Abstract
Objective:
The susceptibility of tissue to ischemia–reperfusion (I/R) injury is a major obstacle to tissue regeneration and cellular survival. In this study, we investigated the possible renoprotective effect of dabigatran in an experimental renal I/R model.
Method:
A total of 25 rats were randomly divided into 5 equal groups. The control group was used to obtain basal values of oxidant and antioxidant biomarkers. The sham group was used to obtain renal prolidase and malondialdehyde (MDA) levels after renal ischemia (for 45 minutes) and reperfusion (for 1 hour). A standard diet was followed. Oral 15 mg/kg dabigatran etexilate was administrated to group I, intraperitoneal 250 U/kg enoxaparin sodium was administrated to group II, and intraperitoneal 250 U/kg bemiparin sodium was administrated to group III for 1 week before the renal I/R was performed. Renal tissue samples were obtained during the first hour of reperfusion to detect renal prolidase and MDA levels in these groups, after which the rats were euthanized.
Results:
Renal prolidase levels were significantly higher in the sham group compared with the control group (1834.2 ± 982.3 U/g protein vs 238.8 ± 43.6U/g protein; P = .001). Lower prolidase levels were observed in groups II (838.7 ± 123.8 U/g protein) and III (1012.9 ± 302.3 U/g protein), and the lowest prolidase levels occurred in group I (533.8 ± 96.2 U/g protein; P < .05) when compared with the sham group. The MDA levels were significantly lower (P < .05) in groups I, II, and III (163.9 ± 41.5, 185.4 ± 51.0, and 138.2 ± 22.6 μmol/g protein, respectively) compared with the sham group.
Conclusion:
Dabigatran etexilate, a univalent direct thrombin inhibitor, may protect the renal tissue more effectively when compared to low-molecular-weight heparins.
Introduction
Because thrombosis can result in partial or total occlusion in vessel lumina, which leads to end-organ ischemia, antithrombotic management is important for avoiding disrupted blood supply due to the thrombosis. 1,2 However, thromboses can be developed under antithrombotic treatment 3 ; therefore, anticoagulant treatment is as important for its organ protective effects as it is for thromboprophylaxis. 4
Heparin and its derivatives are frequently used for management of anticoagulation. The use of unfractionated heparin (UFH) is limited to parenteral administration only, and affectivity control has to be monitored; therefore, UFH treatment requires hospitalization. The UFH has various hematological and systemic side effects, such as heparin-induced thrombocytopenia, bleeding, and osteoporosis. 5 Low-molecular-weight heparins (LMWHs) are fragments of commercial-grade heparin produced by either chemical or enzymatic depolymerization. The LMWHs are potentially more advantageous than heparin due to their reduced risk of bleeding, greater bioavailability at low doses, and longer half-life .They can be used once or twice daily, and those treatments do not require hospitalization. 6 Dabigatran etexilate, a novel oral anticoagulant drug and the prodrug of dabigatran, does require routine coagulation monitoring or dose adjustment. 7 Due to its specific blockage of the coagulation cascade via the thrombin molecule and due to its molecular structure, which differs from that of UFH, dabigatran etexilate has fewer hematological and systemic side effects than UFH does. 8 The protective effects of heparin derivatives and those of enoxaparin sodium, bemiparin sodium, and rivaroxaban, which are classified as factor Xa inhibitors, have been compared in previous reports. 4 Caliskan et al conducted a comparison between rivaroxaban and other factor Xa inhibitors but not between factor Xa inhibitors and thrombin inhibitors. 4 Thus, the aim of this study was to compare the antioxidant effects of dabigatran etexilate with those of LMWHs in an experimental renal ischemia/reperfusion (I/R) model. Enoxaparin sodium and bemiparin sodium which are produced by benzylation followed by alkaline depolymerization were the LMWHs selected for comparison, in accordance with previous reports that have shown the beneficial systemic effects against I/R injury of these agents. 4
Materials and Methods
Ethical approval was obtained from the Animal Research Committee of the University, and all procedures were performed according to Animal Welfare Act and the Guide for the Care and Use of Laboratory Animals prepared by the ethics committee.
