Abstract
Protein Z (PZ), a cofactor for PZ-dependent protease inhibitor, is known to play an important role in inhibiting the coagulation cascade. The aim of the study was to investigate whether PZ G79A polymorphism is a risk factor for puerperal cerebral venous thrombosis (CVT). A total of 71 patients with puerperal CVT and 98 healthy controls were genotyped for PZ 79GA polymorphism by polymerase chain reaction-restriction fragment length polymorphism method. In patients, the genotype distribution for GG, GA, and AA genotypes was 22.5%, 43.7%, and 33.8%, and in controls, 25.5%, 40.8%, and 33.7%, respectively. The risk associated with carrying the mutant genotype (GA and AA) versus the wild GG genotype was found to be 1.11 (95% confidence interval: 0.52-2.35; P = .909). There was no significant difference in the clinical features of the patients with and without the polymorphism. We therefore conclude that PZ G79A polymorphism is not a risk factor for puerperal CVT in Indian women.
Introduction
Thrombosis of the cerebral veins and sinuses occurring during the puerperal period is one of the dreaded nonobstetrical complications. Puerperal cerebral venous thrombosis (CVT) is 10 to 12 times more frequent in India compared to developed countries 1 and is associated with a risk of maternal mortality and morbidity. The etiology of puerperal CVT still remains unclear. In the last few decades, substantial progress has been made to understand the role of genetic thrombophilias as risk factors for CVT, 2 –5 but there are few studies 6,7 that specifically explore the role of genes in CVT occurring during puerperium. The thrombophilic changes occurring during pregnancy and puerperium could be translated into a risk of thrombosis in women with underlying inherited conditions that increase susceptibility to thrombosis. 8
Protein Z (PZ) is a single-chain, vitamin K-dependent, plasma glycoprotein with a molecular weight of 62 kDa. 9 The PZ gene spans about 14 kb with 9 exons and is localized on chromosome 13q34. 10 The amino acid sequence of PZ shows wide homology with many coagulation factors, such as VII, IX, X, and protein C, but it is not a zymogen of a serine protease, unlike many other vitamin K-dependent coagulation factors. 11 It instead acts as a cofactor for PZ-dependent protease inhibitor (ZPI), along with calcium. The ZPI along with PZ, calcium, and activated factor X (FXa) forms a ternary complex, which is known to inhibit FXa on the phospholipid membrane. 12 On the contrary, PZ also promotes assembly of thrombin on phospholipid surfaces, thus enhancing coagulation. 13
Several polymorphisms in PZ gene have been described. 14 –17 Many of these polymorphisms are known to influence the plasma PZ concentration either individually or in combination. 18 The commonly known protein Z G79A (PZ G79A) polymorphism is a point mutation caused by the transition of guanine to adenine nucleotide at intron F of PZ gene. It has been reported that individuals with PZ G79A polymorphism have reduced plasma PZ levels. 16 Protein Z deficiency is associated with prothrombotic events. 19 Studies have shown that an inverse relationship exists between plasma PZ levels and the risk of thrombotic events such as arterial or venous thrombosis (VT). 20,21 The present study aimed at investigating the risk of CVT occurring during puerperium in individuals with PZ G79A polymorphism.
Materials and Methods
The study was approved by the Human Ethics Committee of the National Institute of Mental Health and Neuro Sciences (NIMHANS), Bangalore, India. Seventy-one first ever patients with puerperal CVT who were treated at NIMHANS were recruited for the study. The following diagnostic criteria have been considered for the selection of patients: (1) symptoms suggestive of raised intracranial pressure or acute encephalopathy including headache, seizures, altered mental status, and focal neurological deficits referable to the encephalon or brain stem; (2) patients in the postpartum period, till 4 weeks after childbirth; and (3) magnetic resonance imaging/magnetic resonance venography of brain showing evidence of CVT. Patients were excluded from the study when: (1) neuroimaging studies were inconclusive; (2) CVT was secondary to head trauma, invasive procedures, sepsis, neuroinfection, or malignancy; (3) clinical and laboratory evidence of renal or hepatic dysfunction and blood cell disorders were present; or (4) previous history of venous thrombosis (deep vein thrombosis/CVT) or thrombophilia was present.
