Abstract
Introduction:
Transplant-associated thrombotic microangiopathy (TA-TMA) is a rare entity with no standard of care and high mortality, despite the use of plasma exchange.
Methods:
Using specific search terms, all cases having TA-TMA treated with eculizumab and indexed in MEDLINE (English language only) by November 2014 were reviewed.
Results:
A total of 26 cases, 53% men, had a median age of 33 years (range 2-61). Transplant-associated thrombotic microangiopathy occurred after stem-cell transplant (35%) or solid-organ transplant (65%), frequently associated with the use of cyclosporine or tacrolimus (96%). A disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS 13) level was always >10%. After TA-TMA diagnosis, the following drug adjustments were made: discontinuation of cyclosporine or tacrolimus in 45%, dose reduction in another 27%, continuation of the drugs in 23%, and switch from cyclosporine to tacrolimus in remaining 5%. Plasma exchange was performed in ∼43%. The median interval between transplant and initiation of eculizumab was 63 days (range 11-512). A median of 5.5 doses (range 2-21) of eculizumab was utilized with 92% response occurring after a median of 2 doses (range 1-18). At a median follow-up of 52 weeks (range 3-113), the survivors (92%) were doing well.
Conclusion:
Within the limits of this retrospective analysis, our study demonstrates that eculizumab use may result in high response rate and 1-year survival in patients with TA-TMA refractory to discontinuation of calcineurin inhibitor and plasma exchange.
Keywords
Introduction
Thrombotic microangiopathy (TMA), which may occur in relation to hematopoietic stem cell transplant (SCT) or solid-organ transplant, refers to inflammatory and thrombotic diseases of the microvasculature characterized by otherwise unexplained microangiopathic hemolytic anemia and thrombocytopenia, along with evidence of organ damage. 1 Transplant-associated TMA (TA-TMA) is multifactorial, and risk factors include chemotherapy and radiotherapy used in conditioning regimen, exposure to calcineurin inhibitors (CNIs), unrelated donor, human leukocyte antigen mismatch, graft-versus-host disease (GVHD), and viral infections. 2 –5 Although its pathophysiology is poorly understood, dysregulation of complement system has been observed. 6 The treatment options are not clearly defined and suboptimal with high associated mortality rates. 4,7,8 Eculizumab is a monoclonal antibody against compliment protein C5, approved by the Food and Drug Administration for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uremic syndrome (aHUS). 9 A number of reports describe its successful use in patients with TA-TMA; however, small sample size and a lack of review of these cases have prevented better understanding of the efficacy of eculizumab use in TA-TMA. In this report, we have analyzed all published cases of eculizumab use in TA-TMA with a goal to determine its efficacy.
Methods
A thorough search of relevant articles published from inception to November 2014 was performed in MEDLINE (via PubMed). Following strategy was utilized to perform the search:
((((((((thrombotic microangiopathy) OR thrombocytopenic thrombotic purpura) OR TTP) OR TMA) OR microangiopathic hemolytic anemia) OR MAHA)) AND ((((transplant) OR solid organ transplant) OR stem cell transplant) OR organ transplant)) AND ((((eculizumab) OR complement) OR complement blockade) OR complement inhibition). Bibliographies of the relevant studies were hand-searched to identify additional studies. The diagnosis of TA-TMA as reported by the authors in these studies was accepted. Inclusion criteria included TA-TMA occurring after solid-organ transplant or SCT and use of eculizumab. Patients with a confirmed diagnosis of aHUS and PNH were excluded. Only reports published in English language were included (Figure 1). The primary efficacy outcome was resolution of laboratory and clinical manifestations of TMA with or without persistent renal impairment (response), as reported by the authors. The secondary efficacy outcome was overall survival. Descriptive statistics were used to calculate frequency.