Animals
Twenty-five male Sprague-Dawley rats, 8 to 12 weeks of age and weighing 230 ± 30 g (mean ± standard deviation, SD) were purchased from the Laboratory Animal Production Unit of the University. All of the rats were maintained in standard humidity (50% ±5%) and temperature (22 ± 2°C) controlled cages with a 12-hour light–dark cycle for 1 week prior to the start of the study.
Anesthesia Protocol and Acute Renal I/R Creation
The rats were anesthetized with 130 mg/kg ketamine (Ketalar; Pfizer, Täby, Sweden) and 20 mg/kg xylazine (Rompun; Bayer, Gothenburg, Sweden) via an intraperitoneal line. A middle abdominal incision was made and both renal arteries were isolated as described by Takeda et al 9 and clamped for 45 minutes. Reperfusion was created by declamping the arteries, and blood flow restoration was confirmed. One hour after reperfusion, the rats were euthanized and both kidneys were excised. Maintenance of anesthesia was provided with ketamine hydrochloride (50 mg/kg).
Group Creation and Drug Utilization
Twenty-five rats were randomly divided into 5 equal groups, as follows:
Control group (n = 5): created to obtain basal serum renal prolidase and malondialdehyde (MDA) levels without any intervention. The other rats were separated for I/R groups.
Sham group (n = 5): I/R only, standard diet.
Group I (n = 5): Dabigatran etexilate (Pradaxa; Boehringer Ingelheim Pharma GmbH & Co KG, Biberach, Germany) was administered orally via gavage at a dose of 15 mg/kg/d beginning 1 week prior to the start of study, as described by Blum et al.
10
I/R was induced after 1 week of drug administration.
Group II (n = 5): Enoxaparin sodium (Oksapar 2000 ANTI-XA IU/0.2 mL; Koçak Farma, Tekirdağ, Turkey) was administered intraperitoneally at a dose of 250 U/kg/d beginning 1 week prior to the start of the study.
Group III (n = 5): Bemiparin sodium (Hibor 3500 IU/0.2 mL; Laboratorios Farmaceuticos Rovi SA, Madrid, Spain) was administered intraperitoneally at a dose of 250 U/kg/d beginning 1 week prior to the start of the study.
Laboratory Analysis
Before the animals were euthanized, both kidneys from all groups were dissected at 4°C and quickly frozen in liquid nitrogen. Renal tissues were homogenized in 9 mL of 0.25 mol/L sucrose and 10 mmol/L EDTA (pH 7.4) using a Teflon homogenizer (Potter-Elvehjem glass-teflon tissue grinder, Thomas Scientific, Philadelphia, PA) to obtain a 10% suspension. They were then centrifuged at 3800g for 30 minutes to obtain tissue extracts, which were stored at –70°C until further analysis.
Measurement of MDA and Prolidase in Tissue Extracts
Tissue lipid peroxidation levels were measured by determining the MDA levels by the quantity of thiobarbituric acid-reactive products. Advanced glycation end product and MDA levels were determined following a previously reported procedure. 11 Tissue MDA levels were expressed as μmol/L/g protein. Tissue prolidase levels were evaluated using a method described by Myara et al that is based on measuring proline, a prolidase product. 12 Proline levels were expressed as U/g protein and detected at an absorbance at 515 nm.
Statistical Analysis
SPSS software version 15.0 (SPSS Inc., Chicago, IL) was used for statistical analysis. Results are expressed as mean ± SD. One-way analysis of variance was used to compare continuous variables, and Tukey honestly significant difference test was used as a post hoc test. A P < .05 was considered statistically significant.
