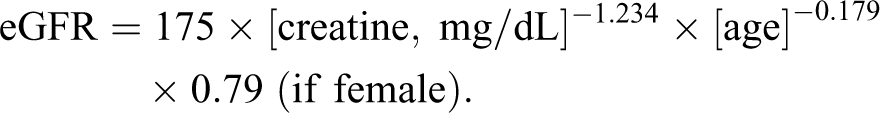Abstract
We investigated the risk of stroke associated with renal dysfunction and the impact of warfarin therapies in Chinese patients with nonvalvular atrial fibrillation (NVAF). Information was collected on age, sex, height, weight, type of atrial fibrillation, and serum creatinine within the previous 6 months, together with the variables needed to calculate the CHADS2 score. For patients not taking warfarin, reasons why not were recorded. Three thousand seventeen eligible patients with NVAF, mean (1 standard deviation [SD]) age of 67.7 (13.0) years, from 50 Chinese hospitals were included from May 2012 to October 2012, with a mean (1SD) CHADS2 score of 2.0 (1.5). Of these, 58.3% were male and 86.2% were at high risk of stroke with a CHADS2 score ≥1. Only 42.6% were on warfarin, and 22.5% of the patients had moderate or severe renal impairment (estimated glomerular filtration rate < 60 mL/min/1.73 m2). After adjustment for the CHADS2 score, renal dysfunction remained moderately but significantly associated with the risk of stroke/TIA (odds ratio = 1.005, 95% confidence interval: 1.002-1.009, P = .002). There was, however, no significant difference in anticoagulant usage between patients with or without impaired renal dysfunction. The most common anticoagulant concerns were the low proportion of patients with regular international normalized ratio monitoring (43.0%) and the risk of bleeding (33.3%). Renal impairment was common and independently associated with the risk of cerebrovascular embolism in Chinese patients with NVAF but not independently related to underuse of anticoagulant treatment.
Anticoagulation therapy is strongly recommended by Chinese expert consensus 1 for patients at moderate to high risk of stroke, defined by their CHADS2 score and CHA2DS2-VASc score in European guidelines. 2 The first step in the prevention of atrial fibrillation (AF)-related stroke is effective risk stratification. Although CHADS2 and CHA2DS2-VASc have been well adopted internationally, they have limited discriminatory power 3,4 and account for little more than half the attributable risk, indicating that other important predictive factors remain undefined. Atrial fibrillation and chronic kidney disease (CKD) frequently coexist. Patients with CKD have increased risk of cardiovascular (CV) events and mortality, including stroke. 5,6 Observational studies have showed that renal dysfunction is an independent risk factor for mortality and intracranial bleeding but not for thromboembolic (TE) events after adjustment of established risk factors. 7 –9 Furthermore, there was no evidence to show the association between renal failure and subtypes of stroke, which is probably important for the prevention of cardioembolic stroke in patients with nonvalvular atrial fibrillation (NVAF). Therefore, it is uncertain whether including moderate to severe CKD as a risk factor adds predictive value for cardioembolic stroke events in patients with NVAF.
Renal dysfunction is a key factor influencing the choice of anticoagulant for the prevention of stroke and TE events. A cross-sectional study showed that half of the patients with increased risk of venous thromboembolism had moderate or severe renal failure, but most of them did not receive the optimal type or dose of antithrombotic treatment adjusted by renal function. 10 Patients with impaired renal function have a high risk of major bleeding. For patients on dialysis requiring anticoagulation, warfarin would appear to be a good choice as it is metabolized by the liver and not by the kidneys. Warfarin, however, did not reduce the risk for stroke in patients with AF on dialysis. 11 There are no data to indicate whether there is a degree of anticoagulation at different levels of renal dysfunction at which the risk of bleeding may outweigh the benefit. On the other hand, new oral anticoagulants (NOACs) have been shown to be at least as effective and safer compared with warfarin in large-scale phase 3 trials. 12 Renal function should be monitored carefully in all patients taking NOACs, irrespective of the type of NOACs. There are few data available for Chinese patients with renal dysfunction and NVAF.
This study reports the first analysis examining the association between renal dysfunction, as measured by estimated glomerular filtration rate (eGFR), and the risk of TE events in a large real-world Chinese population with NVAF. We also investigated the impact of renal dysfunction on the use of anticoagulation treatment and reasons for underuse of anticoagulant therapy.
Methods
Setting
The study was conducted in outpatients from 50 hospitals in China between June 2012 and October 2012. Hospitals were selected according to the economic regions defined by China government to ensure a representative population distribution. The study was approved by the Peking University People’s Hospital Review Committee, and all participants gave written informed consent.
Study Population and Data Collection
Outpatients aged 18 years or older were included if they had diagnosed NVAF. Exclusion criteria were patients without serum creatinine data, patients with rheumatic or infective valvular heart disease, and patients presenting with AF due to a transient cause, for example, perioperative, electrolyte disturbance, and infection.
