Abstract
The alterations of the inflammatory and thrombotic components in patients with cancer are not clearly understood. The purpose of this study was to profile markers of inflammation and thrombotic activation specifically in the patients with bladder cancer undergoing radical cystectomy. For this study, 134 samples were collected from patients undergoing radical cystectomy. Antiphospholipid antibodies (immunoglobulin G subtype), microparticles, and antiglycosaminoglycan antibodies were measured with a commercially available enzyme-linked immunosorbent assay kits. These biomarkers were compared in patients with bladder cancer and normal individuals (n = 20). Patients had an average value of 6.7 ± 11.9 ng/mL (median: 2.8, confidence interval: 4.69-8.75, and P value: .0038) of antiphospholipid antibodies versus normal individuals 1.96 ± 0.9 ng/mL (median: 1.8 and confidence interval: 1.5-2.35). Microparticles level in patients was 8.31 ± 6.14 ng/mL, (median: 6.1, confidence interval: 7.26-9.37, and P value: <.0001) versus normal individuals 3.57 ± 2.34 ng/mL (median: 2.85 and confidence interval: 2.476-4.664). The antiglycosaminoglycan antibodies in patients had an average value of 0.22 ± 0.1 optical density (OD; median: 0.2, confidence interval: 0.21-0.24, and P value: .0213) compared to normal individuals 0.25 ± 0.08 OD (median: 0.25 and confidence interval: 0.22-0.23). The correlation of antiglycosaminoglycan antibodies with antiphospholipid antibodies showed Spearman r value = .2364 (95% confidence interval: 0.05-0.4 and P value .009). The correlation of antiglycosaminoglycan antibodies versus microparticles showed Spearman r = −.195 (95% confidence interval: 0.37-0.01 and P value .0321). These data suggest that patients with bladder cancer have subclinical activation of thrombotic and inflammatory processes that may be further exacerbated by surgical procedures and lead to venous thromboembolism-related complications.
Keywords
Introduction
Bladder cancer (BC) is the fifth most common cancer in the United States. The National Cancer Institute estimates that there will be 74 000 new cases of BC in 2015, which represents 4.5% of all new cancer cases in the United States. It is the ninth leading cause of cancer death in the United States. The mortality rate is 4.4 per 100 000 men and women per year. In 2015, 16 000 deaths related to BC are estimated which represent 2.7% of deaths related to all types of cancer. Among cancers in the United States, BC has the highest per patient treatment costs, and the fifth highest overall cost, 1 estimated at US$3.4B annually with US$2.9B in direct treatment-related costs and of US$184 762 to US$461 907 per patient indirect costs. 2 The major complications related to cancer are metastasis, reoccurrence, and thrombotic complications. Thrombotic complications may be the first manifestation of underlying cancer. Venous thromboembolism (VTE) is the second leading cause of cancer-associated mortality. 3 While patients with BC have a 5-fold higher risk of VTE than the general population, but the risk of VTE increases further if patients undergo surgery. 4,5 Since there are no confirmed biomarkers available for the diagnosis of the VTE in BC, these patients are not placed on any prophylactic anticoagulation.
It is well documented that microparticles (MPs) are highly procoagulant, and several studies suggested that MPs can be used as a marker for vascular endothelial damage. 6,7 However, the role of MPs in thrombotic events related to BC still needs to be explored.
Similarly, the circulating antiphospholipid (APL) antibodies are considered to be associated with an increase in thrombotic and vascular diseases. 8 –13 But the pathologic role of APL in thromboembolism development in malignancy is still unclear and is contradictory. 14 –17 It is also in the literature that binding of heparin–platelet factor 4 (PF4) antibodies to platelet results in their activation and formation of platelet MPs. These platelet MPs also lead to thrombosis. Heparin Induced Thrombocytopenia (HIT) can be severe enough to lead death, 18,19 but there is lack of information regarding HIT antibodies and patients with cancer. The aim of this study is to find the prevalence of MPs, APL antibodies, and antiglycosaminoglycan antibodies (AGAs) in patients with bladder pathology in relation to VTE complication.
