Abstract
Heart failure (HF) is the leading cause of in-hospital morbidity and mortality in the elderly population. Coexistence of HF and atrial fibrillation (AF) increases the risk of thromboembolic events. Oral anticoagulant therapy reduces the risk of thromboembolic events in patients with AF. Novel oral anticoagulants (NOACs) have been introduced as an alternative drug for prevention from thromboembolic events in patients with nonvalvular AF. The primary aim of this study is to investigate the clinical effects of warfarin, dabigatran, and rivaroxaban in patients with nonvalvular AF. The secondary aim of this study is to reveal the predictors of all-cause mortality in patients with nonvalvular AF undergoing NOACs therapy. The study population consisted of 171 patients with nonvalvular AF. Patients were divided into 3 groups according to the usage of oral anticoagulant therapy including coumadin (51 patients), dabigatran (52 patients), and rivaroxaban (68 patients). Although CHA2DS2-VASc score was similar between groups, HAS-BLED score was significantly higher in patients using rivaroxaban. Dyspepsia and itching were more common in patients using dabigatran. Heart failure and vascular disease were more common in the nonsurviving group (10 patients) than in the surviving group (110 patients) in patients using NOACs. Among age, sex, HF, hypertension, vascular disease, and CHA2DS2-VASc, which were included in the regression model, only the presence of HF was an independent predictor of all-cause mortality in patients using NOACs. In conclusion, the mortality rate is significantly higher in patients with HF using NOACs. Moreover, HF is an independent predictor of all-cause mortality in patients using NOACs.
Introduction
Heart failure (HF) is the leading cause of in-hospital morbidity and mortality in the elderly population. 1,2 Atrial fibrillation (AF) is the most common cardiac arrhythmia in HF, as HF and AF often trigger each other. 3 Due to increased risk of cardiovascular morbidity and mortality, treatment of patients with HF and AF is crucial. Coexistence of HF and AF increases the risk of thromboembolic events. 4,5 According to clinical stroke risk scoring system including CHA2DS2-VASc, life-long oral anticoagulation should be recommended in patients with HF having AF. 6 Evidence-based data suggest that oral anticoagulant therapy reduces the risk of thromboembolic events in patients with AF. 5 Therefore, oral anticoagulant therapy is recommended to prevent from thromboembolic events in patients with AF at moderate to high risk for thromboembolic events. Thromboembolic events are also more common in patients with HF. 7 Decreased mobilization, impaired hemodynamic status, and chronic inflammation are among the reasons for thromboembolic events in those patients with HF. Previous study showed that every 5% deterioration in left ventricular ejection fraction (LVEF) leads to an 18% increased risk of cerebrovascular event. 8 Current guidelines do not suggest the oral anticoagulant therapy in patients having HF without AF, although several trials tested the hypothesis. 9
Warfarin has been the gold standard for thromboembolism prophylaxis in patients with AF. However, difficulties in the careful monitoring of warfarin-based anticoagulation and increased interactions with various food and drugs are important handicaps for patients with AF, especially in the elderly population. 10 Novel oral anticoagulants (NOACs) including dabigatran, rivaroxaban, and apixaban have been introduced as an alternative drug for prevention from thromboembolic events in patients with nonvalvular AF. 11 –13 Novel oral anticoagulants have minimal food and drug interactions, rapid onset of effect, and a more stable anticoagulant effect without the need for laboratory monitoring. The overall benefits of NOACs for stroke or systemic embolism prevention, and major and intracranial bleeding, relative to warfarin in the Randomized Evaluation of Long-Term Anticoagulation Therapy (RE-LY) trial were consistent in patients with and without HF. 14 Although there are randomized trials comparing NOACs to warfarin, single-center experience can inform us on individual risk patterns, characteristics of NOACs use in real-life practice.
The primary aim of this study is to investigate the clinical effects of warfarin, dabigatran, and rivaroxaban in patients with nonvalvular AF. The secondary aim of this study is to reveal the predictors of all-cause mortality in patients with nonvalvular AF undergoing NOACs therapy.
Methods
Study Population
Two-hundred fifty consecutive patients with newly diagnosed AF were enrolled into the study. We included those patients in whom full data were available to evaluate the risk of thromboembolic and bleeding complications according to the current scores, along with information on the prescribed anticoagulant treatment. Patients with valvular AF, history of anticoagulant usage, history of malignancy, and no follow-up data were excluded from the study. Forty-seven patients were excluded for the presence of valvular heart disease, 10 patients were excluded for history of previous anticoagulant usage, 5 patients were excluded for malignancy, and 17 patients were excluded for the lack of follow-up data. After exclusion criteria, 171 patients with nonvalvular AF were included into the analysis.