An anonymous questionnaire was designed to collect the following information: age, sex, height, weight, type of AF, and serum creatinine within the previous 6 months. The variables needed to calculate the CHADS2 score were also collected, including heart failure, hypertension, diabetes, and stroke or transient ischemic attack (TIA). At the same visit, concomitant medications and contraindications for the use of warfarin were recorded. The CHADS2 score was calculated with a score of 1 for congestive heart failure, hypertension (blood pressure consistently >140/90 mm Hg or on antihypertensive medication), age ≥75 years, diabetes mellitus, previous stroke, or TIA and 2 for thromboembolism.
New-onset AF was defined as AF occurring during the previous 6 weeks. Paroxysmal AF was defined as an episode terminating spontaneously, usually within 48 hours but within not more than 7 days. Persistent AF was defined as an episode lasting >7 days or requiring termination by cardioversion. Permanent AF was considered present when interventions to restore sinus rhythm were no longer considered appropriate (regardless of the reason).
The 10 options provided as reasons why patients were not taking warfarin at the survey visit were low stroke risk, high bleeding risk, had an indication of antiplatelet treatment other than AF, international normalized ratio (INR) monitoring unavailable, unwilling to take an INR test regularly, poor INR control, patient decision, history of bleeding on warfarin, contraindication of warfarin, and other. Each patient was interviewed by a trained investigator in the outpatient clinic at each site.
Renal Function Assessment
The eGFR was estimated from serum creatinine values with Modification of Diet in Renal Disease (MDRD) equation. The result is expressed in mL/min/1.73 m2. 13
Renal dysfunction was categorized using the current International Society of Nephrology’s Kidney Disease Improving Global Outcomes (KDIGO) recommendations 14 as normal renal function (CKD stages 1 and 2, defined as eGFR > 60 mL/min/1.73 m2), moderate dysfunction (CKD stage 3, defined as eGFR > 30 but ≤60 mL/min/1.73 m2), severe dysfunction (CKD stage 4, defined as eGFR > 15 but ≤30 mL/min/1.73 m2), and end-stage renal disease (CKD stage 5, defined as eGFR ≤ 15 mL/min/1.73 m2).
Statistics
Means and standard deviations (SDs) or medians and interquartile ranges (IQRs) were calculated for continuous variables as appropriate. Frequencies and percentages were calculated for categorical variables. T tests were used for continuous variables with normal distribution or for log-transformed variables with a nonnormal distribution. Categorical variables were analyzed using χ2 tests. Logistic regression (LR; backward LR) was used to identify independent risk factors related to the use of warfarin and for reduced renal function. A P value of <.05 was considered as statistically significant. All analyses were performed using SPSS Statistics version 19.0 (SPSS, Inc, Chicago, Illinois).
Results
Patient Characteristics
Three thousand two hundred seventy-nine eligible patients were recruited from 50 hospitals in Mainland China. Of these, 57 were excluded because one or more of the following essential data items was unavailable: age, sex, type of AF, and type of antithrombotic treatment. A further 105 patients were excluded because a serum creatinine measurement was not available during the previous 6 months (Figure 1). Of the 3017 (92%) patients analyzed, most of them (2526 of the 3017) were from tertiary hospitals. The patient population was roughly homogeneously distributed across China, with 59.2% in the east regions, 8.9% in the northeast regions, 14.7% in the west regions, and 17.3% in the central regions.
