Abstract
The objective:
This study was undertaken to detect characterization of the different gene polymorphisms in Human killer cell immunoglobulin-like receptor (KIR2) gene and multi-drug resistance (MDR1) gene, among childhood ITP Egyptian patients. In addition to assess the potential role of these polymorphisms in relation to types of ITP and response to different treatment modalities.
Patients and Methods:
A total of 48 pediatric patients with immune thrombocytopenia (ITP; 24 newly diagnosed and 24 chronic) and 35 healthy controls were investigated via polymerase chain reaction-restriction fragment length polymorphism analysis for multidrug resistance (MDR) 1 and killer cell immunoglobulin-like receptor (KIR) 2 genes.
Results:
The frequency of MDR1 gene in patients and control was not significant (P = .090). The CT genotype was the highest distribution among all ITP cases (62.50%, n = 30) and control (48.60%, n = 17). There was a significant difference in age at diagnosis of MDR1 gene with the CC genotype had the eldest age and lowest initial platelets count (P = .029 and P = .004). The distribution of KIR2 gene among all patients with ITP and controls was significant (P = .026) with (KIRDL2−/KIRDS2−) genotype was the most prevalent among patients.
Conclusion:
The frequency of MDR1 polymorphisms was not associated with susceptibility to the development and clinical progression of the disease. However, KIR2 gene polymorphisms were independently associated with childhood ITP in Egyptian patients with highest prevalence among (KIRDL2−/KIRDS2−) genotypes.
Keywords
Introduction
Childhood immune thrombocytopenia (ITP) is a common pediatric hematologic disorder characterized by increased destruction of antibody-sensitized platelets with normal to increased megakaryocytes in the bone marrow, as well as the presence of thrombocytopenia with otherwise normal red cells and leukocytes, absence of splenomegaly, and the absence of other causes of thrombocytopenia. 1 The decrease in platelets is caused by increased autoantibodies against self-antigens, particularly immunoglobulin G (IgG) antibodies against glycoprotein IIb/IIIa. The production of these autoantibodies by B cells depends on a number of cellular mechanisms that form a network of modulation, with T cells playing a pivotal role in pathophysiology. 2 Previous studies painted a broader picture of immune dysregulation leading not only to accelerated platelet destruction but also to abnormalities in megakaryocyte growth and development and poorly compensated thrombopoiesis. 3 Although the etiology of ITP remains unclear, it is generally accepted that both environmental and genetic factors play an important role in the development of the disease. 4 Many studies have focused on the association between some noncytokine gene polymorphisms and susceptibility to ITP. 5 Gene polymorphisms of noncytokines are associated with several diseases including ITP. 6
Recently, it has been shown that the conservative mutations in the regions, where noncytokines are encoded, and the nucleotide changes in the regulatory region might be the causes of individual differences in noncytokine production and that these genetic polymorphisms affect the release of noncytokines both in vivo and in vitro media. It was reported that there was an association between the noncytokine gene polymorphisms affecting the noncytokine production and secretion and infectious diseases, allergic diseases, autoimmune diseases, and malignant diseases both at the stage of formation of disease and in the course of disease and their responses to treatment. 4 In principle, ITP biology and care will benefit greatly from modern methods to understand the patterns of gene expression and genetic markers associated with fundamental parameters of the disease including predictors of remission, risk factors for severity, determinants of response to various therapies, and possibly biological subtypes. 7
Multidrug resistance 1 (MDR1) is characterized by the over function of P-glycoprotein (P-gp), a pump molecule that decreases intracellular drug concentration by effluxing them from the intracellular space. P-gp is expressed in the apical membrane of cells with excretory functions, such as those in the liver, kidney, small intestine, stomach, and the blood–brain barrier. 8 Functional P-gp is found in several types of human leukocytes and stem cells. Among hematological cells, expression of P-gp is highest in natural killer (NK) cells, CD4+ and CD8+ lymphocytes, and bone marrow progenitor cells. 9 Exon 26 3435CNT polymorphism was found to correlate significantly with intestinal P-gp expression levels as well as the bioavailability of some drugs. 10 High P-gp-mediated MDR1 (P-gp/MDR1) activity in lymphocytes from patients with ITP may affect disease outcome. The ITP treatment includes glucocorticoids that are substrates of P-gp. 11 There is, however, little information regarding the MDR1 polymorphism in ITP disease and response to treatment. 1
The activation of T cells is mediated through T-cell receptors (TCRs), and this activation can be modulated by killer cell immunoglobulin-like receptors (KIRs). 12,13 The KIR are members of the immunoglobulin superfamily and are expressed in NK cells and subsets of T cells. Depending on their structure, they can generate activating or inhibitory signals. 14 Besides the modulation of TCR-mediated activation of T cells, expression of KIR may affect the role of NK cells in autoimmune diseases, where these cells may exert a pathogenic function through inappropriate activation or suppression function through lysis of dendritic cells or activated T cells. 14 Therefore, genes that control expression of KIR may possibly influence normal and pathological immune responses. Further, genetic associations of activating KIRs have been associated with a number of autoimmune conditions, including psoriatic arthropathy, Sjogren syndrome, and scleroderma. 15 –17
A previous study by Seymour et al 18 suggested that there was an overrepresentation of KIR2DS3 (known to be in linkage disequilibrium with KIR2DS2 and 2DL2) and underrepresentation of KIR2DS5 (also protective against other immune mediated disorders) in adult ITP. In addition, KIR2 genes and polymorphisms of FCGR3a were associated with autoimmunity as Nourse et al 19 reported that KIR2DS2/DL2 genotype was associated with adult persistent/chronic and relapsed ITP independently of FCGR3a-158 polymorphisms. Of note, KIRs are polymorphic both in terms of alleles and haplotype gene content and there is particular variation within populations in the number of activating KIRs carried by different individuals. 17
It is worth noting that risk susceptibility studies cannot be extrapolated from one ethnic group to the other due to well-known variability of the relative frequency of the different alleles among different population. Therefore, in order to evaluate a possible role of MDR1 and KIR2 genes as well as of their polymorphisms in childhood ITP, we analyzed the allelic and genotypic frequencies of both gene polymorphisms in healthy and Egyptian patients with ITP. In addition, we assessed the potential role of these polymorphisms in relation to types of ITP, progression of disease, and response to different treatment modalities.
Patients and Methods
This is a case–control study which was conducted at Pediatric hematology Clinic Children’s Hospital, Ain Shams University, over 10 months period between June 2013 and April 2014. Those with primary ITP and less than 16 years of age were included in the study. Exclusion criteria were those more than the determined age limit, patients with proven secondary ITP (as cases initiated by or associated with infections due to human immunodeficiency virus [HIV associated], Helicobacter pylori-associated, and hepatitis C virus [HCV]-associated secondary ITP). Moreover, concomitant autoimmune disorders such as systemic lupus erythematosus (SLE) and other diseases as common variable immunodeficiency or selective IgA deficiency were also excluded.
The ITP diagnosis was made after a detailed physical examination and history, testing for the presence of thrombocytopenia (<100 × 109/L) with otherwise normal red cells and leukocytes, an evaluation of a Giemsa-stained peripheral blood smear and bone marrow aspiration, and negative serological tests for infectious causes and autoimmune diseases by performing antinuclear antibodies, serologic evaluation for HIV, HCV, detection of H pylori infection, as well as normal values of quantitative baseline immunoglobulin level testing (IgG, IgA, and IgM). We have used the definition of the ITP disease phases 20 as follows.
