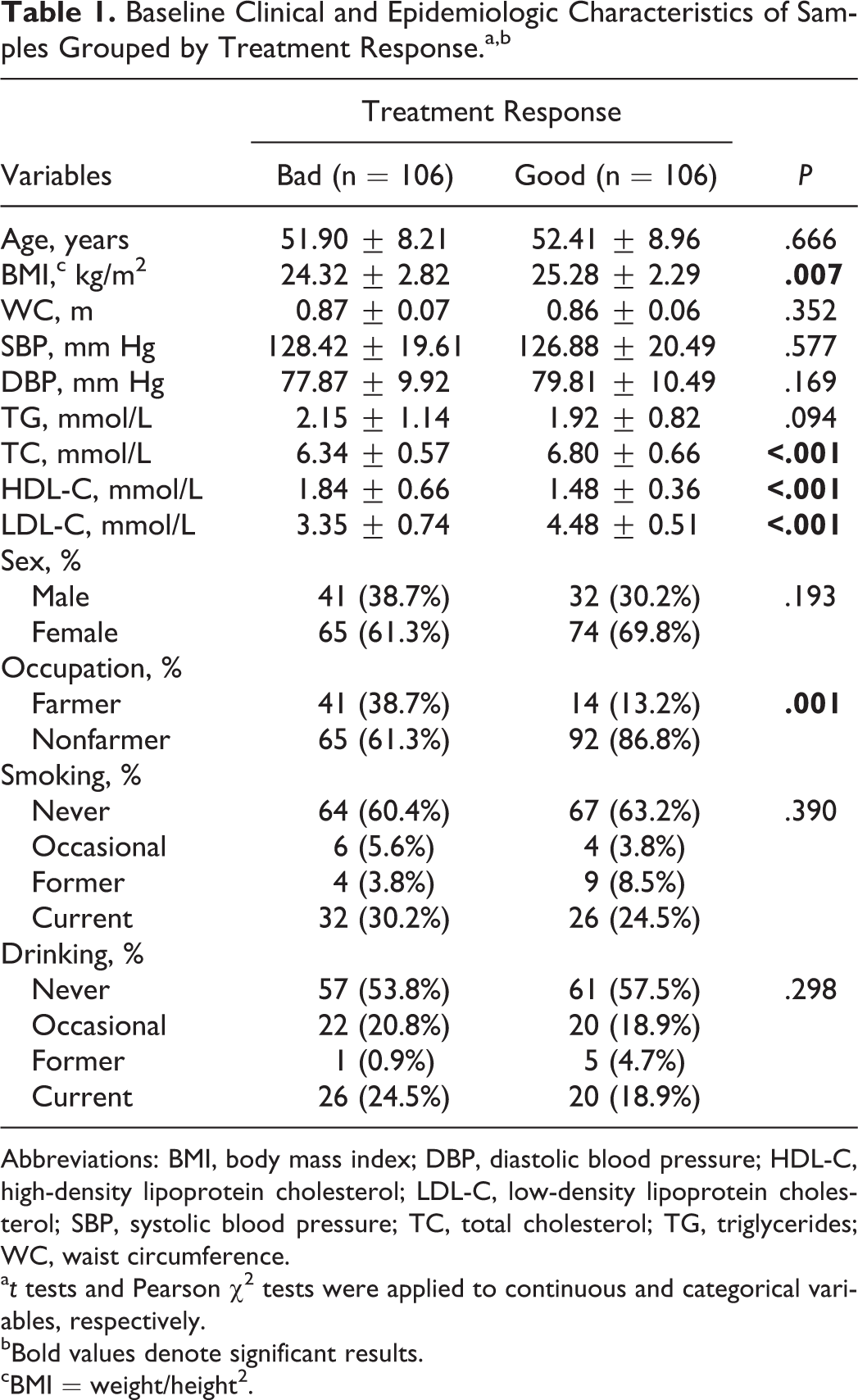
Abbreviations: BMI, body mass index; DBP, diastolic blood pressure; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SBP, systolic blood pressure; TC, total cholesterol; TG, triglycerides; WC, waist circumference.
a t tests and Pearson χ2 tests were applied to continuous and categorical variables, respectively.
bBold values denote significant results.
cBMI = weight/height2.