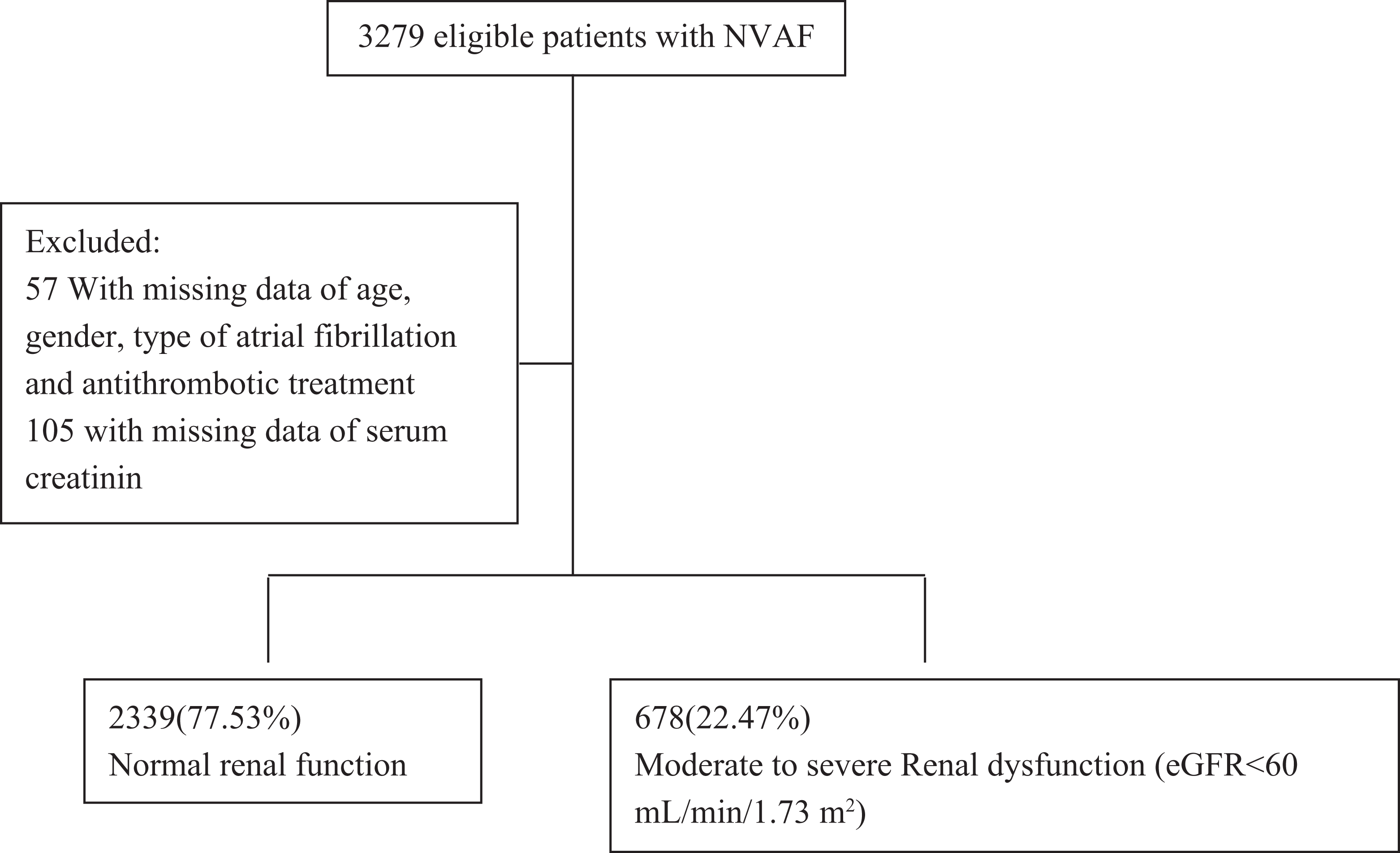
Scheme of the study population.
Mean age was 67.7 ± 13.0 years, and 58.3% were male. The proportion of the patients with a history of stroke/TIA was 24.8%. New-onset AF accounted for 6.6%, paroxysmal AF for 36.9%, persistent AF for 30.3%, and permanent AF for 26.2%. The mean CHADS2 score was 2.0 ± 1.5, with 58.7% of the patients having a score ≥2 and just 7.0% with a score >5. In total, 42.1% of the patients were taking warfarin and half were taking antiplatelet therapy. Median duration of AF was 3 (IQR 0-6) years.
Renal Dysfunction and Relation With Risk of Stroke/TIA
Of these patients with NVAF, 35.8% had normal renal function (eGFR > 90 mL/min/1.73 m2) and 22.5% (678) had impaired renal function (eGFR < 60 mL/min/1.73 m2). There were 19.1% (577) with CKD stage 4 and 3.4% (101) with an eGFR < 30 mL/min/1.73 m2. Patient clinical characteristics are listed in Table 1 according to the severity of their renal impairment. At more severe CKD categories, patients tended to be older (71.6 ± 13.2 in eGFR < 30 group vs 66.3 ± 13.2 in eGFR > 60 group) and more often female (49.5% in eGFR < 30 group and 53.6% in eGFR 30-60 group vs 37.9% in eGFR > 60 group). There was a higher proportion of persistent AF and permanent AF in patients with severe renal dysfunction compared to those without renal dysfunction (49.5% in eGFR < 30 group and 53.6% in eGFR 30-60 group vs 37.9% in eGFR >60 group). In patients with permanent AF, the prevalence of moderate and severe renal dysfunction was 38.6% and 34.0%, respectively, compared to 24.2% in those without renal dysfunction. Both the prevalence of risk factors of stroke and the proportion of patients with a CHADS2 score of 2 or higher increased with worsening of CKD stages.