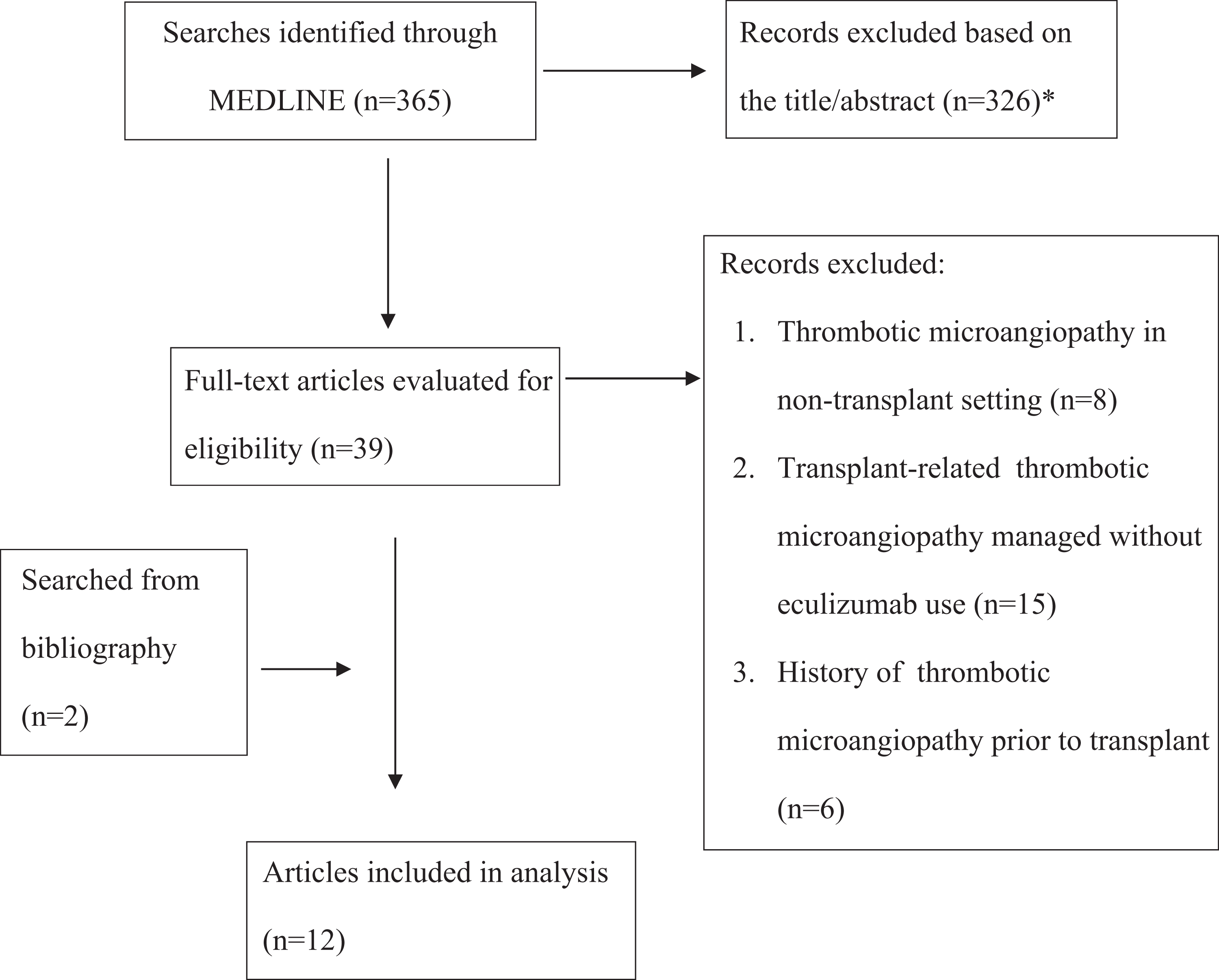
Flow diagram for selection of the articles. *These articles were excluded because of one of the following reasons: Not related to thrombotic microangiopathy, not related to transplant, review article, nontherapeutic study, preclinical study, and eculizumab use in other conditions, for example, atypical hemolytic uremic syndrome, paroxysmal nocturnal hemoglobinuria, and antiphospholipid syndrome.