Methods
This study was carried out retrospectively in conjunction with an institutional review board-approved protocol. Banked baseline blood samples (N = 134) were collected from patients with BC presenting at Loyola Medical Center for radical cystectomy. Residual blood samples from routine preoperative laboratory draws in tubes containing 3.2% (0.109 mol/L) sodium citrate were used. Samples were centrifuged for preparation of platelet poor plasma at 3000 rpm, at room temperature, for 20 minutes. Plasma samples were divided into 3 different aliquots to avoid repeated thawing and freezing. Samples were stored at −80°C until analysis was completed. Microparticles, APL, and AGAs were determined using commercially available enzyme-linked immunosorbent assay (ELISA) kits. The APL antibodies were measured using an ELISA-based method for immunoglobulin G-specific antibodies (American Diagnostica, Stamford, Connecticut). The MPs were measured utilizing an annexin capture chromogenic substrate method to quantitate functional procoagulant activities of MPs (Hyphen Biomedical; Paris, France). Due to insufficient quantity of plasma, 121 samples were used to measure AGA antibodies. The AGA antibodies were measured using a commercially available method where a polysulfonate polymer is used as a capture probe for these antibodies (GTI; Madison, Wisconsin). We used George King Biomedical (Overland Park, Kansas) samples from healthy volunteers as our controls. These were 20 citrated plasma samples collected from nonsmoking, drug-free individuals in the age range of 18 to 35 years.
Statistical Analysis
GraphPad Prism program (version 4, 2003) was used for statistical analysis. Nonparametric tests were used. The Mann-Whitney U test was used to identify significant differences between patients and controls. The Spearman r test was used to demonstrate correlations between 2 variables. The 2-tailed P value of <.05 was considered significant.
Results
A statistically significant difference in these biomarkers between patient group and normal group was noted. Patients showed higher values for APL antibodies, MPs, and AGAs when compared to normal individuals.
Patients’ APL antibodies had an average value of 6.7 ± 11.9 ng/mL (median: 2.8, confidence interval: 4.69-8.75, and P value: .0038) compared to normal individuals 1.96 ± 0.9 ng/mL (median: 1.8, confidence interval: 1.5-2.35; Figure 1).
