Abstract
Abstract
The soluble form of CD40L (sCD40L) is a platelet-derived mediator that links inflammation, hemostasis, and vascular dysfunction. Indeed, blockade of CD40L by neutralizing antibodies or genetic disruption in mice prevents atherosclerosis and atherothrombosis. Until recently, it was believed that CD40 and αIIbβ3 were the only receptors on platelets responsible for binding sCD40L, leading to platelet activation and initiation of thrombotic events. Recent findings showed α5β1 integrin as a novel platelet sCD40L receptor, with an unknown function. For the first time, using anti-α5β1 blocking antibodies, we show that sCD40L/α5β1 interaction leads to platelet activation as evaluated in the human whole blood. Establishing α5β1 integrin’s role in platelet activation, and therefore thrombosis will help further shed light on the etiology of thrombotic disease.
Introduction
Platelets are blood cells originating as fragments of cytoplasm from megakaryocytes in bone marrow. They enter the blood as anucleated, lens-shaped cells with a short life span of 5 to 9 days. Aged and damaged cells are cleared in the spleen by macrophages and in the liver by hepatocytes and Kupffer cells. 1 Hemostasis is a classic, and perhaps the most studied, function of platelets. Injury or disruption of the endothelium causes platelets to attach, activate, and change shape leading to aggregation, initiation of the coagulation cascade, and fibrin deposition. Formation of a fibrin “clot” prevents bleeding and allows for injury repair and healing. Indeed, a reduction in platelet formation or increased platelet lysing due to disease or xenobiotics (thrombocytopenia) is associated with bleeding disorders, while abnormal platelet activation or aggregation can lead to thrombosis. Platelet function, however, extends beyond hemostasis, painting a picture that is multifactorial and complex. It has been shown that platelets regulate aspects of inflammation, 2,3 adaptive 4 –6 and innate 4,7 immunity, tissue repair, 8,9 atherosclerosis, 10 and even cancer. 11,12 Significant advances have been made in understanding specific roles of platelet receptors, ligands, and mediators and how these orchestrate the aforementioned dynamics and pathologies.
A key mediator of platelet function is CD40 ligand (CD40L; also known as CD154). Human CD40L is a 33-kDa type II transmembrane protein, a member of the tumor necrosis factor (TNF) gene superfamily. Following platelet activation, soluble CD40L (sCD40L) is released from the cell membrane by proteolytic cleavage of membrane CD40L by metalloproteases (MMPs) MMP-2 and MMP-9. Similar to other TNF family members, both CD40L and sCD40L form homotrimers, a configuration that is crucial for their function. 13,14 Over 95% of the plasma sCD40L originates from activated platelets. 15 In fact, platelet activation level is associated with elevated blood sCD40L levels, 16 in particular in patients with thrombotic disorders. 17 In addition, in line with the inflammatory cytokine activity, sCD40L is elevated in immune disorders such as systemic lupus erythematosus and rheumatoid arthritis. 18,19 Upon platelet activation, CD40L is rapidly expressed on the cell surface from stores in α-granules. 20 In addition, CD40L messenger RNA (along with the functional translational mechanism) has been detected in platelets, 21 indicating that platelets can generate CD40L de novo, a critical functional remnant from megakaryocyte differentiation. Platelets can be activated by a number of immobilized and soluble agonists such as adenosine-5′-diphosphate (ADP), thromboxane A2, thrombin, and collagen; as part of the amplification feedback loop, sCD40L activates platelets as well. 22 The role of sCD40L as an autocrine, paracrine, and endocrine cytokine that enhances platelet activation, aggregation, and inflammatory response as part of atherothrombotic etiology has been highlighted. 23,24 Besides T cells and platelets, CD40L is expressed on natural killer cells, dendritic cells, cells of monocyte and macrophage lineage, endothelial, smooth muscle, fibroblast, and epithelial cells. 10,25
CD40 is a classic receptor for CD40L; it is expressed constitutively or is induced in a number of immune and vascular cells. The CD40L/CD40 interaction is required for B-cell activation and immunoglobulin isotype switching; the significance of this interaction is observed in hyperimmunoglobulin-M syndrome, where a mutation in the CD40L gene leads to the impairment of CD40 engagement, resulting in deficiency of immunoglobulin (Ig) G, IgA, and IgE. 26 CD40 is constitutively expressed on platelets, 22 and it was originally thought to be a sole receptor for sCD40L responsible for platelet activation. Contradicting evidence emerged showing that CD40L−/− but not CD40−/− mice exhibited unstable thrombotic plaques and delayed vessel occlusions; authors further showed that these events were reversed upon dosing with sCD40L wild type but not with sCD40L mutation lacking αIIbβ3 binding. 27 This had brought up the debate around the primary and secondary effects of platelet activation by sCD40L/CD40. For example, Inwald et al argued that sCD40L activation of platelets is driven via CD40 and that it secondarily induces αIIbβ3 integrin to high enough levels under high shear stress to cause platelet aggregation. 