Results
Twelve reports, describing a total of 26 patients with TA-TMA managed with eculizumab, met the eligibility criteria. 1,10 –20 The median age was 33 years (range 2-61), and 47% were females (Table 1). Thirty-five percent (n = 9) of cases occurred after SCT 1,15,17,18 —predominantly allogeneic SCT (67%, n = 6) 1,15,17 but also autologous SCT (22%, n = 2) 15 and tandem autologous-allogenic SCT (11%, n = 1). 18 The rest (65%, n = 17) of the cases occurred after solid-organ transplant, 10 –14,16,19,20 which included renal transplant (41%, n = 7), 10,11,14,19 small bowel (29%, n = 5), 16 liver (12%, n = 2), 16 simultaneous pancreas and kidney (12%, n = 2), 12,20 and combined lung and kidney (6%, n = 1) 13 transplants. In SCT, 56% of donors were matched and 44% were unmatched. The donor status was not known in 53% of solid-organ transplants, and among the remainder, 37% were matched and 67% were unmatched. Calcineurin inhibitors such as cyclosporine or tacrolimus were used as prophylaxis in 96% of total cases. All cases of SCT received CNIs. Among those, 89% (n = 8) of cases received cyclosporine only 15,17,18 and 11% (n = 1) received tacrolimus only. 1 In solid-organ transplant cases, 94% (n = 16) had CNI prophylaxis but 6% (n = 1) did not. 14 Patients on CNIs received tacrolimus only (44%, n = 7), 10 –12,19,20 cyclosporine only (12%, n = 2), 11,13 or unspecified agent (44%, n = 7). 16 Acute GVHD occurred in 83% (n = 5) 15,17,18 of allogeneic SCT and graft dysfunction or rejection occurred in 29% (n = 5) 11,12,16,20 of solid-organ transplants.
Characteristics of Patients Receiving Eculizumab for Transplant-Associated Thrombotic Microangiopathy.a
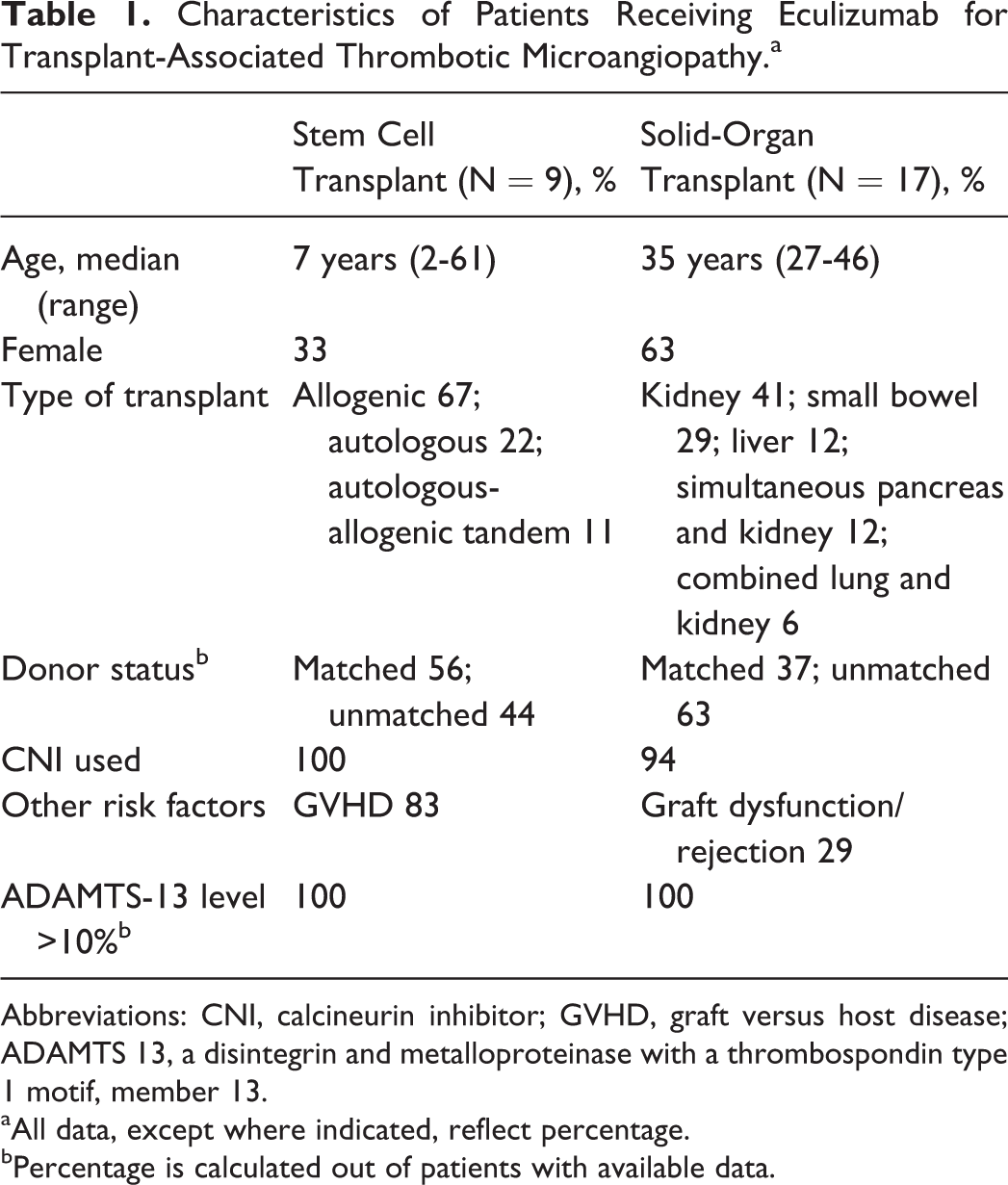
Abbreviations: CNI, calcineurin inhibitor; GVHD, graft versus host disease; ADAMTS 13, a disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13.