Newly diagnosed ITP: within 3 months from diagnosis. Persistent ITP: between 3 and 12 months from diagnosis. Includes patients not reaching spontaneous remission or not maintaining complete response of therapy. Chronic ITP: disease lasting for more than 12 months.
Enrolled patients were divided into 2 groups: Group 1: Patients with newly diagnosed ITP received different treatment protocols (a prospective study arm) and were followed up for 3 months to detect the remission or progression into the persistent course of the disease. Group 2: Patients with chronic ITP who were regularly attending the clinic on different treatment protocols (a retrospective study arm).
Thirty-five healthy volunteers with no obvious medical disorder and not receiving any medication served as a control group. This group included children recruited from the same region as the case patients; most were classmates or acquaintances of the case patients. They were age- and gender-matched healthy individuals with patients with ITP (P = .84 and P = .87, respectively).
The study protocol was approved by the ethical committee of Ain Shams University, and all the patients, control participants, and parents of study participants gave their written informed consent after explaining the study design and tests.
Methods
All patients underwent all of the following.
Detailed Questionnaire
Information on demographic characteristics, duration of disease, bleeding symptoms, life-threatening bleeding (if patients had clinically significant symptoms of severe bleeding such as intracranial bleeding, internal hemorrhage, gastrointestinal, or genitourinary bleeding), any history of proceeding viral infection, recent vaccination, and medications use was obtained during history taking.
Clinical Assessment
Physical examination includes anthropometric measures, weight in kg and height in cm. Severity of bleeding was also assessed using specific bleeding score from grades 1 to 4 which is as follows
21,22
: Grade 1: minor bleeding, few petechiae (≤100 total) and/or ≤5 small bruises (≤3-cm diameter); no mucosal bleeding. Grade 2: mild bleeding, many petechiae (>100 total) and/or >5 large bruises (>3-cm diameter); no mucosal bleeding. Grade 3: moderate bleeding, overt mucosal bleeding, troublesome lifestyle. Grade 4: mucosal bleeding or suspected internal hemorrhage.
Specific treatment was received to newly diagnosed patients according to Provan et al 22 either:
Corticosteroids: a course of prednisone 2 mg/kg/d for 2 weeks then tapered over 21 days in patients with a platelet count between 10 and 29 × 109/L and no evidence of mucosal hemorrhage or
Intravenous immunoglobulin (IVIG): low-dose IVIG given 1 to 2 days of 0.8 to 1.0 g/kg.
The chronic ITP group was divided into 2 subgroups according to the line of treatment received either:
1. Patients received steroid therapy a. High-dose dexamethasone: used in a child who had mucous membrane bleeding and more extensive cutaneous symptoms which was given on a dose of 28 mg/m2/d over a very short course (maximum 4 days). b. High-dose methyl prednisolone: it was given at a dose of 30 mg/kg/d for 3 days and 20 mg/kg/d for 4 days, consecutively and intravenously. The maximum dose given was 1 g or
2. Patients received nonsteroid therapy. This comprised the following lines of treatment:
(1) Intravenous immunoglobulin:
It was given as a single dose of 0.8 to 1g/kg.
(2) Anti-D immunoglobulin:
It was given to Rh (D)-positive individuals as a short infusion (50-75 µg/kg).
(3) Thrombopoietin receptor agonists:
Romiplostim was given (Nplate 250 µg; Amgen Europe BV, Breda, the Netherlands). The initial starting dose was 1 μg/kg based on actual body weight. The weekly dose adjusted by increments of 1 μg/kg until the platelet count was 50 × 109/L or higher. The maximum weekly dose was 10 μg/kg. When platelet counts were greater than 200 × 109/L for 2 consecutive weeks, the dose reduced by 1 μg/kg weekly. When platelet counts exceeded 400 × 109/L, the dose withheld until the count had fallen below 200 × 109/L. The dose resumed with a reduction of 1 µg/kg weekly.
(4) Immunosuppressive and cytotoxic drugs (azathioprine):
They used less commonly, mainly to salvage severely affected and highly refractory patients on a dose of 2 mg/kg postoperatively daily.
We used for assessment the criteria recommended in the evaluation of response to ITP treatment 20 as follows.
Complete response (CR); any platelet count of at least 100 × 109/L and absence of bleeding. Response (R): any platelet count ≥30 × 109/L and at least 2-fold increase the baseline count and absence of bleeding. No response (NR): any platelet count <30 × 109/L or less than 2-fold increase in baseline platelet count or bleeding. Loss of CR or R: platelet count below 100 × 109/L or bleeding (from CR) or below 30 × 109/L or less than 2-fold increase in baseline platelet count or bleeding (from R). Time to response: time from starting treatment to time of achievement of CR or R.
Investigations
Full blood count using Coulter GenS systems (Beckman Coulter Inc, Miami, Florida) and careful film examination.
Bone marrow aspiration report was recorded (if done).
Gene polymorphism using polymerase chain reaction (PCR) for multidrug resistance gene 1 (MDR1) which includes: (1) CC genotype: represents the wild genotype, (2) TT genotype: represents the mutant genotype, and (3) CT genotype: represents the heterozygote genotype of MDR1 gene.
Gene polymorphism using PCR for human KIR2 gene. To detect KIR inhibitory gene KIR2DL2 and the noninhibitory KIR2DS2 of the Human KIR2 gene.
DNA Extraction
The work is carried out in accordance with “The Code of Ethics of the World Medical Association” (Declaration of Helsinki) for experiments in Human. DNA was extracted from peripheral blood leucocytes by spin column method of GeneJET Genomic DNA purification kit (#K0722-0721; PureExtreme Fermentas Life Sciences, Thermo Scientific, Lithuania, European Union). The eluted DNA was stored at −20°C till application.
A. Genotyping of the human MDR gene (Figure 1; MDR1; ABCB1) was identified by PCR-restriction fragment length polymorphism (PCR-RFLP) assay using primers sequence for in vitro amplification of the gene according to Hoffmeyer et al.
