Abstract
Background and Objectives:
Raised levels of von Willebrand factor (VWF) and reduced levels of a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13 (ADAMTS13) activity are associated with thrombosis. We aimed to investigate the relationships between plasma levels of VWF and ADAMTS13, their ratios, and the occurrence of cerebral infarction and to understand the roles of VWF and ADAMTS13 in cerebral infarction.
Methods:
Ninety-four patients with cerebral infarction and 103 controls were analyzed. Plasma levels of VWF antigen (VWF: Ag), VWF ristocetin cofactor activity (VWF: Rcof), and VWF collagen binding activity (VWF: CB) were measured by enzyme-linked immunosorbent assay (ELISA). The ADAMTS13 activity (ADAMTS13) was measured with FRETS-VWF73. The relationship between plasma levels and ratios of VWF and ADAMTS13 and the occurrence of cerebral infarction were analyzed.
Results:
Patients with cerebral infarction displayed higher VWF: Ag and VWF: Rcof levels and lower ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof levels compared to controls (P < .01). The highest quartiles of VWF: Ag (odds ratio [OR] = 5.11, 95% confidence interval [CI], 1.49-17.50) and VWF: Rcof (OR = 5.04, 95% CI, 1.62-15.66) and the lowest quartiles of VWF: CB/VWF: Ag (OR = 5.91, 95% CI, 1.95-17.93), ADAMTS13/VWF: Ag (OR = 9.11, 95% CI, 2.49-33.33), and ADAMTS13/VWF: Rcof (OR = 3.73, 95% CI, 1.39-10.03) are associated with cerebral infarction.
Conclusions:
An association was found between reduced levels of VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof ratios and cerebral infarction. Our data suggest that increased levels of VWF and reduced levels of ADAMTS13 activity may contribute to the pathogenesis of cerebral infarction.
Introduction
Von Willebrand factor (VWF) is a large multimeric plasma glycoprotein that plays an important role in primary hemostasis. 1 It is synthesized in vascular endothelium and megakaryocytes, then stored in Weibel-Palade bodies of endotheliocytes and α granules of platelets, or released into the circulation. 2 Upon vascular wall damage, plasma VWF binds to collagen in the exposed subendothelial matrix, and platelet glycoprotein Ibα (GPIbα) triggers platelet aggregation and thrombus formation. 3 In vitro, the VWF/GPIb interaction can be studied using ristocetin as agonist. 4 As a result, von Willebrand factor ristocetin cofactor activity (VWF: Rcof) and von Willebrand factor collagen binding activity (VWF: CB) can be used to reproduce in vitro the ability of VWF to bind to platelet GPIb and collagen. It is ultralarge VWF (ULVWF) that produces a marked effect in recruiting circulating platelets to the site of endothelial activation and injury. But ULVWF is not usually found in the circulation. Indeed, a metalloprotease called a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13 (ADAMTS13), cleaves ULVWF into smaller, less active multimers. 5 Proteolysis by ADAMTS13 determines VWF activity, and plasma ADAMTS13 activity can be used to reflect VWF activity. The level of ADAMTS13 in the blood may thus influence thrombosis disease.
Cerebrovascular disease (CVD) has high morbidity and mortality worldwide, and cerebral infarction is the most common CVD manifestation. 6 Thrombus formation is a major factor in the pathophysiology of cerebral infarction. Supporting the pivotal role of VWF in platelet adhesion and thrombus formation, large population studies have revealed that increased VWF levels and reduced ADAMTS13 activity are associated with a risk of ischemic stroke. 7,8 Moreover, in recent years, an increasing number of studies have considered VWF an emerging target in stroke therapy. 9,10 However, none have revealed a specific threshold value of VWF as a diagnostic indicator to detect the VWF multimeric pattern changes in cerebral infarction.
In this study, we aimed to measure plasma levels of VWF antigen (VWF: Ag), VWF: Rcof, VWF: CB, and ADAMTS13 activity (ADAMTS13) in patients with cerebral infarction and controls and to analyze the ratios of VWF: Rcof/VWF: Ag, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, ADAMTS13/VWF: Rcof, and ADAMTS13/VWF: CB. We will use this information to assess their relationship with cerebral infarction.
Patients and Methods
Study Design
The participant group comprised 94 patients (median age 70 years, interquartile range [IQR], 63-77 years) who were diagnosed with cerebral infarction based on clinical symptoms, neurologic examination, and computed tomography or magnetic resonance imaging in the Department of Neurology of the First Affiliated Hospital of Soochow University, and 103 control participants (median age 60, IQR, 54-67 years) who were volunteers and matched by sex and index year and having no history, signs, or symptoms of cerebral infarction. All the participants in this study were recruited at random from residents of Suzhou, China, and its adjacent communities. The study was approved by the institutional ethics committee at the First Affiliated Hospital of Soochow University, China. All participants gave written informed consent.
Blood Sampling and Procedures
Whole blood samples from patients with cerebral infarction were collected within 24 hours of admission. Overnight-fasted patient blood samples were collected into evacuated tubes containing trisodium citrate (1:9, vol/vol). Plasma was immediately centrifuged at 1500g for 10 minutes at room temperature and stored at −80°C until analysis.