Thromboembolic risk was evaluated using the CHA2DS2-VASc score, and bleeding risk was evaluated using the HAS-BLED score. 15,16 Valvular AF was defined as AF in patients who previously underwent any intervention, either percutaneous or surgical, due to valvular disease or patients in whom intervention was currently indicated for valvular disease. The CHA2DS2-VASc score was calculated as follows: presence of each following items including congestive HF, hypertension, diabetes, age 65 to 74 years, female gender, coronary artery disease, and peripheral vascular disease are 1 point and presence of each following items including age ≥75 years and prior ischemic stroke are 2 points. The CHA2DS2-VASc score was categorized into 3 levels according to total points of patients as 0 (low risk), 1 (intermediate risk), and ≥2 (high risk). The HAS-BLED bleeding risk score was calculated as follows: presence of each following items including hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile international normalized ratio (INR), elderly population, and drugs/alcohol concomitantly are 1 point. Heart failure was defined according to the current guideline. 17 Patients were followed from the time of anticoagulant initiation and continued until 1 year.
The investigation complied with the principles outlined in the Declaration of Helsinki. The study was approved by the local ethics committee, and all participants gave written informed consent.
Statistical Analysis
Statistical analyses were performed using SPSS 16.0 statistical package for Windows. Continuous data were expressed as the mean ± standard deviation. The χ2 test was used for comparison of the categorical variables. Statistical comparisons of quantitative data were performed using Student t test, Kruskal-Wallis, or Mann-Whitney U test. Logistic regression analyses were performed to determine the predictors of all-cause mortality in patients using NOACs. A value of P < .05 was considered statistically significant.
Results
The study population consisted of 171 patients with nonvalvular AF. Patients were divided into 3 groups according to the usage of oral anticoagulant therapy including coumadin (51 patients), dabigatran (52 patients), and rivaroxaban (68 patients). Comparison of clinical characteristics of study population is shown in Table 1. Age and sex distribution were similar between the groups (P = .076 and .079, respectively). Biochemical parameters and cardiovascular risk factors were also similar between the groups. Although the CHA2DS2-VASc score was similar between the groups, the HAS-BLED score was significantly higher in patients using rivaroxaban (P = .014). Comparison of medical therapy of study population is shown in Table 2. Medical therapies were similar among the 3 groups.
Comparison of Clinical Characteristics of Study Population.a
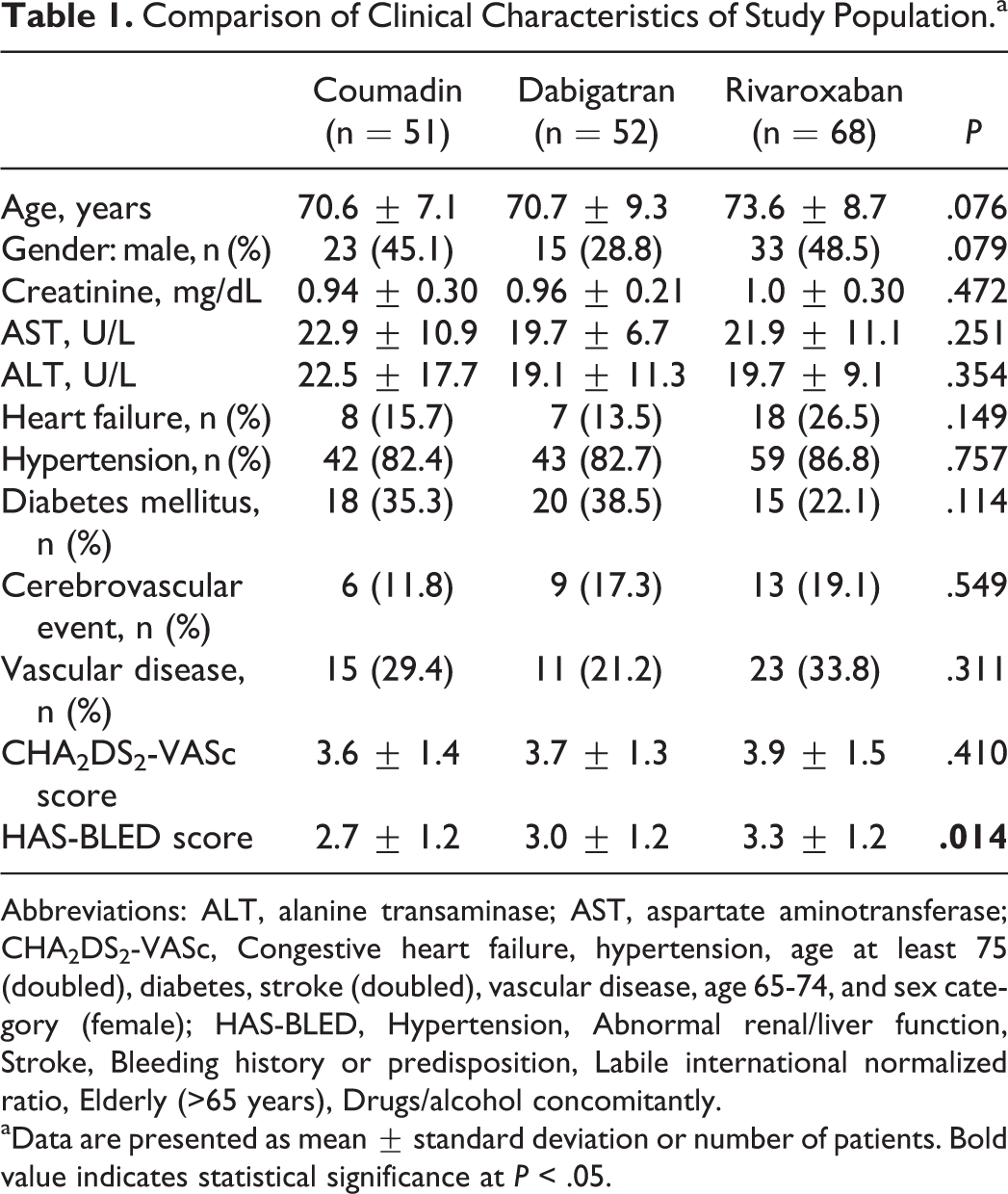
Abbreviations: ALT, alanine transaminase; AST, aspartate aminotransferase; CHA2DS2-VASc, Congestive heart failure, hypertension, age at least 75 (doubled), diabetes, stroke (doubled), vascular disease, age 65-74, and sex category (female); HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly (>65 years), Drugs/alcohol concomitantly.