22 Yacoub et al showed that enhanced levels of sCD40L potentiate platelet aggregation and exacerbate inflammatory thrombus formation as it was observed by dosing mice with high concentration of sCD40L prior to inducing vascular injury. 28 Debate over the mechanism of sCD40L platelet activation and coinciding evidence that CD40−/− mice develop normal thrombogenesis 27 has recently lead to identification of new sCD40L receptors. As noted earlier, CD40L binds to αIIbβ3, an integrin exclusively expressed on platelets and megakaryocytes. It has been shown that sCD40L can bind both active and inactive forms of the αIIbβ3 integrin leading to outside-in signaling, platelet spreading, and aggregation under high shear stress. 29 The engagement of αIIbβ3 by CD40L enhances thrombus formation, platelet spreading, and it leads to an increase in CD40L surface expression, contributing to autoamplification loop. 23,29,30 Some evidence suggests that CD40 and αIIbβ3 may represent high and low affinity CD40L receptors, respectively. 28 Subsequently, another integrin, α5β1, was shown to act as a receptor for CD40L in U937 cell line model. 31 Authors have shown that sCD40L/α5β1 activation leads to intracellular activation of extracellular signal-regulated kinase (ERK) phosphorylation cascade, as observed with αIIbβ3 engagement. This binding was sCD40L dose dependent and specific, and the assays were performed under serum-free conditions, therefore eliminating possible nonspecific binding and/or coactivation. Interestingly, unlike αIIbβ3 which CD40L can bind in active and inactive conformation, α5β1 is engaged by CD40L only in its inactive form. 31,32 Binding experiments further showed distinct residues on CD40L for binding to CD40, αIIbβ3, and α5β1, suggesting these receptors can be engaged by CD40L separately or as a combination of trimers 33 conceivably reflecting a range of effects on platelets. Although it has been suggested that CD40L/α5β1 interaction could play a role in platelet activation/aggregation and thrombosis, 32 no platelet CD40L/α5β1 interaction has been documented. The objective of this initial study was to evaluate the role of α5β1 integrin in sCD40L-mediated platelet activation using human whole blood flow cytometry-based approach. For the first time, our data show that sCD40L platelet activation can be significantly inhibited by blocking its interaction with α5β1 integrin.
Materials and Methods
Materials
Anti-CD61-PE, anti-PAC-1-FITC, anti-CD62p-APC, IgG1k isotype control, IgM k isotype control, and IgG1k isotype control were purchased from BD Biosciences (San Jose, California). Integrin antibodies, JBS5 (anti-α5β1), P1D6 (anti-α5β1), BHA2.1 (anti-α2β1) and CBL544 (anti-αVβ3), were purchased from EMD Millipore (Billerica, Massachusetts), and CA3 (anti-αIIbβ3) was purchased from Enzo Life Sciences (Farmingdale, New York). Recombinant Human sCD40-Ligand was purchased from PeproTech (Rocky Hill, New Jersey). To minimize lot-lot sCD40L trimer variability, we ran the same lot for all experiments (lot#0408145). Adenosine-5′-diphosphate was purchased from Bio/Data Corporation (Horsham, PA). BD Vacutainer Buffered Na Citrate tubes were purchased from Becton Dickinson (Franklin Lakes, New Jersey). HEPES-buffered Tyrode’s solution was purchased from Boston BioProducts (Ashland, Massachusetts).
Human whole blood
Human blood was collected from healthy volunteers as per Johnson & Johnson policy on employee sample donations. Informed consent was obtained from each donor prior to blood collection. The first 2 mL of blood was discarded followed by 4 mL of blood drawn into sodium-citrated tubes and then gently inverted.
Washed platelets
Blood was drawn into a Becton Dickinson Vacutainer containing acid citrate dextrose (ACD) (yellow cap), mixed gently, and spun at 200 ×
Assay protocol
Assay was performed at room temperature. Fluorescence activated cell sorting antibody solution was prepared per manufacturer’s instructions; briefly 82.5 µL CD61 PE for platelet gating, 165 µL CD62p-APC and 165 µL PAC-1-FITC (to measure platelet activation), and 1155 µL Tyrode’s solution with an additional 9.9 µL of 150 mmol/L CaCl2. To this solution, 60 µL of the whole blood was added and gently inverted to mix. Whole blood–antibody solution was incubated with blocking antibodies to integrins to a final concentration of 0.1, 1, and 10 µg/mL for 25 minutes in the dark after which sCD40L was added to the blood–antibody solution to a final concentration of ∼2 µmol/L and incubated at room temperature in the dark for 15 minutes. The solution was then fixed with 1% formalin, and flow cytometric analysis was preformed within 1 hour. Adenosine-5′-diphosphate at 20 µmol/L was used as a positive control for platelet activation.
Flow cytometry
Flow cytometric analysis was performed on FACScalibur instrument (Becton Dickinson, San Jose, California) using BD CellQuest Pro Version 6.0 software. Platelets were identified by their characteristic forward scatter/side scatter and gated for CD61 expression. CD61+ cells (platelets) were evaluated downstream for the ability to activate following treatment. CD61+ PAC-1+ (activated αIIbβ3 integrin) and/or CD61+ CD62p+ (P selectin expressing) population was noted as activated.
