Abstract
Background:
The risk of venous thromboembolism is high in patients undergoing fracture-related major orthopedic surgery, but data on pharmacological thromboprophylaxis are limited. This analysis evaluated the effectiveness and safety of rivaroxaban after fracture-related orthopedic surgery in routine care compared with other pharmacological thromboprophylaxis (standard of care [SOC]).
Methods:
The study population comprised a subset of patients with lower-limb fracture from XArelto in the prophylaxis of post-surgical venous thromboembolism after elective Major Orthopaedic Surgery of hip or knee (XAMOS; a phase IV noninterventional study) and a XAMOS extension study (XAMOS-Extra). The study participants included patients who underwent surgery for hip/femur or lower-leg fractures (below-knee lower-leg fractures, eg, the tibia or foot). All adverse events were recorded, including symptomatic thromboembolic events and bleeding events.
Results:
Data from 790 patients were available for analysis (n = 350 for rivaroxaban and n = 440 for SOC). The incidence of symptomatic thromboembolic events 3 months postsurgery was 0.57% (2 of the 350) in the rivaroxaban group and 1.14% (5 of the 440) in the SOC group (odds ratio [OR]: 0.50; 95% confidence interval [CI]: 0.10-2.59). Treatment-emergent major bleeding events occurred in 0.29% (1 of the 350) of patients receiving rivaroxaban and 0.45% (2 of the 440) of patients receiving SOC (OR: 0.63; 95% CI: 0.06-6.95). There were no cases of fatal or critical bleeding in either treatment group. The incidences of wound complications and any other adverse events were numerically lower with rivaroxaban compared with SOC.
Conclusions:
These data from routine practice demonstrate that rivaroxaban can provide effective thromboprophylaxis after fracture-related orthopedic surgery of the lower limb with a favorable safety profile.
Introduction
Hip fracture is a common injury, particularly in the elderly population, and trauma, immobilization, and subsequent surgery are associated with the risk of potentially serious venous thromboembolism (VTE). 1 –3 The incidence of venographically detected deep vein thrombosis (DVT) without thromboprophylaxis after hip fracture surgery (HFS) is estimated at approximately 50% (of which 27% is proximal DVT). 4 Although routine pharmacological thromboprophylaxis is effective in reducing the risk of VTE after HFS, patients remain at risk. Objectively confirmed symptomatic VTE has been reported in 1.3% to 8.2% of patients (within 3 months of HFS) receiving routine thromboprophylaxis. 4 Fatal pulmonary embolism is also more likely after HFS than after elective hip or knee replacement surgery and ranges in incidence from 0.4% to 7.5% within the first 3 months after HFS. 4
Lower-leg injuries comprise a heterogeneous presentation and include fractures below the knee, Achilles tendon ruptures, and cartilage injuries of the knee and ankle. 5 Clinical data in these patients are particularly scarce; however, it is known that the risk of DVT increases with the proximity of the fracture to the knee. 5 A high incidence of symptomatic VTE related to lower-limb surgery was found after internal fixation (IF) of pelvic fracture (12%; 95% confidence interval [CI]: 5.0-26.0), after knee replacement surgery (3.7%;95% CI: 2.8-5.0), after IF of proximal tibia fracture (3.8%; 95% CI: 2.3-6.3), and after IF of ankle fracture (3.6%; 95% CI: 2.9-4.4). 1
Compared with elective hip or knee replacement surgery, data on pharmacological thromboprophylaxis after fracture-related orthopedic surgery of the lower limbs are relatively limited. Evidence from randomized clinical trials of patients with hip or other lower-leg fractures shows a reduction of approximately 50% in thromboembolic events after low-molecular-weight heparin (LMWH) prophylaxis compared with placebo, 6,7 and thromboprophylaxis after HFS is recommended in guidelines published by the American College of Chest Physicians (ACCP) and the International Union of Angiology (IUA). 5,8 The IUA further recommends considering routine prophylaxis with LMWH for isolated limb trauma in the absence of contraindications. 8
Several studies have investigated the efficacy and safety of anticoagulants for thromboprophylaxis (eg, fondaparinux and LMWH) in the setting of HFS or lower-leg fracture surgery. 9 –13 Although outcomes from these studies have been generally positive, 9,11 –14 some data suggest that an appropriate benefit–risk balance is not always achievable in clinical practice with the currently available parenterally administered anticoagulants and that the residual risk of VTE remains high. 10,15
To date, no randomized controlled trials have been conducted with the direct oral factor Xa inhibitors rivaroxaban and apixaban, or the thrombin inhibitor dabigatran, for thromboprophylaxis after fracture-related orthopedic surgery of the hip or lower limb. In most countries, rivaroxaban is approved for the prevention of VTE after elective hip or knee replacement surgery based on the outcomes from the phase III REgulation of Coagulation in ORthopaedic surgery to prevent Deep vein thrombosis and pulmonary embolism (RECORD) program. 16 –19 However, in some countries (eg, Switzerland, Colombia), it is also approved for VTE prevention in patients undergoing major orthopedic surgery of the lower limbs, including HFS.
The effectiveness of rivaroxaban versus standard of care (SOC) has been investigated in XArelto in the prophylaxis of post-surgical venous thromboembolism after elective Major Orthopaedic Surgery of hip or knee (XAMOS), a phase IV noninterventional study that assessed thromboprophylaxis after major orthopedic surgery of the hip or knee in unselected patients in routine clinical practice. 20 A subset of countries that participated in XAMOS also included patients undergoing fracture-related orthopedic surgery. The objective of this analysis was to evaluate the data collected on the effectiveness and safety of rivaroxaban for thromboprophylaxis in this patient group in routine clinical practice compared with SOC pharmacological thromboprophylaxis.
Patients and Methods
Study Population
The XAMOS study is an international, multicenter, phase IV, noninterventional, open-label cohort study. Eligible patients were aged ≥18 years, with planned hip or knee replacement surgery (or fracture-related orthopedic surgery in countries where this was part of the rivaroxaban [Xarelto] label), and in whom a decision on pharmacological thromboprophylaxis had already been made (Supplementary Figure 1). Exclusion criteria were based on the contraindications in the approved local product information (summary of product characteristics), and written informed consent was provided in countries where necessary.
This analysis included a subset of patients who underwent fracture-related orthopedic surgery from XAMOS and a XAMOS extension study (XAMOS-Extra), which used the same study protocol (Figure 1). These patients (from 25 countries and 76 centers) underwent surgery for hip/femur fractures or for below-knee lower-leg fractures (eg, knee, tibia, and foot). These fractures were predominantly trauma related.