23
Template DNA (500 ng) was amplified in the presence of 25 μL Dream Taq Green PCR master mix (2×; #K1081; Thermo Scientific/Fermentas, Vilnius, Lithuania) containing 4 mmol/L MgCl2, 0.4 mmol/L each dNTPs, and 2.5 U/μL Taq DNA polymerase in addition to 1 μmol/L for each of the forward and reverse primers. The PCR amplification was performed on thermal cycler (HVD Life Science, S24 thermal cycler; Quanta Biotech Manufacturing, Surrey, England, United Kingdom) as shown in Table 1. Analysis of the amplified products was done on agarose gel electrophoresis 1.6% stained with ethidium bromide to detect the corresponding amplified fragment (248 bp). The restriction enzyme MboI FastDigest (#FD1004 Thermo Scientific/Fermentas) was used for detecting C to T transition at position (3435C>T) within exon 26 of MDR1 gene located on chromosome 7. The endonuclease cleavage was performed by mixing 20 μL of amplified PCR products and 2 units of the enzyme in the corresponding appropriate buffer for 2 hours at 37°C. Digested products (Table 2) were separated on a 3% agarose gel. Polymerase chain reaction (PCR)-amplified fragments of multidrug resistance (MDR) gene on 3% agarose gel electrophoresis stained with ethidium bromide (after restriction digestion and RFLP analysis by MboI). Lane 1: a 50-bp DNA molecular weight marker. Lane 2: wild-type DNA (w/w) yields fragments of 172, 60, and 16 bp (16 bp is a faint band below). Lane 3: the heterozygote genotype (w/m) yielded 4 fragments of the following lengths: 232, 172, 60, and 16 bp. Lane 4: the C3435T mutation destroys 1 restriction site and MboI digestion yields a 232- and 16-bp fragments (m/m). Primer Pairs and PCR Conditions Used in Genotyping of MDR1 (ABCB1) and KIRs (KIR2DL2 and KIR2DS2). Abbreviations: KIR, killer cell immunoglobulin-like receptor; MDR, multidrug resistance; PCR, polymerase chain reaction. Genotyping of the MDR1 by PCR-RFLP Assay. Abbreviations: MDR, multidrug resistance; PCR-RFLP, polymerase chain reaction-restriction fragment length polymorphism; w/w, homozygote wild type; m/w, heterozygote mutant/wild type; m/m, homozygote mutant type; SNP, single-nucleotide polymorphism.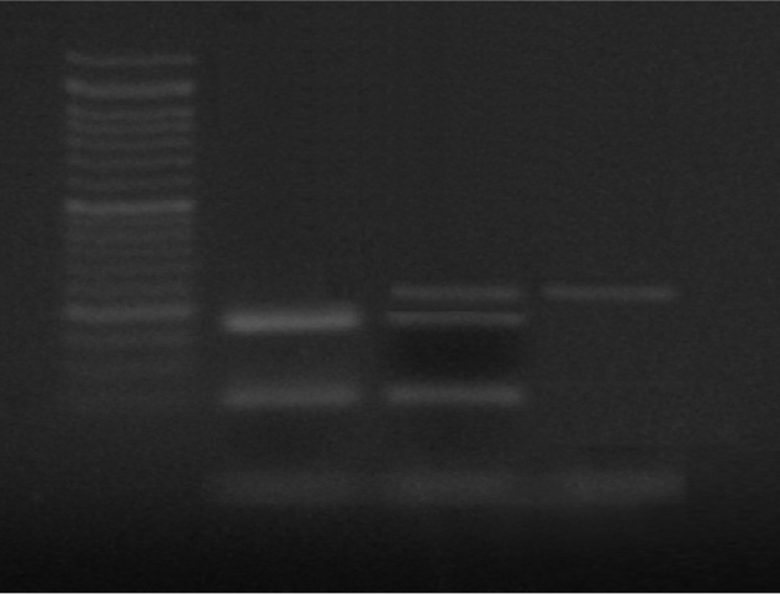

B. Genotyping of the human KIRs (Figure 2; KIR inhibitory gene KIR2DL2 and the noninhibitory KIR2DS2). Genotypes were identified by PCR according to Nourse et al
19
and Uhrberg et al.
24
In vitro amplification was performed on gradient thermal cycler (HYBAID Express; HYBAID Limited, Ashford, Middlesex, United Kingdom; Table 1). Analysis of the amplified products was done on agarose gel electrophoresis 2.5% stained with ethidium bromide in 1× Tris-EDTA-borate buffer to detect the corresponding amplified fragments (1800 bp for KIR2DL2 gene, 1750 bp for KIR2DS2 gene, and 82 bp confirming successful PCR for the internal control) under ultraviolet illumination. Amplified fragments of the KIR2DS2 gene by 2% agarose gel electrophoresis. Lane 1: 100 bp ladder (100-2000 bp). Lane 2: upper band: KIR2DS2 +ve at 1750 bp. Lower band: internal control band at 82 bp.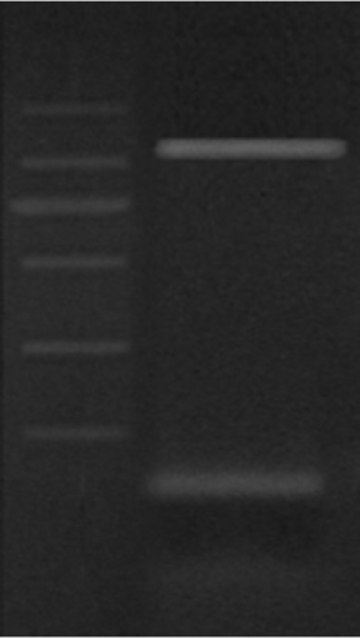
C. The PCR products and digested fragments were all documented by Gel Documentation System and Software for DNA analysis (InGenius Syngene, Cambridge, United Kingdom) using GelPilot Mid Range Ladder 100 bp (cat no. 239125; QIAGEN GmbH, Hilden-Geschäftsführer, Germany) and molecular weight GeneRuler 50 bp DNA ladder (#SM0373; Thermo Scientific/Fermentas) for confirmation of proper PCR product length.
D. The distribution of polymorphisms, genotypes, and allele frequencies was all statistically compared in all patients versus healthy controls.
Statistical Methodology
The data were coded, entered, and processed on computer using Statistical Program for IBM SPSS statistics (V. 21.0, IBM Corp, 2012). Data were expressed as median and percentiles for quantitative nonparametric measures in addition to both number and percentage for categorized data. Quantitative variables were described in the form of mean ± standard deviation, and qualitative variables were described as number and percent. Chi-square (χ2) test was used to compare between groups, regarding nonnumerical variables. Wilcoxon rank sum test was used to compare 2 independent groups for nonparametric data. Kruskal-Wallis test was used to compare more than 2 patient groups for nonparametric data. P Value of less than .05 was considered the cutoff value for significance.
Results
Of a total of 65 children with ITP were recruited, 9 declined to participate in this study and sign the informed consent. Further, we excluded 8 of the 65 patients for the following reasons: 3 children proved to have Evans syndrome and 5 had SLE.
A total of 48 (24 males and 24 females) children with ITP between the age of 2 and 13 years (7.75 ± 2.9) were included in the study. This is along with 35 healthy children and adolescent (16 [45%] males and 19 [55%] females) who served as a control group with a mean age of 7.6 ± 4.3 years range (3-13 years) with matching of cases and control as regards age and sex (t = 0.190, P value = .849 for age and χ2 = 0.027, P value = .870 for sex).
Clinical Characteristics of Studied Population
Patients with ITP were divided into 2 groups:
Group 1 (a prospective study arm): –21 patients with newly diagnosed ITP. This group comprised of 12 (57%) males and 9 (43%) females, their ages ranged from 2 to 8 years with a mean of 5.8 ± 2.6 years.
Group 2 (a retrospective study arm): –27 patients with chronic ITP.
This group comprised of 12 (45%) males and 15 (55%) females, their ages ranged from 5 to 13 years with a mean of 9.7 ± 3.3 years, their disease duration ranged between 2 and 7 years with a mean of 4.1 ± 2.9 years. The control were matched with newly diagnosed patients with ITP regarding age and sex (t = 1.734, P value = .088 for age and χ2 = 0.081, P value = .776 for sex). This was also applicable for chronic patients with ITP and control (t = 1.922, P value = .056 for age and χ2 = 0.025, P value = .875 for sex respectively).
Distribution of MDR1 Genotype in the Studied Population
The frequency of the genotype MDR1 gene in all patients with ITP and control was not significant (P = .090). The distribution of CC, CT, and TT genotypes in all patients were 8.3%, 62.5%, and 27.1%, respectively, and in control was 28.60%, 48.60%, and 22.9%, respectively. This makes CT genotype the highest distribution among all patients with ITP and among control.