Graphs and statistics were generated using GraphPad Prism6 software: “Two-way ANOVA followed by Dunnett multiple comparisons test was performed using GraphPad Prism version 6.00 for Windows, GraphPad Software, La Jolla, California, www.graphpad.com.”
Results
sCD40L-Mediated Platelet Activation and Donor Variability
Nine donors were evaluated on 3 separate occasions (n = 3) for platelet activation by ADP (positive control) and sCD40L (2 µmol/L). All donors responded to ADP treatment, resulting in ∼90% platelet activation. Following treatment with sCD40L, most donors responded above baseline phosphate-buffered saline (PBS) treated whole blood, however, only the most robust responders (donors 037, 003, 346, and 087) were selected for downstream assessment (Figure 1). The arbitrary cutoff of 10% and 20% of platelet activation was set for PAC-1 and CD62p, respectively. Selecting only “high-responders” with a broad dynamic range would allow for most effective assessment of inhibition of sCD40L-mediated platelet activation. In addition, platelet activation of ≤20% is within our traditional assay variability, thus only activation above cutoff was considered.
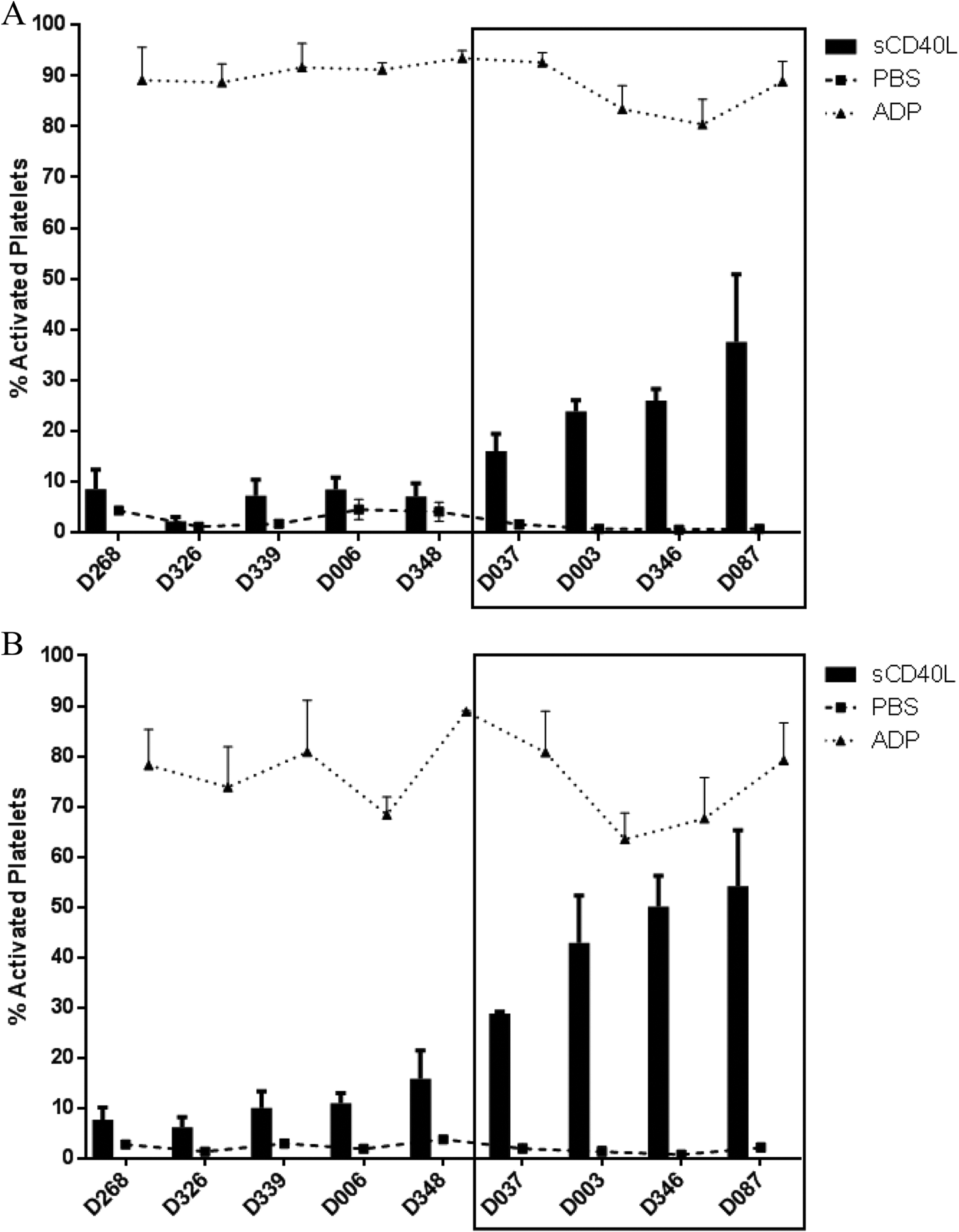
Platelet activation response between donors. Nine human whole blood donors were evaluated 3 times (n = 3) in the flow cytometry-based platelet activation assay, using PAC-1 (A) or CD62p (B) as markers of platelet activation. All donors showed consistent platelet activation following treatment with the positive control, Adenosine-5′-diphosphate (ADP; 20 µmol/L). Response to soluble CD40LsCD40L (soluble CD40L; 2 µmol/L) was donor specific and consistent; the assay was repeated 3 times at different days with similar results (error bars). Based on arbitrary platelet activation cutoff (10% for PAC-1 and 20% for CD62p), 4 donors were identified as “robust” responders and were subsequently followed up for the downstream mechanistic evaluation of sCD40L-mediated platelet activation. Data presented as mean and standard deviation (error bars).