Effect of Simvastatin Treatment on Serum Lipid Levels
The effect of simvastatin treatment on serum TG, TC, HDL-C, and LDL-C levels is shown in Table 2. Among all patients, the baseline lipid levels for TG, TC, HDL-C, and LDL-C were 2.03 ± 1.00 mmol/L, 6.57 ± 0.66 mmol/L, 1.66 ± 0.56 mmol/L, and 3.91 ± 0.85 mmol/L, respectively. Simvastatin therapy resulted in a significant reduction in total TG, TC, HDL-C, and LDL-C levels either after 29 or 57 days of treatment in both the good and bad response groups.
Effect of Simvastatin Treatment on Serum Lipid Levels After 29 and 57 Days Stratified by Treatment Response.a
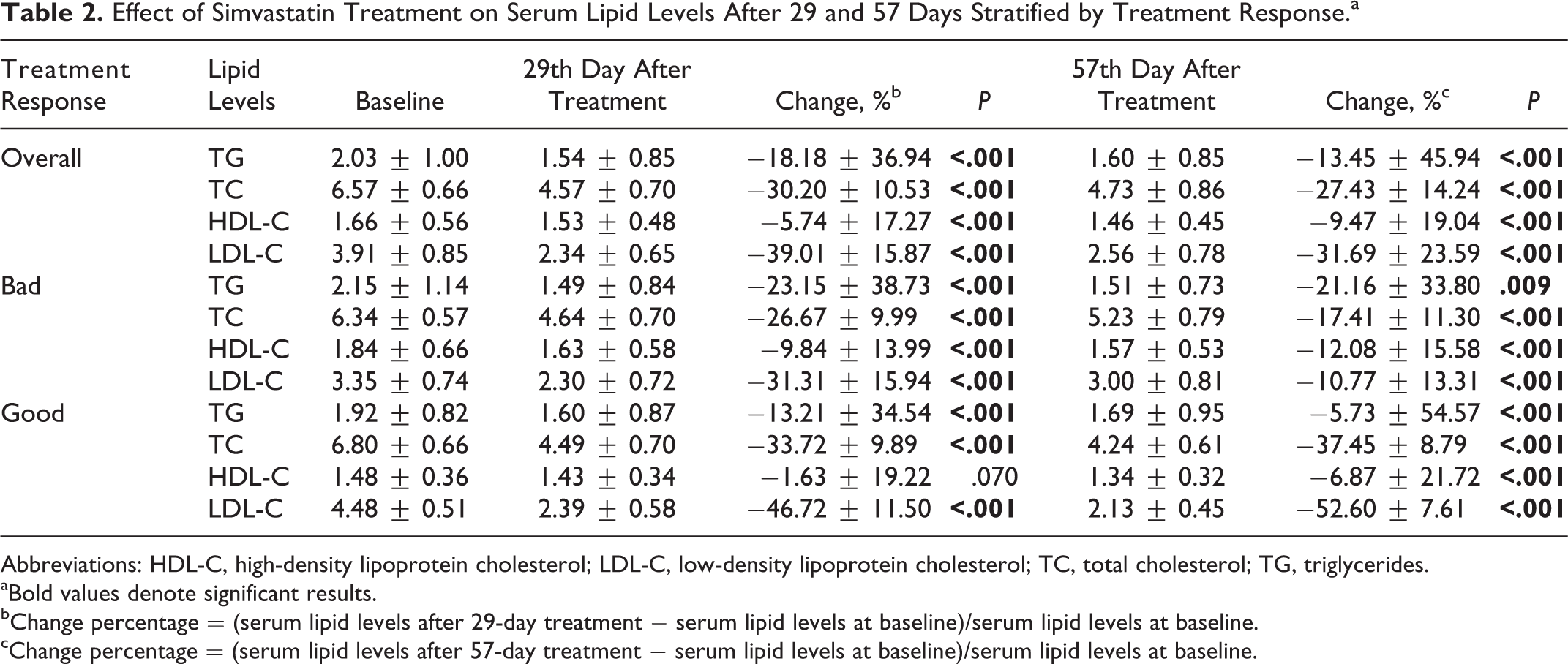
Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; TC, total cholesterol; TG, triglycerides.