The allele frequencies of T and C were 32.41% and 67.59% in patients compared to 52.86% and 47.14% in control, respectively (P = .1; Figure 3). When dividing the patients into newly diagnosed and chronic ITP, the distribution of MDR1 genotype CC, CT, and TT in newly diagnosed patients with ITP was 9.5%, 61.9%, and 28.6%, respectively, and in chronic patients was 11.1%, 63.0%, 25.9%, respectively, and was not significant (P = .832). The allele frequencies of T and C were 62.5% and 70.59% in newly diagnosed patients compared to 37.5% and 29.41% in chronic patients, respectively (P = .51).
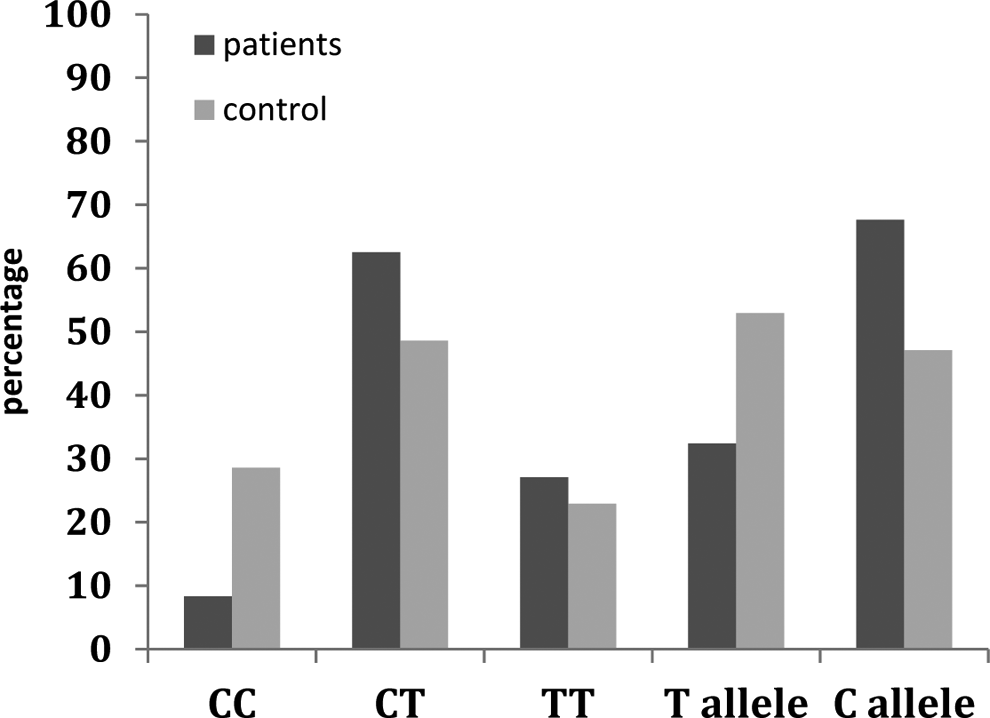
Genotype and allele frequencies of the multidrug resistance (MDR) 1 gene exon 26/C3435T polymorphism in the enrolled patients.
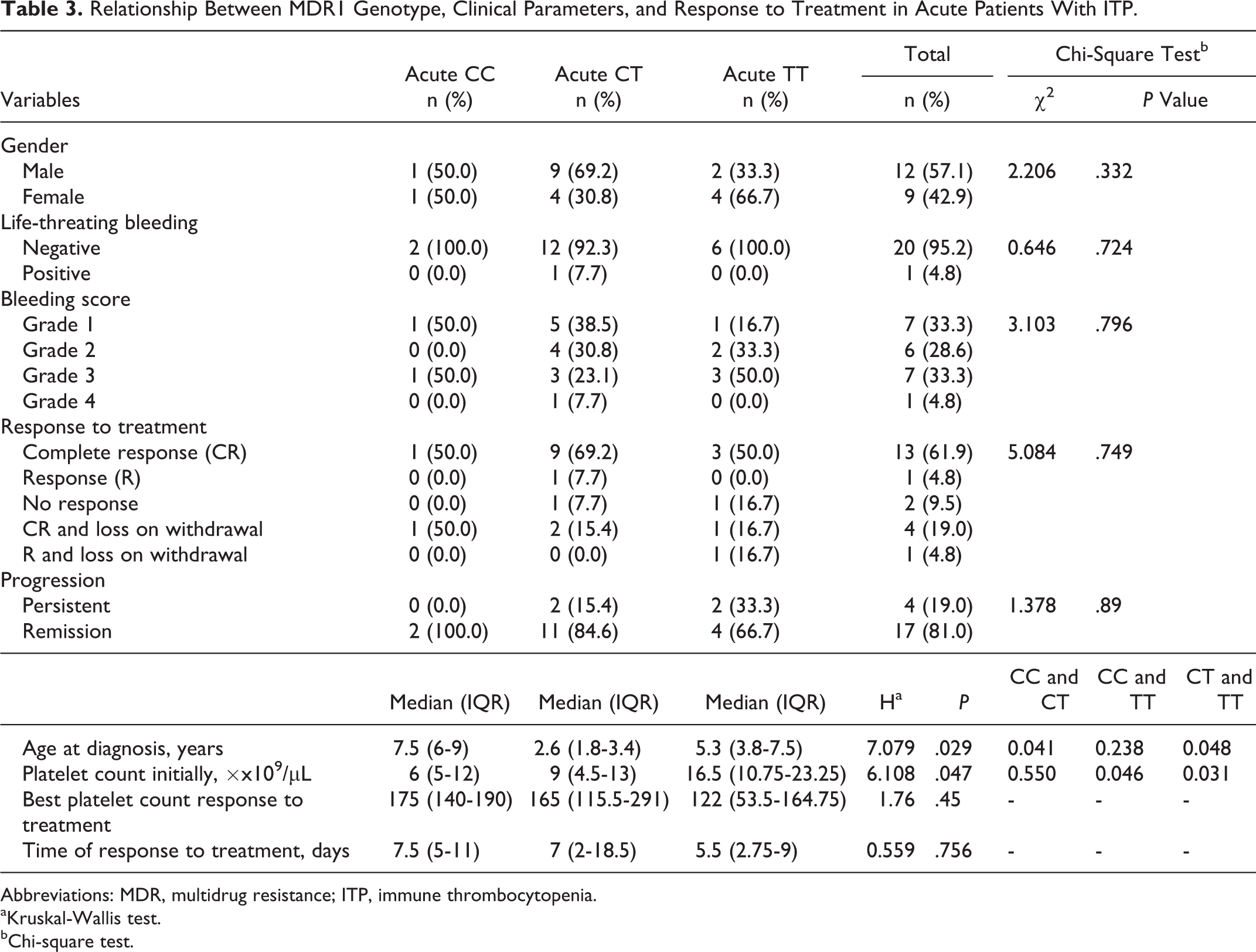
Relationship Between MDR1 Genotype, Clinical Parameters, and Response to Treatment
There were no significant differences in gender, bleeding score, life-threatening bleeding, response to different lines of treatment, time of response to treatment, and posttreatment platelet counts between CC, CT, and TT genotypes of the MDR1 gene in newly diagnosed patients with ITP (P = .33, P = .79, P = .72, P = .74, P = .75, and P = .45, respectively). Further, there was no significant difference (P = .89) between CC, CT, and TT genotypes of the MDR1 gene as regards the progression of the disease whether had remission or had persistent course of the disease (Table 3).