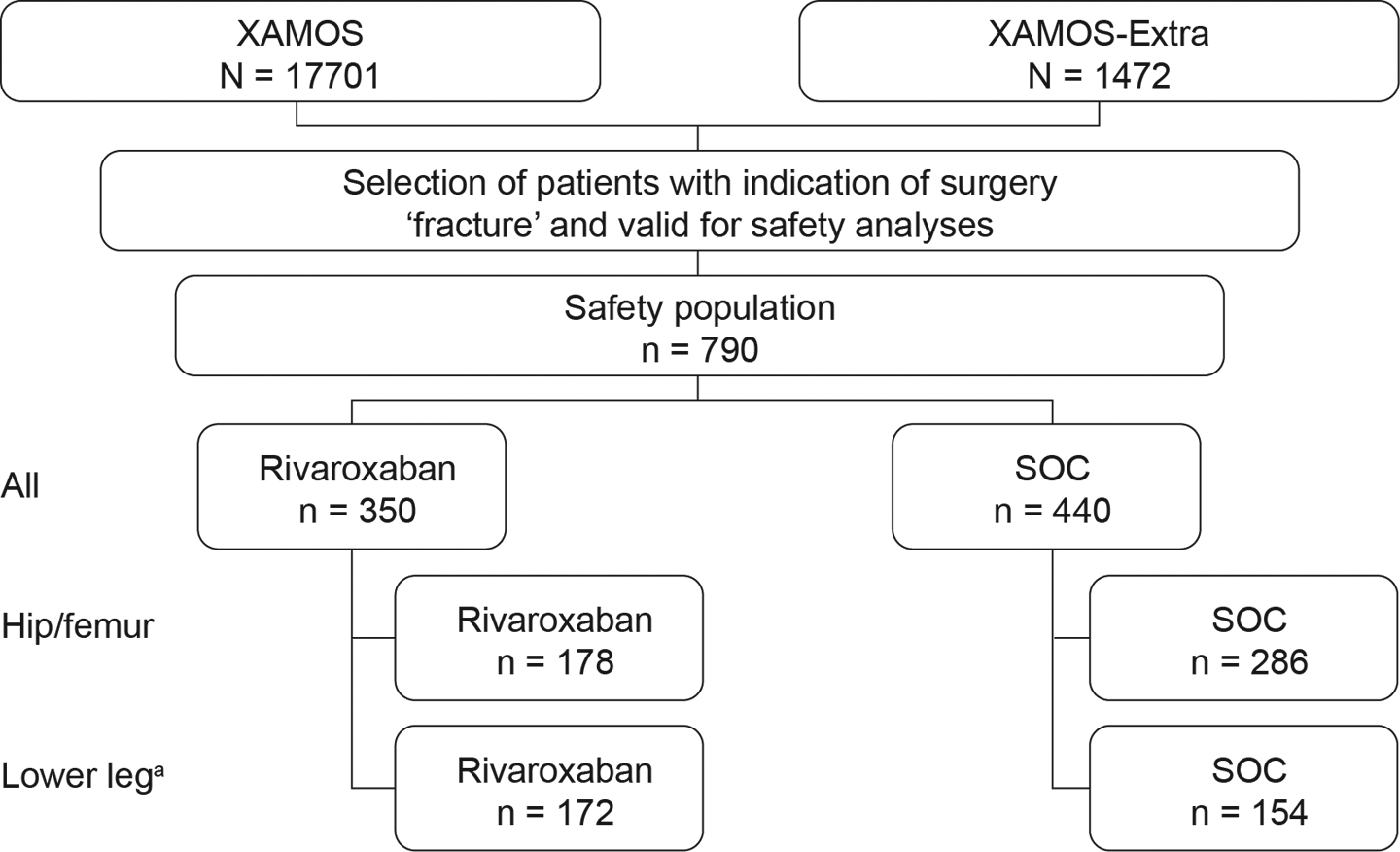
Study flow chart showing breakdown of patients undergoing fracture-related orthopedic surgery in XAMOS and XAMOS-Extra. aBelow-knee lower-leg fractures: indication for surgery not located at the hip or femur, for example, in the knee, tibia, and foot. SOC indicates standard of care; XAMOS, XArelto in the prophylaxis of post-surgical venous thromboembolism after elective Major Orthopaedic Surgery of hip or knee.
Thromboprophylaxis
Decisions about the type, dose, and duration of VTE prophylaxis were determined by the attending physician. The type of prophylaxis given was rivaroxaban 10 mg once daily or any other pharmacological prophylaxis (SOC—mostly LMWHs but also unfractionated heparin [UFH], fondaparinux, dabigatran etexilate, and acetylsalicylic acid). The safety population included patients who received at least 1 dose of rivaroxaban or SOC.
Clinical Outcomes
Data on adverse events, including symptomatic thromboembolic events, bleeding events, wound complications, mortality, and other adverse events, were collected and coded according to the standardized Medical Dictionary for Regulatory Activities (version 14.0). Symptomatic thromboembolic events included arterial and venous events occurring during the entire study period (3 months after surgery), and objective diagnostic confirmation of thromboembolic events was not mandated by the study protocol. Major bleeding events, as defined in the RECORD studies (clinically overt bleeding that was fatal, occurred in a critical organ, necessitated reoperation, was outside the surgical site, was associated with a fall in hemoglobin of ≥2 g/dL, or required a transfusion of ≥2 units of blood), were analyzed. In addition, major bleeding events were analyzed according to the European Medicines Agency (EMA) definition, which was similar to the aforementioned RECORD major bleeding definition but with the inclusion of bleeding warranting treatment cessation, occurring at the surgical site, or leading to any unusual medical intervention. Wound complications included postoperative wound infection, postprocedural wound discharge, and wound hemorrhage. Treatment-emergent events were defined as events starting on or after the day of the first dose and up to 2 days after the last dose of a prophylactic drug for VTE.
Data Collection and Analyses
Data were collected at the start of VTE prophylaxis, at hospital discharge, 1 week after completion of VTE prophylaxis, and 3 months after surgery. All adverse events were documented immediately, irrespective of the set general data collection time points. Serious adverse events were followed up until a final outcome was available. Symptomatic thromboembolic events occurring during the entire study period were identified and adjudicated in a treatment-blinded fashion. Cumulative crude incidences are presented in the rivaroxaban and SOC groups (safety population). The analysis is descriptive, and therefore no statistical significance of the findings can be derived. Odds ratios (ORs) and 95% CIs are given in the context of the overall results to explore directional trends.