aBold values denote significant results.
bChange percentage = (serum lipid levels after 29-day treatment − serum lipid levels at baseline)/serum lipid levels at baseline.
cChange percentage = (serum lipid levels after 57-day treatment − serum lipid levels at baseline)/serum lipid levels at baseline.
Different Frequency Distributions of Genotypes in Extreme Response Groups of Simvastatin Treatment
The prevalence of LEP G2548A and LEPR Q223R polymorphisms in patients with good or bad responses to simvastatin treatment is shown in Table 3. There were no patients carrying the QQ genotype for the LEPR Q223R gene. The distribution of LEPR Q223R genotypes was significantly different between the 2 groups (P = .046). However, LEP G2548A was not associated with therapeutic response to simvastatin.
Prevalence of LEP G2548A and LEPR Q223R Polymorphisms in Patients With Good or Bad Responses to Simvastatin Treatment.a
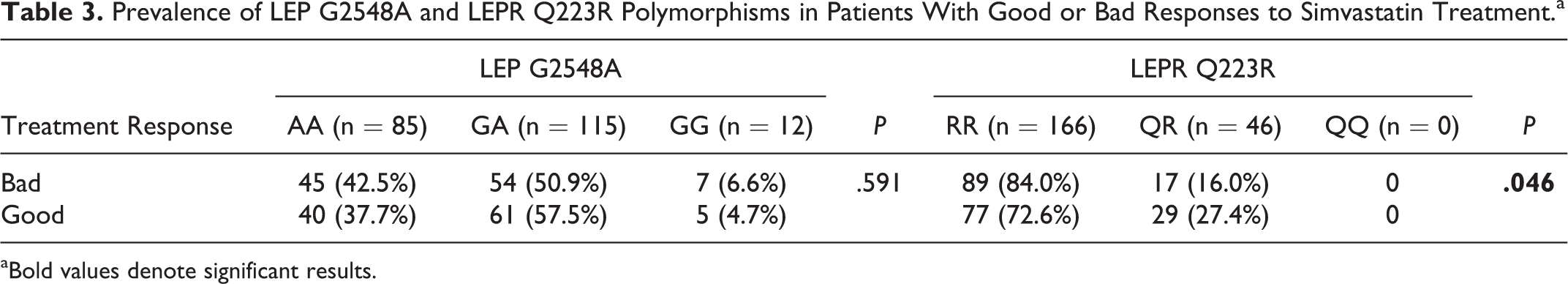
aBold values denote significant results.
Effect of LEP G2548A and LEPR Q223R Polymorphisms on Serum Lipid Levels at Baseline
The results are shown in Table 4. Multiple linear regression analysis demonstrated that there were no statistically significant differences in TG, TC, HDL-C, LDL-C, BMI, and WC levels among those polymorphisms in the bad simvastatin treatment response group. However, among the good response group, the LEPR Q223R polymorphism was significantly associated with HDL-C levels. That is, patients carrying the RR genotype of the LEPR Q223R gene had significantly higher HDL-C levels than those with QR genotype at baseline (P = .034).