Demographics and Clinical Characteristics of Patients With NVAF in China.a
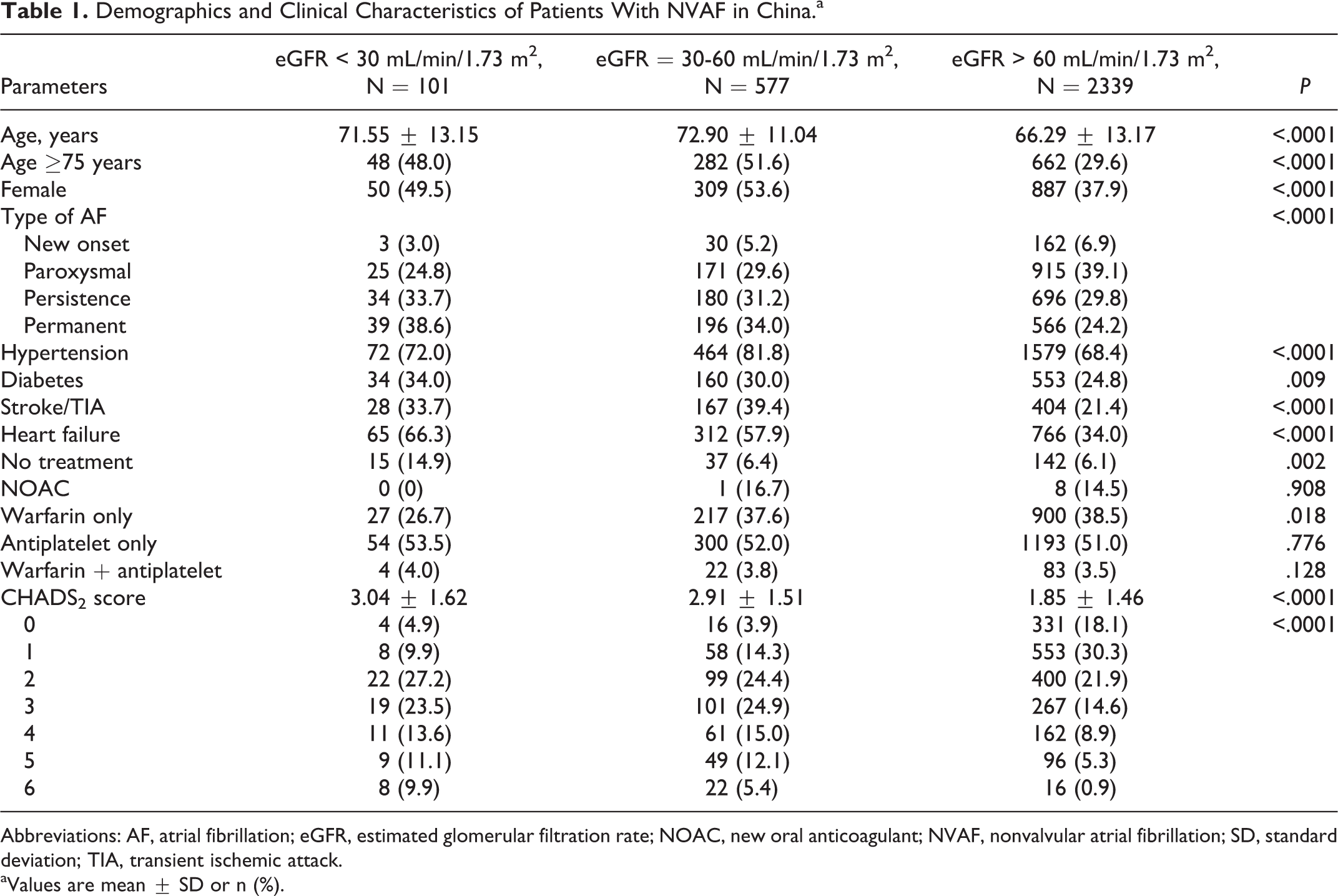
Abbreviations: AF, atrial fibrillation; eGFR, estimated glomerular filtration rate; NOAC, new oral anticoagulant; NVAF, nonvalvular atrial fibrillation; SD, standard deviation; TIA, transient ischemic attack.
aValues are mean ± SD or n (%).
Independent risk factors associated with impaired renal function were age ≥75 years, female sex, heart failure, hypertension, diabetes, and history of stroke/TIA (Table 2). Renal dysfunction was moderately but significantly associated with the risk of stroke or TIA after adjustment for the CHADS2 score (odds ratio: 1.005, 95% confidence interval [CI]: 1.002-1.009, P = .002).
Clinical Characteristics Associated With eGFR < 60 mL/min/1.73 m2.