Results
Patient Demographics and Clinical Characteristics
Of the 790 patients who underwent fracture-related orthopedic surgery, 350 patients received rivaroxaban and 440 received SOC pharmacological thromboprophylaxis (LMWH: n = 367; fondaparinux: n = 20; UFH: n = 9; dabigatran: n = 4; acetylsalicylic acid: n = 15; other: n = 25). In the rivaroxaban and SOC groups, 178 and 286 patients had hip/femur fractures, respectively, and 172 and 154 patients had below-knee lower-leg fractures, respectively. Baseline patient demographics (age, sex, and body mass index) were similar between the rivaroxaban and SOC groups (Table 1). The median patient age was 73 years in the hip/femur fracture group, whereas patients with lower-leg fractures were younger (median age of 48 years). Patients with hip/femur fracture in the rivaroxaban and SOC groups received prophylaxis for a median duration of 25 days and 21 days, respectively, whereas patients with lower-leg fracture received prophylaxis for a longer period in both treatment groups (Table 1). In the rivaroxaban group, 26.9% of patients received their first postoperative dose ≤6 hours after surgery and 40.0% and 19.1% at >6 to ≤10 hours and >10 to ≤24 hours after surgery, respectively. There were a small number of patients who received their first rivaroxaban dose before/during surgery (0.6%) or >24 hours after surgery (7.1%). Presurgery (from the day of admission to the day of surgery) heparin was used in 92 of the 350 patients in the rivaroxaban group (for a median period of 4.0 days) and in 46 of the 440 patients in the SOC group (for a median period of 6.0 days).
Baseline Demographics of Patients Undergoing Fracture-Related Orthopedic Surgery Receiving Rivaroxaban or SOC (Safety Population).a
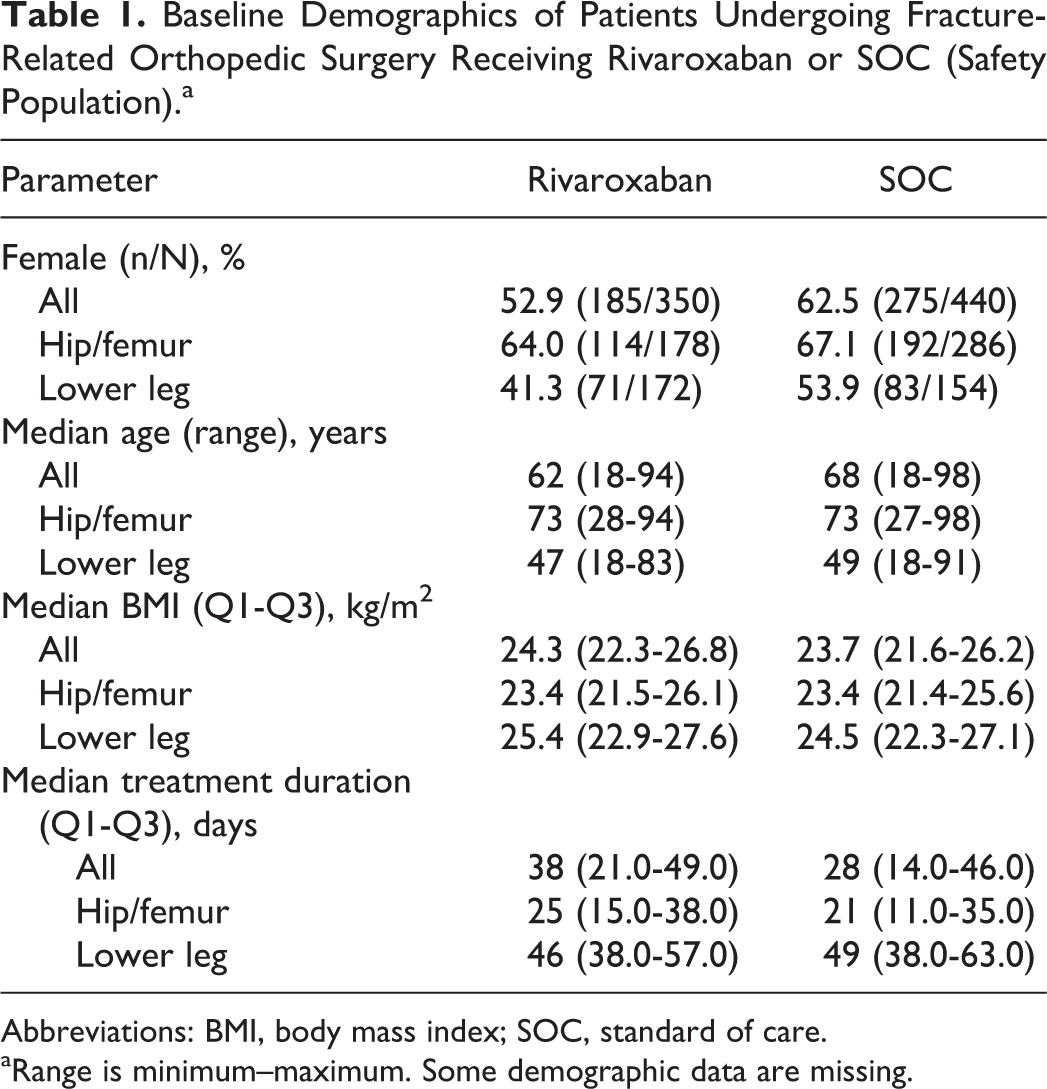
Abbreviations: BMI, body mass index; SOC, standard of care.
aRange is minimum–maximum. Some demographic data are missing.
Clinical Outcomes
Thromboembolic events
For all fracture types, any symptomatic thromboembolic events occurred in 2 of the 350 patients in the rivaroxaban group and in 5 of the 440 patients in the SOC group (0.57% vs 1.14%; OR: 0.50; 95% CI: 0.10-2.59). All of these were venous thromboembolic events, and no symptomatic arterial thromboembolic events were documented (Figure 2).