Relationship Between MDR1 Genotype, Clinical Parameters, and Response to Treatment in Acute Patients With ITP.
Abbreviations: MDR, multidrug resistance; ITP, immune thrombocytopenia.
aKruskal-Wallis test.
bChi-square test.
There were a significant difference in age at diagnosis between CC, CT, and TT genotypes of MDR1 gene (P = .029), with the youngest age at 2.6 years at CT genotype and eldest at CC genotype with a mean of 7.5 years. Also there was a significant difference in platelets count at initial diagnosis between CC, CT, and TT genotypes of MDR1 gene (P = .047) with the highest initial platelet count at TT genotype with a median of 16.5 × 109/L (IQR 10.75-23) and lowest at CC genotype with a median of 6 × 109/L (IQR 5-12).
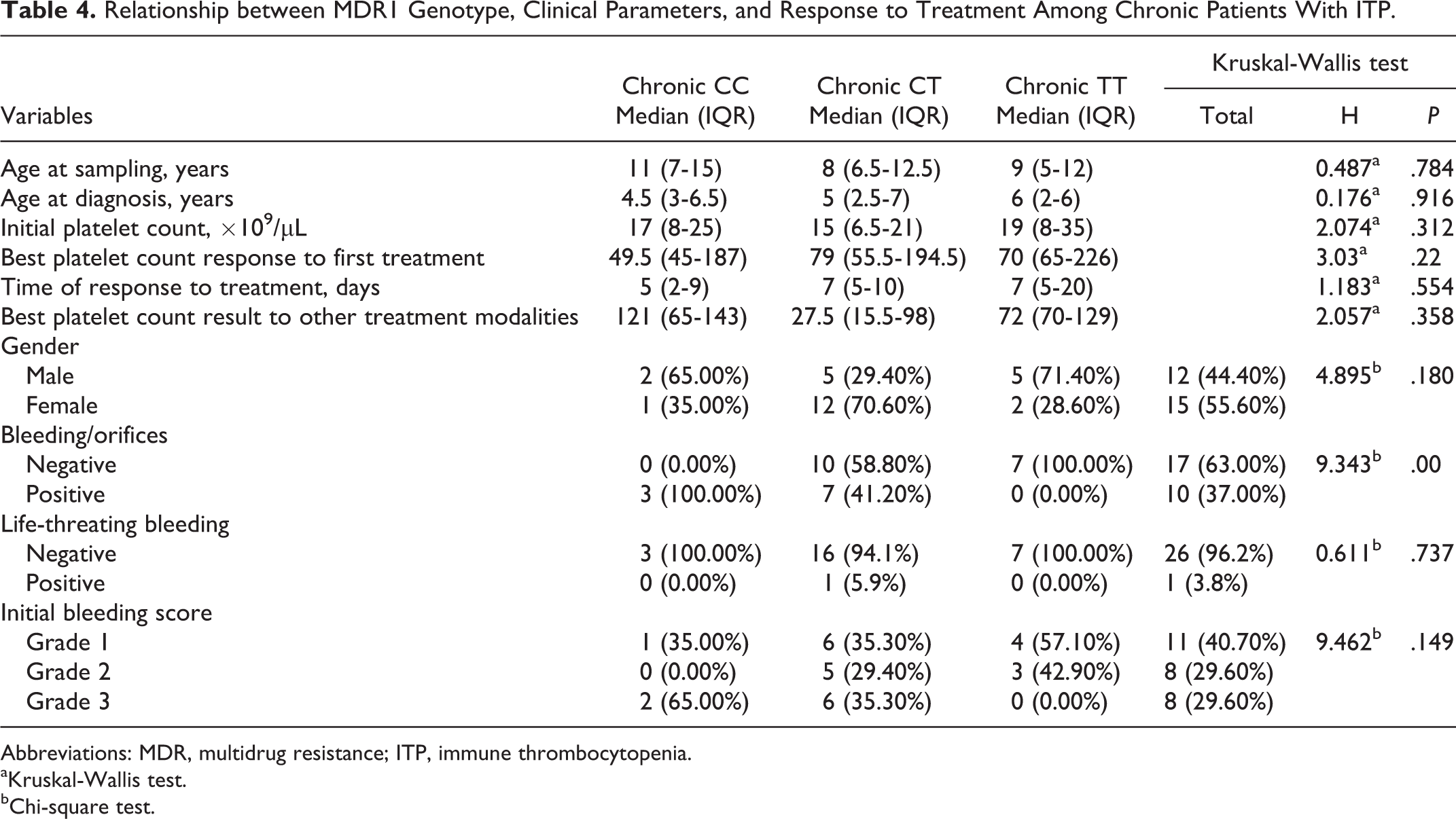
There were no significant differences in age at sampling, age at diagnosis, gender, life-threatening bleeding, initial platelets count, bleeding score, best first platelets response, time of response, and other different treatment modalities in platelets response between CC, CT, and TT genotypes of MDR1 gene in chronic cases of ITP (P = .78, P = .91, P = .18, P = .73, P = .31, P = .14, P = .22, P = .55, and P = .35, respectively; Table 4). However, there was a significant difference in bleeding/orifices as a clinical manifestation of chronic patients with ITP between CC, CT, and TT genotypes of MDR1 gene (P = .00). Patients who did not exhibited any bleeding/orifices represented (66.7%) with highest prevalence among TT genotype (100.0%) then CT (58.8%) and finally CC genotypes (0%).
Relationship between MDR1 Genotype, Clinical Parameters, and Response to Treatment Among Chronic Patients With ITP.
Abbreviations: MDR, multidrug resistance; ITP, immune thrombocytopenia.
aKruskal-Wallis test.
bChi-square test.
The Distribution of KIR2 Genotype in the Studied Population
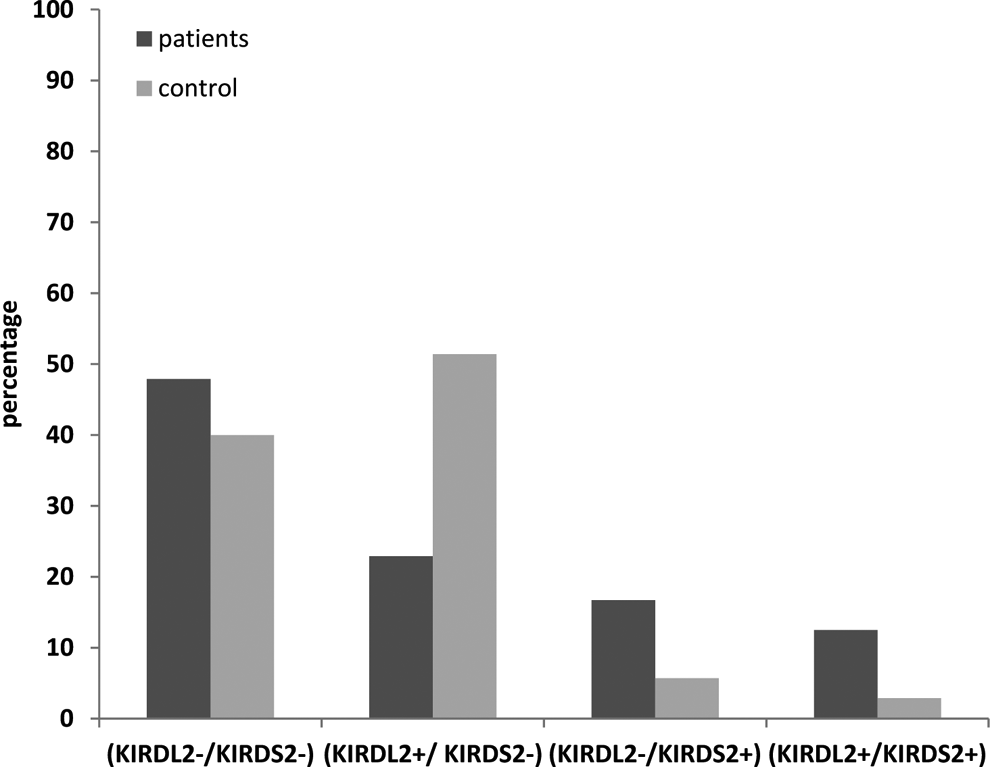
The distribution of KIR2 gene among all patients with ITP and controls was significant (P = .026). The (KIRDL2−/KIRDS2−) genotypes was the most prevalent among patients representing 47.90% vs 40% followed by KIRDL2+/KIRDS2− representing 22.9% vs 51.40% then KIRDL2−/KIRDS2+ representing 16.70% vs 5.70%, and finally KIRDL2+/KIRDS2+ representing 12.5% vs 2.90%; Figure 4. Moreover, the frequency of KIR2 genotypes between newly diagnosed and chronic patients with ITP was not significant (P = .93).