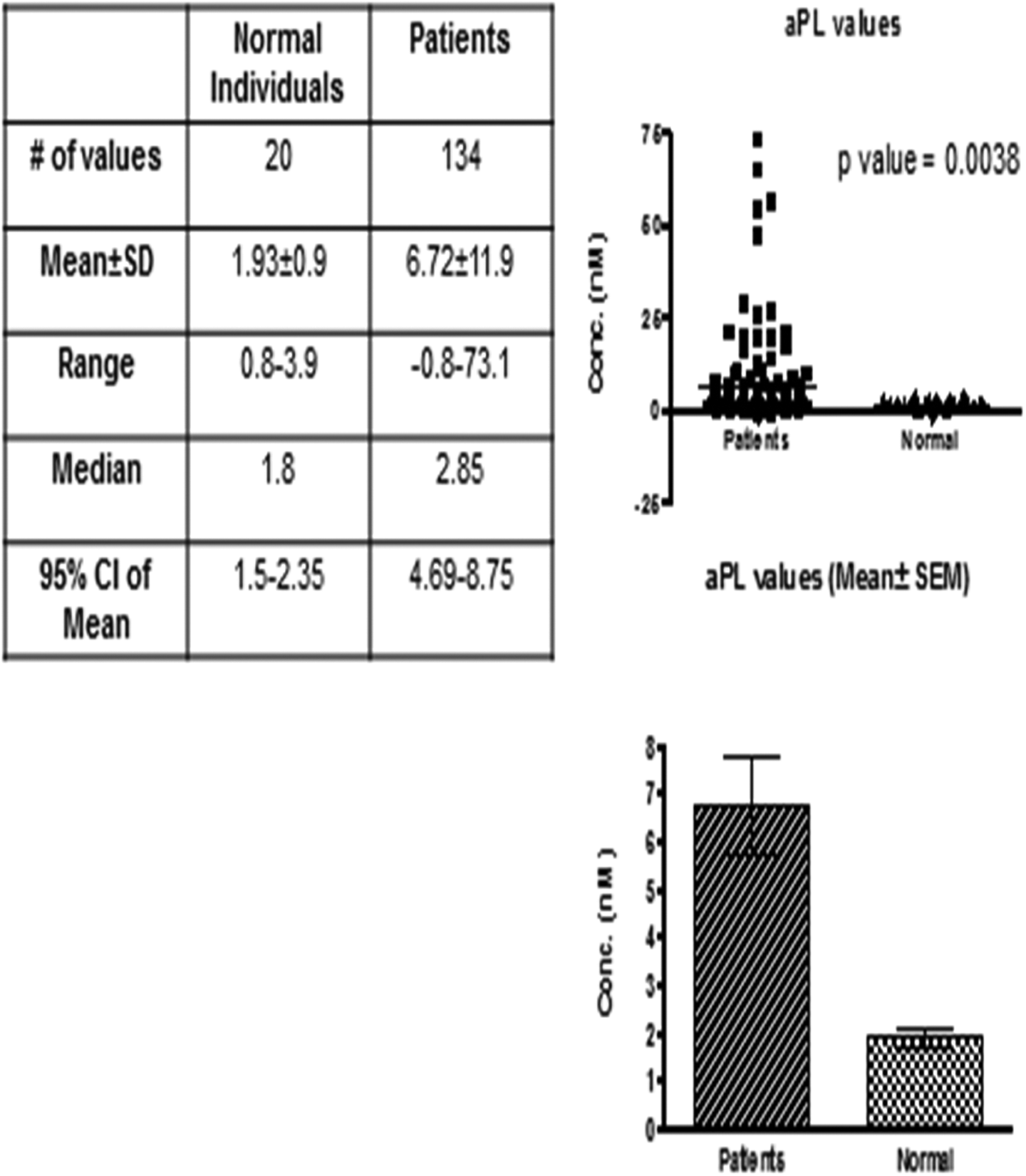
Antiphospholipid antibody titers in normal and patients with bladder tumor. Top right is a scattergram plotted against antiphospholipid antibody levels in patients versus normal individuals. Bottom right is a bar graph for the mean ± standard error of the mean (SEM) of patient group versus control group. The table shows statistical analysis (nonparametric Mann-Whitney U test) between patient and control groups.
Patients’ MPs had an average value of 8.31 ± 6.14 ng/mL (median: 6.1, confidence interval: 7.26-9.37, and P value: <.0001) compared to normal individuals 3.57 ± 2.34 ng/mL (median: 2.85 and confidence interval: 2.476-4.664; Figure 2).
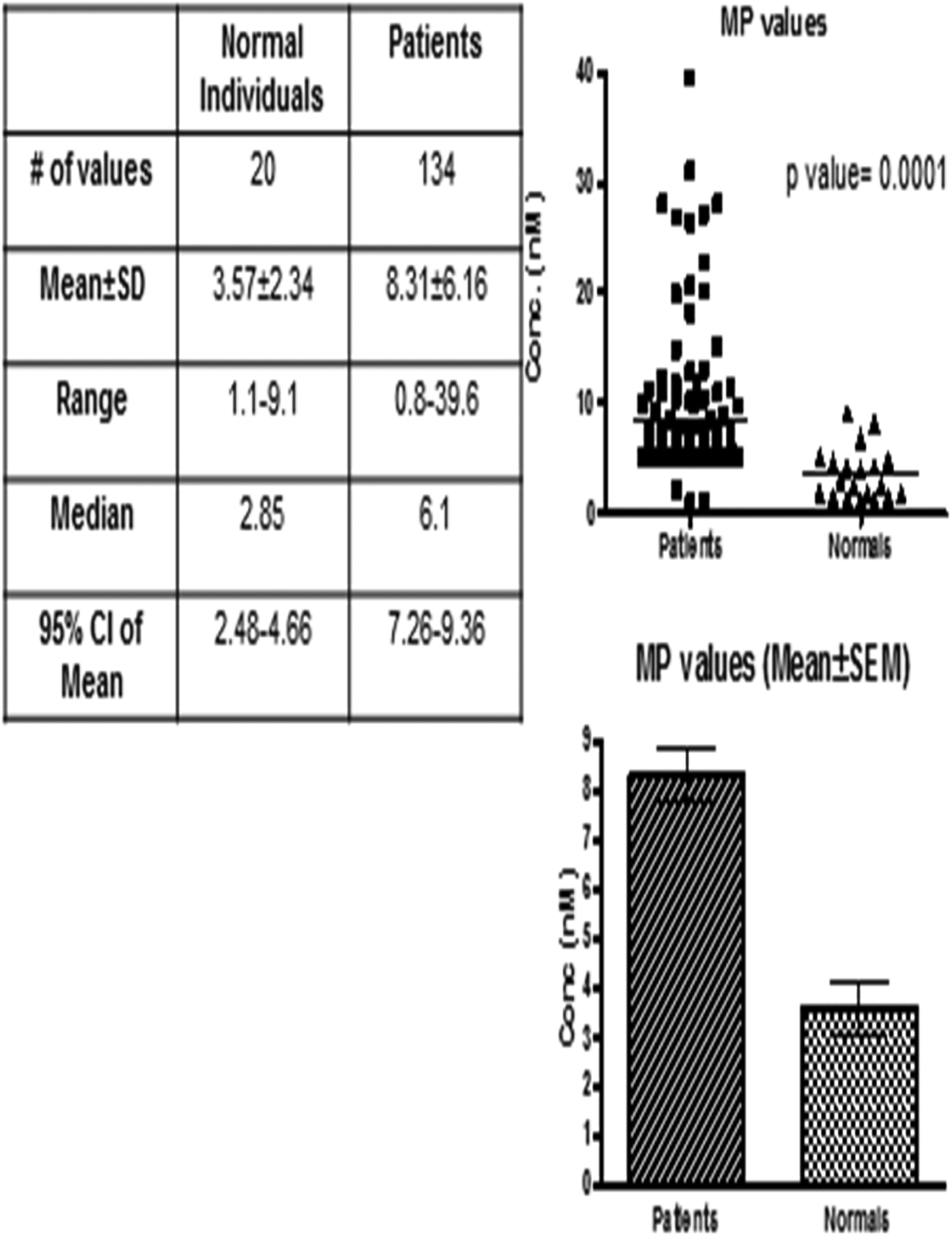
Microparticle titers in normal and patients with bladder tumor. Top right is a scattergram plotted against microparticle levels in patients versus normal individuals. Bottom right is a bar graph for the mean ± standard error of the mean (SEM) of patient group versus control group. The table shows statistical analysis (nonparametric Mann-Whitney U test) between patient and control groups.