Association Between LEP G2548A and LEPR Q223R Polymorphisms and Serum Lipid Levels at Baseline.a
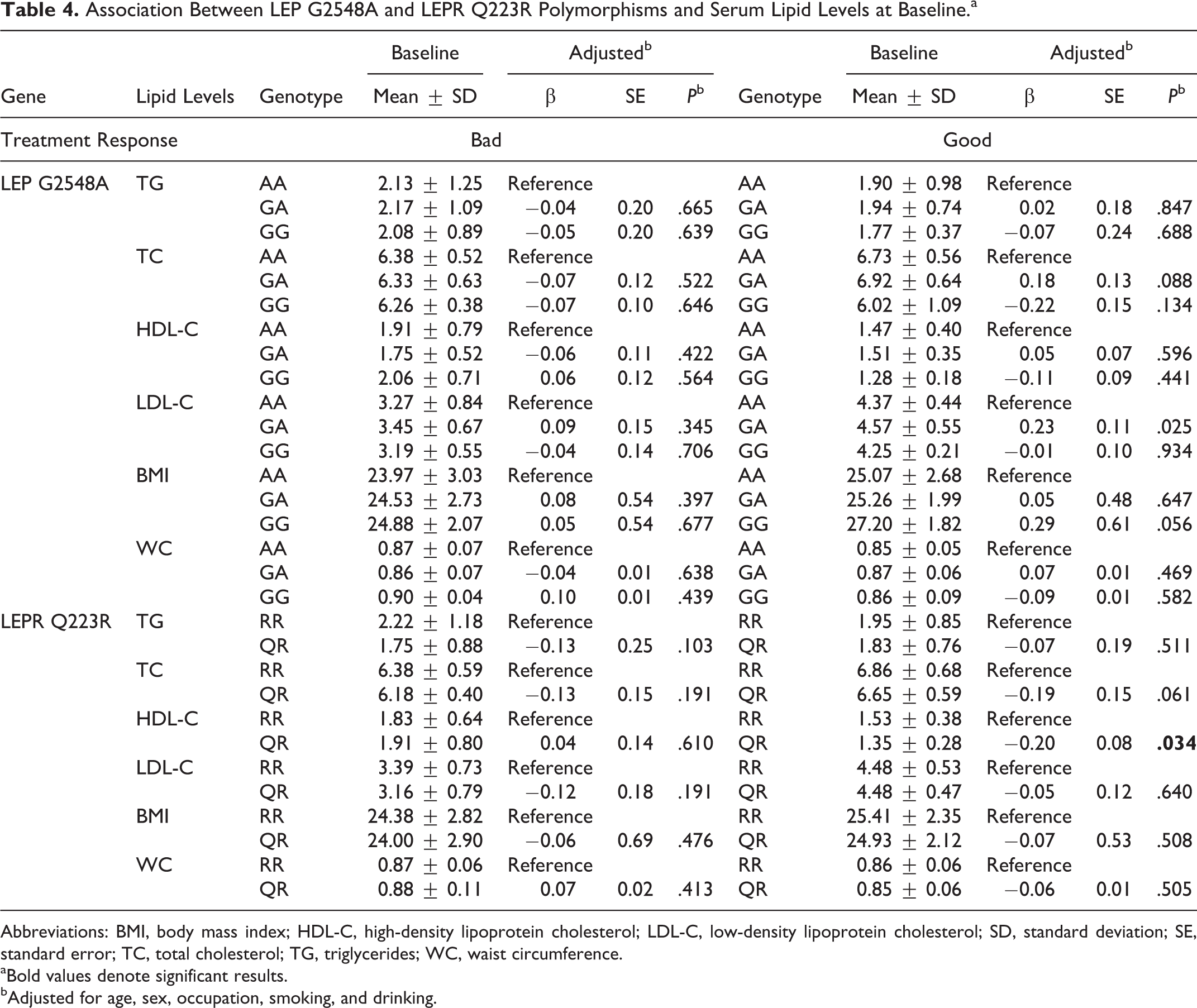
Abbreviations: BMI, body mass index; HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SD, standard deviation; SE, standard error; TC, total cholesterol; TG, triglycerides; WC, waist circumference.
aBold values denote significant results.
bAdjusted for age, sex, occupation, smoking, and drinking.