sCD40L and Integrins αIIbβ3 and α5β1
Treatment with the sCD40L activated 25% and 40% of platelets, as evident by the induction of PAC-1 and CD62p, respectively (Figure 2A). Pretreating platelets with anti-αIIbβ3 blocking antibody (CA3) reduced sCD40L-mediated platelet activation in a dose-dependent manner, from 25% to 11%, as measured by PAC-1. Platelet activation marker CD62p revealed similar pattern of inhibition. Pretreatment with CA3 antibody attenuated sCD40L-mediated platelet activation dose dependently from 40% to 17%, as measured by CD62p. Generally, blocking αIIbβ3 integrin resulted in a statistically significant inhibition of sCD40L-mediated platelet activation ≤60%.
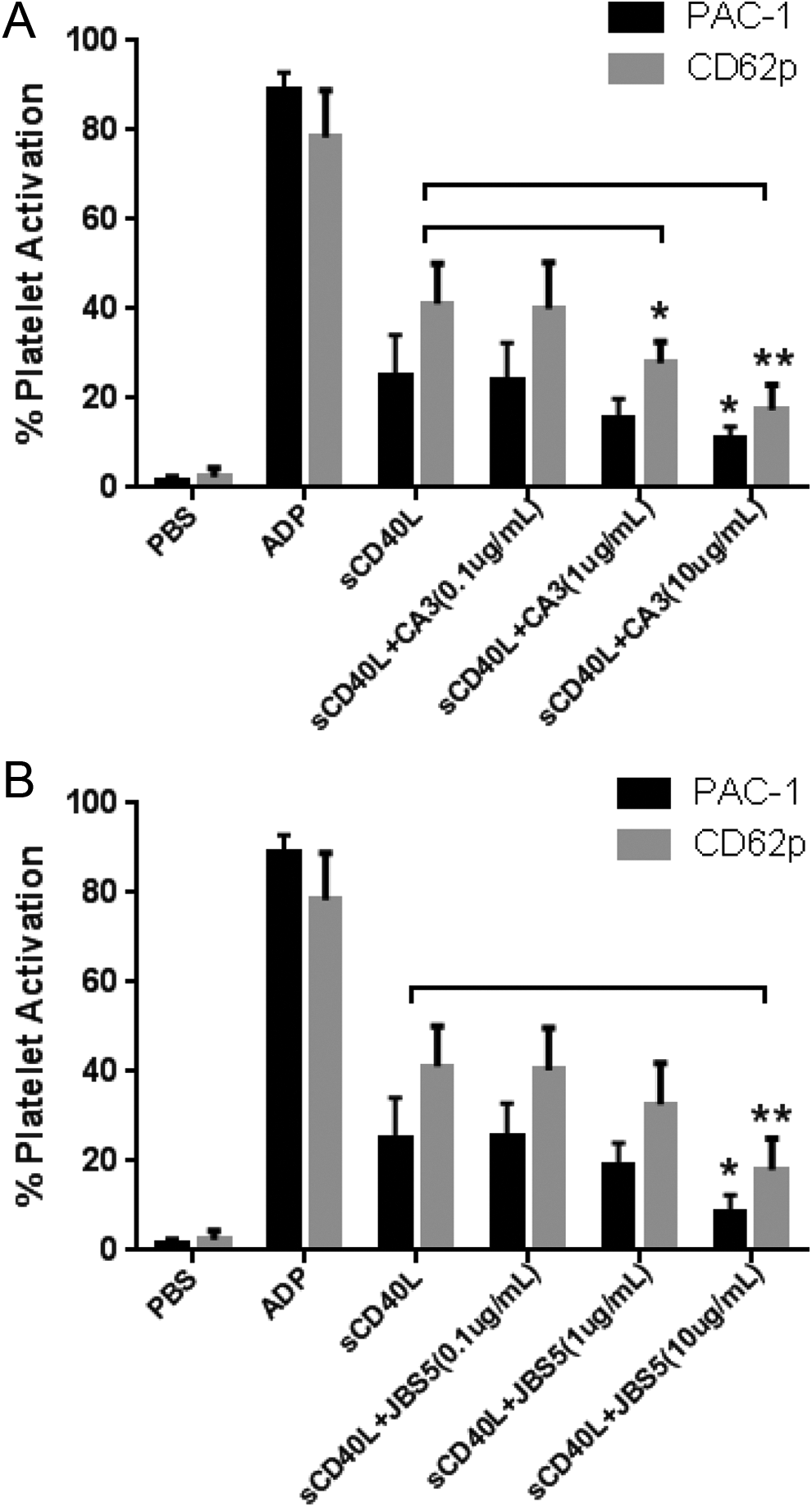
Blocking αIIbβ3 and α5β1 integrins attenuates soluble CD40L (sCD40L)-mediated platelet activation. sCD40L-mediated platelet activation was attenuated by preincubating platelets with αIIbβ3 blocking antibody (CA3) (A) and α5β1 blocking antibody (JBS5) (B). Platelet activation markers PAC-1 and CD62p resulted in a similar response. Error bars represent n = 4. *
Blocking the α5β1 integrin using JBS5 blocking antibody also resulted in a significant decrease in sCD40L-mediated platelet activation (Figure 2A). The reduction in platelet activation was JBS5 concentration dependent. By evaluating PAC-1 expression, pretreatment of platelets with anti-α5β1 blocking antibody attenuated sCD40L-mediated platelet activation from 25% to 9%. Similarly, by evaluating