Patients’ AGAs had an average value of 0.22 ± 0.1 optical density (OD; median: 0.2, confidence interval: 0.21-0.24, and P value: .0213) compared to normal individuals 0.25 ± 0.08 OD (median: 0.25 and confidence interval: 0.22-0.23; Figure 3).
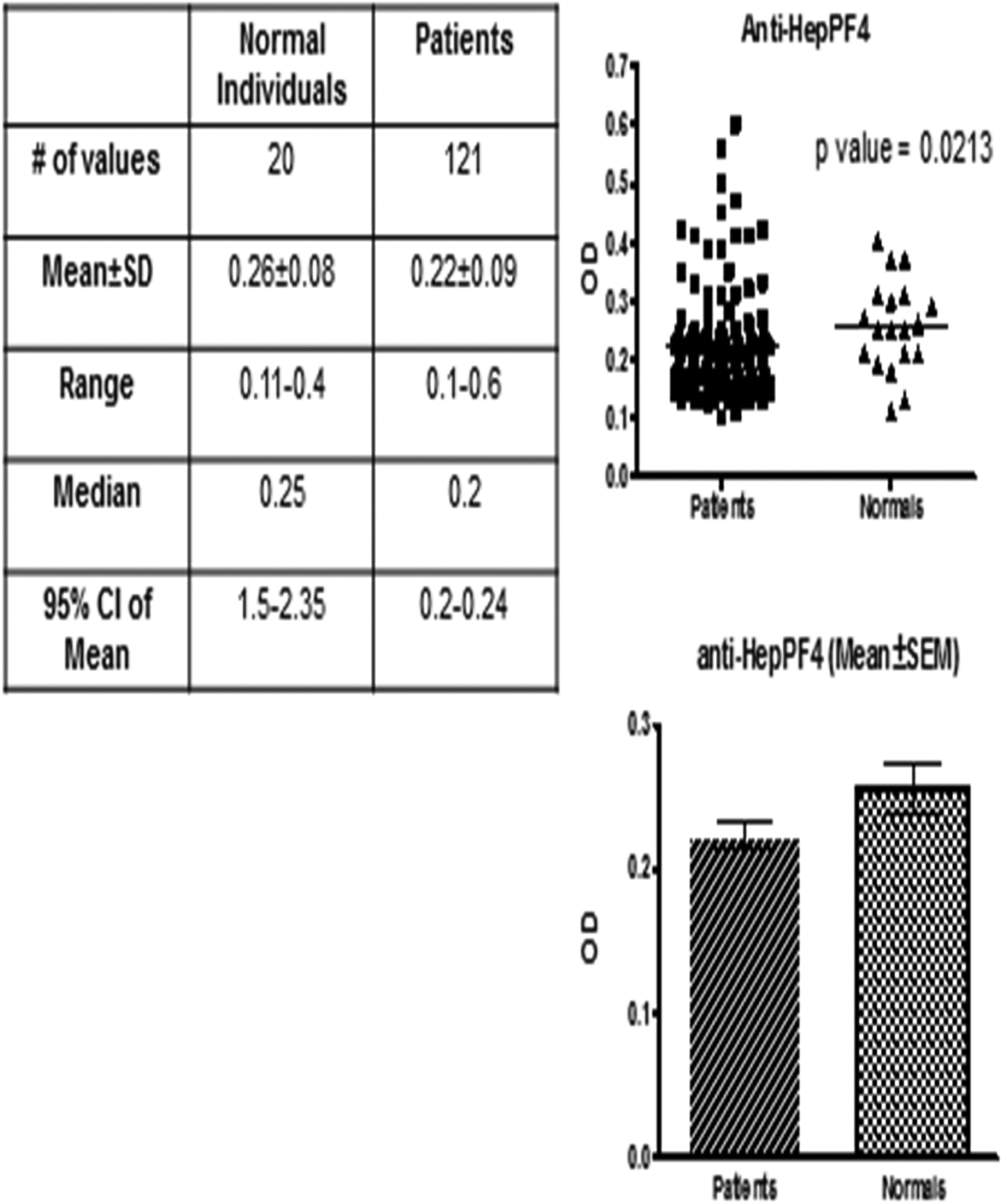
Antiglycosaminoglycan antibody titers in normal and patients with bladder tumor Top right is a scattergram plotted against antiglycoaminoglycan antibody levels in patients versus normal individuals. Bottom right is a bar graph for the mean ± standard error of the mean (SEM) of patient group versus control group. The table shows statistical analysis (nonparametric Mann-Whitney U test) between patient and control groups.
Antiglycosaminoglycan antibodies showed statistically significant correlation with MPs and APL antibodies. Antiglycosaminoglycan antibodies versus APL antibodies showed Spearman r value of .2364 (95% confidence interval: 0.05-0.4 and P value .009). Antiglycosaminoglycan antibodies versus MPs showed Spearman r of −0.195 (95% confidence interval: 0.37-0.01, P value .0321; Figure 4). However, no correlation was noted between MPs and APL antibodies.