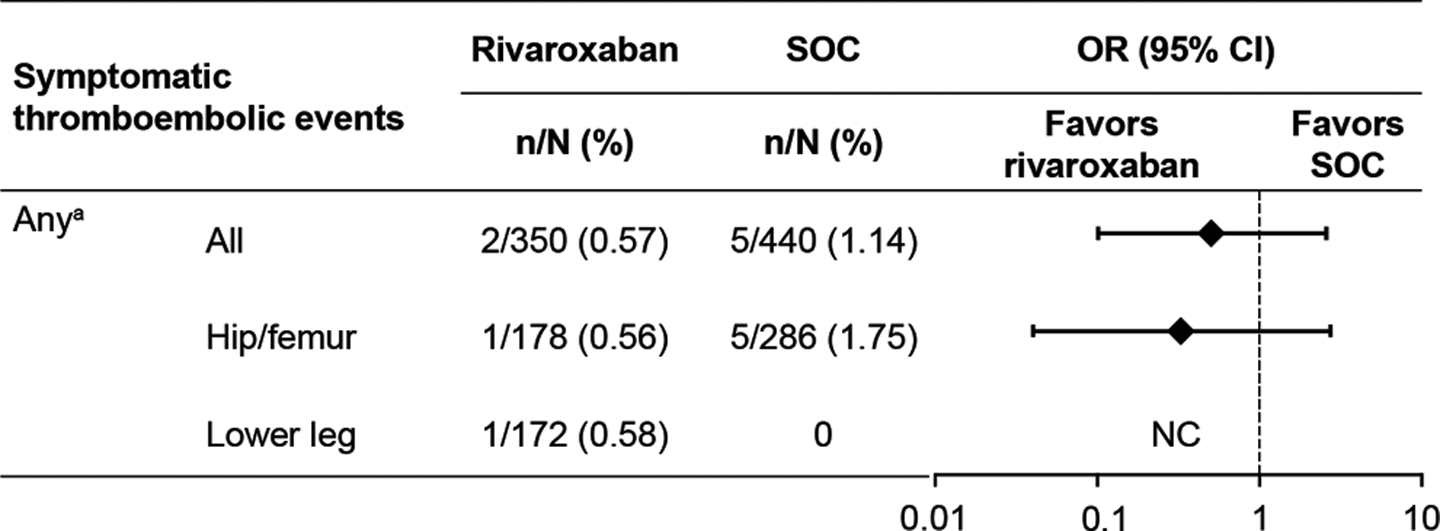
The incidence of symptomatic thromboembolic events in patients undergoing fracture-related orthopedic surgery (safety population). aAll symptomatic thromboembolic events were venous in origin. CI indicates confidence interval; NC, could not be calculated; OR, odds ratio; SOC, standard of care.
In patients who received anticoagulation treatment after hip/femur fracture surgery, any symptomatic thromboembolic events occurred in 1 of the 178 patients in the rivaroxaban group and in 5 of the 286 patients in the SOC group (0.56% vs 1.75%; OR: 0.32; 95% CI: 0.04-2.74; Figure 2). The thromboembolic event that occurred in the rivaroxaban group was attributed to pulmonary embolism. Six events in the SOC group were identified in 5 patients and classified as DVT (n = 3), pelvic thrombosis (n = 1), venous thrombosis (n = 1), and thrombophlebitis (n = 1).
In patients who received anticoagulation treatment after lower-leg fracture surgery, any symptomatic thromboembolic events occurred in 1 of the 172 patients in the rivaroxaban group (recorded as a venous thrombosis). No symptomatic thromboembolic events were reported in the SOC group (Figure 2).
Treatment-emergent bleeding events
For all fracture types, major bleeding events using the RECORD definition occurred in 1 of the 350 patients receiving rivaroxaban and in 2 of the 440 patients receiving SOC (0.29% vs 0.45%; OR: 0.63; 95% CI: 0.06-6.95). With the EMA major bleeding definition, bleeding events occurred in 5 of the 350 patients receiving rivaroxaban and in 4 of the 440 receiving SOC (1.43% vs 0.91%; OR: 1.58; 95% CI: 0.42-5.93; Figure 3). Any bleeding events occurred in 9 of the 350 patients receiving rivaroxaban and in 7 of the 440 patients receiving SOC (2.57% vs 1.59%; OR: 1.63; 95% CI: 0.60-4.43).
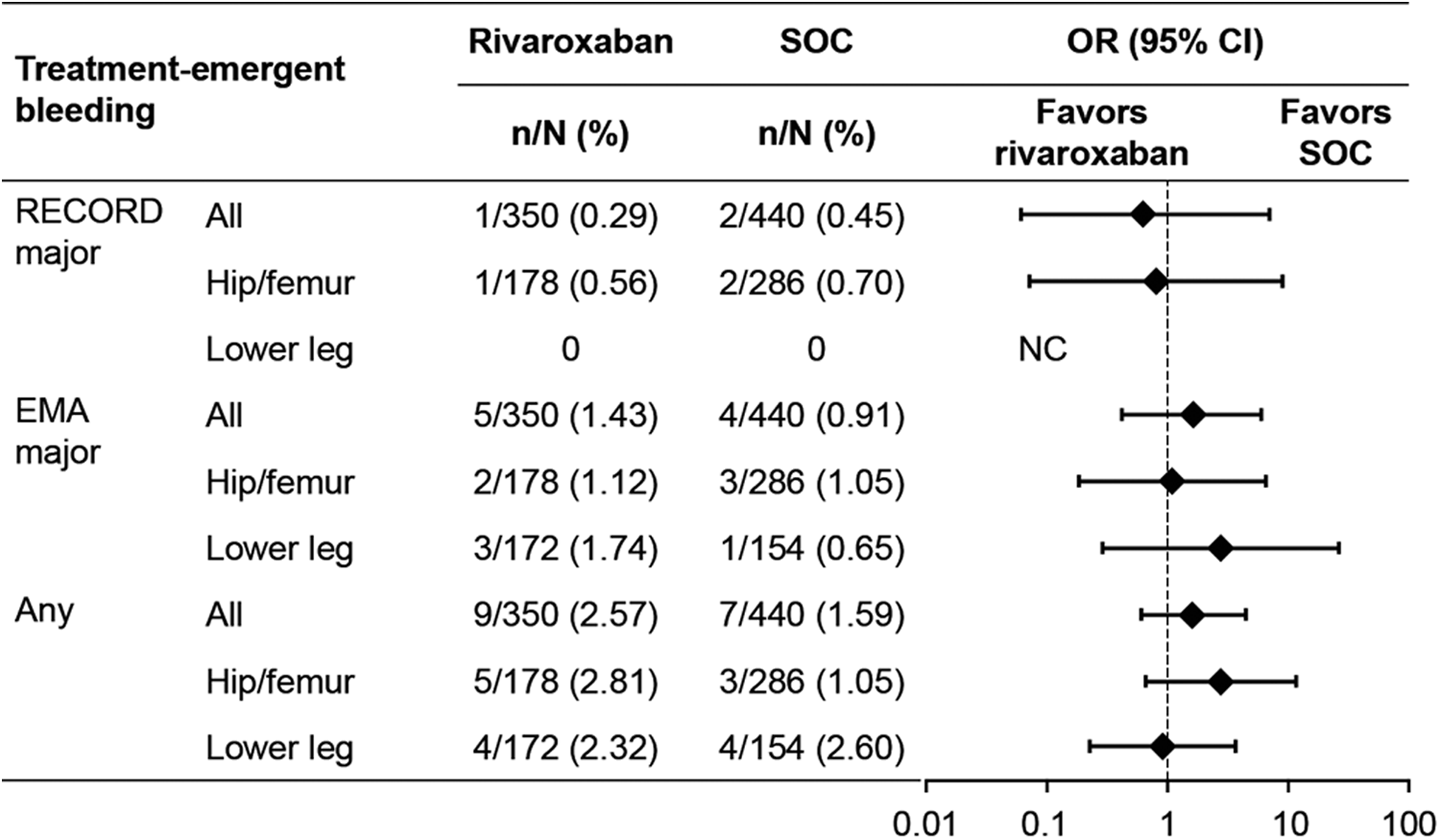
The incidence of treatment-emergent bleeding events in patients undergoing fracture-related orthopedic surgery (safety population). CI indicates confidence interval; EMA, European Medicines Agency; NC, could not be calculated; OR, odds ratio; SOC, standard of care; RECORD, REgulation of Coagulation in ORthopaedic surgery to prevent Deep vein thrombosis and pulmonary embolism.