aData are presented as mean ± standard deviation or number of patients. Bold value indicates statistical significance at P < .05.
Comparison of Medical Therapy of Study Population.
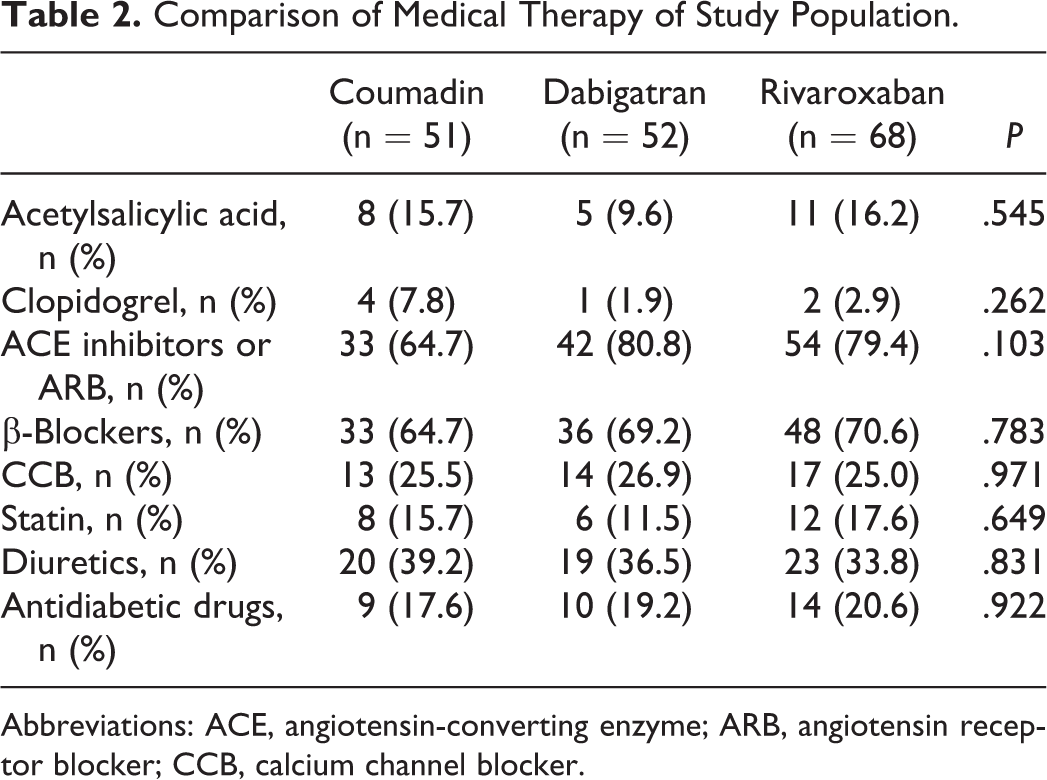
Abbreviations: ACE, angiotensin-converting enzyme; ARB, angiotensin receptor blocker; CCB, calcium channel blocker.