Effect of LEP G2548A and LEPR Q223R Polymorphisms on the Effectiveness of Simvastatin Treatment
To detect the effect of LEP G2548A and LEPR Q223R polymorphisms on the lipid-lowering efficacy of simvastatin, we investigated the relationship between genotypes and the changes in serum lipid levels after 29 and 57 days of simvastatin treatment among patients with primary hyperlipidemia. After 29 consecutive days of treatment with simvastatin, no statistically significant differences in changes were observed in TG, TC, HDL-C, and LDL-C levels among different LEP G2548A polymorphisms either in bad or good response groups. Comparing LEPR Q223R polymorphism in the bad response group, those carrying the RR genotype had a significantly larger decrease in TG and TC levels (change: −0.74 ± 0.92, P = .036; change: −1.77 ± 0.68, P = .023) compared to those patients carrying the QR genotype (Table 5). After 57 consecutive days of treatment with simvastatin, serum lipid levels had no significant changes among those genotypes either in bad or good response groups (Table 6).
Association Between LEP G2548A and LEPR Q223R Polymorphisms and Lipid-Lowering Response After 29 Consecutive Days of Treatment With Simvastatin.a,b
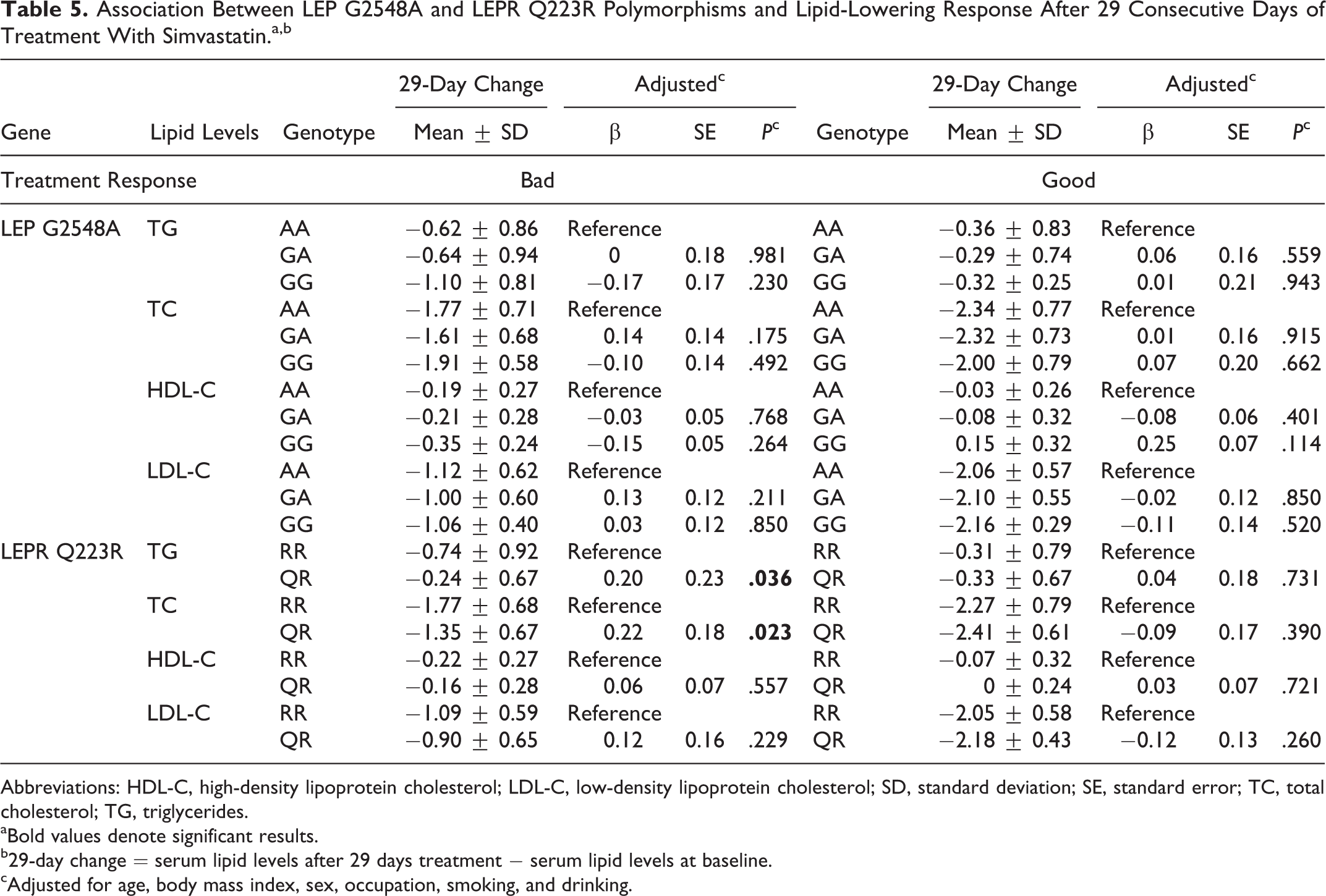
Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SD, standard deviation; SE, standard error; TC, total cholesterol; TG, triglycerides.