CD62p, pretreatment with JBS5 α5β1 blocking antibody attenuated (in a dose-dependent manner) sCD40L platelet activation from 41% to 18%. Blocking α5β1 integrin resulted in the statistically significant reduction in sCD40L-mediated platelet activation ≤65%.
Combination of α5β1 and αIIbβ3 Integrin Block and sCD40L Platelet Activation
To evaluate possible additive effects of blocking α5β1 and αIIbβ3 integrin simultaneously, whole blood platelets were pretreated with the JBS5 (10 µg/mL) and CA3 (10 µg/mL) prior to adding sCD40L (Figure 3). Simultaneously blocking α5β1 and αIIbβ3 integrin reduced sCD40L-mediated platelet activation by approximately 65% for CD62p and PAC1 platelet activation markers. Blocking both integrins did not result in an additive inhibitory effect, as the level of inhibition was similar to the individual integrin inhibition.
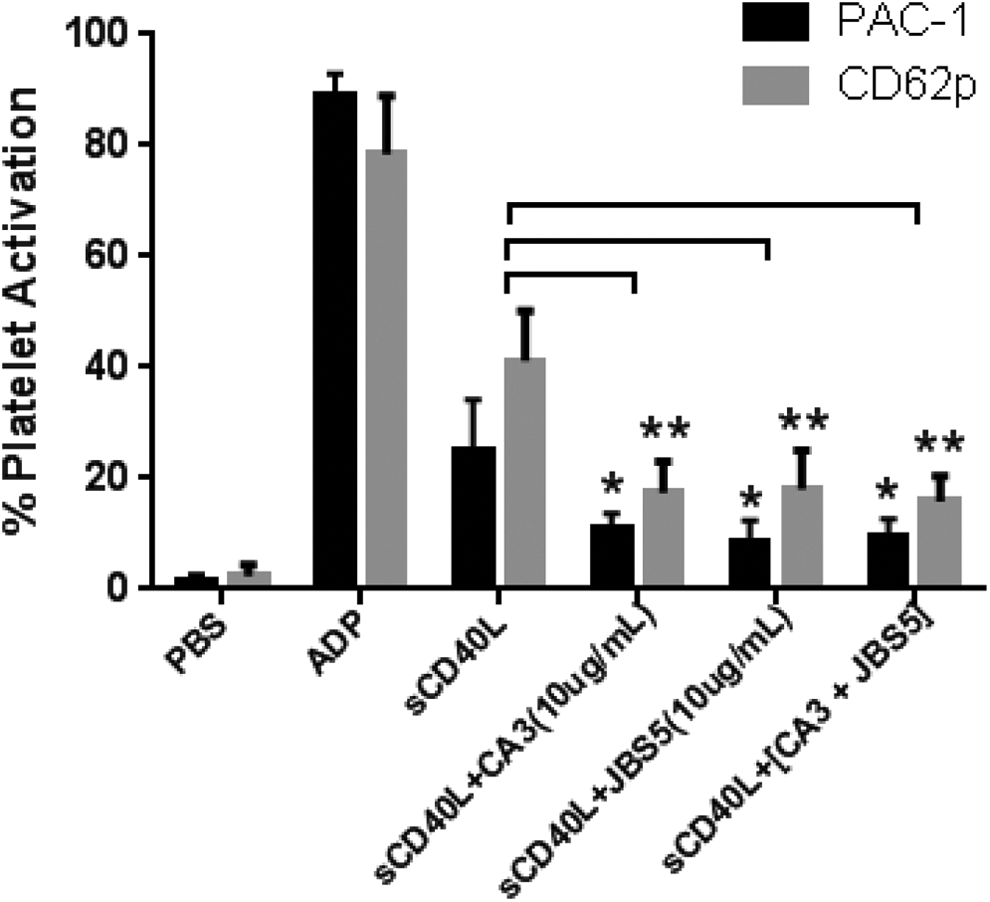
Blocking α5β1 and αIIbβ3 does not result in additive attenuation of soluble CD40L (sCD40L) platelet activation. Error bars represent n = 4. *
Blocking α2β1 and αVβ3 Integrins
To assess the specificity of integrin blocking on platelets, α2β1 and αVβ3 integrins were blocked using BHA2.1 (10 µg/mL) and CBL544 (10 µg/mL) blocking monoclonal antibodies (mAbs), respectively. Both integrins are expressed on platelets 34,35 but are not CD40L target receptors and were therefore used as internal standards in the assay. Blocking α2β1 or αVβ3 had no effect on sCD40L-mediated platelet activation (Figure 4).