aAll data, except where indicated, reflect percentage.
bPercentage is calculated out of patients with available data.
All SCT cases (n = 9) met the diagnostic criteria recommended by Cho et al based on their validation study. 2 Although 53% (n = 9) of cases of solid-organ transplant utilized biopsy findings to support the diagnosis, 10 –12,14,19,20 the remainder of cases either utilized clinical criteria alone or did not specify diagnostic criteria utilized (n = 8). 13,16 A disintegrin and metalloproteinase with a thrombospondin type 1 motif, member 13 (ADAMTS 13) levels were normal or only mildly decreased (>10%) in all 17 cases, where data were available. 1,15 –19 The median interval between transplant and TA-TMA diagnosis was 41 days (range 3-390; Table 2). After TA-TMA diagnosis, in cases of SCT, cyclosporine or tacrolimus was discontinued in 67% (n = 6), 1,15,18 the drugs were continued in 22% (n = 2), 15 and cyclosporine was switched to tacrolimus in remaining 11% (n = 1) of cases. 17 Nineteen percent (n = 3) 10,13 of cases in solid-organ transplant did not mention about drug adjustments. Where mentioned, the following drug adjustments were made: discontinuation of cyclosporine or tacrolimus in 31% (n = 4), 12,16,19,20 dose reduction in 46% (n = 6), 16 and continuation of drugs in 23% (n = 3) 11 of cases. Sirolimus was initiated after discontinuation of tacrolimus in 2 cases with TA-TMA after solid-organ transplant. 12,20 Additionally, plasma exchange was performed in 42% (n = 11) of total cases including 6 SCT cases 1,15 and 5 solid-organ transplant cases. 11,12,19,20 Dialysis was done in 7% (n = 2) 13,16 and both plasma exchange and dialysis in 4% (n = 1) 14 of cases.
Treatment Received and Outcomes of Patients With Transplant-Associated Thrombotic Microangiopathy.a
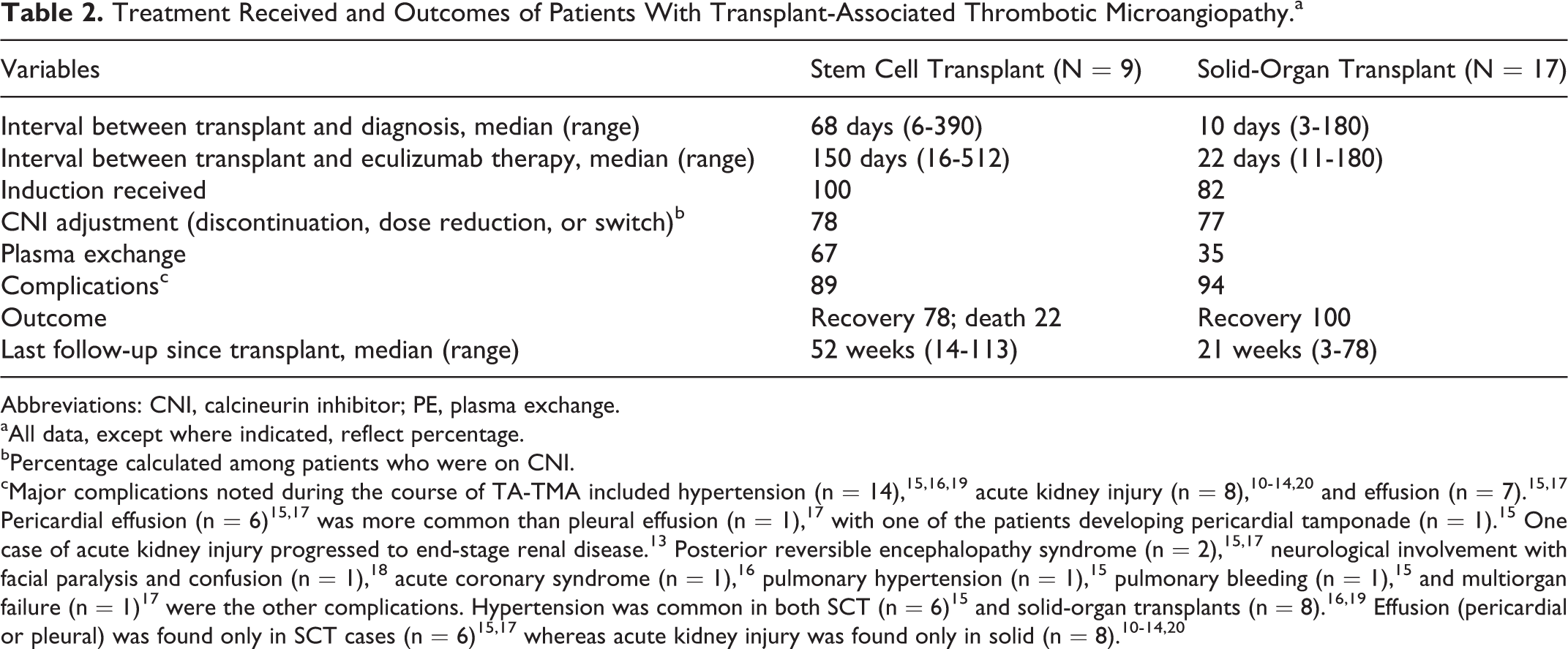
Abbreviations: CNI, calcineurin inhibitor; PE, plasma exchange.
aAll data, except where indicated, reflect percentage.
bPercentage calculated among patients who were on CNI.