Ninety-eight age-matched, clinically healthy women from the same geographic and socioeconomic group with no postpartum complications were selected as controls for the study. Demographic data, medical history, and details regarding recognized risk factors for CVT were recorded.
Venous blood was drawn from the study patients after obtaining written informed consents. Extraction of human genomic DNA from whole blood was carried out using the protocol of Miller et al . 22 The method described by Lichy et al was followed for the detection of PZ G79A polymorphism. 23 Polymerase chain reaction (PCR) was carried out with the following forward and reverse primers, respectively: 5′-TAACACCATAGACAGAGTCCGATATTCGC-3′ (Sigma-Aldrich, St Louis) and 5′-ATGAACTCGGCATTAGAACATGGTTGGAA-3′ (Sigma-Aldrich). The HpaI restriction digested fragments were separated on 2% agarose gel (Figure 1). Depending on the presence or absence of polymorphism, a specific band pattern was obtained on the gel, which was used for identification of different genotypes. In the wild type (GG genotype), a single band of 320 bp was observed. In the heterozygous mutants (GA genotype), bands of 320, 221, and 99 bp were present. In homozygous (AA genotype), bands of 221 and 99 bp were seen. The results of genotyping were confirmed by sequencing of the PCR products.
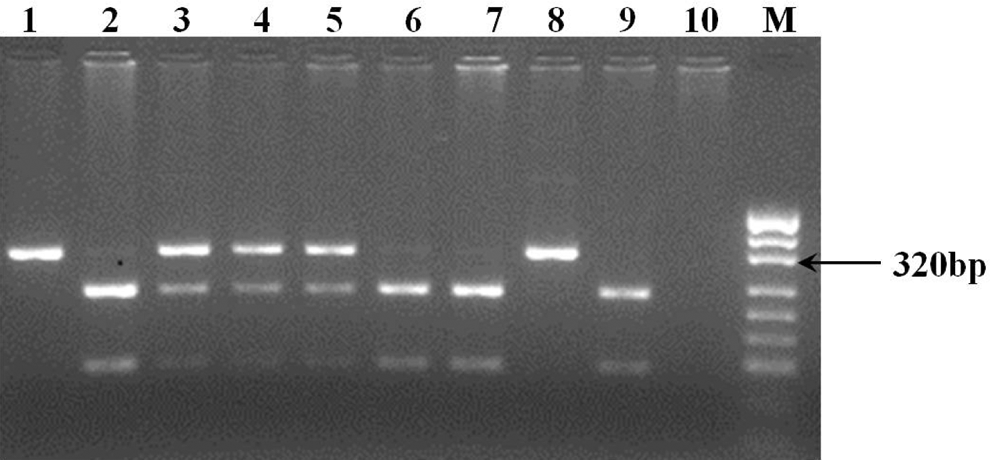
Restriction fragment length polymorphism (RFLP) analysis and representative band patterns for the protein Z intron F G79A polymorphism. Lanes 1 and 8 are the samples of patients with GG wild genotype; lanes 2, 6, 7, and 9 are the samples of patients with mutant AA genotype; and lanes 3, 4, and 5 are the samples of patients with heterozygous GA genotype, M: DNA marker: pUC19 DNA/MspI (HpaI; Fermentas).
The statistical analysis of data was performed using SPSS version 11. Student t test was used for the comparison of mean age between patients and controls. The odds ratio and 95% confidence interval (CI) were done to find risk associated with the mutation. Chi-squared test or Fisher exact test (2 × 2 tables, when the sample number was ≤5) was used to find the distribution of risk factor between controls and patients and between patients with and without polymorphism. The value of P < .05 on 2-sided tests was considered as significant. Genotype frequency and allelic frequency were calculated by Hardy-Weinberg formula.