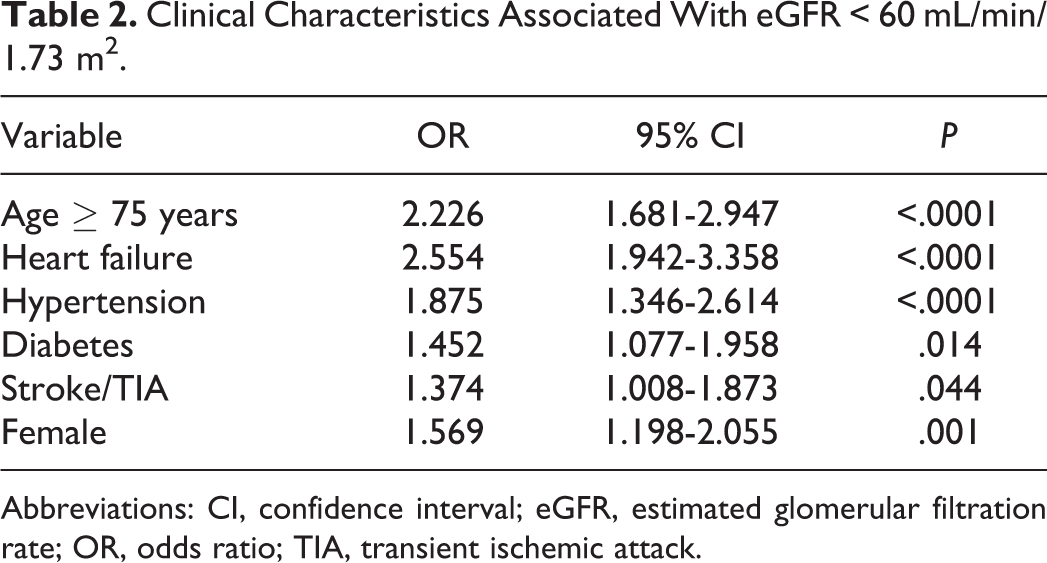
Abbreviations: CI, confidence interval; eGFR, estimated glomerular filtration rate; OR, odds ratio; TIA, transient ischemic attack.
Clinical Factors Associated With Warfarin Underuse
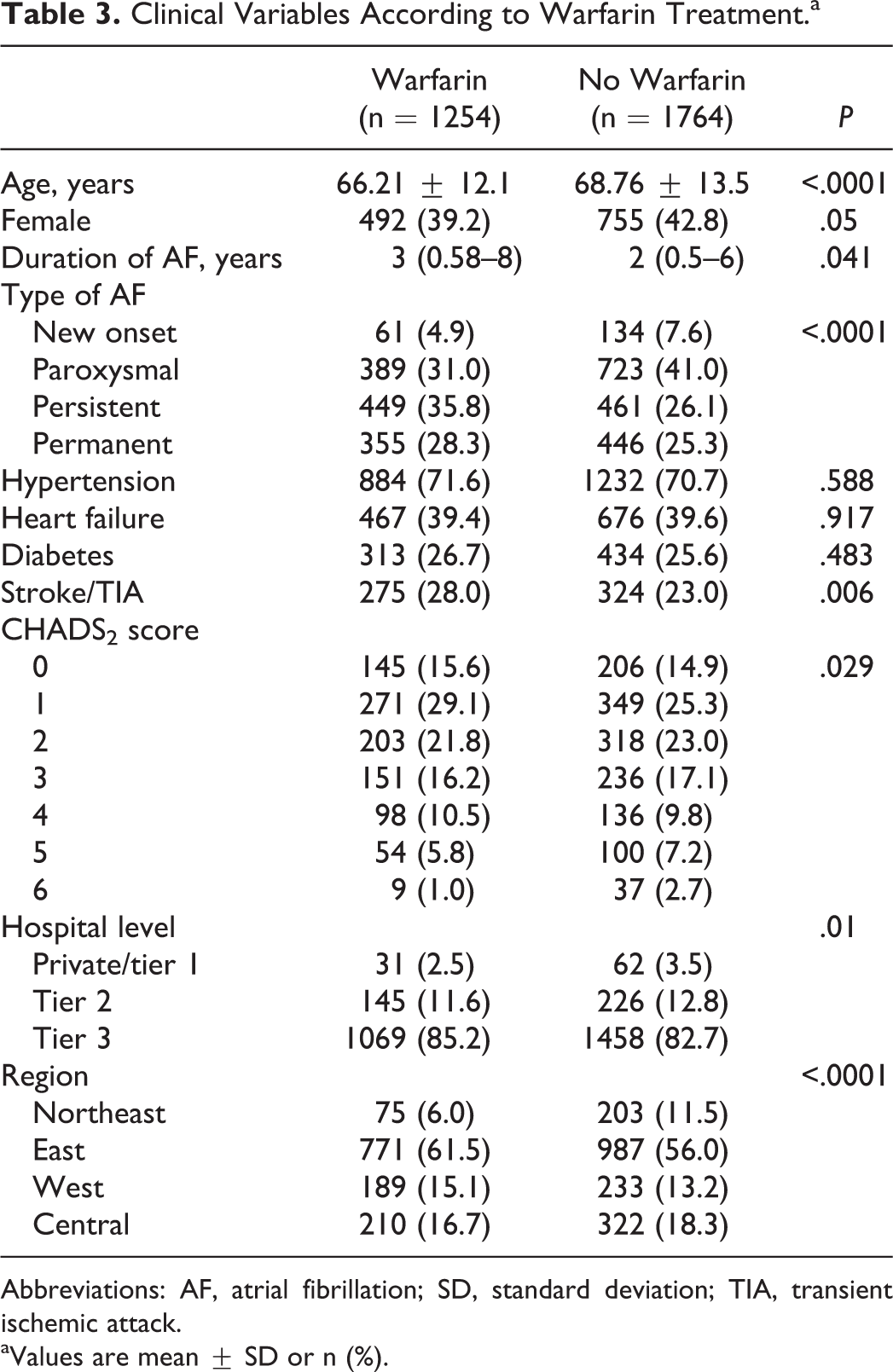
Of these patients with NVAF, 86.2% were at high risk of stroke with a CHADS2 score ≥1, 42.6% of whom were taking warfarin. The proportion of patients taken warfarin was significantly lower in the northeast regions, which is the most underdeveloped region in China, compared to other regions. The rate of warfarin treatment was marginally higher in patients from tier 3 hospital compared with those from tier 2 and tier 1 hospitals. (Table 3).