Results
The basal renal prolidase and MDA levels were 238.8 ± 43.6 U/g protein and 33.11 ± 6.48 μmol/L/g protein, respectively, in the control group (determined as normal ranges). The renal prolidase levels increased to 1834.2 ± 982.3 U/g protein, and the renal MDA levels increased to 448.4 ± 111.3 μM/g protein after renal I/R was performed on the sham group.
In group I (dabigatran group), renal prolidase and renal MDA levels were 533.8 ± 96.2 U/g protein and 163.9 ± 41.5 μmol/L/g protein, respectively. In group II (enoxaparin group), renal prolidase and renal MDA levels were 838.7 ± 123.8 U/g protein and 185.4 ± 51.0 μmol/L/g protein, respectively. Lower renal prolidase (1012.9 ± 302.3 U/g protein) and renal MDA activities (138.2 ± 22.6 μM/g) were found in group III (bemiparin group). The mean ± SD values of prolidase and MDA of each group are summarized in Figure 1.
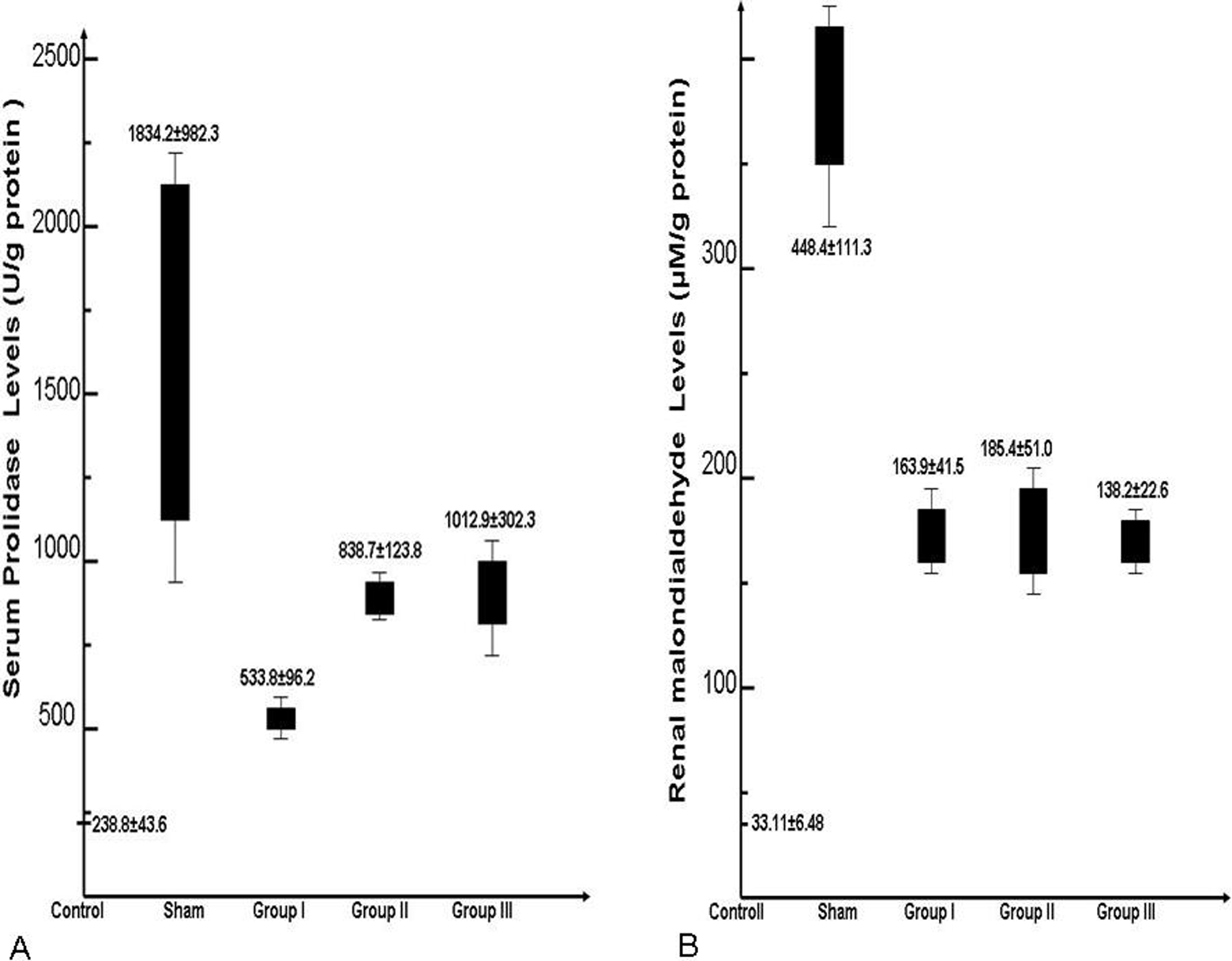
The mean ± standard deviation values of prolidase and malondialdehyde (MDA) in each groups.