aBold values denote significant results.
b29-day change = serum lipid levels after 29 days treatment − serum lipid levels at baseline.
cAdjusted for age, body mass index, sex, occupation, smoking, and drinking.
Association Between LEP G2548A and LEPR Q223R Polymorphisms and Lipid-Lowering Response After 57 Consecutive Days of Treatment With Simvastatin.a
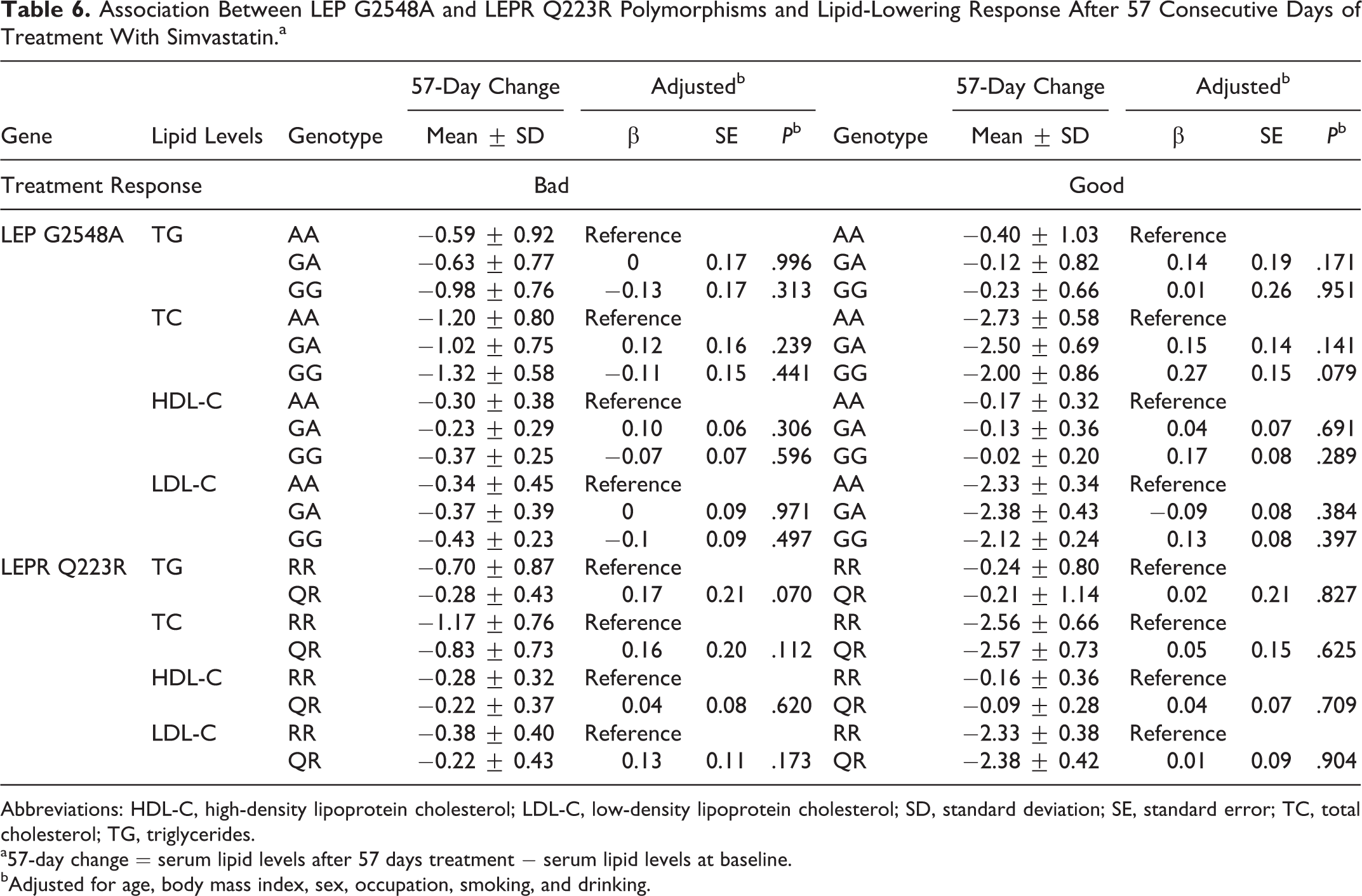
Abbreviations: HDL-C, high-density lipoprotein cholesterol; LDL-C, low-density lipoprotein cholesterol; SD, standard deviation; SE, standard error; TC, total cholesterol; TG, triglycerides.