Clinical Variables According to Warfarin Treatment.a
Abbreviations: AF, atrial fibrillation; SD, standard deviation; TIA, transient ischemic attack.
aValues are mean ± SD or n (%).
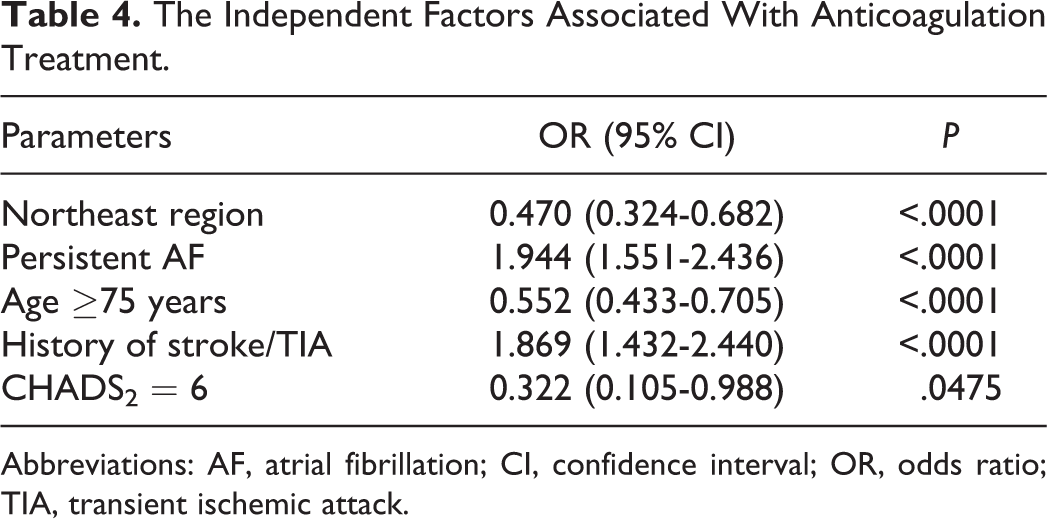
In univariate analyses, warfarin treatment rates were significantly lower in patients with severe renal dysfunction (<30 ml/min/1.73 m2) compared with those without renal dysfunction (Table 4). For patients with eGFR < 30 to 60 mL/min/1.73 m2, the rate of warfarin use was not significantly different from patients with normal renal function (Table 1). Factors associated with patients not taking warfarin identified by a LR model were CHADS2 ≥ 6, advanced age (≥75 years), new-onset AF or paroxysmal AF, and history of stroke/TIA (Table 4).
The Independent Factors Associated With Anticoagulation Treatment.
Abbreviations: AF, atrial fibrillation; CI, confidence interval; OR, odds ratio; TIA, transient ischemic attack.