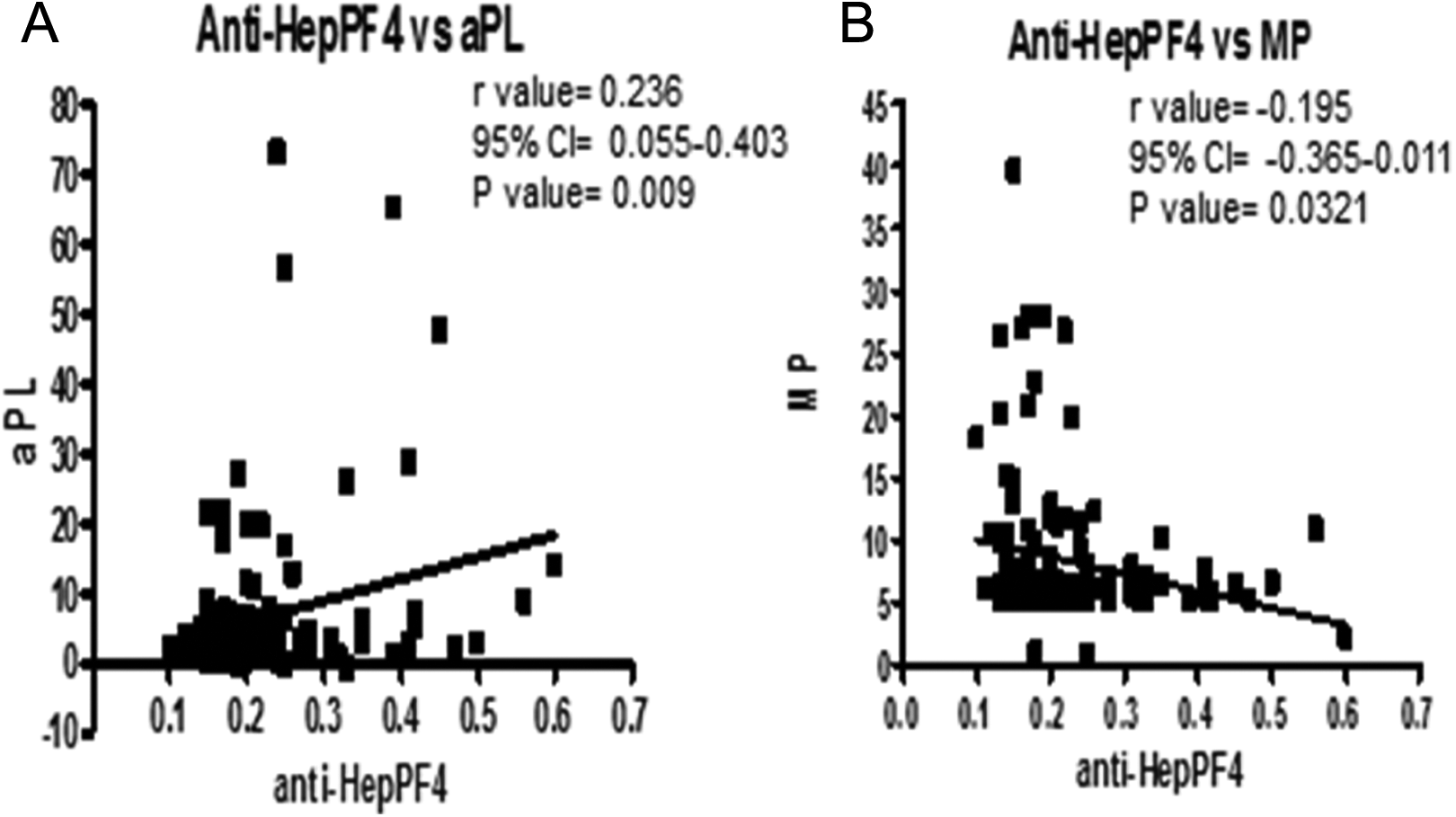
Panel A depicts the correlation between antiglycosaminoglycan antibody and antiphospholipid antibody, whereas Panel B depicts the correlation between antiglycosaminoglycan antibody and microparticle. Spearman r test was performed for the statistical analysis for the correlation.
Discussion
This study represents a retrospective analysis of banked plasma samples from the patietns with BC and commercially obtained normal plasma samples. The primary object of this study was to demonstrate the relative elevation in Tissue factor-microparticles (TF-MPs), AGAs, and antiphospholipid antibodies in the patients with BC in comparison to the normal individuals. The data are represented in terms of cumulative mean and intervals to determine the relative increase and distribution profile. Since the plasma samples were deidentified individuals, no additional analysis in terms of age, gender other clinical parameters, and outcome measures were performed. The primary objective was to demonstrate the relative elevation in biomarkers and any potential correlation between the biomarkers that are elevated in patients with BC.