Measurements of VWF: Ag, VWF: Rcof, VWF: CB, and ADAMTS13
Previously, we developed improved sensitivity and reliability enzyme-linked immunosorbent assay methods for testing VWF antigen levels, VWF ristocetin cofactor activity, and VWF collagen binding activity. 11 In this study, we used these assays to measure plasma VWF: Ag, VWF: Rcof, and VWF: CB.
Activity of ADAMTS13 was measured with FRETS-VWF73 (Peptides International, Louisville, Kentucky), a chemically synthesized fluorogenic peptide containing 73 amino acid residues of VWF D1596 to R1668. 12 The normal plasma pool (NPP) was composed of 20 normal individuals with different ABO blood groups as mixed standard plasma. The NPP (diluted from 1:12.5 to 1:400) and test samples (diluted 1:25) were diluted in a pH 6.0 buffer containing 5 mmol/L Bis-Tris, 25 mmol/L CaCl2, and 0.005% Tween 20. Diluted samples of 50 μL and 6% FRETS-VWF73 in the assay buffer were added to each well of an EIA/RIA plate (Corning, New York). After 5 minutes, fluorescence was measured 31 times at 30°C with a 485-nm excitation filter and a 530-nm emission filter every 2 minutes.
Statistical Analysis
The levels of VWF: Ag, VWF: Rcof, VWF: CB, and ADAMTS13 and ratios of VWF: Rcof/VWF: Ag, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, ADAMTS13/VWF: Rcof, and ADAMTS13/VWF: CB were compared in cases and controls with Student t test and Mann-Whitney U test. Then, unconditional logistic regression modes were utilized to test the relation between high- and low-level variables and risk of cerebral infarction. They were divided into quartiles (Q1-Q4) based on the distribution in the control groups, and the highest quartiles (Q4) formed reference groups for the odds ratio (OR), except in the cases of VWF: Ag, VWF: Rcof, and VWF: CB, for which the lowest quartiles (Q1) were the reference groups. We adjusted for stratification variables (eg, sex, index year, and area of residence) when determining the first model, OR1, and additionally adjusted for several risk factors for vascular diseases (eg, age, diabetes mellitus, hypertension, hyperlipidemia, and C-reactive protein) in the second model, OR2. The receiver–operating characteristic (ROC) curve assessed the relation between the previous values and cerebral infarction, and the area under the curve (AUC) was used to compare the diagnostic value of each variable. Finally, to assess the value of cerebral infarction associated with the presence of multiple variables (eg, VWF: Ag, VWF: Rcof, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof), a joint analysis was calculated. The joint categories were classified by representing exposures to any of these factors, and no exposure was used as reference category. All statistical analyses were performed using software SPSS version 20.0 and MedCalc version 13.2.2.0.
Results
A total of 94 patients with cerebral infarction and 103 control participants are represented in this study, and their characteristics are shown in Table 1. As expected, there were significant differences between patients and controls with established risk factors for vascular diseases, such as age, diabetes mellitus, hypertension, hyperlipidemia, and C-reactive protein (P < .05).
Baseline Characteristics of Participants in the Case-Control Study.a
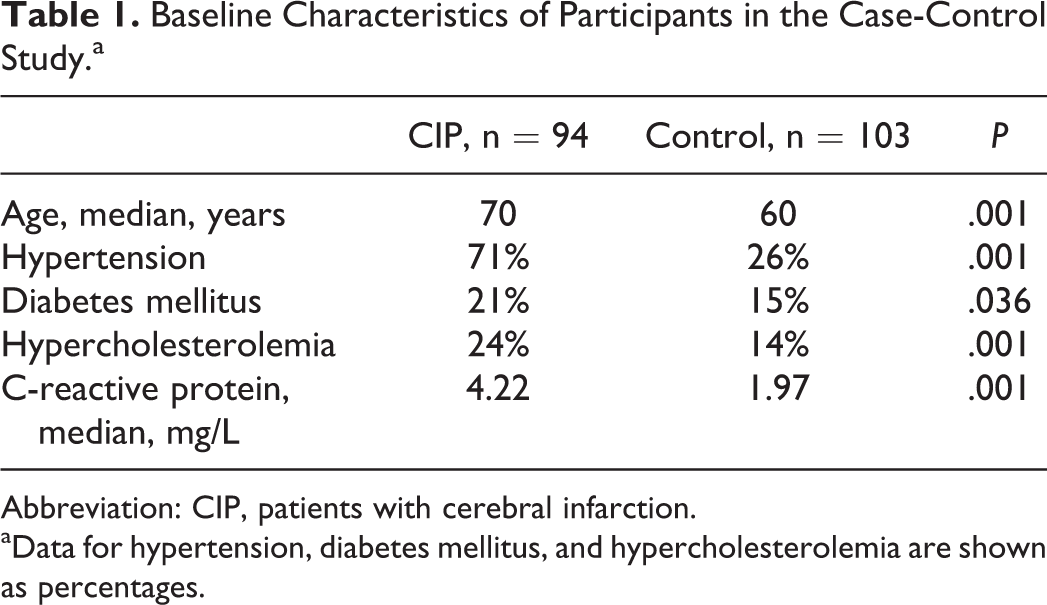
Abbreviation: CIP, patients with cerebral infarction.
aData for hypertension, diabetes mellitus, and hypercholesterolemia are shown as percentages.