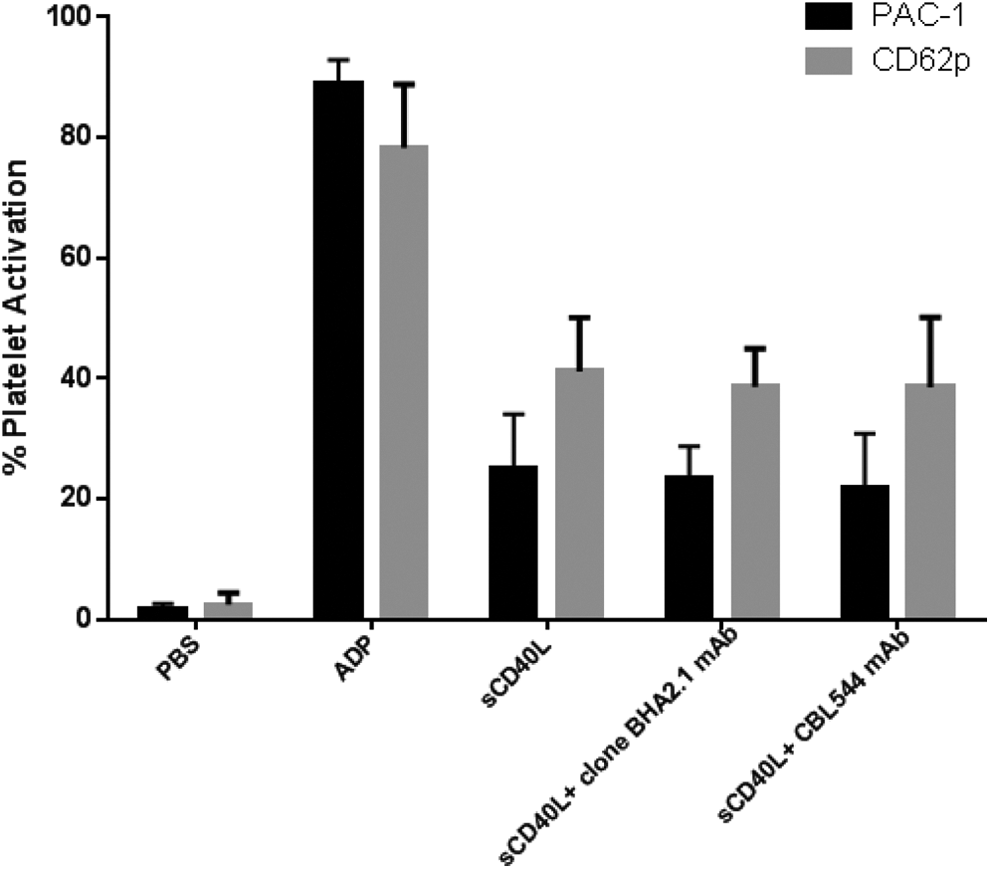
Blocking of α2β1 and αVβ3 integrins. Blocking α2β1 (clone BHA2.1) or αVβ3 (CBL544) integrin receptors did not affect soluble CD40L (sCD40L; 2 µmol/L) mediated platelet activation, n = 4. Data presented as mean and standard deviation (error bars).
Blocking of α5β Integrin in Washed Platelets
To eliminate the possibility that fibronectin (α5β ligand) may be interfering with the binding of the sCD40L to α5β1, we have performed an activation assay using washed platelets. Three highest sCD40L “responder” donors were evaluated. By normalizing to sCD40L, JBS5 (10 µg/mL) inhibited sCD40L platelet activation by 45% and 73%, based on CD62p and PAC1, respectively (Figure 5). This inhibition was comparable to the whole blood. In addition to JBS5, we have tested another α5β blocking antibody, P1D6. Cell protein binding data have shown that P1D6 has lower affinity (∼2×) for α5β than JBS5, 36 which was reflective in the assay. At the same concentration as JBS5 (10 µg/mL), P1D6 did trend toward inhibition of sCD40L-induced activation 5% to 15%. sCD40L and positive control ADP activated platelets similarly as in the whole blood (data not shown).