In patients who received anticoagulation treatment after hip/femur fracture surgery, major bleeding events using the RECORD definition occurred in 1 of the 178 patients receiving rivaroxaban and in 2 of the 286 patients receiving SOC (0.56% vs 0.70%; OR: 0.80; 95% CI: 0.07-8.91). The patient in the rivaroxaban group experienced a bleeding event leading to reoperation. The 2 patients who had major bleeding events in the SOC group experienced bleeding leading to reoperation and clinically overt extrasurgical site bleeding leading to transfusion. There were no cases of fatal or critical bleeding in either treatment group. Major bleeding using the EMA definition occurred in 2 of the 178 patients receiving rivaroxaban and in 3 of the 286 patients receiving SOC (1.12% vs 1.05%; OR: 1.07; 95% CI: 0.18-6.48; Figure 3). Any bleeding events occurred in 5 of the 178 patients receiving rivaroxaban and in 3 of the 286 patients receiving SOC (2.81% vs 1.05%; OR: 2.73; 95% CI: 0.64-11.55).
In patients who received anticoagulation treatment after lower-leg fracture surgery, no major bleeding events were documented when using the RECORD definition. Major bleeding events using the EMA definition occurred in 3 of the 172 patients in the rivaroxaban group and in 1 of the 154 patients in the SOC group (1.74% vs 0.65%; OR: 2.72; 95% CI: 0.28-26.39). Any bleeding events occurred in 4 of the 172 patients receiving rivaroxaban and in 4 of the 154 patients receiving SOC (2.33% vs 2.60%; OR: 0.89; 95% CI: 0.22-3.63; Figure 3).
Treatment-emergent wound complications
For all fracture types, the incidence of wound complications was 2.00% in the rivaroxaban group and 2.73% in the SOC group (OR: 0.73; 95% CI: 0.28-1.87). In patients who received anticoagulation treatment after hip/femur fracture surgery, wound complications occurred in 1 of the 178 patients in the rivaroxaban group and in 2 of the 286 patients in the SOC group (0.56% vs 0.70%; OR: 0.80; 95% CI: 0.07-8.91). For patients who were treated for lower-leg fractures, wound complications occurred in 6 of the 172 patients in the rivaroxaban group and in 10 of the 154 patients in the SOC group (3.49% vs 6.49%; OR: 0.52; 95% CI: 0.18-1.47; Table 2).
Adverse Events in Patients Undergoing Fracture-Related Orthopedic Surgery Receiving Rivaroxaban or SOC.
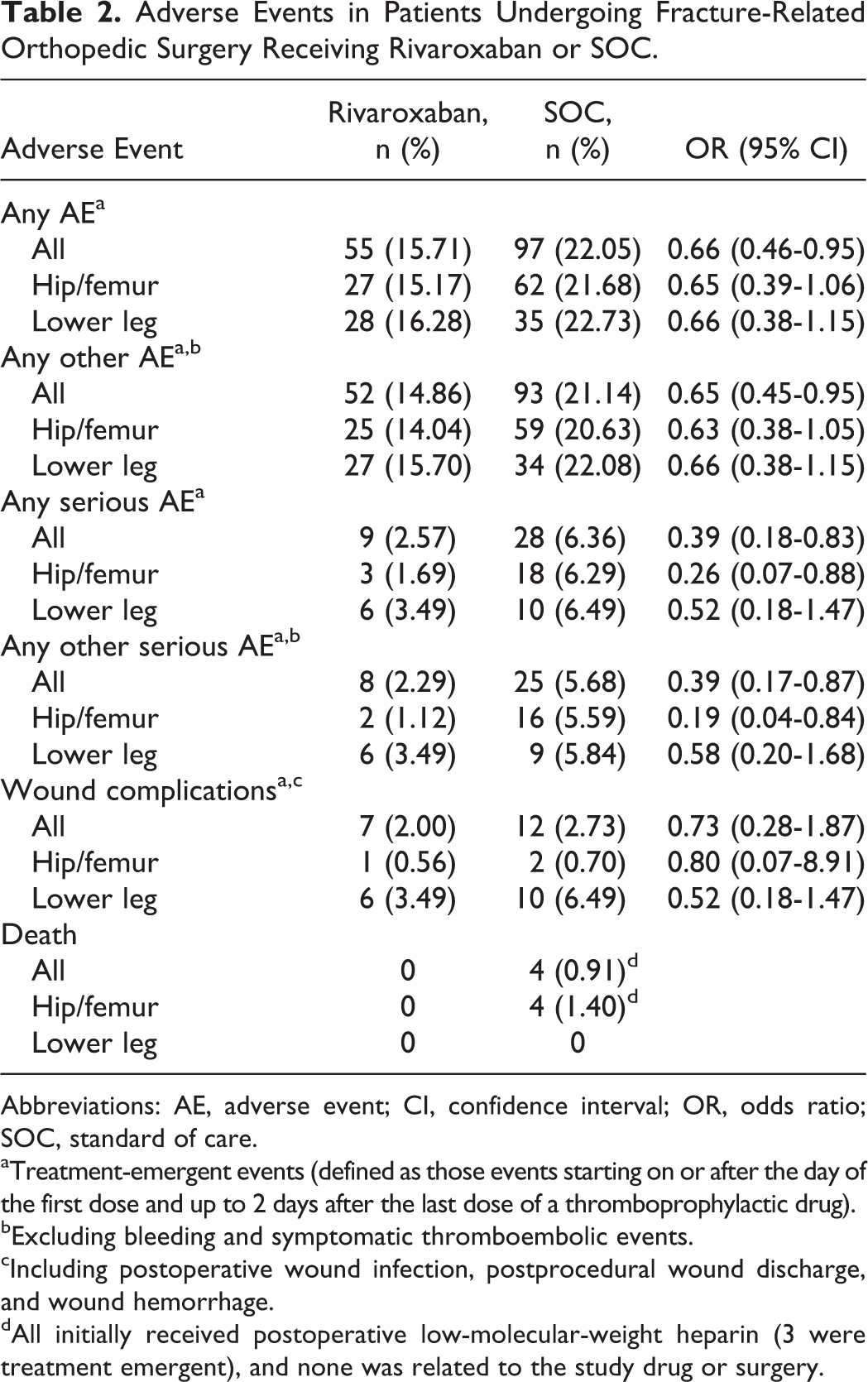
Abbreviations: AE, adverse event; CI, confidence interval; OR, odds ratio; SOC, standard of care.
aTreatment-emergent events (defined as those events starting on or after the day of the first dose and up to 2 days after the last dose of a thromboprophylactic drug).
bExcluding bleeding and symptomatic thromboembolic events.
cIncluding postoperative wound infection, postprocedural wound discharge, and wound hemorrhage.
dAll initially received postoperative low-molecular-weight heparin (3 were treatment emergent), and none was related to the study drug or surgery.
Treatment-emergent adverse events and serious adverse events
Overall, the incidence of any treatment-emergent adverse event was 15.71% in the rivaroxaban group and 22.05% in the SOC group (OR: 0.66; 95% CI: 0.46-0.95). In patients who underwent surgery for hip/femur fractures, adverse events occurred in 27 of the 178 patients in the rivaroxaban group and in 62 of the 286 patients in the SOC group (15.17% vs 21.68%; OR: 0.65; 95% CI: 0.39-1.06). In patients with lower-leg fractures, adverse events occurred in 28 of the 172 patients in the rivaroxaban group and in 35 of the 154 patients in the SOC group (16.28% vs 22.73%; OR: 0.66; 95% CI: 0.38-1.15; Table 2).