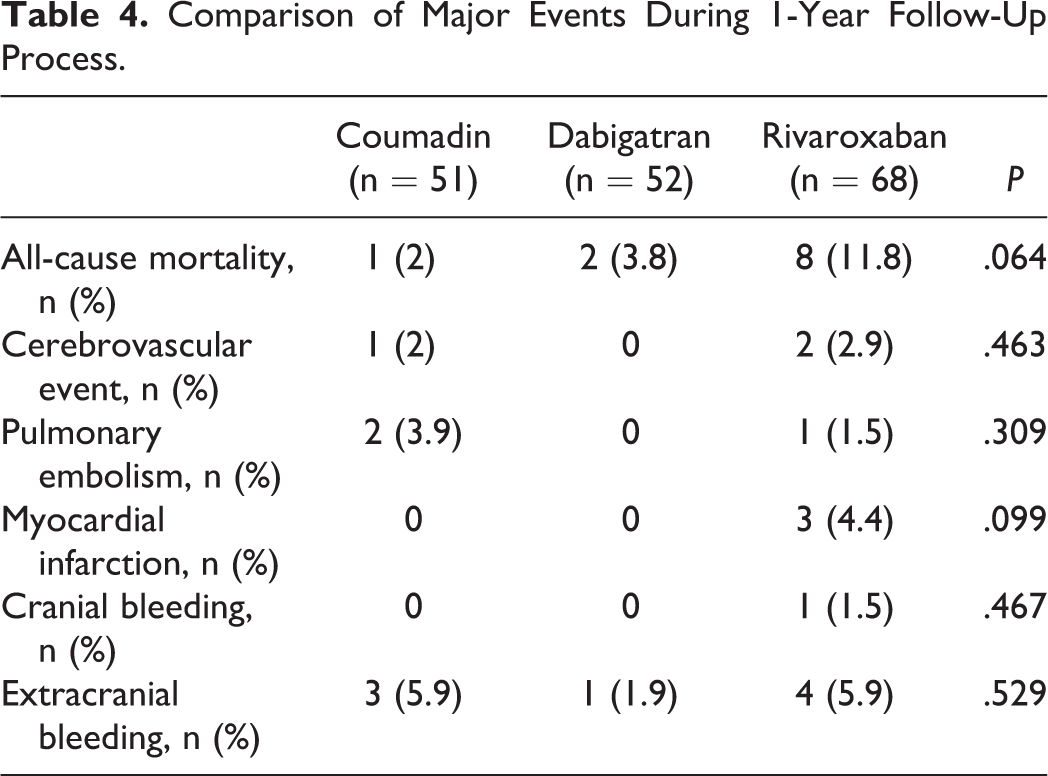
The study population was followed up approximate a year via telephone calling. Comparison of minor events during 1-year follow-up process is shown in Table 3. Dyspepsia was significantly more common in patients using dabigatran (P = .029). Itching was also more common in patients using dabigatran without reaching statistical significance (P = .054). Comparison of major events during 1-year follow-up process is shown in Table 4. Patients were divided into 2 groups according to the presence of HF. Comparison of clinical characteristics of the study population is shown in Table 5. Age distribution was similar among the groups. Creatinine and alanine transaminase levels were significantly higher in patients with HF. The presence of vascular disease was significantly more common in patients with HF (48.5% vs 23.9%, P = .005). Although the HAS-BLED score was similar between the groups, the CHA2DS2-VASc score was significantly higher in patients with HF (4.5 ± 1.3 vs 3.6 ± 1.4, P = .001).
Comparison of Minor Events During 1-Year Follow-Up Process.a
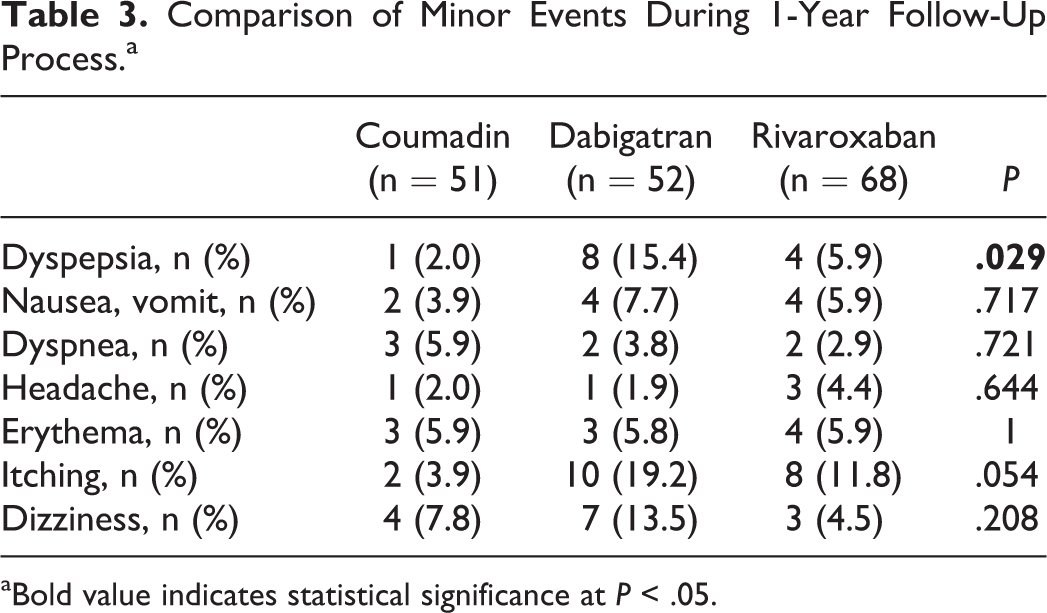
aBold value indicates statistical significance at P < .05.