cMajor complications noted during the course of TA-TMA included hypertension (n = 14), 15,16,19 acute kidney injury (n = 8), 10 –14,20 and effusion (n = 7). 15,17 Pericardial effusion (n = 6) 15,17 was more common than pleural effusion (n = 1), 17 with one of the patients developing pericardial tamponade (n = 1). 15 One case of acute kidney injury progressed to end-stage renal disease. 13 Posterior reversible encephalopathy syndrome (n = 2), 15,17 neurological involvement with facial paralysis and confusion (n = 1), 18 acute coronary syndrome (n = 1), 16 pulmonary hypertension (n = 1), 15 pulmonary bleeding (n = 1), 15 and multiorgan failure (n = 1) 17 were the other complications. Hypertension was common in both SCT (n = 6) 15 and solid-organ transplants (n = 8). 16,19 Effusion (pericardial or pleural) was found only in SCT cases (n = 6) 15,17 whereas acute kidney injury was found only in solid (n = 8). 10 –14,20
The median interval between transplant and initiation of eculizumab was 63 days (range 11-512). A median of 5.5 doses (range 2-21) of eculizumab was utilized with the first response occurring after a median of 2 doses (range 1-18). The dosing of eculizumab largely followed the recommended dosing for aHUS. Ninety-five percent (n = 18) of adults 1,10,11,13,14,16,18 –20 received induction, whereas the remaining 5% (n = 1) did not. 12 Maintenance therapy, as used for aHUS, 21 was given in 77% (n = 20) 1,10,11,14 –18 of all patients, while the others (n = 6, 23%) 12,13,15,19,20 received no maintenance doses. Jodele et al 15 also infused the first dose of eculizumab according to recommendations for children with aHUS. The subsequent doses were adjusted to maintain a trough concentration >99 mg/mL since this drug level has been considered therapeutic in aHUS. 21 Although 4 of the 6 patients had complete resolution of severe TA-TMA after achieving steady therapeutic eculizumab levels, the remaining 2 patients did not achieve therapeutic levels even after dose escalation and died. For the entire cohort in our study, 92% (n = 24) 1,10 –20 of the patients recovered with the use of eculizumab but the rest (n = 2) 15 died. At a median follow-up of 52 weeks (range 3-113), the survivors were doing well.