Results
The characteristics of patients and healthy controls are given in Table 1. The age of the patients ranged between 18 and 37 years with mean age of 23.4 ± 3.67 years. None of the study patients were found to be smokers. Only 1 patient reported a history of alcohol consumption . History of intrauterine fetal deaths was reported in 8.5% of patients with puerperal CVT. The most common period for the onset of symptoms was found to be the third postpartum week; 45.1% of patients had onset of symptoms during this period, followed by the second postpartum week (31%). In a single case, onset of symptoms occurred within 48 hours of delivery. The most common symptoms were headache 63 (88.7%), seizures 58 (81.7%), and altered sensorium 44 (61.9%). Other clinical features included limb weakness 25 (35.2%), papilledema 21 (29.6%), and fever 19 (26.7%). Routine laboratory investigations showed that serum venereal disease research laboratory test, antinuclear antibodies, lupus erythematosus cells, and rheumatoid arthritis tests were negative in all patients. Anemia was present in 74%.
Characteristics of Patients With Puerperal Cerebral Venous Thrombosis and Healthy Controls.
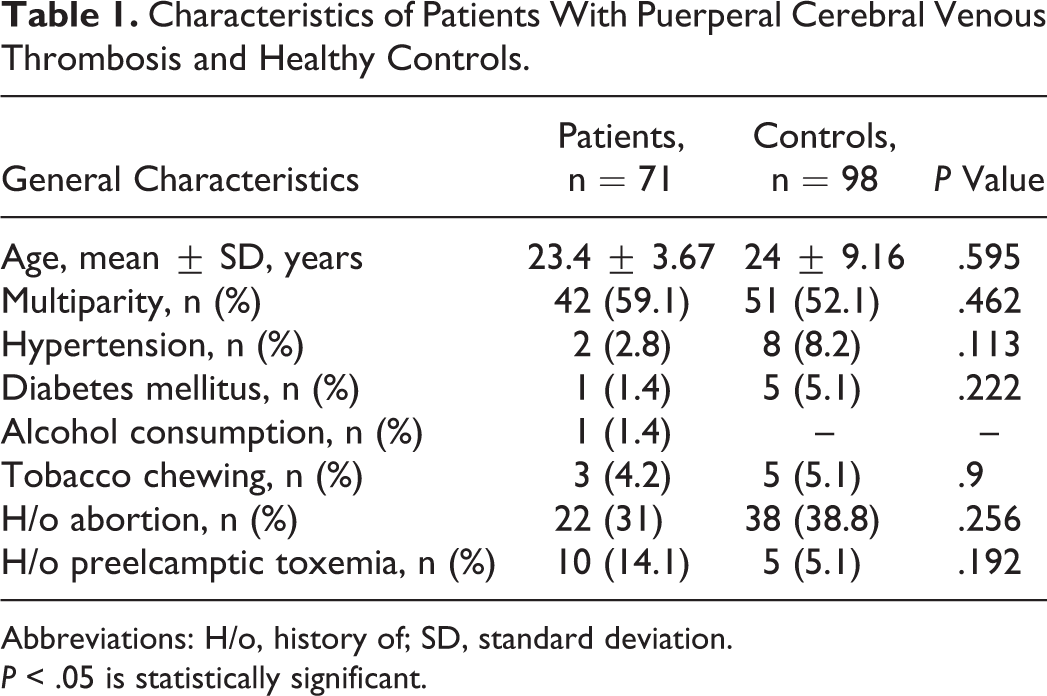
Abbreviations: H/o, history of; SD, standard deviation.
P < .05 is statistically significant.
The results of the PZ genotyping are shown in Table 2. Wild genotype with GG alleles was found in 22.5% of patients and 25.5% of controls. In all, 43.7% of patients and 40.8% of controls were found to be heterozygous having GA alleles. Homozygous genotype, having AA alleles, was found in 33.8% of patients and 33.7% of controls. The risk of CVT associated with PZ G79A polymorphism was found to be 1.11 (95% CI = 0.52-2.35; P = .909), suggesting that the presence of this polymorphism does not increase the risk of CVT during puerperium. There was no significant difference in the clinical features of the patients with and without the polymorphism.
Protein Z Intron F G79A Genotype Distribution Among Patients and Controls.