Comparison of Major Events During 1-Year Follow-Up Process.
Comparison of Clinical Data Between Patients With and Without HF.a
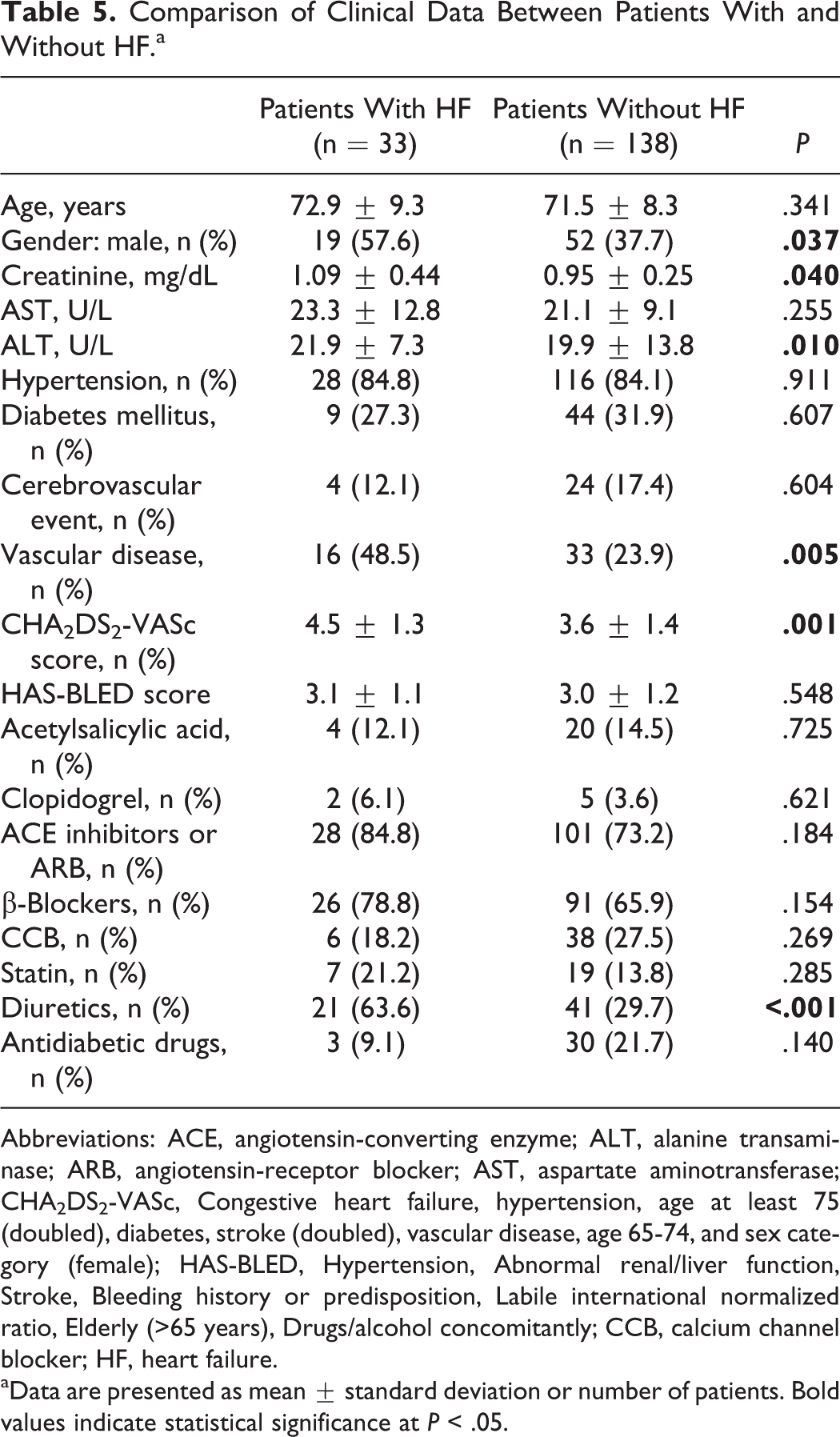
Abbreviations: ACE, angiotensin-converting enzyme; ALT, alanine transaminase; ARB, angiotensin-receptor blocker; AST, aspartate aminotransferase; CHA2DS2-VASc, Congestive heart failure, hypertension, age at least 75 (doubled), diabetes, stroke (doubled), vascular disease, age 65-74, and sex category (female); HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly (>65 years), Drugs/alcohol concomitantly; CCB, calcium channel blocker; HF, heart failure.
aData are presented as mean ± standard deviation or number of patients. Bold values indicate statistical significance at P < .05.