a57-day change
bAdjusted for age, body mass index, sex, occupation, smoking, and drinking.
Discussion
Our main findings showed that the LEPR 223RR genotype was significantly associated with good response to simvastatin treatment (P = .046). Further multiple regression analyses demonstrated that patients carrying the RR genotype of the LEPR Q223R gene had significantly higher HDL-C levels than those with QR genotype at baseline (P = .034). After 29 consecutive days of treatment with simvastatin, patients in the bad response group carrying the RR genotype had a significantly larger decrease in TG and TC levels compared with those carrying the QR genotype. It indicated that LEPR Q223R, but not LEP G2548A, was associated with serum lipids and further altered the efficacy of simvastatin treatment in Chinese patients with primary hyperlipidemia.
Hyperlipidemia is a major risk factor for CVD. Statins are widely used to prevent CVD because statins can significantly decrease TC, especially LDL-C levels, and then reduce cardiovascular risks. 9 Our results showed that simvastatin treatment significantly decreased the serum TG, TC, HDL-C, and LDL-C levels (all P < .001). A previous study 26 indicated that atorvastatin, simvastatin, and fenofibrate all resulted in a significant reduction in TG, TC, and LDL-C levels in patients with dyslipidemia after 6 and 12 weeks of treatment, but only fenofibrate markedly increased the HDL-C levels. The other 2 statins had no significant effect on HDL-C levels. In our previous study, Jiang et al 23 provided similar evidence that 8 weeks of simvastatin treatment was effective in reducing TG, TC, LDL-C, and HDL-C levels in patients with hyperlipidemia. Nonetheless, these findings need to be replicated and expanded to a large population with primary hyperlipidemia.
Numerous studies found that SNPs of the genes involved in statins and lipid metabolism, such as LEP G2548A and LEPR Q223R, could be major genetic factors for affecting the individual lipid-lowering efficacy. However, limited data showed that the effect of LEP G2548A and LEPR Q223R polymorphisms on the effectiveness of statins remained controversial.
A study on Korean patients with dyslipidemia having schizophrenia 27 suggested that participants carrying the minor 223R allele had significantly lower HDL-C levels than those without this allele (P = .021), but polymorphisms of LEP G2548A were not associated with lipid levels in the entire cohort. Sahin et al’s study 21 also reported that no association was found between LEP G2548A variant and HDL or TG levels in Turkish obese patients (P > .05), but carriers of AG or AA genotypes exhibited higher serum TC, LDL, and VLDL levels compared to those with the GG genotype (P < .05). Research conducted in diabetic patients in Jordan 28 showed that neither LEP G2548A nor LEPR Q223R polymorphisms were associated with levels of HDL, LDL, TG, and TC. However, in patients taking atorvastatin therapy, those with GG or GA genotypes of LEP G2548A experienced significantly higher levels of LDL (P < .002) compared with AA genotype. 28 In contrast, our results also found that patients carrying the GA genotype of LEP G2548A gene had significantly higher LDL-C levels than those with AA genotype (P = .025). Furthermore, after 4 or 8 weeks of simvastatin treatment, there were no marked changes observed in lipid levels among those with different LEP G2548A polymorphisms either in bad or good response groups.