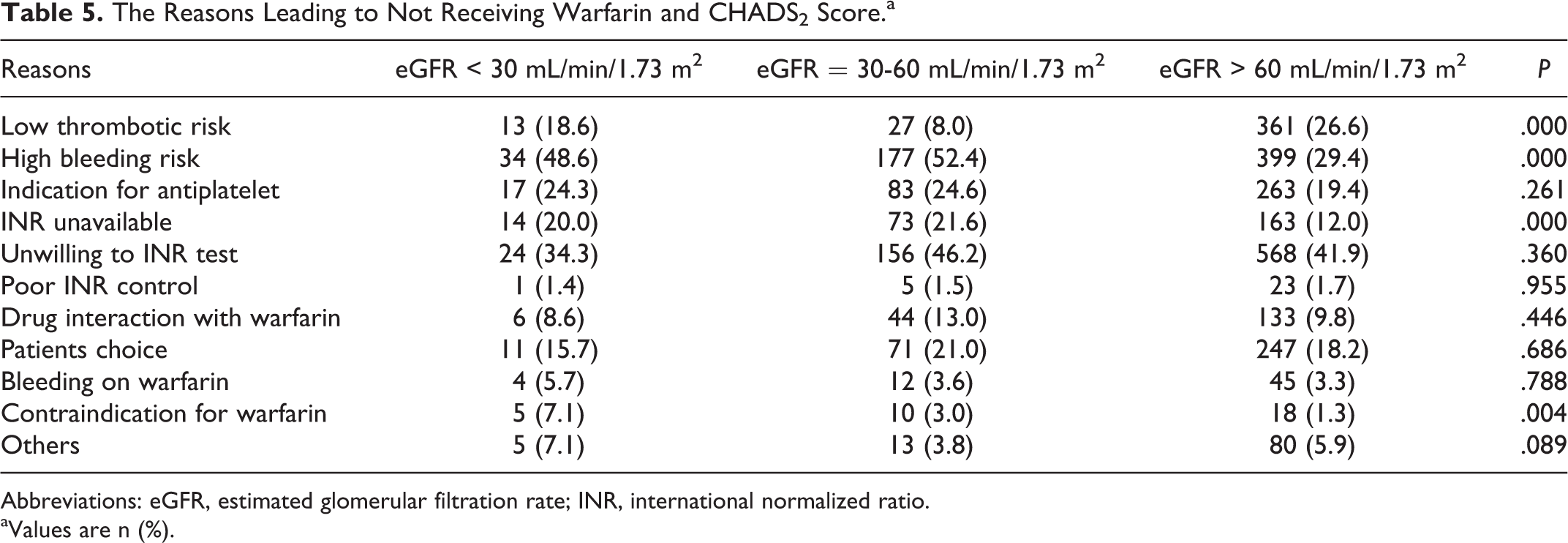
For the 1765 patients not taking warfarin treatment, the 2 commonest reasons were patient unwillingness to undergo regular INR monitoring (43.0%) and high risk of bleeding (33.3%). Of those patients with high risk of bleeding as the main concern (610) about anticoagulant therapy, a higher proportion had renal dysfunction compared to those without renal dysfunction (34.6% vs 17.1%, respectively, P < .0001). The proportion of the patients in whom warfarin was contraindicated was higher in patients with severe renal dysfunction than those with normal renal function (7.1% vs 1.3%, P = .002; Table 5).
The Reasons Leading to Not Receiving Warfarin and CHADS2 Score.a
Abbreviations: eGFR, estimated glomerular filtration rate; INR, international normalized ratio.
aValues are n (%).
Discussion
In this large cohort of “real-world” outpatients with NVAF, we found that 22.5% had moderate to severe renal impairment based on eGFR values calculated using the MDRD Chinese equation. Impaired renal function was independently associated with age, female sex, and comorbid CV disease. Renal dysfunction was moderately but significantly associated with the risk of stroke, even after adjustment for the CHADS2 score. In patients at high risk for stroke with a CHADS2 score ≥1, 42.6% were on warfarin, but only 20.8% of the patients with an eGFR < 30 were taking warfarin. The 2 commonest reasons for not taking warfarin were patient unwillingness to undergo regular INR monitoring (43.0%) and risk of bleeding (33.3%). The degree of renal dysfunction, however, was not related to the use of anticoagulant treatment.
The Incidence of Renal Dysfunction
In our study, 35.8% of the patients with NVAF had normal renal function (eGFR > 90 mL/min/1.73 m2), 20% had moderate CKD, and 3% had severe CKD (eGFR < 30 mL/min/1.73 m2, stage 4). We used the Chinese modification of the MDRD equations for greater applicability in clinical practice in Chinese patients with CKD. 13 The incidence of renal impairment is comparable with previous studies in Western and Chinese patients with NVAF. 15,16 In a cohort of hospitalized patients, 76 of 617 had moderate impaired renal function, and there was an increase over time in stroke or death. 16
It is important to evaluate and monitor renal function in patients with NVAF. First, our study supports previous findings that eGFR decreased with age. 15 Accordingly, it would be prudent to assess renal function every year for patients with NVAF as most of them are elderly patients. Second, there has been an upsurge of interest in the evaluation of renal function in patients with NVAF because NOACs are contraindicated in patients with severe renal dysfunction. The optimal choice of anticoagulation in patients with CKD has been controversial. Deteriorating renal function has been reported to increase the bleeding risk with warfarin in patients with end-stage renal dysfunction. 17 In patients with NVAF, NOACs has been shown to be as least as effective in patients and to be associated with less intracranial hemorrhage compared with warfarin. 18 Finally, renal dysfunction is itself a key factor for bleeding.