Comparison of clinical data between the surviving (110 patients) and the nonsurviving (10 patients) groups in patients using NOACs is shown in Table 6. Age and sex distribution were similar between the groups (P = .241 and .519, respectively). Heart failure and vascular disease were more common in the nonsurviving group than in the surviving group in patients using NOACs (80% vs 15.5% and 60% vs 25.5%, P < .001 and .030, respectively). The CHA2DS2-VASc score and the HAS-BLED score were similar between the groups (P = .192 and .651, respectively). Logistic regression analysis was performed to determine the predictors of all-cause mortality in patients using NOACs (Table 7). Among age, sex, HF, hypertension, vascular disease, and CHA2DS2-VASc, which were included in the regression model, only the presence of HF was an independent predictor of all-cause mortality in patients using NOACs (r 2 of the model = .383; P = .001).
Comparison of Clinical Data Between the Surviving and Nonsurviving Groups in Patients Using NOAC.a
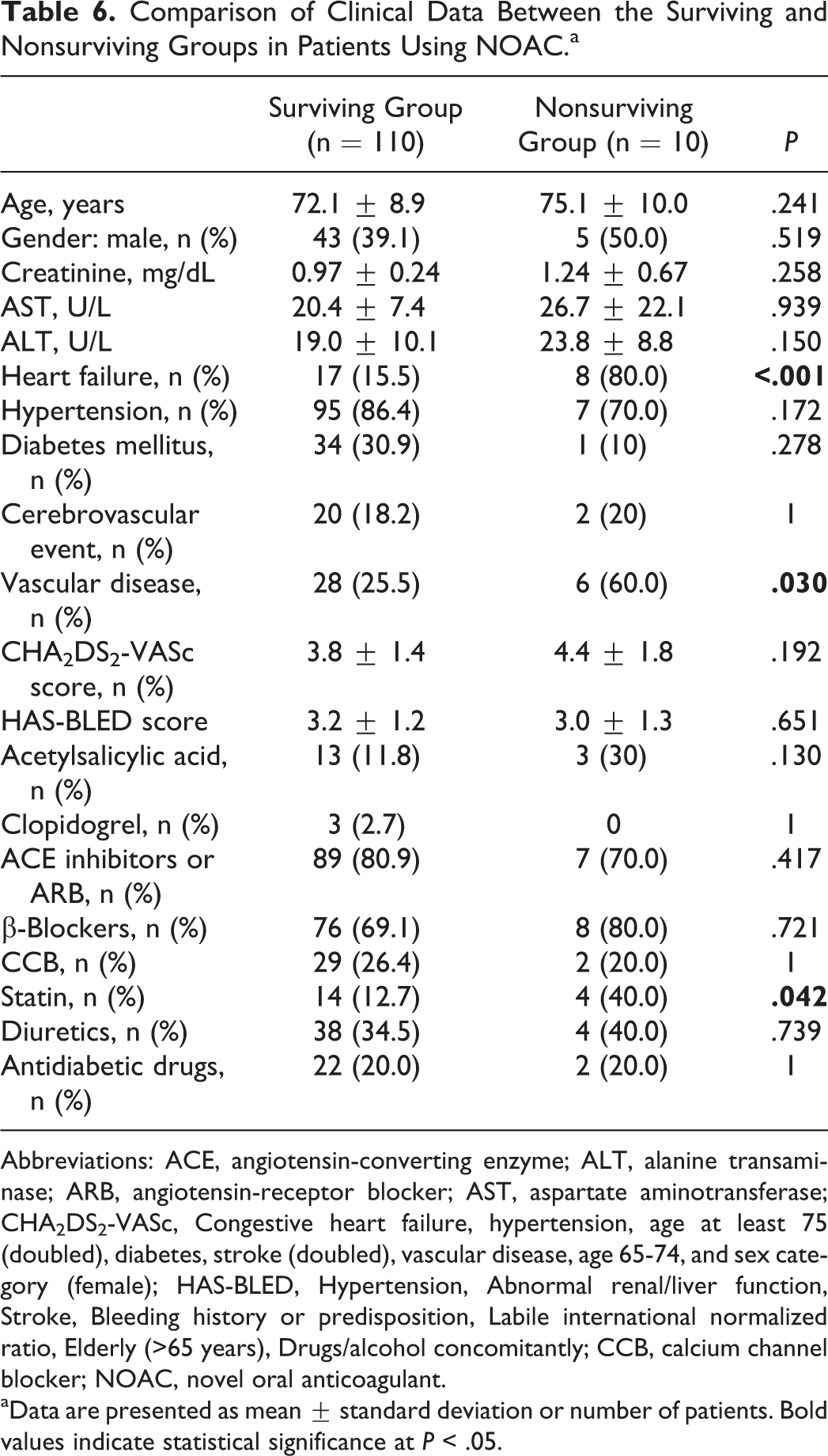
Abbreviations: ACE, angiotensin-converting enzyme; ALT, alanine transaminase; ARB, angiotensin-receptor blocker; AST, aspartate aminotransferase; CHA2DS2-VASc, Congestive heart failure, hypertension, age at least 75 (doubled), diabetes, stroke (doubled), vascular disease, age 65-74, and sex category (female); HAS-BLED, Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile international normalized ratio, Elderly (>65 years), Drugs/alcohol concomitantly; CCB, calcium channel blocker; NOAC, novel oral anticoagulant.
aData are presented as mean ± standard deviation or number of patients. Bold values indicate statistical significance at P < .05.