It is well documented that MPs are highly procoagulant and are responsible for the thrombotic events in diseases such as cancer, sickle cell disease, and endotoxemia. 6,19 The MPs arise from plasma membranes (microvesicles), produced by exosomal pathway (exosomes), or generated from apoptotic cells. Various studies demonstrate that cancer cell also stimulates production of MPs, which are considered to be responsible for various functions, such as cancer cell survival, 20 –23 escapes from immune system, 24 –36 helps cancer in metastasizing, 37 –40 and involved in cancer-associated coagulopathy. 41 –43 In the following studies, the investigators found that TF-MPs in the patients with cancer contribute to thrombus formation and initiate coagulation by various pathways. 44 –47 However, there is a wide variation in the literature regarding the association of MPs with VTE seen in patients with cancer. Several investigators showed that there is significant increase in the MP-TF activity in patients having cancer with VTE. 48 –51 While Pabinger et al showed that there was an increase in MPs level in patients with pancreatic cancer when compared to normal but did not show association of MPs with VTE. 52 It is likely that the tissue factor upregulation in these patients stems from the cancerous cells. Immunohistochemical evaluation of the cancerous lesions may be useful in the staging of the cancer to demonstrate progression and possible regression due to therapeutic interventions. While such analysis was beyond scope of this study, simultaneous immunohistochemical studies were tissue factors and MPs subtype may be useful in the understanding of the pathogenesis of the BC.
On the other hand, circulating APL antibodies also showed increased levels in various thrombotic and vascular diseases 8 and may contribute to a procoagulant state. 53 –57 This role of APL antibodies was discovered from a clinical observation 58 and was confirmed later. 59,60 Several studies have shown the higher prevalence of APL antibodies in patients with cancer. 9,61–62 The increased production of APL antibodies may be due to the response of particular immune system pathway or immunotherapy of cancer. However, it is suggested that APL antibodies are not able to cause thrombosis alone and need a triggering factor (“Two hits Hypothesis”). 12 So even when high levels of APL antibodies are circulating in the body, a clinical manifestation would be rare. Opatrny and Warner found that the heparin–PF4 antibodies were significantly high in patients with cancer when compared to noncancer patients, and there was increase in number of deaths related to thromboembolic events in HIT diagnosed patients with cancer when compared to patients having cancer without positive HIT antibodies. However, there was no significant increase in the overall mortality among 2 groups. 63 The AGAs were measured in the normal individual and patients with BC using ELISA method. The antibody titer was expressed in terms of OD which is directly proportional to these antibodies. The average value in both populations was below assay threshold for positive antibodies. However, 12 of the 121 patients showed positive titers in the patient group, and none of the 20 patients have positive titer. In addition, there was a wide scatter in the patient population in comparison to the normal individuals. One of the limitations of these data are the lower number normal individuals in comparison with patients. Regardless of this, the wide scatter of the AGA, and the presence of nearly 10% of the AGA in the patient group are consistent with the reported data in patients with cancer. 18
In our study, we also showed that there is an inverse relation between MP and AGA levels. This is due to the fact that there is an activation of platelets and formation of platelet MPs. Due to binding of antibodies to the platelet, their circulating levels decrease. On the other hand, more platelets get activated and release more MPs in the blood. We also showed that there is a positive correlation between AGAs and APL antibodies. This may be due to the cross-reactivity between these 2 antibodies. 64 But we are not sure about the extent of the cross reactivity and suggest more thorough research on this area.
Our study supports the concept of that patient with cancer has upregulation of MPs, APL antibodies, and AGAs. Obviously, these patients with BC have a subclinical activation of both thrombotic and inflammatory processes. The procoagulant MPs may be generated due to cellular tissue damage or by cancer cells, whereas the APL antibodies and AGAs represent more of a vascular endothelial damage and activation. The generation of these biomarkers in the patient with BC needs to be investigated thoroughly. This may provide additional information on the adverse thrombotic events which in turn influence the outcome and affect the overall prognosis of patients with BC.
Upregulation of thrombotic and inflammatory mediators is generally associated with progression of cancer and may also be related to the degree of metastasis. Simultaneous evaluation of the metastasis and tumor size along with the quantitation of the biomarkers such as the one reported in this study may provide additional useful information. The relevance of these biomarkers to the clinical outcome has not been addressed, and only limited data are available. Thus, long time survival rate and additional data on the tumor progression and the metastasis may provide further understanding of the pathogenesis and the value of the biomarkers in risk stratification in these patients.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