There was a statistically significant increase in renal prolidase and MDA levels in the sham group compared with the basal levels of the control group (P = .001). Renal prolidase levels were significantly lower in all 3 drug groups when compared to sham group (P < .05). Lowest prolidase levels were observed in group I (P = .001). Renal MDA levels were significantly lower in all drug groups as well (P < .05). However, the greatest renal prolidase decrease was found in group III when compared with the sham group (P = .008). The comparison of groups according to renal prolidase (Table 1) and MDA levels (Table 2) are summarized in the Tables.
Statistical Comparison of Renal Prolidase Levels (U/g Protein) Between Groups.
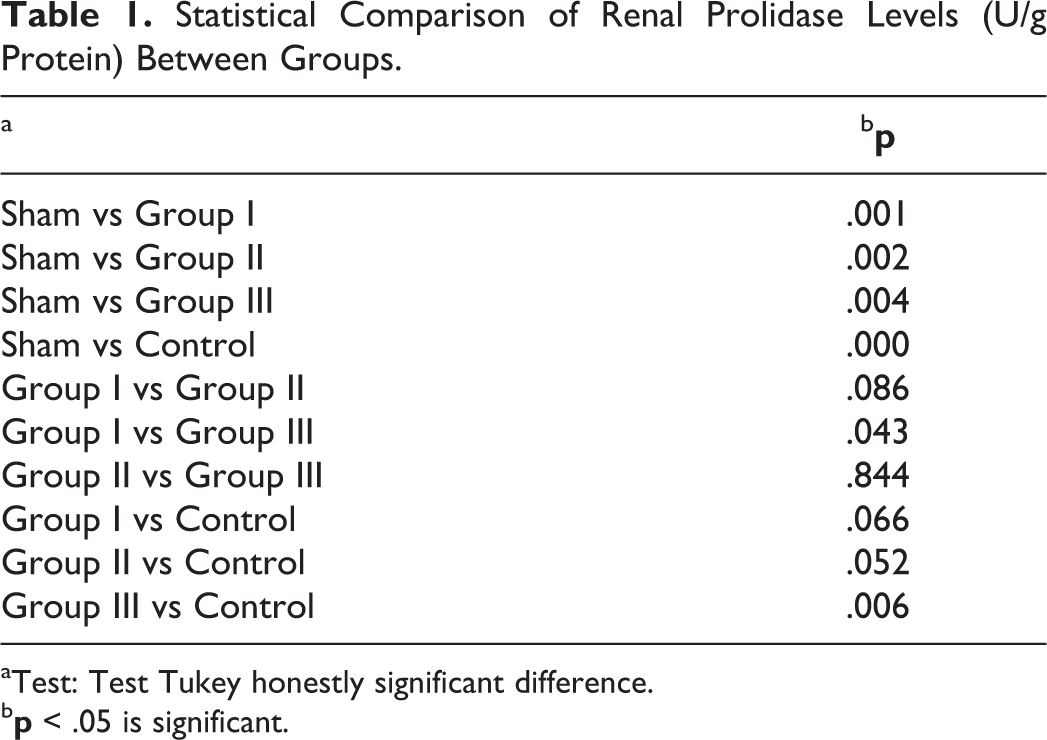
aTest: Test Tukey honestly significant difference.
b
Statistical Comparison of Renal Malondialdehyde (MDA) Levels (μmol/L/g protein) Between Groups.