As shown in Figures 1 and 2, the levels of VWF: Ag and VWF: Rcof in patients with cerebral infarction (median [IQR], 176% [114%-293%] and 197% [105%-381%]) were significantly higher compared with controls (105% [61%-145%] and 100% [60%-175%], P < .001). Levels of ADAMTS13 and ratios of VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof in patients (109% [80%-132%], 0.43 [0.28-0.90], 0.54 [0.32-0.99], and 0.57 [0.29-0.91], respectively) were significantly lower compared with controls (117% [103%-146%], 1.13 [0.72-1.81], 1.23 [0.79-2.16], and 1.18 [0.71-1.79], P < .01, respectively). However, there were no statistical differences between cerebral infarction and controls in VWF: CB (90% [53%-163%] vs 109% [73%-158%], P = .079), VWF: Rcof/VWF: Ag (0.95 [0.66-1.87] vs 1.18 [0.75-1.80], P = .405), or ADAMTS13/VWF: CB (1.05 [0.70-1.69] vs 1.10 [0.751.67], P = .694).
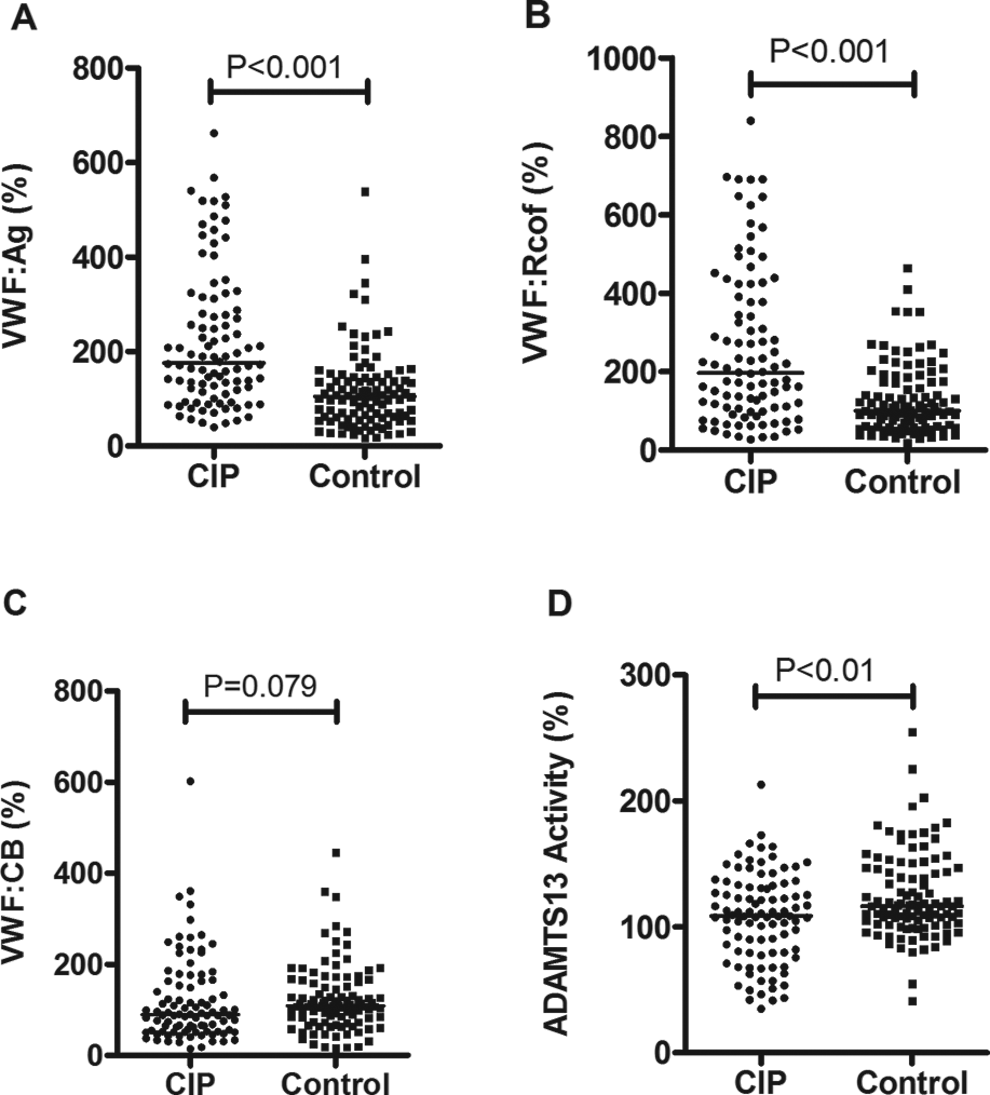
Levels (median) of VWF antigen (A), VWF ristocetion cofactor activity (B), VWF collagen binding activity (C), and a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13 (ADAMTS13) activity (D) in patients with cerebral infarction (CIP, n = 94) and control participants (control, n = 103). The values of pooled plasma from normal individuals were set at 100%. VWF, von Willebrand factor.