Genotype and allele frequencies of the killer cell immunoglobulin-like receptor (KIR) 2 gene polymorphisms in the enrolled patients.
The KIRDL2−/KIRDS2−genotype was the most prevalent among newly diagnosed patients (42.90%), followed by KIRDL2+/KIRDS2−genotype (23.80%), then KIRDL2−/KIRDS2+genotype (19.0%), and finally KIRDL2+/KIRDS2+genotype (14.3%). The KIRDL2−/KIRDS2−genotype was the most prevalent among chronic patients with ITP (51.90%), followed by KIRDL2+/KIRDS2−genotype (22.2%), then KIRDL2−/KIRDS2+genotype (14.80%), and finally KIRDL2+/KIRDS2+genotype (11.1%).
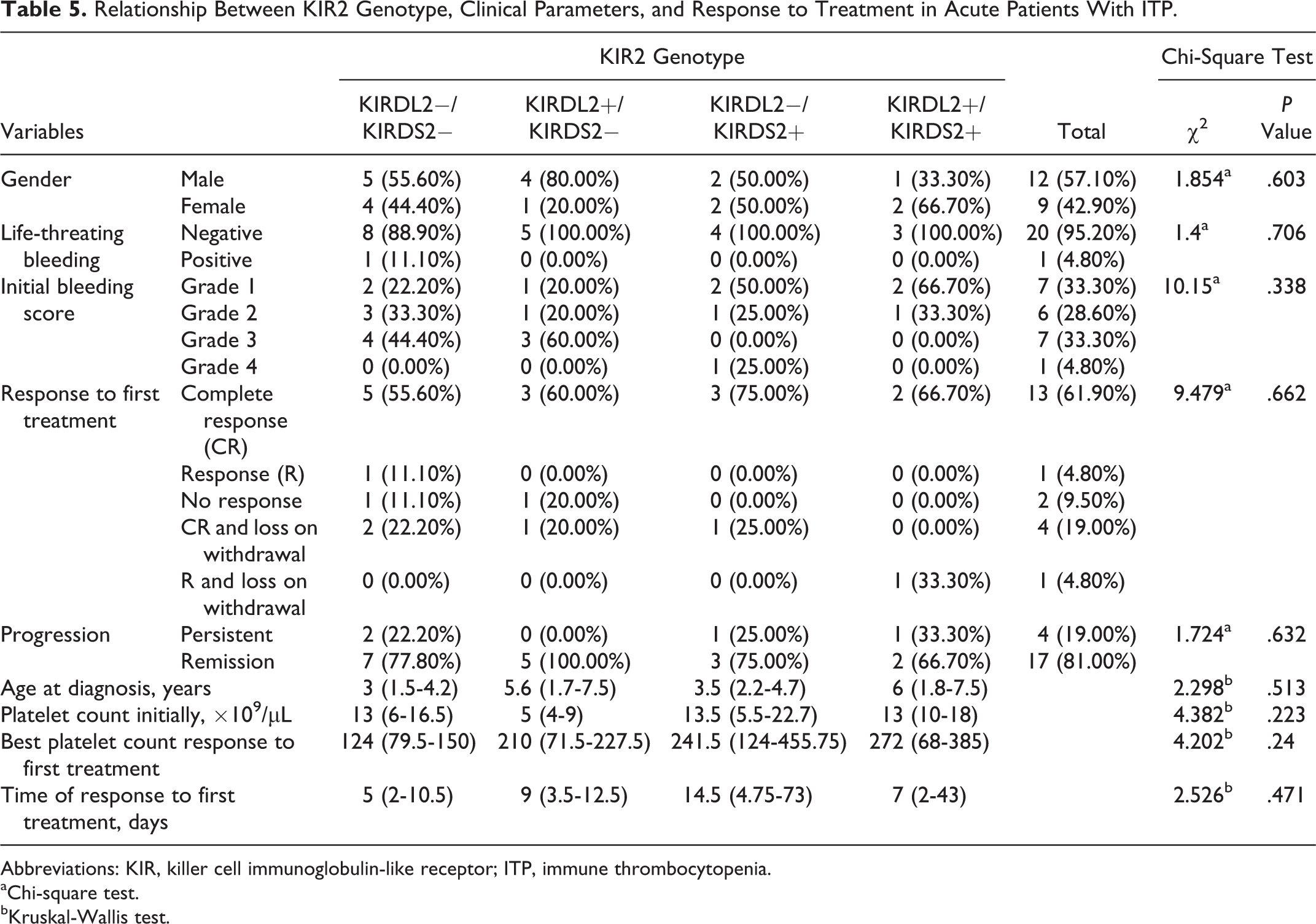
Relationship Between KIR2 Genotype, Clinical Parameters, and Response to Treatment
There were no significant difference in gender, age, bleeding score, life-threatening bleeding, initial platelets count at diagnosis, response to lines of treatment, time of response to treatment, and posttreatment platelet counts between different genotypes of the KIR2 gene KIRDL2−/KIRDS2−, KIRDL2+/KIRDS2−, KIRDL2−/KIRDS2+, and KIRDL2+/KIRDS2+ in newly diagnosed patients with ITP (P = .60, P = .51, P = .33, P = .70, P = .22, P = .66, P = .47, and P = .24, respectively). Further, there was no significant difference (P = .63) between different genotypes of the KIR2 gene as regards the progression of the disease whether had remission or had persistent course of the disease (Table 5).
Relationship Between KIR2 Genotype, Clinical Parameters, and Response to Treatment in Acute Patients With ITP.
Abbreviations: KIR, killer cell immunoglobulin-like receptor; ITP, immune thrombocytopenia.
aChi-square test.
bKruskal-Wallis test.
Also, there were no significant difference in gender, age at sampling, age at diagnosis, bleeding score, bleeding/orifices, life-threatening bleeding, different response to lines of treatment, best first platelets count response, time of response to treatment, and initial platelet count between KIRDL2−/KIRDS2−, KIRDL2+/KIRDS2−, KIRDL2−/KIRDS2+, and KIRDL2+/KIRDS2+ genotypes of the KIR2 gene in chronic patients with ITP (P = .26, P = .36, P = .25, P = .81, P = .73, P = .81, P = .88, P = .70, P = .87, and P = .21, respectively; Table 6).