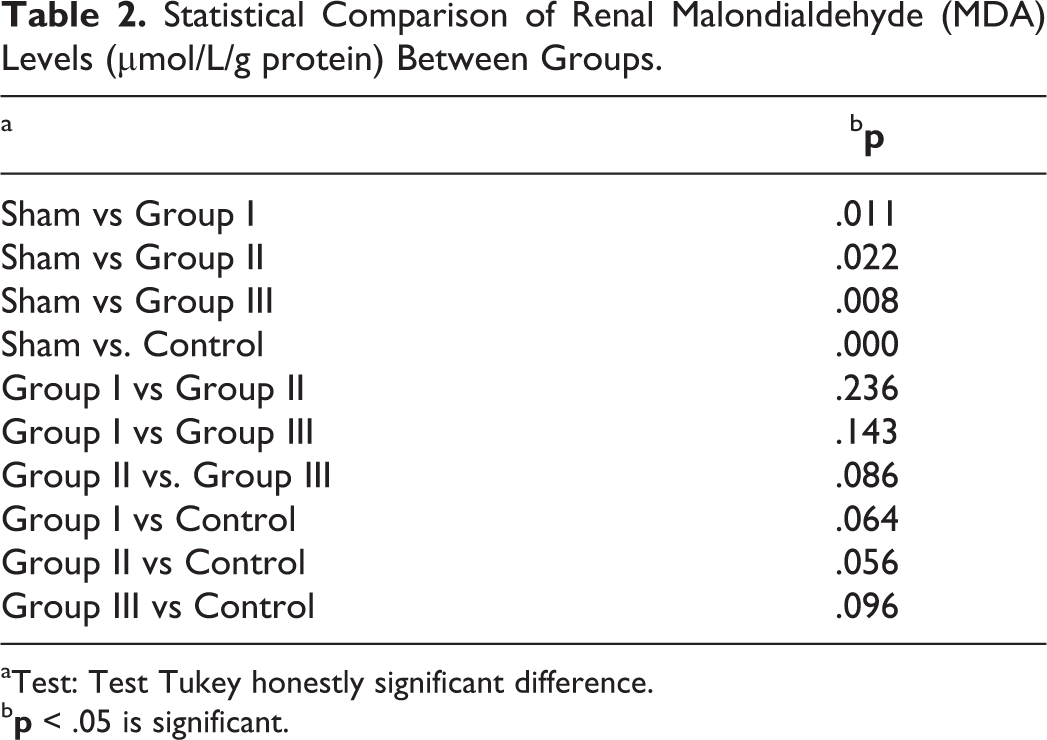
aTest: Test Tukey honestly significant difference.
b
Discussion
Our study revealed that renal prolidase and MDA levels increased after I/R injury in experimentally renal ischemia-induced rats. The thromboprophylaxis seemed to provide partial protection against I/R injury in renal tissues. In group 1 (dabigatran administered group), extremely lower renal prolidase levels were obtained compared with the sham group. The decrease was more prominent in group I when compared with the LMWH groups (group II and III). The bemiparin-administered group (group III) exhibited a greater decrease in MDA than the other drug groups (groups I and II) compared with the sham group. The decrease in renal prolidase and MDA levels was significant in all 3 drug groups. Briefly, the antioxidant effects of each of the 3 agents were similar. Nevertheless, bemiparin had a somewhat better response. However, better amelioration was observed in the dabigatran group in terms of prolidase levels.