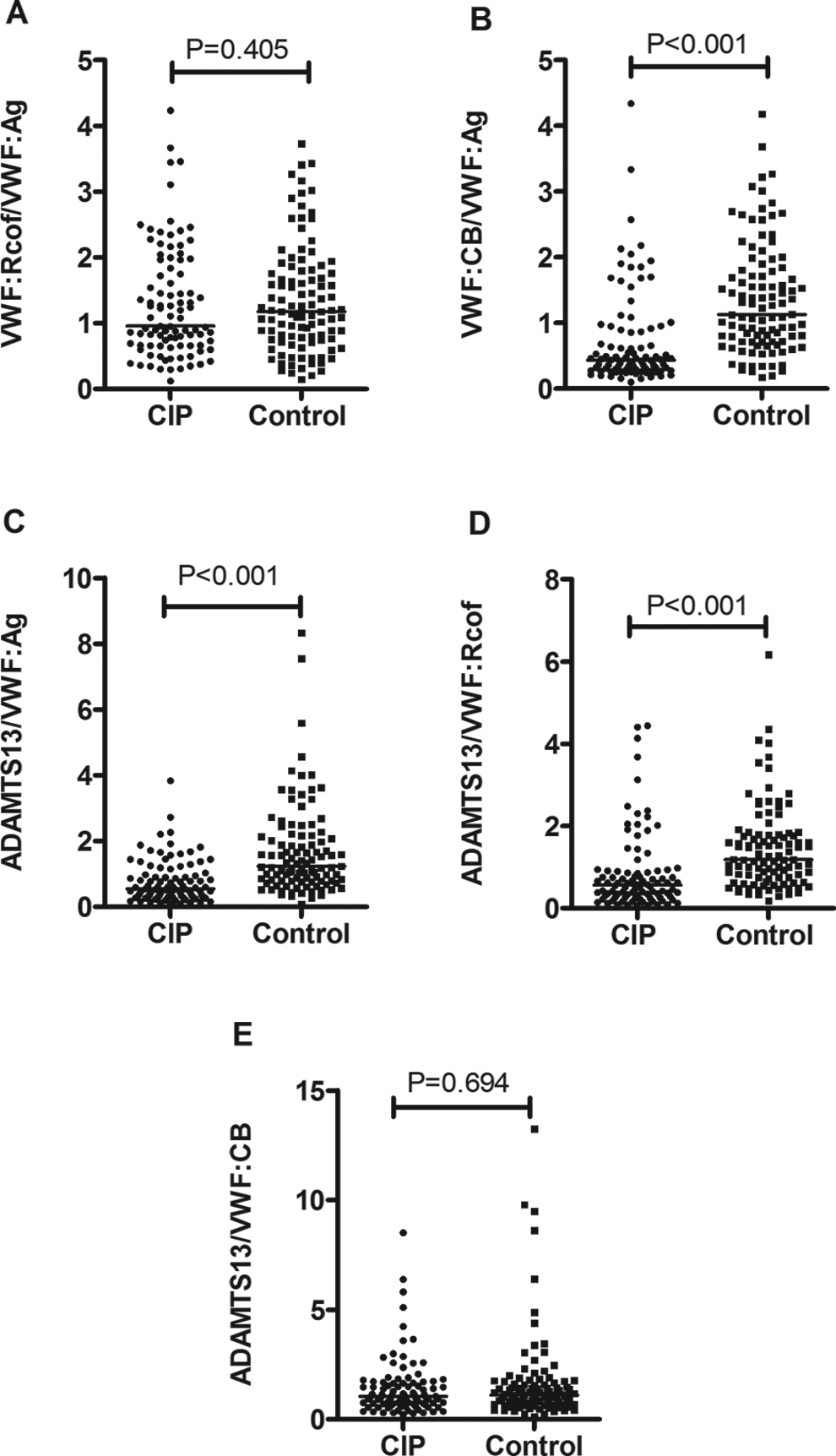
Ratios (median) of VWF: Rcof/VWF: Ag (A), VWF: CB/VWF: Ag (B), ADAMTS13/VWF: Ag (C), ADAMTS13/VWF: Rcof (D), and ADAMTS13/VWF: CB (E) in patients with cerebral infarction (CIP, n = 94) and control participants (control, n = 103). ADAMTS13 indicates a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; VWF, von Willebrand factor.
The ORs of VWF: Ag, VWF: Rcof, ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof, whose Student t test and Mann-Whitney U test results were significantly different between patients with cerebral infarction and controls, were calculated by logistic regression and were adjusted for sex, index year, area of residence, age, diabetes mellitus, hypertension, hypercholesterolemia, and C-reactive protein. Table 2 shows that the highest quartiles (Q4) of VWF: Ag and VWF: Rcof in patients with cerebral infarction displayed a significantly increased OR1 of 11.60 (95% confidence interval [CI], 4.00-33.58) and 5.56 (95% CI, 2.26-13.67) compared with the lowest quartiles (Q1) in controls. The OR1 of cerebral infarction in the lowest quartiles (Q1) of VWF: CB/VWF: Ag 6.30 (95% CI, 2.67-14.85), ADAMTS13/VWF: Ag 14.90 (95% CI, 4.72-47.10), and ADAMTS13/VWF: Rcof 4.31 (95% CI, 1.93-9.60) were also significantly increased compared with controls in the highest quartiles (Q4). The OR1 in the lowest quartile (Q1) for ADAMTS13 was 2.40 (95% CI, 1.06-5.44). Further adjustment (age, diabetes mellitus, hypertension, hyperlipidemia, and C-reactive protein, OR2) changed the estimates appreciably, but they were still associated with cerebral infarction.