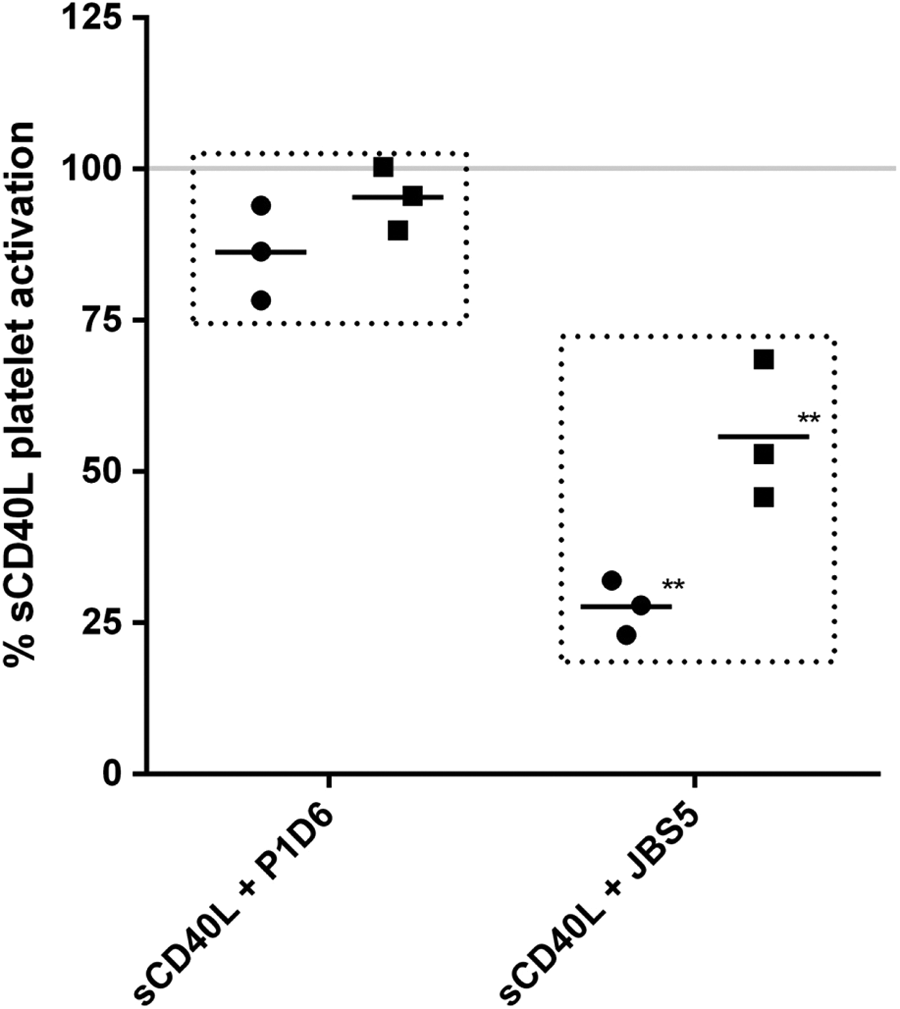
Blocking of α5β1 integrin in washed platelets. Two different α5β1 blocking antibodies (JBS5 and P1D6 at 10 µg/mL) were evaluated for the ability to block soluble CD40L (sCD40L)-mediated platelet activation (circles PAC1 and squares CD62p) in washed platelets, n = 3. The response (y-axis) was normalized to sCD40L activation as 100%. **
Conclusion
Because of its activating, cytokine-like activity, sCD40L has gained interest for its involvement in vascular disease, cancer, and immune-mediated disease pathogeneses. Although >95% of sCD40L originates from platelets, platelets are also one of the major targets of sCD40L, leading to the platelet activation cascade and a plethora of thromboinflammatory effects. In fact, increasing levels of sCD40L have been correlated with the severity of the cardiovascular diseases. 17,37,38 This direct correlation suggests that the range of response may be facilitated by multifaceted high- and low-affinity receptors. Indeed, the responses between donors to the same sCD40L concentration were variable, emphasizing potential receptor-mediated regulation of response. Although we didn’t pursue the cause of donor variability, it could be hypothesized that the level of expression and the combination of types of sCD40L receptor(s) expressed on platelets may play a role. Similarly, others have hypothesized that an active (s)CD40L homotrimer can bind to a number of heterotrimeric receptor variants containing combination of CD40, αIIbβ3, and α5β1 monomers, leading to a possible range of response intensities, 33 further adding to the complexity and the variability noted between donors. Identifying and characterizing receptors that mediate platelet activation are therefore central to deciphering conditions and diseases associated with thrombotic events.