Chronic Kidney Disease and Increased Risk of Thromboembolism
We found that moderate to severe renal dysfunction is associated with an increased risk of stroke after adjustment for the CHADS2 score, although renal dysfunction is independently related to CV disease in this study. A study of nonanticoagulated Chinese patients with AF showed renal dysfunction to be an independent predictor of “ischemic stroke and death” in both males and females. However, the increased risk was driven largely by the impact on all-cause mortality rather than stroke. 11 In a cohort that included patients receiving oral anticoagulants, baseline eGFR was closely related to stroke, CV events, and bleeding events (hazard ratio [HR] = 1.42, 95% CI: 1.11-1.83, P = .006) after 2-year follow-up, 15 although Banerjee et al 19 have shown that adding renal impairment to existing stroke risk stratification systems for thromboembolism (CHADS2 and CHA2DS2-VASc) did not independently increase the predictive value of these scores at 1-year follow-up (HR = 1.06, 95% CI: 0.75-1.49). After excluding patients on anticoagulation treatment at baseline, renal function was not an independent predictor for ischemic stroke/TE. A model that included creatinine clearance (R2CHADS2) improved net reclassification index by 6.2% compared with CHA2DS2-VASc (C statistic = 0.578) and by 8.2% compared with CHADS2 (C statistic = 0.575). 20
In a recent meta-analysis 21 of 18 studies involving 538 479 patients, patients with AF having eGFR < 60 mL/min compared with those having eGFR ≥ 60 mL/min experienced a significantly increased risk of developing TE events (relative risk: 1.62, 95% CI: 1.40-1.87, P < .001]), whether they were receiving anticoagulation. There was a slight but significant improvement in CHADS2 score by the addition of CKD (pooled area under the curve [AUC] difference: 0.03, 95% CI, 0.01-0.05), but the AUCs for CHADS2-CKD and CHA2DS2-VASc scores were not statistically different. The importance of renal dysfunction warrants further investigation in Chinese population.
Anticoagulation Treatment and the Concerns of Renal Function
The overall rate of anticoagulant treatment is about 48% in our study, which is a dramatic increase compared to previously reported data. 22 This may reflect the fact that most of the patients were enrolled from tier 3 hospitals in the central area as we found that the use of anticoagulant treatment was far less in patients from northeast and west regions than those from east and central regions, that is, developed regions. Although the rate of warfarin treatment in Chinese patients with NVAF has increased during the last decade, our findings highlight the need for targeted improvement in stroke prophylaxis in elderly patients and those with new-onset or paroxysmal AF.
There were sparse data on the reasons why patients were not taking anticoagulant treatment. In our analysis, the leading cause was the unwillingness of the patient to undergo regular INR monitoring. Access to clinics and health care professionals also represent a major challenge in China, particularly in rural communities and regions with a low health care expenditure. Patients with severe renal impairment are at very high risk of bleeding (if anticoagulated). 23 The second leading concern was high risk of bleeding, for which patients with impaired renal function account for one-third. Some data suggest a higher incidence of intracranial bleeding in Asian compared with Caucasian patients, with a 15-fold increase in risk for patients on warfarin compared with those not taking warfarin, with only a 3-fold increased risk observed in Caucasians. 24 Higher rates of anticoagulant use have been seen in anticoagulation clinic compared with more traditional outpatient cardiology clinics. 25
The discrimination of stroke subtype may be important in anticoagulation decisions for secondary prevention of stroke. Besides the clinical and imaging characteristics, participants with cardioembolic stroke had a significantly higher level of immune-inflammatory markers because of the large infarction compared to participants with other Trial of Org 10172 in Acute Stroke Treatment (TOAST) subtypes. 26 Whether coagulation and fibrinolytic parameters are helpful for the prediction of cardioembolic risk in patients with NVAF warrant further large sample studies. 27,28
Limitations
The limitations of this study include the inherent limitations of cross-sectional observational data with potential residual confounding and the absence of data on CHA2DS2-VASc scores to further investigate the value of renal dysfunction in prediction for risk of stroke/TIA. Larger prospective cohorts of Chinese patients with NVAF should be evaluated to confirm whether renal dysfunction is independently associated with the risk of TE events.
Conclusions
This study demonstrated that renal dysfunction was common in patients with NVAF and that the incidence is associated with comorbid CV disease. Renal dysfunction is moderately but significantly associated with risk of stroke/TIA, in addition to the CHADS2 score. Although concern of bleeding is one of the main reasons leading to underanticoagulation, renal dysfunction itself was not independently related to underuse of anticoagulant treatment.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.