Risk of Cerebral Infarction in Relation to VWF and ADAMTS13 and Their Ratios by Quartiles (Q1-Q4).a
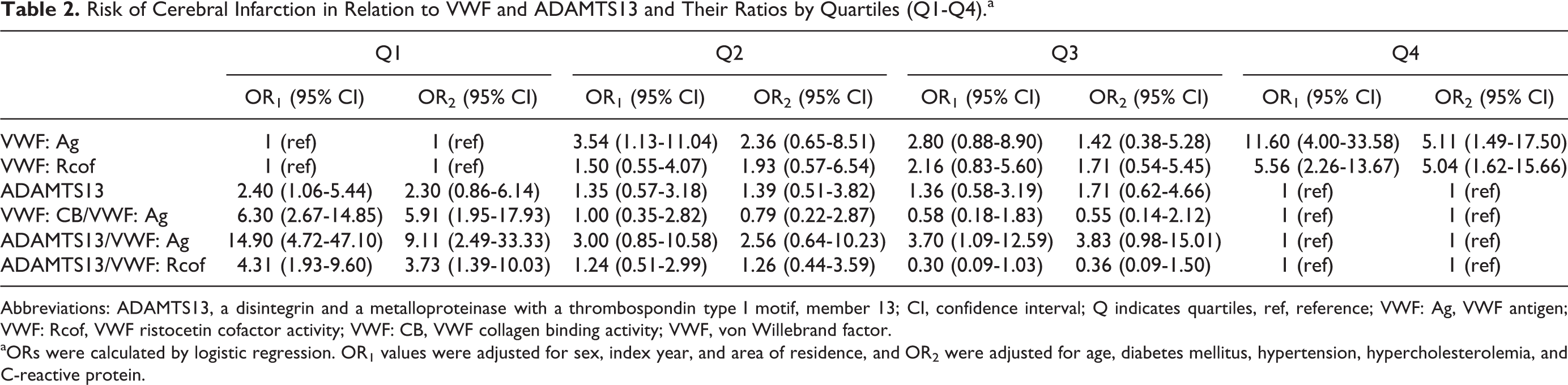
Abbreviations: ADAMTS13, a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; CI, confidence interval; Q indicates quartiles, ref, reference; VWF: Ag, VWF antigen; VWF: Rcof, VWF ristocetin cofactor activity; VWF: CB, VWF collagen binding activity; VWF, von Willebrand factor.
aORs were calculated by logistic regression. OR1 values were adjusted for sex, index year, and area of residence, and OR2 were adjusted for age, diabetes mellitus, hypertension, hypercholesterolemia, and C-reactive protein.
The ROC curves of VWF: Ag, VWF: Rcof, ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof are shown in Figure 3. Tables 3 and 4 show the AUC and their comparison of the previous values predict diagnostic value in cerebral infarction. Analysis of ROC curves detected the optimal cutoff values of VWF: Ag (137.88%, sensitivity 66.0%, and specificity 73.8%), VWF: Rcof (135.88%, sensitivity 66.0%, and specificity 68.0%), VWF: CB/VWF: Ag (0.538, sensitivity 64.9%, and specificity 88.3%), ADAMTS13/VWF: Ag (0.974, sensitivity 75.5%, and specificity 67%), and ADAMTS13/VWF: Rcof (0.946, sensitivity 78.7%, and specificity 64.1%). However, the AUC of ADAMTS13 (0.618) was significantly lower than the others (P < .01) except VWF: Rcof (AUC = 0.706, P = .097). There were no significant differences (P > .05) between VWF: Ag (AUC = 0.748), VWF: Rcof (AUC = 0.706), VWF: CB/VWF: Ag (AUC = 0.771), ADAMTS13/VWF: Ag (AUC = 0.773), or ADAMTS13/VWF: Rcof (AUC = 0.740).