The incidence of any serious treatment-emergent adverse event was 2.57% in the rivaroxaban group compared with 6.36% in the SOC group (OR: 0.39; 95% CI: 0.18-0.83). For patients with hip/femur fracture, serious treatment-emergent adverse events occurred in 3 of the 178 patients in the rivaroxaban group and in 18 of the 286 patients in the SOC group (1.69% vs 6.29%; OR: 0.26; 95% CI: 0.07-0.88). In patients with lower-leg fracture, serious treatment-emergent adverse events occurred in 6 of the 172 patients in the rivaroxaban group and in 10 of the 154 patients in the SOC group (3.49% vs 6.49%; OR: 0.52; 95% CI: 0.18-1.47; Table 2).
No deaths occurred in the rivaroxaban group and a total of 4 deaths occurred in the SOC group, all of which were in patients with hip/femur fracture receiving LMWH (3 were treatment emergent). The respective rates for other treatment-emergent adverse events and other serious adverse events are also presented in Table 2.
Discussion
Rivaroxaban is approved worldwide for the prevention of VTE after elective hip or knee replacement surgery, and in some countries, it is also approved for VTE prophylaxis after major orthopedic surgery of the lower limbs, which includes surgery for fractures. The surgical procedures for total hip replacement and HFS are technically similar. In patients with displaced femoral neck fractures, failed osteosynthesis, or periprosthetic fractures, hemi or total hip replacement is often performed to stabilize the hip fracture in the acute nonelective setting.
This observational study allowed an analysis of data in 790 patients in routine clinical practice undergoing hip/femur fracture or lower-leg fracture surgery. Rivaroxaban was associated with a low incidence of symptomatic thromboembolic events and bleeding events, which were similar to or lower than that seen in patients receiving SOC pharmacological thromboprophylaxis. Rivaroxaban was also associated with a numerically low incidence of wound complications and other adverse and serious adverse events.
The real-life incidence of symptomatic VTE in patients who have a hip fracture has been estimated at 2.2% (with the use of heparin prophylaxis). 21 A retrospective cohort study assessing the incidence of symptomatic VTE after HFS in a real-life setting found that although fondaparinux was associated with a risk reduction of 37% to 61% compared with dalteparin, enoxaparin, and UFH, the residual incidence of VTE remained at 2%. 22 In the PENTasaccharide in HIp-FRActure Surgery (PENTHIFRA) study, the rate of symptomatic VTE was 0.5% in both the fondaparinux and enoxaparin treatment arms. 13 Conversely, although extended thromboprophylaxis with reviparin (35 days) significantly reduced the incidence of symptomatic DVT compared with placebo in patients with lower-leg injury (as a result of leg fracture or Achilles tendon rupture), the residual risk remained high (9% and 19%, respectively). 6 In patients with isolated nonsurgical unilateral below-knee injury, the incidence of symptomatic VTE was 0.3% and 1.1% with fondaparinux and nadroparin, respectively. 23 The overall incidence of symptomatic VTE observed with rivaroxaban in the present analysis was 0.57% compared with 1.14% for SOC, suggesting that rivaroxaban is effective as a thromboprophylactic regimen in such high-risk patient groups.