Relationship Between KIR2 Genotype, Clinical Parameters, and Response to Treatment Among Chronic Patients With ITP.
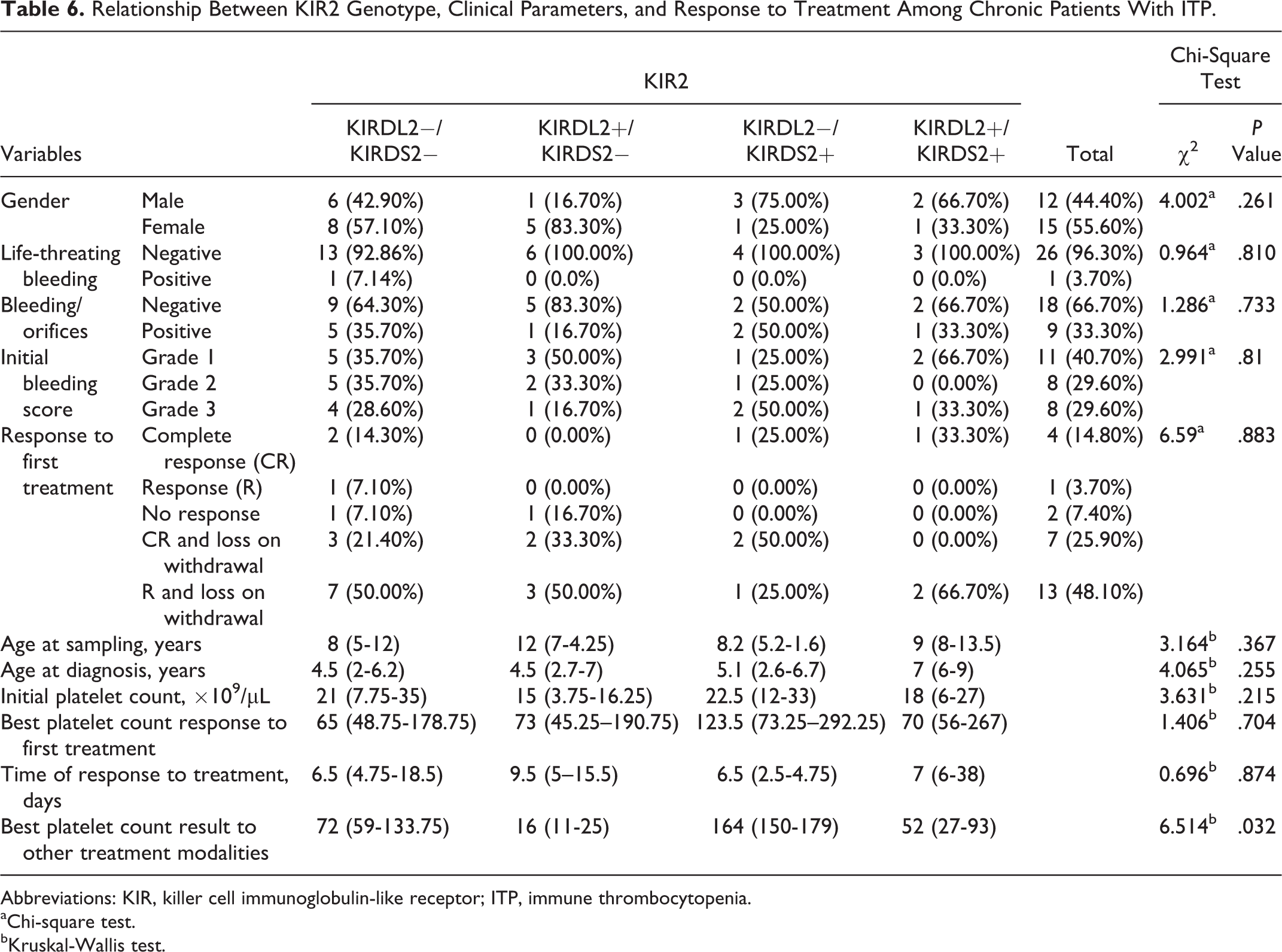
Abbreviations: KIR, killer cell immunoglobulin-like receptor; ITP, immune thrombocytopenia.
aChi-square test.
bKruskal-Wallis test.
There was a significant difference in other different treatment modalities in platelets response between KIR2 genotypes in chronic patients with ITP (P = .018). Forty percent of chronic patients were not responsive to lines of treatment (either to full dose steroids, anti-D therapy, or thrombopoietin receptor agonists) with highest prevalence among KIRDL2+/KIRDS2−genotype representing 100%, then among KIRDL2−/KIRDS2−genotype representing 12.5%. Thirty-three percent of chronic patients with ITP were responsive to treatment with equal percentages between KIRDL2−/KIRDS2− and KIRDL2−/KIRDS2+genotypes representing 50%. There was a high significant difference between best platelets response to treatment and KIR2 genotypes (P = .03) as the highest platelet count was found after treatment among KIRDL2−/KIRDS2+genotypes.
Discussion
Primary ITP is one of the most common bleeding disorders of childhood. In most cases, it presents with sudden widespread bruising and petechiae in an otherwise well child. Thought to be mainly a disorder of antibody-mediated platelet destruction, ITP can be self-limited or develop into a chronic condition. 25
Establishing associations between candidate genes and ITP susceptibility may provide insight into pathogenesis and support the development of therapeutic strategies. 1 However, it is imperative for studies of genetic susceptibility to assign patients definitively to specific ITP phenotypes, as evidence indicates ITP is a biologically heterogeneous condition. 26 In particular, the pathophysiology of primary versus secondary ITP is likely to be different. However, even within primary ITP, there may be age-related biological differences. Pediatric ITP is frequently preceded by viral infection and commonly resolves without treatment. By contrast, a high proportion of newly diagnosed adult cases with ITP will go on to have chronic disease. Furthermore, the variable clinical course of adult ITP raises the possibility that there are differences in pathogenesis within the cases of adult ITP. 27
To the best of our knowledge, this is the first study to assess both MDR1 and KIR2 genotype polymorphisms in childhood patients with ITP compared with healthy control participants, as only a well-defined newly diagnosed/chronic Egyptian patients with ITP were included. The MDR1 gene encodes a membrane P-gp that functions as an adenosine triphosphate (ATP)-dependent exporter of xenobiotics from cells. 28 P-gp is expressed in normal tissue, such as the intestine, liver, and kidneys, which have excretory functions, as well as in capillary endothelial cells of the brain, placenta, and testis and in peripheral blood cells. 28 In the kidney, the P-gp is expressed in the brush border membrane of proximal tubular epithelial cells. 28,29 Various types of structurally unrelated drugs, including steroids, are known to be substrates for P-gp. 30,31 It was previously reported that approximately 50 single-nucleotide polymorphisms (SNPs) have been found in the MDR1 gene. 32,33 Among these SNPs, C1236T, G2677T/A, and C3435T are the most common variants in the coding region of MDR1, and they are in strong linkage disequilibrium. 34
We assessed MDR1 genotypes and allele distribution of childhood patients with ITP that was not statistically different between patients and control as well as in newly diagnosed and chronic patients with ITP. The CT genotype showed the highest distribution among newly diagnosed and chronic patients with ITP with 65% and 51.53%, respectively. In a previous study, 1 it was found that the genotype and allele distribution of childhood newly diagnosed patients with ITP was no different from the healthy controls.