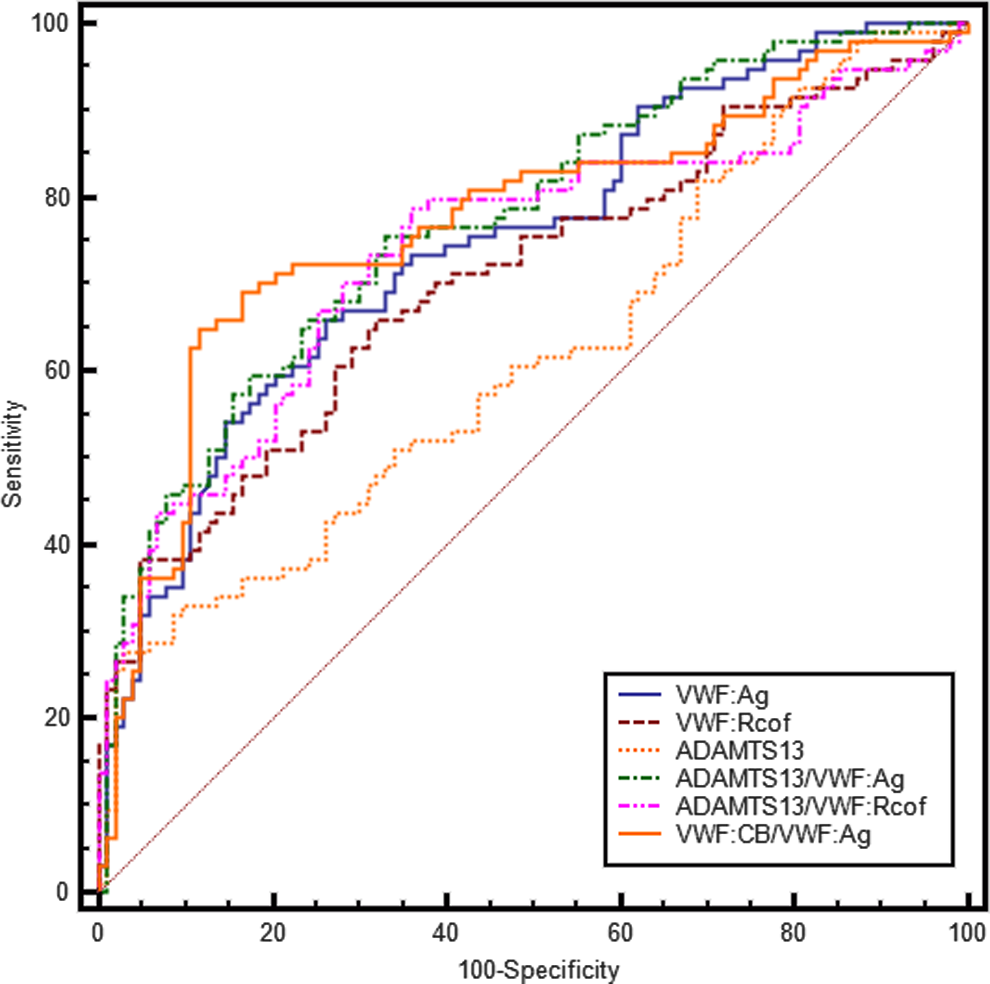
Receiver–operating characteristic (ROC) curve of VWF: Ag, VWF: Rcof, ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof. ADAMTS13 indicates a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; VWF: Ag, VWF antigen; VWF: Rcof, VWF ristocetin cofactor activity; VWF: CB, VWF collagen binding activity; VWF, von Willebrand factor.
AUC Evaluation of VWF: Ag, VWF: Rcof, ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof to Predict Cerebral Infarction Risk Diagnostic Value.
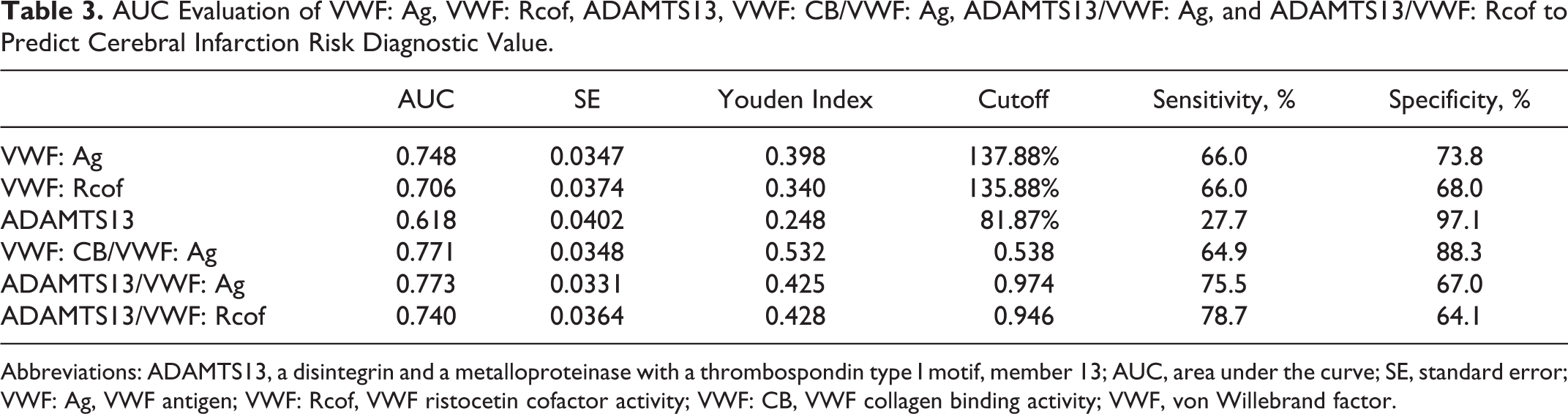
Abbreviations: ADAMTS13, a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; AUC, area under the curve; SE, standard error; VWF: Ag, VWF antigen; VWF: Rcof, VWF ristocetin cofactor activity; VWF: CB, VWF collagen binding activity; VWF, von Willebrand factor.
Comparison of ROC Curves in VWF: Ag, VWF: Rcof, ADAMTS13, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof to Predict Cerebral Infarction Diagnostic Value.a

Abbreviations: ADAMTS13, a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; ROC, receiver–operating characteristic; VWF: Ag, VWF antigen; VWF: Rcof, VWF ristocetin cofactor activity; VWF: CB, VWF collagen binding activity; VWF, von Willebrand factor.
a P values indicate the significance of the test for the differences between the areas under 6 ROC curves previously.
b P < .05.