Discussion
The majority of the patients in our study developed TA-TMA in the setting of CNI and absence of severe deficiency of ADAMTS-13. Patients were initially treated with discontinuation or dose reduction of CNI with or without plasma exchange. Eculizumab was often utilized after the failure of these initial therapies. The use of eculizumab improved the condition in 92% of patients who were alive at 1-year follow-up. These results are promising compared to high mortality traditionally observed in TA-TMA.
Eculizumab has been utilized in TA-TMA based on the successful use of eculizumab in aHUS 21 and the findings of complement dysregulation in prior reports of TA-TMA. 6 A recent study 6 demonstrated deletions in complement factor H (CFH)-related genes 3 and 1 or CFH autoantibodies in 6 children who developed TA-TMA. Conversely, CFH autoantibodies were not detected in 18 children who had undergone SCT and did not develop TA-TMA. This demonstrates the role of alternative complement pathway activation in the pathogenesis of TA-TMA. Direct endothelial tissue damage from triggers such as high-dose chemotherapy, CNI, viruses, and GVHD can lead to classic complement activation as well 15 and C4d staining may be used as a marker in these instances. 22,23 Dysregulation of complement pathways ultimately leads to terminal complement activation and formation of cytolytic membrane attack complex. By blocking C5, eculizumab prevents deployment of terminal complement system and thus formation of membrane attack complex. 12 In a report, however, eculizumab was successful in managing TA-TMA even in the presence of normal level of complement proteins C3, C4, factor H, and factor I and the absence of anti-CFH antibodies. 18 This is not surprising, since complement-mediated TMA is associated with known mutations in only half of the patients. 24 Arteriolar C4d staining in suspected cases of TA-TMA may assist in making the diagnosis. 25 Although C4d may not be sensitive for TA-TMA, C4d staining in kidney tissue from patients with clinical signs of TMA can be considered in future reports, if biopsy is performed. 25,26
The dosing of eculizumab is not well established in cases with TA-TMA and was somewhat variable in our study but largely followed the dosing utilized for aHUS. 21 The dose of eculizumab for the management of adult patients with aHUS includes an induction phase (900 mg weekly for 4 weeks) and maintenance phase (1200 mg for first week, then 1200 mg every 2 weeks). Supplemental dose of 600 mg is given before plasma infusion or within 1 hour after plasma exchange. 21 The recommended dosage for children with aHUS depends on weight and varies from 300 mg for a body weight of 5 to 10 kg to 900 mg for 40 kg or more. Jodele et al 15 proposed that CH50 level, a hemolytic complement assay, may be used to titrate eculizumab doses in children since drug monitoring is not readily available; CH50 level ≤4 complement activity enzyme units correlated with therapeutic eculizumab levels as well as clinical response. 15
Although 2 patients in our study died even after eculizumab therapy, all others responded and were alive at 1-year follow-up. The patients who died were critically ill with severe multiorgan injury at the time of initiation of eculizumab therapy and neither achieved sustained therapeutic eculizumab trough levels or complement blockade, as measured by CH50. 15 In the remaining patients, the first response to eculizumab was seen after 2 doses (median, range 1-18) and the patients received a median of 5.5 doses (range 2-21). Although not definite, these findings suggest that in TA-TMA, eculizumab may be stopped at some point after resolution of TA-TMA. In clinical practice, clinical and laboratory parameters may be used to measure the response and decide drug discontinuation. Such parameters may include improvement in neurological status or other clinical symptoms, increase in platelet counts, decrease in lactate dehydrogenase level, increase in serum haptoglobulin, and possibly improvement of kidney function and proteinuria. However, kidney damage may not always be reversible.