In the present study, there were a significant difference in the age at diagnosis between CC, CT, and TT genotypes of MDR1 gene among newly diagnosed patients with ITP having the youngest age at 2.6 years at CT genotype and eldest at CC genotype with a mean of 7.5 years. Also there was a significant difference in platelets count at initial diagnosis between different genotypes of MDR1 gene in newly diagnosed patients with ITP (P = .047) with the highest initial platelet count at TT genotype and lowest at CC genotype.
In contrast to our study, Akin et al 1 found that there was no significant difference in age as well as in the pre- and posttreatment platelet counts between CC, CT, and TT genotypes of the MDR genotypes. Moreover, the response to treatment did not differ between different MDR1 genotypes in our study. In a previous Turkish study, there was no difference in the high-dose methyl prednisone treatment response between MDR1 gene genotypes among cases with ITP. 1 However, Drozdzik 35 reported that in rheumatoid arthritis, patients with CC genotypes had a weaker methyl prednisone response than did patients with the TT genotype. Also previous studies showed high-intestinal expression of P-gp in patients with the CC genotype and decreased oral drug use effectiveness. 10 Moreover, López-Karpovitch et al 11 found that no significant differences in MDR1 values before and after the prednisone treatment correlate with the notion that glucocorticoids do not significantly modify the function and expression of P-gp in adults with ITP.
Resistance to glucocorticoids remains a tricky problem complicating the therapy of ITP. Recently, ATP binding cassette gene B1 gene ABCB1 (also called MDR1) was reported to be correlated with susceptibility and therapeutic efficacy of autoimmune diseases through P-gp. 36 Multidrug resistance 1 is characterized by drug extrusion through P-gp, reducing the intracellular levels of drugs and diminishing their pharmacological effects. 37 It has been reported that an increased number of lymphocytes are able to extrude P-gp substrates in rheumatoid arthritis, ITP,, and SLE, the patients with poor response to treatment being the ones that exhibit the highest values. 38
Studies of MDR1 gene polymorphism in nephrotic syndrome demonstrated that the homozygous mutant genotype of (SNP) C3435T was not associated with the steroid-resistant phenotype 39 and another one 40 suggested that the MDR1 C1236T polymorphism was also associated with steroid resistance among pediatric patients with idiopathic nephrotic syndrome (INS). In addition, Chiou et al 41 found that the presence of the MDR1 1236 CC genotype or the C allele predicted a better initial response to steroids in INS. The discrepancies of association results may be due to changes in expression of P-gp and function in SNPs of the MDR1 gene among different ethnicities and patients. 23
In addition, the methylation status of certain CpG sites on the MDR1 promoter and/or other epigenetic mechanisms plays a critical role in switch-on or -off of MDR1 gene expression. 42 Although SNP is a synonymous variation, it is associated with altered protein expression. 31 The MDR1 C3435T SNP that affects the expression of P-gp may be through possible linkage disequilibrium with other MDR1 variants that control expression, including the nonsynonymous G2677T/A and synonymous C1236T SNPs. 33 In addition, the MDR1 C3435T SNP leads functional alteration of P-gp by affecting the timing of cotranslational folding and insertion of P-gp into the membrane, thereby altering the structure of substrate and inhibitor interaction sites. 43
The distribution of KIR2 gene among all patients with ITP and control was significant. The KIRDL2−/KIRDS2− genotypes were the most prevalent among all patients with ITP.
Accumulating evidence indicates that KIR2DS2 contributes to the pathogenesis of autoimmune diseases such as type 1 diabetes and psoriasis. 44 A pathogenetic model for the association of KIR2 genotype with ITP is unclear. The KIR proteins are expressed in both NK and effector T cells. The activation of these cells is controlled by a balance between a genetically determined repertoire of activating and inhibitory KIRs. 45 In both cell types, KIRs impact direct cell-mediated cytotoxicity and, in NK cells, they may also influence antibody-dependent cell-mediated toxicity (ADCC). 19
In a previous study, autologous T cells from patients with active chronic ITP were shown to mediate platelet destruction. 46 By contrast, autologous NK cells were incapable of inducing direct platelet cytolysis. Moreover, the inhibitory KIR2DL3, KIR3DL2, and KIR3DL1 were upregulated in the T cells of patients with ITP in remission relative to those with active disease. 46 Along these lines, it may be suggested that KIR2DL2/KIR2DS2 genotype influences T-cell-mediated platelet cytotoxicity.
Moreover, in comparison with healthy controls and a tightly defined cohort of adult patients with ITP, KIR2DS2/KIR2DL2 genotype and FCGR3a-158 polymorphisms were independently associated with ITP. 19 Previous studies concerning FCG3a polymorphisms in patients with ITP showed conflicting results between adult and childhood ITP. In American, Canadian, and Dutch pediatric ITP cohorts, the FCGR3a-V158 polymorphism was over represented. 47 However, studies examining this in the adult setting have shown no increase in the FCGR3a-V158 allele 47 and only a decrease in FCGR3a-F/F158 genotype frequency. 48
The induction of autoimmunity and the induction of a therapeutic antibody-mediated response are likely to have some similarities. Interestingly, the FCGR3a-V158 allele most likely influences response to rituximab monotherapy in a variety of clinical settings. 49 Rituximab has activity “upfront” as well as for chronic adult ITP. Therefore, the identification of new candidates that potentially influence the pharmacogenetics of rituximab might be of clinical value in this disease setting. 50 Alternatively, KIR2DL2/KIR2DS2 may influence indirect destruction of autoantibody bound platelets by NK cells. However, the role of NK-cell-mediated ADCC in ITP has yet to be determined.
There are some limitations of the present study that need to be addressed. First, these polymorphisms were investigated in a relatively small number of patients, so larger multicenter studies will be needed to confirm our findings. Furthermore, this study was conducted upon Egyptian patients. So it is necessary to evaluate the role of those 2 genes expression in different ethnic groups.
We couldn’t examine the long-term influence of these polymorphisms on functional outcome of ITP disease as the follow-up period of the prospective study arm is relatively short, thus limiting generalization of our results. Our data should therefore be considered preliminary, awaiting further confirmatory studies on an expanded patient base and longer follow-up duration. Further, there is no space for comparison as earlier studies of these genes are scarce and hard to find; other studies were on adult patients and different diseases as nephrotic syndrome and rheumatoid arthritis.
In conclusion, we found that the MDR1 C3435T polymorphisms did not play an important role as a genetic risk factor for the development and progression of childhood ITP. However, patients carrying MDR1 (CC) genotypes had a higher risk to develop childhood ITP at an older age and having lower initial platelet count than patients carrying other genotype. Also, we found that KIR2 gene polymorphisms were independently associated with childhood ITP in Egyptian patients with highest prevalence among KIRDL2−/KIRDS2−genotypes. Moreover, KIRDL2+/KIRDS2− genotype was not responsive to treatment in chronic patients with ITP. Further studies involving larger patient populations are required to establish the basis for these observations and their impact on immune-thrombocytopenia pathogenesis, progression, and therapies.
Footnotes
Acknowledgments
We would like to thank Dr Ghada Elnady, Medical Genetic Diseases Research Center for helping in the practical part of genetic analysis. Also we wish to express our sincere appreciation to all the patients and their families following in the Pediatric Hematology/Oncology Clinic, Children’s Hospital, Ain Shams University for their cooperation during the study period.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