To further identify the diagnostic value of the previous thresholds, we evaluated whether the ORs of cerebral infarction were increased by the concomitant presence of these indicators. The result of the ORs in Table 5 had shown that individuals who were exposed to 5 factors (eg, VWF: Ag > 137.88%, VWF: Rcof > 135.88%, VWF: CB/VWF: Ag ≤ 0.538, ADAMTS13/VWF: Ag ≤ 0.974, and ADAMTS13/VWF: Rcof ≤ 0.946) yielded a significantly higher OR1 of 182.00 (95% CI, 22.03-1503.37) for cerebral infarction than individuals exposed to 0, 1, 2, 3, or 4 factors. These associations were also obtained after adjustment for potential confounders (see OR2) and suggested that joint analysis of VWF: Ag, VWF: Rcof, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof acts as an independent indicator of cerebral infarction.
ORs of Cerebral Infarction Following Combination of 5 Exposures.a
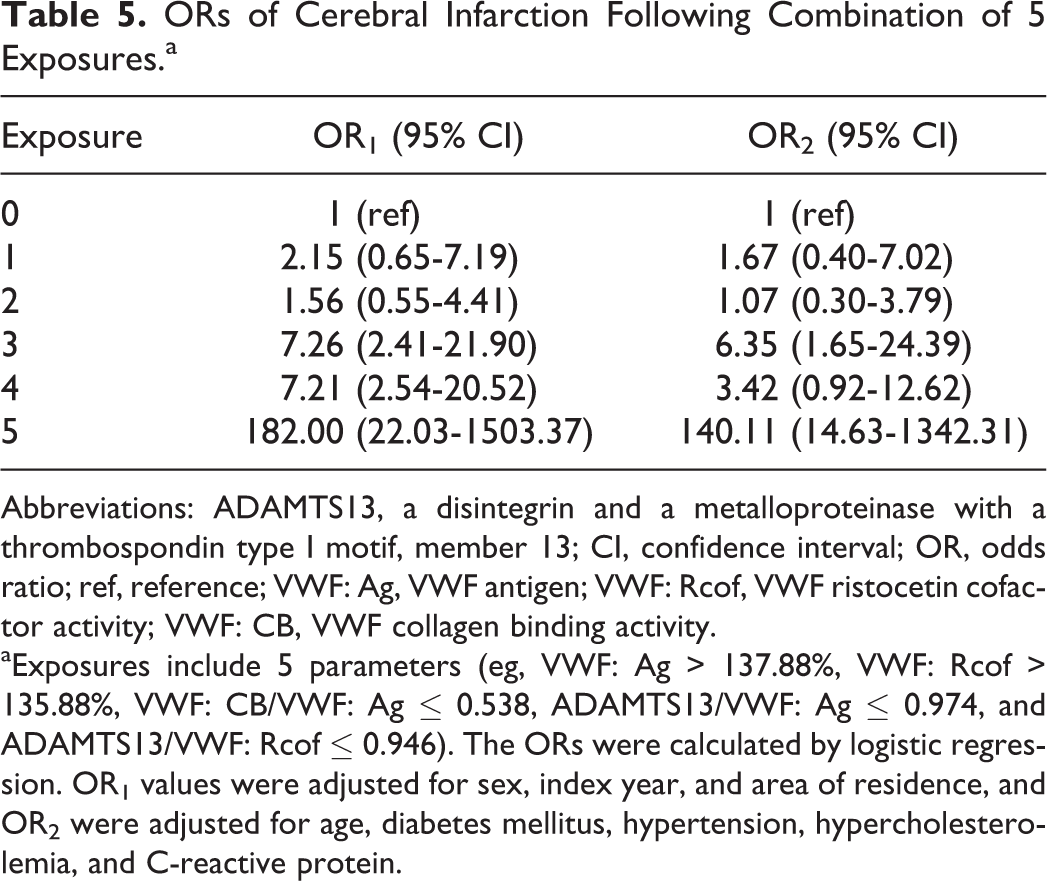
Abbreviations: ADAMTS13, a disintegrin and a metalloproteinase with a thrombospondin type I motif, member 13; CI, confidence interval; OR, odds ratio; ref, reference; VWF: Ag, VWF antigen; VWF: Rcof, VWF ristocetin cofactor activity; VWF: CB, VWF collagen binding activity.
aExposures include 5 parameters (eg, VWF: Ag > 137.88%, VWF: Rcof > 135.88%, VWF: CB/VWF: Ag ≤ 0.538, ADAMTS13/VWF: Ag ≤ 0.974, and ADAMTS13/VWF: Rcof ≤ 0.946). The ORs were calculated by logistic regression. OR1 values were adjusted for sex, index year, and area of residence, and OR2 were adjusted for age, diabetes mellitus, hypertension, hypercholesterolemia, and C-reactive protein.
Discussion
Von Willebrand factor as an adhesive plasma glycoprotein mediates platelet adhesion to damaged vascular subendothelium, which is followed by platelet aggregation and finally thrombus formation. 13 In this study, cerebral infarction was associated with the content of VWF and the activity of VWF and ADAMTS13, including VWF: Ag, VWF: Rcof, VWF: CB, ADAMTS13 and their ratios of VWF: Rcof/VWF: Ag, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, ADAMTS13/VWF: Rcof, and ADAMTS13/VWF: CB. The purpose is to comprehend relationships between these parameters and the occurrence of stroke events.