Using a flow cytometry-based platelet activation assay, we show that sCD40L activates platelets, a finding that is aligned with others. 22,28,29,39,40 Differences in assay design (aggregation, flow cytometry-based PRP, or the whole blood assay), end points, and concentration of sCD40L contribute to slight discrepancies in the level and type of platelet activation between investigations. As mentioned, sCD40L trimerization is essential for its activating function, this is particularly important for the exogenous sCD40L, as it can be challenging to control its trimeric, dimeric, or monomeric concentrations in the solution. We have noted significant differences between sources and lots of sCD40L and their ability to activate platelets (data not shown). In addition, Anand et al highlighted some of the conflicting findings between groups when using different sources of sCD40L, 14 stressing the need to validate different batches. Beyond nuances of specific assays, a common message has emerged; at low (20 nmol/L) concentrations, sCD40L seems to potentiate platelet activation, while at high concentrations (2 µmol/L), it acts as an activator. This phasic effect may be in part due to differential affinities to combinations of low and high affinity receptors.
To evaluate receptors responsible for sCD40L platelet activation, we used an activating concentration of 2 µmol/L of sCD40L, a concentration also used by Prasad et al. 29 It was argued that this concentration may be more relevant to the concentrations within thrombi and its immediate microenvironment. In our assay, we show a significant induction of PAC-1 and CD62p signal in select donors following sCD40L treatment, indicating αIIbβ3 activation and P-selectin expression, respectively. Interestingly, similar to others, we also see a greater induction of P-selectin than αIIbβ3 activation, 22 although both are induced in our assay. In regard to the difference in αIIbβ3 activation and P-selectin expression, it is hypothesized that sCD40L engagement does not induce a significant calcium influx or dense granule secretion necessary for αIIbβ3 integrin activation on platelets, 28 which may explain attenuated PAC-1 versus CD62p levels.
Since CD40 is a classic CD40L receptor, we focused our efforts on less common integrin receptors–sCD40L interactions and their roles in platelet activation; in particular as some have been implicated in platelet activation. For example, preliminary evidence has shown that αIIbβ3–CD40L interaction can be sufficient to activate platelets. 41 Work by Leveille et al had shown that in a U937 cell line model, sCD40L can bind to α5β1 and activate downstream signaling cascade. 31 The expression of α5β1 on the platelets then invokes the question if α5β1 plays a role in sCD40L-mediated platelet activation. For the first time, we show that sCD40L can activate platelets by engaging α5β1 integrin. Anti-α5β1 (JBS5) blocking antibody was able to abrogate sCD40L-mediated platelet activation in the whole blood (≤65%) and washed platelets (≤73%). Additional α5β1 blocking antibody (P1D6) was tested in the washed platelets model, resulting in lower attenuation of sCD40L platelet activation ≤15%. In line with our findings, compared to JBS5, P1D6 antibody has lower affinity for α5β1 (in K562 cell model) as well as a unique α5β1 N-terminal binding site. 36 Perhaps because of its lower affinity, the P1D6 concentrations used by Leveille et al in their U937 cell assay were significantly higher ∼300× than used in the present model. Interestingly, both antibodies seemed to inhibit αIIbβ3 integrin activation (PAC1) to the greater extent than P-selectin expression (CD61p). These data therefore supports adding a third platelet receptor (besides CD40 and αIIbβ3), exhibiting functional activation and relevance to thromboinflammatory effects via sCD40L engagement.
We also showed that blocking αIIbβ3 and α5β1 integrins but not α2β1 or αVβ3 (2 integrins also expressed on platelets) can inhibit sCD40L-mediated activation. Further, the effect of blocking αIIbβ3 and α5β1 was not additive suggesting engagement of additional receptors, most probably CD40, although we cannot rule out the possibility that anti-α5β1 and anti-αIIbβ3 mAbs added simultaneously could hinder each other’s binding to the target(s). Although αIIbβ3 is highly expressed on platelets, blocking its binding with sCD40L resulted in a comparative level of inhibition as anti-α5β1, and as mentioned, blocking both integrins simultaneously did not result in additive effect. One possibility is that the binding of sCD40L to α5β1 and αIIbβ3 integrins may be heterotrimeric (combination of α5β1 and αIIbβ3), a hypothesis also brought up by others. 33,42 In fact, a recent finding has shown that CD40L receptors, including α5β1 and αIIbβ3, upon stimulation with CD40L activate same downstream pathways, p38 and ERK1/2 phosphorylation, further related response to sCD40L stimulation. 42 It is therefore possible that the level of α5β1 on platelets can be rate limiting, governing the overall level and activity of (s)CD40L platelet heterotrimeric integrin receptors. Although current work was focused on sCD40L-integrin–mediated platelet activation, it will be interesting to frame this interaction in the context of CD40 and how these receptors orchestrate (s)CD40L-mediated platelet activation. In particular, it will be pertinent to evaluate the trimeric interplay between these receptors to regulate the type (potentiation or activation) and the level of platelet activation especially since (s)CD40L can bind to all 3 receptors concomitantly. 33 Interestingly, by screening limited number of donors, we were able to further confirm that it is not just the level of sCD40L that can orchestrate platelet activation, the differences in platelet response may be mediated via differential expression and regulation of the repertoire of sCD40L high and low affinity receptors. In fact, this could be one of the key drives of the sCD40L differential response between donors noted above. Future work would evaluate the type, level of expression, and activity of these receptors based on overall donor response (to sCD40L) and/or underlying thromboinflammatory condition.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