All anticoagulant therapies are associated with an increased risk of bleeding, and this remains a major concern. Studies of fondaparinux in HFS showed the incidence of major bleeding to be approximately 2% up to 3 weeks after surgery. 12,13 Studies with LMWHs have shown a clinically relevant thromboprophylactic effect in HFS, 7,9,24,25 but data have shown a significant increase in the need for blood transfusions with LMWH treatment. 7
In this observational study, the incidence of treatment-emergent major bleeding (RECORD definition—including surgical site bleeding requiring intervention but excluding surgical site bleeding without intervention) was low, occurring in 1 patient in the rivaroxaban group and 2 patients in the SOC group (0.29% vs 0.45%). Differences in bleeding definitions are common between studies of anticoagulants and make direct comparisons difficult. When using the EMA definition of major bleeding, events occurred in 5 patients in the rivaroxaban group and in 4 patients in the SOC group (1.43% vs 0.91%). This definition also includes surgical site bleeding and bleeding leading to treatment cessation. It should be noted that the EMA no longer regards bleeding leading to treatment cessation as a sole criterion for major bleeding. 26
In patients undergoing HFS, the type of surgery is one of the several independent predictors of blood loss. 27 Undisplaced neck fractures are most often managed by inserting simple percutaneous annulated screws, 28 whereas treatment for unstable trochanteric fractures and subtrochanteric fractures is more challenging. Excess time in the operating room and blood loss associated with open reduction may put the patient at further risk of systemic response to injury. 29 It has been suggested that total blood loss after HFS is much greater than that observed intraoperatively and can be associated with complications and longer hospital stays. 27
The first administration of an oral direct factor Xa inhibitor (such as rivaroxaban) can be delayed in cases in which surgery cannot take place within the first 12 to 24 hours. From clinical experience with the pentasaccharide fondaparinux, preoperative dosing may be an option if surgery is delayed for more than 24 hours. Such a strategy would cover the risk period from admission to surgery, as long as the level of anticoagulation is low when surgery is planned. An alternative would be to administer LMWH up to 12 hours before planned surgery and then to transition to an oral factor Xa inhibitor after surgery when hemostasis is achieved. In this analysis, presurgery heparin was used in both the rivaroxaban group (26.3%) and the SOC group (10.5%). Another observation is that patients with lower-leg fractures received prophylaxis for a long period (ie, median of 46 days with rivaroxaban and 49 days with SOC)—the reason for such an extended period of thromboprophylaxis is not known.
Wound infections are a well-known complication after fracture-related surgery, and these can contribute to a substantial increase in the length of hospital stay. 30,31 The incidence of superficial and deep wound infections has been estimated at 1.1% and 1.2%, respectively, in patients undergoing HFS, 30 whereas the incidence of infection ranges from 10% to 22% in patients with fractures located at the knee, lower leg, or ankle. 31 In the present analysis, the incidence of wound complications was numerically lower in the rivaroxaban group compared with the SOC group (0.56% vs 0.70% for patients with hip/femur fracture and 3.49% vs 6.49% for patients with lower-leg fracture). As expected, owing to the known higher risk of fracture infection, the incidence was higher in patients with lower-leg fractures. 31
Limitations of this study include the size of the study population. However, it is worth noting that the results of this analysis are consistent with those from the overall XAMOS study of rivaroxaban and SOC (symptomatic thromboembolic events: 0.89% vs 1.35%, respectively; major bleeding [RECORD definition]: 0.40% vs 0.34%, respectively) in patients undergoing major orthopedic surgery, for which the majority of patients (>90%) underwent elective hip/knee replacement. 20 These data are also supported by the RECORD studies, which compared rivaroxaban with enoxaparin (symptomatic VTE and all-cause mortality: 0.5% vs 1.0%, respectively; major bleeding [RECORD definition]: 0.3% vs 0.2%, respectively), 32 suggesting that outcomes with rivaroxaban are consistent between patients undergoing elective and fracture-related (nonelective) orthopedic surgery. Further studies in a larger population would be of value to confirm the effectiveness and safety of rivaroxaban in this setting. In addition, although noninterventional, observational studies provide valuable information on patients in routine clinical practice, biased estimates of treatment effects may exist because of systematic differences in patient baseline characteristics and other risk factors in these patients. 33
There are currently no ongoing randomized studies with direct oral anticoagulants for VTE prophylaxis after fracture-related orthopedic surgery, such as HFS. However, a number of studies have assessed the efficacy and safety of rivaroxaban in this setting. A single-center nonrandomized study of rivaroxaban in patients with pelvic trauma has shown that it is effective and well tolerated, with no increase in intraoperative or postoperative bleeding in surgical wounds. 34 Furthermore, a retrospective cohort study of >2000 patients with lower-limb fractures (including fractures of the femur, patella, tibia, fibula, ankle, and foot) showed a significantly lower incidence of symptomatic VTE with rivaroxaban compared with LMWH (4.9% vs 8.6%; P = .008). This study also showed a numerically lower incidence of major bleeding (0.2% vs 0.6%) and a significantly shorter length of hospital stay (12.2 days vs 13.1 days; P = .016). 35 Data on VTE prophylaxis based on direct oral anticoagulants are also emerging for lower-leg injuries other than those caused by fractures and joint replacement. A study on rivaroxaban in patients with knee arthroscopy, for instance, showed that extended prophylaxis (21 days) resulted in no symptomatic thromboembolic events, a good safety profile, and improved treatment adherence compared with bemiparin. 36
Conclusions
Data on pharmacological thromboprophylaxis in patients undergoing HFS or lower-leg fracture surgery are relatively limited compared with elective hip or knee surgery. This analysis of data from daily practice shows that thromboprophylaxis with rivaroxaban can be used effectively for both fracture-related and elective major orthopedic surgeries, while presenting a favorable safety profile. Although rivaroxaban is currently not generally approved for thromboprophylaxis after hip fracture or lower-leg fracture, these data demonstrate the potential of rivaroxaban for thromboprophylaxis in this high-risk patient group.
Footnotes
Authors’ Note
The information contained in this paper is presented on behalf of the XAMOS Investigators.