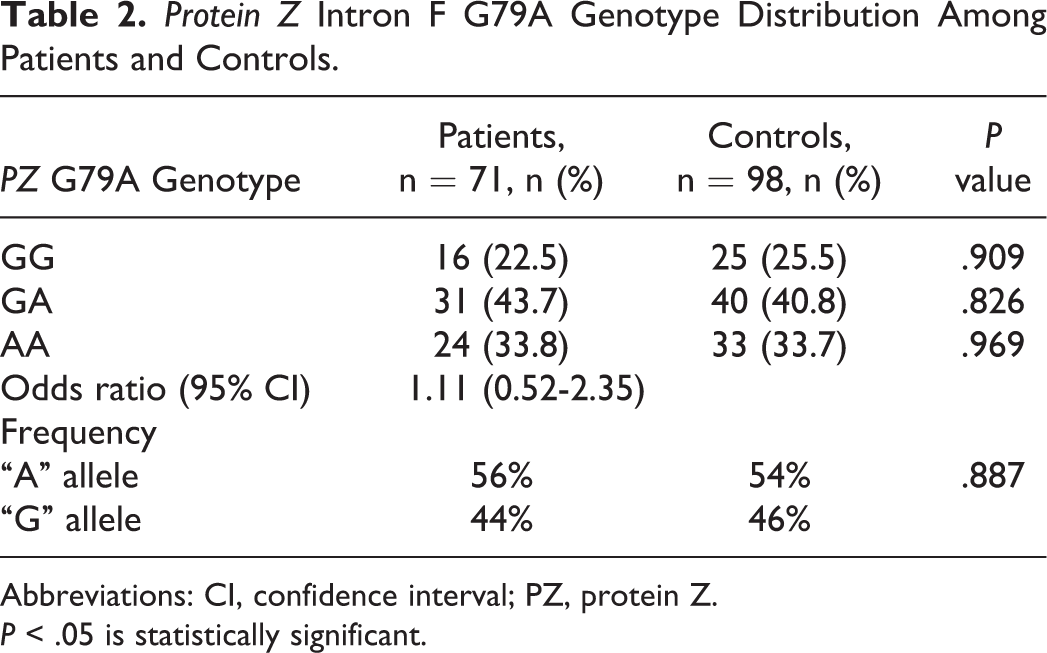
Abbreviations: CI, confidence interval; PZ, protein Z.
P < .05 is statistically significant.
Discussion
Thrombosis is considered as a multicausal disease. The major contributors to thrombotic risk are abnormal levels of some coagulation factors and defects in the natural anticoagulant proteins. 24 Common polymorphisms in the coagulation factor genes such as factor V Leiden and prothrombin G20210A and the less common thrombophilias like deficiencies of protein S, protein C, and antithrombin III have been found to play significant roles in causing CVT. 25 In addition, pregnancy and puerperium raise the risk of thrombotic events, and these risks are likely to be increased in women who are carriers of genetic thrombophilic conditions.
In this study, we have evaluated the risk associated with G79A polymorphism of the PZ gene in the patients with CVT occurring during puerperium. No significant difference (P = .887) was observed between patients with puerperal CVT and healthy controls. Also, no statistical significance was seen when the general characteristics and clinical symptoms were compared between patients with and without the polymorphism.
There are very few studies on PZ G79A polymorphism in CVT. These case–control studies report contradictory findings. In a retrospective, case–control study on 54 patients with CVT, Le Cam-Duchez et al found that PZ G79A polymorphism was an independent risk factor for CVT. 26 On the other hand, another study on 77 patients with CVT concluded that PZ G79A polymorphism was not significantly associated with CVT. 3 Our data are consistent with the later study suggesting that PZ G79A polymorphism is not a risk factor for CVT.
Recently, a meta-analysis on vascular thrombosis has concluded that the deficiency in plasma PZ levels was a risk factor for the thrombotic events such as arterial thrombosis, pregnancy complications, and VT. 14 The drawback of our study is that the relationship between plasma levels of PZ and PZ G79A polymorphism was not evaluated because our findings indicated the insignificance of this polymorphism in causing CVT during puerperium.
To the best of our knowledge, this is the first study investigating the influence of the PZ G79A polymorphism as a risk factor for puerperal CVT. Large prospective studies remain to be done to investigate its possible role in CVT.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Indian Council of Medical Research, New Delhi, India.