After analyzing the principle of statistics, we found that increased levels of VWF: Ag and VWF: Rcof and decreased levels of ADAMTS13 were correlated with cerebral infarction; this was consistent with those previous results. 14 –16 However, VWF: CB was not significantly different between patients and controls.
Our study differs from previous studies in that it includes not only the common content and activity indicators of VWF but also involves the ratios that are more precise in the respective effect estimates. Our results show that low levels of VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof were relevant with cerebral infarction, and the ratios of VWF: Rcof/VWF: Ag and ADAMTS13/VWF: CB were not significantly different between cerebral infarction and controls. Among these parameters, the lowest quartile of ADAMTS13/VWF: Ag is the most significant (OR1 = 14.90, 95% CI, 4.72-47.10) and showed almost 2- or 3-fold increase compared with the ORs of other parameters.
Von Willebrand factor is synthesized by endothelial cells and megakaryocytes and stored in the endothelial Weibel-Palade bodies and platelet α-granules. These storage granules contain larger, more hemostatic forms of VWF that are released upon stimulation by agonists such as thrombin, epinephrine, and fibrin. However, the ultralarge multimers (ULVWF) are not usually found in the circulation because of the cleaving function of ADAMTS13. 17 The VWF: CB represents the levels of ULVWF in plasma. 18 The result of VWF: CB in patients and controls suggest that ULVWF did not increase in cerebral infarction. Indeed, ADAMTS13 activity was lower in patients with cerebral infarction compared with controls, and the lowest quartile of ADAMTS13 (OR = 2.3, 95% CI, 0.86-6.14) was not statistically significant compared with individuals in the highest quartile. It is reasonable to assume that the higher plasma level of VWF antigen in patients with cerebral infarction cause VWF ristocetin cofactor activity to increase while VWF collagen binding activity is not affected by the increased VWF antigen levels, and the level of ADAMTS13 is functionally decreased since larger, more hemostatic forms of VWF are released to the damaged vascular subendothelium in cerebral infarction. This hypothesis is compatible with the previous reports of reduced plasma ADAMTS13 activity in acute myocardial infarction. 19 As a result, the plasma ULVWF with reduced ratio of ADAMTS13/VWF: Ag is not easy to be cleaved into the normal VWF multimeric pattern. Therefore, the patients with decreased ratio of ADAMTS13/VWF: Ag easily have thrombosis. In addition, the VWF: CB/VWF: Ag and ADAMTS13/VWF: Rcof ratios appear to be strongly influenced by increased VWF: Ag levels.
The ROC curves analysis showed that the threshold of VWF: Ag > 137.88%, VWF: Rcof > 135.88%, VWF: CB/VWF: Ag ≤ 0.538, ADAMTS13/VWF: Ag ≤ 0.974, and ADAMTS13/VWF: Rcof ≤ 0.946 have better diagnostic accuracy because AUC > 0.7 and ADAMTS13 ≤ 81.87% have lower accuracy because its AUC is < 0.7 and has low sensitivity. 20,21 As VWF: Ag and VWF: Rcof represent normal VWF multimeric pattern and VWF: CB and ADAMTS13 reflect the level of ULVWF in circulation, the combination of VWF, ADAMTS13, and their ratios, such as joint analysis of VWF: Ag, VWF: Rcof, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof, can improve accuracy in predicting cerebral infarction (as in Table 5) and reflect the changes in VWF multimeric distribution.
Lasek-Bal et al 22 and Chang et al 23 found that the elevated FVIII concentration plays a significant part in brain ischemia, but the mechanism is still unclear. It is known that VWF not only supports primary hemostasis but also serves as a carrier for factor VIII (FVIII), protecting this coagulation factor from proteolysis by the activated protein C system. Our findings that high levels of VWF were found in cerebral infarction and were compatible with the elevated FVIII concentration as described previously may provide a reasonable interpretation of the high levels of plasma FVIII in ischemic stroke. High levels of VWF protect more FVIII from proteolysis, and elevated FVIII promotes thrombosis when activated.
Plasma levels of VWF: Ag were higher in persons of blood group non-O compared to those in O group. Hence, blood group is a potential confounding factor of the relationship between VWF and cerebral infarction. A larger cohort would be desirable to improve the power of the study, and clinical interviews including new cases of stroke or transient ischemic attack, death from any cause, complications and dropouts should also be subsequently performed.
In conclusion, increased levels of VWF: Ag and VWF: Rcof and reduced levels of VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof are associated with cerebral infarction, and ADAMTS13/VWF: Ag is the optimal indicator to represent the VWF multimeric pattern changes in cerebral infarction. The combination of VWF: Ag, VWF: Rcof, VWF: CB/VWF: Ag, ADAMTS13/VWF: Ag, and ADAMTS13/VWF: Rcof may present a clear relationship between VWF and cerebral infarction and provide references for the clinical application of VWF target antithrombotics in the future.
Footnotes
Acknowledgment
We thank Zaixiang Tang for giving valuable advice during the preparation of this manuscript.
Authors’ Note
Le Qu and Miao Jiang contributed equally to this work.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Scientific Foundation of China (NSFC) (81270593 and 81100347), the National Key Technology R&D Program of China (2012BA118B02), Jiangsu Provincial Special Program of Medical Science (BL2012005), and Jiangsu Province’s Key Medical Center (ZX201102), China.