Patients with TA-TMA are at risk of several complications. In our study, therefore, it was not possible to ascertain toxicities related to eculizumab. Nonetheless, the vast majority of patients did not have any known long-term complications. This suggests that eculizumab may be safe in the treatment of TA-TMA but this requires further prospective, larger studies to validate. Experiences with eculizumab in other conditions have shown an increased susceptibility to meningococcal infection and infections with other encapsulated organisms; therefore, prophylaxis against meningococcal infection is recommended with its use. 27
Plasma exchange along with reduction or withdrawal of offending agent and supportive care are commonly used management options for TA-TMA. Noncomparative studies have reported the response to plasma exchange in up to one-third of patients, although it may possibly be influenced by the timing of clinical intervention, the presence of concomitant acute GVHD, and the disappearance of circulating endothelial cells. 28 –31 In a recent review, 37% to 55% of patients of TA-TMA responded to plasma exchange; however, the overall mortality was still 80%. 32 Since the use of CNIs constitutes one of the important risk factors for TA-TMA, change of immunosuppressive regimen such as discontinuation of CNIs, dose reduction, or conversion is considered pivotal in its management. 1,33 This may lead to improvement in TMA process in some patients. Response rates of 63% have been observed when these drugs are withheld, 1 which is better than the rates of response to plasma exchange. 31 The majority of the patients in our study developed TA-TMA in the setting of CNIs and absence of severe deficiency of ADAMTS-13 (<10% activity). Before the use of eculizumab, 58% of patients underwent discontinuation or dose reduction of CNI (31% along with plasma exchange and 27% without plasma exchange) but none responded. Our report also highlights that severe deficiency of ADAMTS-13 is rare in TA-TMA, which may arguably be a reason for low success rate with plasma exchange.
Rituximab, a monoclonal antibody directed toward CD20 antigen, has also been used in a few patients with TA-TMA as monotherapy or in combination with plasma exchange or defibrotide. 34 –37 Au et al described 5 cases of TA-TMA in allogeneic SCT recipients. Four (80%) patients were successfully treated with rituximab. Although the nonresponder died from multiorgan failure, 75% (n = 3) of responders remained in remission at a 10-month follow-up period (the fourth patient died of sepsis on day +250 while still in remission from TA-TMA). 34 In 6 pediatric patients with TA-TMA (3 allogeneic SCT and 3 autologous SCT), 6 4 patients (67%) responded to rituximab with or without plasma exchange. Nonresponders progressed to end-stage renal disease. Four other patients with TA-TMA have been reported to respond to rituximab as upfront therapy or with plasma exchange. 36,38 –40 Although the exact mechanism is unknown, rituximab is speculated to alter immune regulation, antibody production, or possibly complement activation. 34 Rituximab can be an alternative therapeutic agent in TA-TMA. Whether eculizumab is superior to rituximab is unknown; similarly, the patient population most likely to benefit from one or the other therapy is unclear.
Our retrospective review has several potential limitations. Unsuccessful cases are less likely to be reported than successful cases, thus the review may significantly overestimate the benefit of eculizumab. Some of the information such as ADATMS-13 level was not available in a few reports. The original reports were case reports/series without any control group, thus definite conclusions may not be drawn. Nonetheless, to our knowledge, this is the first review analysis of all published cases of eculizumab use in TA-TMA, which provides information useful for therapy selection in this rare disease. Until recently, a diagnosis of TA-TMA was associated with a high mortality with the 3-year survival rate reported to be 11% in one study. 41 The results of our study are promising compared to high mortality traditionally observed in TA-TMA. Within the limits of this retrospective analysis, our study demonstrates that eculizumab may be effective in the management of TA-TMA and may improve survival. However, larger prospective studies are necessary to validate the results. Given the rarity of the disease, prospective registry-based studies or phase II trials may be more practical. Future studies should focus on determining the optimal sequencing of available therapies as well as optimal dosing and duration of eculizumab therapy in TA-TMA. Since complement mutations are not seen in half of the patients with complement-mediated TMA, 24 the role of other tests to detect complement activation such as C4d or C5b-9 deposition in skin or affected organ should be investigated. 1,25,26 Whether recipient’s or donor’s DNA should be utilized for assessing complement gene mutation is unclear 6 and a subject of future research. Larger studies may also identify new toxicities of eculizumab in transplant recipients. Since TA-TMA frequently occurs within the first 3 months of transplant, meningococcal vaccination may not be practical; hence the strategy for meningococcal prophylaxis such as the use of prolonged prophylactic antibiotic needs further evaluation in this patient population. It is hoped that the outcomes of future research will guide the optimal therapy of TA-TMA and prevent premature deaths.
Footnotes
Authors’ Note
This article was presented in an abstract form at the National American College of Physician annual meeting 2015, Boston, MA, held on April 30 to May 2, 2015.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported in part by the University of Nebraska Medical Center, College of Medicine, Physician-Scientist Training Program Grant 2015-2016.