Acknowledgments
The authors would like to acknowledge Yong-Ling Liu, who provided editorial assistance with funding from Bayer HealthCare Pharmaceuticals and Janssen Scientific Affairs, LLC, and Kai Vogtländer and Verena Haupt of Bayer HealthCare Pharmaceuticals for assistance with data management and statistical analysis. The authors thank the following investigators who recruited patients to the XAMOS nonelective study. Patients were recruited to this subanalysis in addition to those previously reported in the main XAMOS study. Australia (3 centers)—P. Blombery (Avenue Cardiovascular Center); A. Khalafallah (Launceston Public Hospital); S. Sommerville (The Wesley Research Institute). Austria (2 centers)—S. Thor (Orthopädisches Krankenhaus Der Stadt Wien Gersthof); C. Woisetschlager (Orthopädisches Spital Wien). Bosnia and Herzegovina (1 center)—I. Gavrankapetanovic (KCU Sarajevo). Chile (1 center)—S. Bittelman (Hospital San José). China (16 centers)—J. Chen (The PLA General Hospital); J. Yao (Xi’an Red Cross Hospital); Q. Zhu (Xijing Hospital); Z. Yang (The 2nd Hospital of Shanxi Medical University); X. Ma (Tianjin Hospital); W. Zhang (Ruijin Hospital Shanghai); Q. Jiang (Nanjing Drum Tower Hospital); W. Xu (Wuhan Union Hospital); G. Zhou (Foshan Hospital of TCM); J. Li (Qi Lu Hospital of Shandong University); Y. Wang (Affiliated Hospital of Qingdao Medical University); Z. Shi (The South Hospital of Southern Medical University); W. He (1st Hospital of GZ University of Chinese Medicine); X. Su (Huarui Hospital 3rd Hospital of Southern Medicine University); D. Cai (The 3rd Hospital of Sun Yat-Sen University); H. Liu (Xiangya Hospital of Central South University). Colombia (5 centers)—C. Sabbag (Clinica de Fracturas); J. Garcia (Unidade Musculoesquelética UMEC); A. Llinas (Fundación Santa Fe de Bogotá); L. Sardi (Clínica Farallones); B. Aguilera (Centro Médico Imbanaco). Denmark (3 centers)—A. Borgwart (Frederiksberg Hospital); M. Lassen (Hørsholm Hospital); M. Hornsleth (Bispebjerg Hospital). Finland (2 centers)—V. Remes (HUS Peijas Hospital); M. Pesola (Keski-Suomen Keskussairaala). France (4 centers)—Y. Matuszczak (Clinique de Cèdre); J. M. Debue (Clinique les Maussins Nollet); Y. Eoche (Clinique Océane); P. Banco (Clinique de l’Union). Germany (5 centers)—H. J. Bail (Charite Virchow KL); P. Mouret (Stadtische Kliniken Frankfurt am Main); J. Grifka (Asklepios Klinikum Bad Abbach); C. Schulze-Pelenga (Orthopädische Uniklinik St. Josef Hospital); H. Joneidi-Jafari (Uniklinik Bergmannsheil). Greece (2 centers)—A. Mpeldekos (KAT); I. Feroussis (Asklipieio Voulas). Hong Kong (1 center)—W. L. Lau (The Macau Orthopaedic Association). Korea (8 centers)—J. M. Lee (National Medical Center); S. Y. Kim (Kyungpook National University Hospital); D. S. Hwang (Chungnam National University Hospital); T. Yoon (Chonnam National University Hwasun Hospital); S. B. Han (Korea University Anam Hospital); Y. C. Ha (Chung-Ang University Hospital); J. D. Chang (Hangang Sacred Heart Hospital); S. J. Yim (Soon Chung Hyang University Hospital Bucheon). Lebanon (2 centers)—N. Sonji (Rafic Hariri University Hospital); Y. Yaghi (Hamoud Hospital). Lithuania (2 centers)—J. Belickas (Kaunas Red Cross Hospital); V. Maciulis (Klaipeda Regional Hospital). Macedonia (2 centers)—J. Nanceva (University Clinic for Orthopaedic Surgery); L. Bogojevski (Hospital St. Erazmo). Mexico (1 center)—J. C. Muno (Centro Medico Puerta de Hierro). Netherlands (1 center)—S. Haarsma (Laurentius Ziekenhuis Roermond). Norway (1 center)—O. Lian (Kristiansund Hospital). Philippines (1 center)—A. Tanchuling (St. Luke’s Medical Center). Singapore (1 center)—W. C. Chang (Gleneagles Medical Center). Spain (1 center)—J. A. A. Tercedor (Clínica el Ángel de Málaga). Switzerland (9 centers)—P. Hoffmeyer (University Hospital Geneva); M. Jakob (University Hospital Basel); H. Simmen (University Hospital Zürich); A. Platz (Stadtspital Triemli); M. Schuler (Kantonsspital Münsterlingen); T. Ilchmann (Kantonsspital Liestal); E. Grossen (Limmattalspital); C. Sommer (Kantonsspital Graubünden); C. Ryf (Spital Davos). United Kingdom (1 center)—S. Scott (Aintree University Hospital). Venezuela (1 center)—E. Dominguez (Uyapar IVSS).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M. R. Lassen has been a consultant for Sanofi-Aventis, Bristol-Myers Squibb, Medtronic, Depuy-Synthes, and Pfizer and has been paid for educational presentations from Bayer HealthCare Pharmaceuticals and Janssen Pharmaceutical Research & Development, LLC. S. Haas has been a consultant for Bayer HealthCare Pharmaceuticals, Bristol-Myers Squibb, Pfizer, Boehringer Ingelheim, and Daiichi Sankyo. R. Kreutz has been a consultant for Merck and Trommsdorff, Servier, Daiichi Sankyo Pharma, Berlin-Chemie, Bristol-Myers Squibb, Menarini, and Bayer HealthCare Pharmaceuticals. L. G. Mantovani has been a consultant for Bayer HealthCare Pharmaceuticals and has received grants from Boehringer Ingelheim, Pfizer, Bristol-Myers Squibb, and Daiichi Sankyo. G. Holberg is an employee of Bayer HealthCare Pharmaceuticals. A. G. G. Turpie has been a consultant for Bayer HealthCare, Janssen Pharmaceutical Research & Development, LLC, Astellas, Portola, and Takeda.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was sponsored by Bayer HealthCare Pharmaceuticals with support from Janssen Research & Development, LLC. A Steering Committee (Alexander G. G. Turpie, Reinhold Kreutz, Michael R Lassen, Lorenzo Mantovani, Sylvia Haas, Martin van Eickels, Gerlind Holberg) provided advice and recommendations on the design and effective operation of this study, reviewed study progress, assisted in the analyses and interpretation of the data (but not data collection), and clarified logistical and ethical issues relating to the study when required. All thromboembolic events were adjudicated in a treatment-blinded fashion by the academic Steering Committee members.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