Prolidase, a cytosolic peptidase that plays an important role in collagen metabolism, was previously reported in I/R studies evaluating tissue response. 4 In particular, prolidase has been shown to have a catabolism function, such as catalyzing the hydrolysis of dipeptides. 13 Elevated prolidase levels have been reported in experimentally induced ischemia studies. 14 The MDA is used for evaluating enzymatic and oxygen radical-induced lipid peroxidation as a biomarker of peroxidative damage in reperfused organs. 4 Therefore, we used those 2 parameters to evaluate the beneficial effects of dabigatran, enoxaparin, and bemiparin after I/R.
The beneficial effects of factor Xa inhibitors (LMWH) on end organs were described in previous studies. 4 Caliskan et al suggested that thromboprophylaxis with factor Xa inhibitors can improve the systemic response against I/R injury. 4 They also found a partial increase in systemic prolidase levels in the LMWH-treated rats after I/R injury. 4 In the current study, we found decreased prolidase levels in the dabigatran and LMWH-treated groups, which might indicate a means of reducing tissue catabolism. In particular, previous studies in the literature have claimed that renal tissues have the highest prolidase activity in the human body. 15 Previous studies also reported increased serum prolidase activities in degenerative valve disease and lung fibrosis due to the increased collagen turnover. 16,17 Significantly elevated MDA levels have been reported in previous I/R studies. 4,18,19 Studies investigating beneficial treatment strategies after I/R injury reported lower MDA levels for protection against oxidative stress injury. 19,20 The protective effects of LMWHs against I/R injury have also been reported. 4 The cellular regulatory effects of antithrombotic agents were previously reported as growth factor regulation and cell adhesion, in addition to preventing new thrombosis formation. 21,22 It has been claimed in other reports that the multifaceted renoprotective role of LMWHs might be dependent on the protection of cellular integrity by inhibiting inflammation (anti-inflammatory effect) and by antioxidant features (increased superoxide dismutase and catalase activity). 23 However, the cytoprotective effects of dabigatran remain unclear. According to our results, MDA levels were lower in the groups administered thromboprophylaxis with dabigatran, enoxaparin, and bemiparin compared with the sham group. We suggest that the decrease in tissue prolidase activity in the drug-administered groups might be associated with this decreased oxidative stress, which can alleviate prolidase activity or reduce collagen catabolism due to the decrease in renal tissue injury. These results indicate that routine prophylaxis with these factors Xa or thrombin inhibitors might be potentially beneficial against oxidative stress induced by I/R conditions and that they might prevent end-organ injury in patients with prothrombotic risk factors and prevent new thrombosis formation.
In summary, our results indicate that anticoagulants have renoprotective abilities against I/R. According to our findings, enoxaparin and bemiparin exhibited antioxidant properties while providing anticoagulation. The novel oral anticoagulant dabigatran showed similar effects with these conventional LMWHs. The renoprotective features of dabigatran appeared to be more effective than those of the LMWHs. Further studies are required to identify the renoprotection effectivity of dabigatran in I/R injury and oxidative stress conditions.
Limitations of Study
There are 2 major limitations that need to be acknowledged regarding the present study. The primary limitation concerns the experimental I/R model that was created for this study in animals. Therefore, these results reflect animal findings and they should be confirmed in humans. Second limitation has to do with sample size. The sample sizes are partially few for statistical power. However, the number of rats was determined by local ethical committee, and the study was designed as a pilot study for further investigations.
Footnotes
Authors’ Note
Suleyman Yazici contributed to study concept and design and was the main researcher. Oguz Karahan contributed to study concept and design, text writing, and study supervision. Mehmet Kerem Oral contributed to drafting of the manuscript. Zehra Bayramoğlu contributed to data analysis and recording. Mehmet Unal contributed to administrative, technical, or material support. Baris Caynak contributed to analysis and interpretation of the data. Ertan Sagbas was the moderator and coordinator of the study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