Okada et al 29 and Endo et al 30 found no effect of LEPR Q223R polymorphisms on serum lipid levels in Japanese obese children. In addition, a study in Japanese adults also showed that serum lipid profiles were not associated with Q223R polymorphisms. 31 However, Takahashi-Yasuno et al displayed that the LEPR Q223R polymorphism in Japanese men was associated with significant elevation in serum TC and LDL-C levels. 32 Becer et al 15 demonstrated that homozygous RR participants had significantly higher TG levels than the heterozygous QR and homozygous QQ in obese participants, but the nonobese RR participants showed significantly higher TC (P < .001) and LDL-C (P < .05) levels than QQ and QR. A previous study conducted by Sun et al 6 showed that patients with the QQ genotype had significantly higher TC levels and lower HDL-C levels than those with the RR genotype (P < .05) among Chinese patients with coronary heart disease. After 12 weeks of simvastatin treatment, they found the ability of simvastatin to increase HDL-C levels was significantly lower in patients with the QQ genotype than those with the RR genotype (P < .05). Similar to their findings, our results found that patients carrying the RR genotype of LEPR Q223R had significantly higher HDL-C levels (P =.034) than those with the QR genotype at baseline among the good response group. However, after 29 consecutive days of treatment with simvastatin, compared with patients carrying QR genotype, those carrying the RR genotype had a significantly larger decrease in TG and TC levels (change: −0.74 ± 0.92, P = .036; change: −1.77 ± 0.68, P = .023) in the bad response group.
The association of LEP G2548A and LEPR Q223R polymorphisms with BMI and WC was also examined in our study, but no statistically significant association was observed, and the results could be inconsistent among different research. Similar to our result, Mergen et al 33 also showed no significant association between Q223R polymorphisms and BMI in a Turkish population. On the contrary, Thompson et al 34 reported an association between Q223R polymorphisms and BMI in Pima Indians. Additionally, another study performed in Turkish obese patients also demonstrated that the G2548A polymorphism was related to BMI, and AA or GA genotypes tended to have higher BMI (P < .001), which supported the hypothesis for leptin involvement in the etiopathogenesis of obesity. 21 Although Becer et al 15 reported that Q223R polymorphism had no correlation with BMI in obese or nonobese participants, it was associated with WC in obese participants, and those with the RR genotype had higher WCs (P = .04) compared to QQ genotype. However, a meta-analysis reported no statistically significant association between Q223R polymorphisms and WC. 35
A strength of the study was an extreme sampling approach that effectively detected differences between the groups. However, a limitation of our study should be mentioned. It was a relatively small sample. There was less power to detect significant associations, and parameters were estimated with less precision. In addition, our primary outcome was a statistically significant result, but the secondary exploratory analyses were statistically insignificant after Bonferroni correction. The observation of an association between LEPR Q223R and both TG and TC after 4 weeks of treatment, but not 8 weeks, was also a limitation. It is uncertain whether these results are transient or persist after a long course of treatment. So, these findings need to be replicated and expanded within a larger sample of patients with primary hyperlipidemia.
In conclusion, our study clearly demonstrated that patients with primary hyperlipidemia can be efficiently treated with simvastatin. The LEPR Q223R polymorphism, but not LEP G2548A, modulated serum lipids and the efficacy of simvastatin treatment in this sample.
Footnotes
Acknowledgments
The authors gratefully acknowledge the assistance and cooperation of the faculty and staff of the Anhui Medical University and thank all of the participants in our study.
Authors’ Note
This study was conducted in accordance with the current regulations of the People’s Republic of China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (Grant No. 81373484, 81141116, and 30700454) and the Academic Leader and Reserve Candidate of Anhui Province (No. 05010543).