Multivariate Logistic Regression Analysis to Determining the Independent Predictors of Mortality in Patients Using NOAC.a
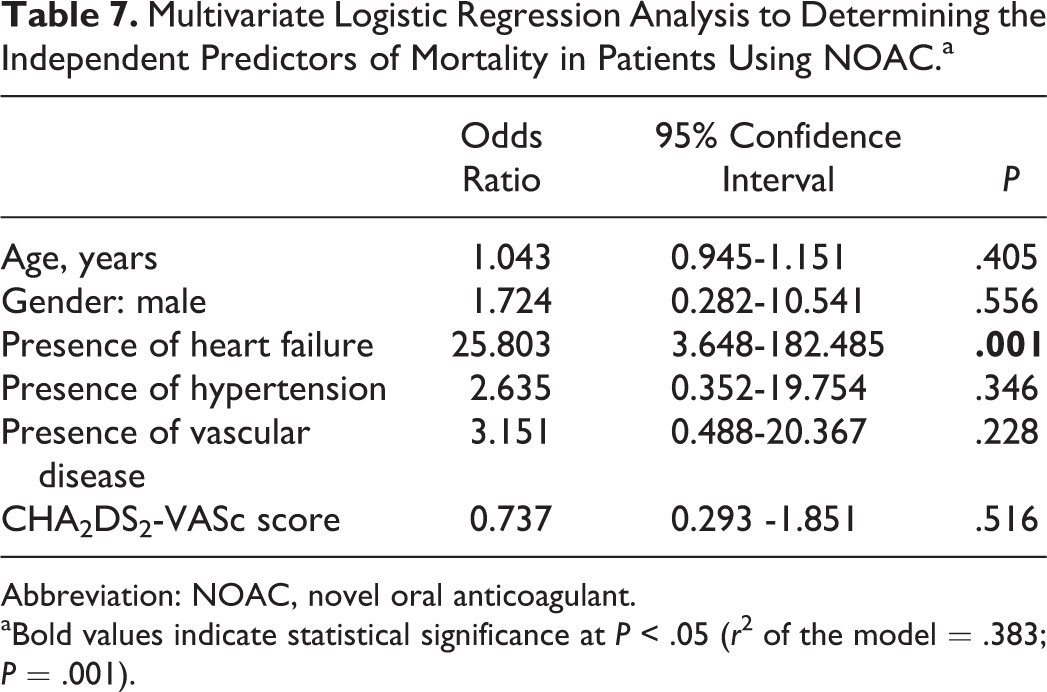
Abbreviation: NOAC, novel oral anticoagulant.
aBold values indicate statistical significance at P < .05 (r 2 of the model = .383; P = .001).
Discussion
In this study, we investigated single-center experience of NOACs use in major tertiary care center from Istanbul. Our aim is to analyze the outcome, characteristics of patients, and factors affecting outcome after NOACs use in developing countries. Although there are randomized trials comparing NOACs to warfarin, single-center experiences can elucidate individual risk patterns, characteristics of NOACs use in real-life practice. For instance, we observed nearly 10% mortality in 1 year in patients who are started on NOACs. In addition, HF and vascular disease were more common in the nonsurviving group than in the surviving group in patients using NOACs. Moreover, we first demonstrated that the presence of HF is an independent predictor of all-cause mortality in patients using NOACs.
Novel oral anticoagulants have been introduced as an alternative treatment choice to warfarin. Numerous studies have demonstrated the efficacy and safety of NOACs in large population subsets. 18 –22 Although patient compliance is better with NOACs than warfarin, lack of long-term follow-up data of patients using NOACs and insufficient clinical experience in developing countries with heavy patient loads are problems, especially in the elderly population. The clinical usage of NOACs started with the favorable results of the 2 randomized studies. 11,12 In RE-LY study, 11 dabigatran 110 mg/150 mg versus warfarin, both dabigatran doses were found to be noninferior to warfarin with respect to the primary efficacy outcome of stroke or systemic embolism. Dyspeptic symptoms were also more common with both dabigatran doses when compared to warfarin. In the Rivaroxaban Once Daily Oral Direct Factor Xa Inhibition Compared with Vitamin K Antagonism for Prevention of Stroke and Embolism Trial in Atrial Fibrillation (ROCKET AF) study, 12 rivaroxaban 15 mg/20 mg versus warfarin, rivaroxaban was found to be noninferior to warfarin for the prevention of stroke or systemic embolism.
In light of the published trials, there are several explanations about our results. First, our study was observational, and baseline differences in patient groups can explain the differences. For instance, patients using rivaroxaban were older than others. Second, the HAS-BLED score was significantly higher in patients using rivaroxaban than in patients using warfarin and dabigatran. In addition, HF was also more common in patients using rivaroxaban without reaching statistical significance (26.5% for rivaroxaban, 13.5% for dabigatran, and 15.7% for warfarin, P = .149). Therefore, all-cause mortality was more common in patients using rivaroxaban, likely due to baseline differences. In a previous study, Hohnloser et al 23 reported that there was a nonsignificant increase in myocardial infarction with dabigatran compared with warfarin. Annual rates of myocardial infarction was 0.82% and 0.81% with dabigatran 110 or 150 mg compared to 0.64% with warfarin (P = .09 and 0.12, respectively). A previous randomized study 24 demonstrated that the twice-daily 2.5 mg dose of rivaroxaban reduced the rates of death from cardiovascular causes (2.7% vs 4.1%, P = .002) and from any cause (2.9% vs 4.5%, P = .002).
Heart failure is a common cause of cardiovascular morbidity and mortality in the elderly population. Due to increased risk of thromboembolic events in patients with HF, anticoagulant therapy may be crucial. Antiplatelet Therapy in Chronic HF (WATCH) study 25 demonstrated that warfarin is associated with fewer nonfatal strokes than aspirin or clopidogrel in patients with systolic HF (LVEF: ≤35% and sinus rhythm). In another study, Homma et al 26 showed that treatment with warfarin did not affect the primary outcome of ischemic stroke, intracerebral hemorrhage, or death from any cause as compared with 325 mg dose of aspirin in patients with reduced LVEF and sinus rhythm. However, treatment with warfarin resulted in a significant reduction in ischemic stroke, although the rate of major bleedings was almost 2-fold higher in the warfarin group, with no difference in intracerebral and intracranial bleedings. According to subgroup analysis of ROCKET AF study, the risk of major or nonmajor clinically relevant bleeding was similar in patients with HF (62.5% of the study population) and without HF. A reduction in hemorrhagic stroke was observed with rivaroxaban in patients with HF. The efficacy of rivaroxaban was comparable among patients with LVEF <40% or ≥40% and patients with different severity of HF symptoms. According to subgroup analysis of RE-LY study, dabigatran demonstrated similar clinical effects on development of stroke or systemic embolism and major bleeding among patients with and without HF. In our study, 33 of the 171 patients had HF. The mortality rate was significantly higher in patients with HF (24.2% vs 2.2%, P < .001). We also investigated the predictors of all-cause mortality in patients with nonvalvular AF using NOACs. We found that only the presence of HF was an independent predictor of all-cause mortality in patients using NOACs.
Approximately 80% of cardiovascular death occurs in low- and middle-income countries. Yet, most clinical trials are performed in developed countries, and practice patterns may differ significantly among populations. Current guidelines which are based on randomized controlled trials recommend NOACs as they do not require INR follow-up. Yet, such guidelines do not benefit every patient. Recent studies question the “one size fits all” principle particularly in complex traits such as thromboembolism in AF. Evaluation of the individual patient with his or her environment, detailed medical history, and compliance to treatment is crucial in preventing complications from antithrombotic embolization in AF.
Study Limitations
Our study is a single-center study and comes with several limitations including small sample size and short follow-up period of 1 year which is not sufficient to draw definite conclusions in a chronic condition such as AF. The study was observational, and baseline differences in patient groups can at least partially explain the observed differences. Several center- and country-specific factors can affect the study outcome. For instance, our study population did not receive apixaban therapy. Second, NOACs are prescribed with the implication that these agents do not require INR follow-up. In several countries, this implication can dangerously be translated into a message that patients do not require close follow-up after being started on NOACs. Furthermore, in Turkey, outpatient clinics have a tremendous patient load, enabling the physician less than 10 minutes to conclude the examination. Lack of specialty clinics such as HF clinic in major institutions can also affect long-term outcome. This may be particularly detrimental for patients with coexistent comorbidities such as HF.
Conclusions
Developing countries face the challenges of containing costs and health expenditure with novel therapeutics such as NOACs. On the other hand, public present an increasing demand for the accessible and high-quality health care. Current guidelines which are based on randomized controlled trials direct the cardiovascular diagnosis and treatment. Yet, patient outcomes from individual centers can lead the clinician a more informed view. Our findings of nearly 10% mortality after start of NOACs and increased mortality in patients with HF should be evaluated in this context. In subspecialty clinics (ie, anticoagulation and HF), allowance of longer time for physicians to educate their patients may help developing countries to embrace novel treatment options in a more effective manner. Further prospective large scale studies are needed to demonstrate the exact efficiency and safety of NOACs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
