Abstract
Therapeutic drug monitoring of enoxaparin with antifactor Xa levels (AXALs) is recommended in some populations; however, the approach to dose titration is poorly described. Our study at a large, tertiary teaching facility examined the dose response to titration of enoxaparin based on AXAL. Patients from 2008 to 2012 receiving enoxaparin were included, provided 2 or more steady state AXAL were obtained within 30 days and that the enoxaparin was prescribed for treatment rather than prophylaxis. The primary outcome was the percentage of dose change required to obtain goal range AXAL following dose titration. Eighty-seven patients were available for analysis with the following key characteristics: renal dysfunction during treatment 72%, obesity 8%, and solid organ transplant 26%. Initial goal AXAL was attained in 27 (31%) patients, and ultimately 54 (62%) patients achieved goal AXAL. Of the 31 patients who had initial AXAL above goal, 13 (42%) patients reached goal with a median dose decrease of 24%. In the 29 patients who had an initial AXAL below goal, 11 (38%) achieved therapeutic AXAL with a median dose increase of 16%. The AXAL monitoring can guide enoxaparin titration with subtherapeutic or supratherapeutic AXAL and an increase or decrease of roughly 20% is suggested as an initial change.
Background
Low-molecular-weight heparins (LMWHs), including enoxaparin, are the preferred anticoagulants in many settings. These include the treatment of venous thromboembolism (VTE) or acute coronary syndrome as well as stroke prevention in patients with atrial fibrillation and/or a mechanical heart valve undergoing cardioversion when stroke risk is high. 1 –4 Enoxaparin holds several advantages over unfractionated heparin (UFH) including more predictable pharmacokinetics, greater ease of administration, better safety margin, lack of requirement for therapeutic drug monitoring, and, in some settings, increased efficacy. 1 These properties have resulted in the recommendation by expert groups to use LMWHs over UFH. 1 –3
In some situations, however, therapeutic drug monitoring using antifactor Xa levels (AXALs) is warranted. Specifically, AXAL monitoring is recommended in patients for whom pharmacokinetics may be less predictable, such as those with renal insufficiency, pregnancy, or low or high body weight. 2 In these circumstances, monitoring of AXAL may lead to a greater percentage of patients who achieve therapeutic dosing, thus reducing the risk of bleeding and thrombotic complications. Previously published case series in pregnancy and solid organ transplantation have demonstrated that standard weight-based dosing regimens have often resulted in an above goal AXAL. 5,6 In addition, studies in patients with renal failure suggest that enoxaparin requires dose reduction (either once-daily dosing at 1 mg/kg or twice-daily dosing at less than 1 mg/kg/dose) to achieve goal AXAL. 7 –9 Finally, evidence in patients with morbid obesity suggests that even a conservative dosing strategy can lead to above goal AXAL. 10
Despite these contributions to the literature, there is currently a paucity of evidence to guide titration of LMWH based on AXAL, and additional research is needed to determine the most effective and safest dosing strategy for LMWHs in the aforementioned at-risk patients. One report in children (n = 12) using a LMWH not available in the United States has been published in abstract form only. 11 At least 2 other studies have examined a dose titration strategy based on AXAL in patients with varying degrees of renal failure. 9,12
Despite these contributions to the literature, additional research is needed to determine the most effective and safest dosing strategy for LMWHs in the aforementioned at-risk patients. The purpose of our study was to describe an inpatient patient population that underwent AXAL monitoring to determine the average dose change required to achieve goal values for AXAL following dose titration.
Methods
This was a retrospective cohort study of patients at a large tertiary referral center. Due to the retrospective nature of this study, the requirement for informed consent was waived by the institution. Patients treated between January 1, 2008, and April 31, 2012, with an order for enoxaparin in addition to at least 2 AXAL levels drawn within 30 days were included. Eligible patients were queried from the Medical Informatics Department and electronic charts were reviewed. Those patients with missing height or weight parameters and any use of enoxaparin for prevention of VTE rather than treatment were excluded. The effects of dose titration were analyzed among patients with 2 interpretable peak, steady-state AXAL, provided more than 3 sequential, appropriately timed doses of enoxaparin corresponding to the AXAL were administered. Since 2008, our institution has utilized an AXAL curve specific to enoxaparin.
Patients were divided based upon achievement of both initial and then subsequent AXAL goal values: achieved goal AXAL, AXAL higher than goal, and AXAL lower than goal. After dosage adjustment, the same categories were applied. The percentage of dose adjustment was compared among the groups. Additional factors with the potential to impact AXAL were examined including renal function, comorbidities, age, and dosing strategy.
Definitions utilized in this study include the following: steady state AXAL = at least 3 doses of enoxaparin provided, peak AXAL = 3 to 5 hours after charted dose, and goal AXAL = steady state value of 0.6 to 1 units/mL (every 12-hour dosing) or 1 to 2 units/mL (once-daily dosing). 1,4 Creatinine clearance estimates were determined using the Cockcroft-Gault equation for comparisons among groups, and renal dysfunction during treatment was defined as a serum creatinine >1.4 g/dL.
Results
The initial informatics query identified 87 patients with AXAL data available for evaluation during the study period. Baseline characteristics of the 87 patients who received therapeutic drug monitoring of enoxaparin are listed in Table 1. Notably the majority of patients were either overweight or obese, 24% had a history of solid organ transplant, and 19.5% of the patients experienced renal dysfunction during treatment. Monitoring of AXAL occurred most frequently among patients on the medicine service and in the intensive care unit. Table 2 describes the dose-related characteristics of patients in the overall cohort. A trend of lower enoxaparin requirements in mg/kg/dose was noted with increases in serum creatinine or body weight.
Baseline Characteristics Among all Patients with Enoxaparin Therapeutic Drug Monitoring.
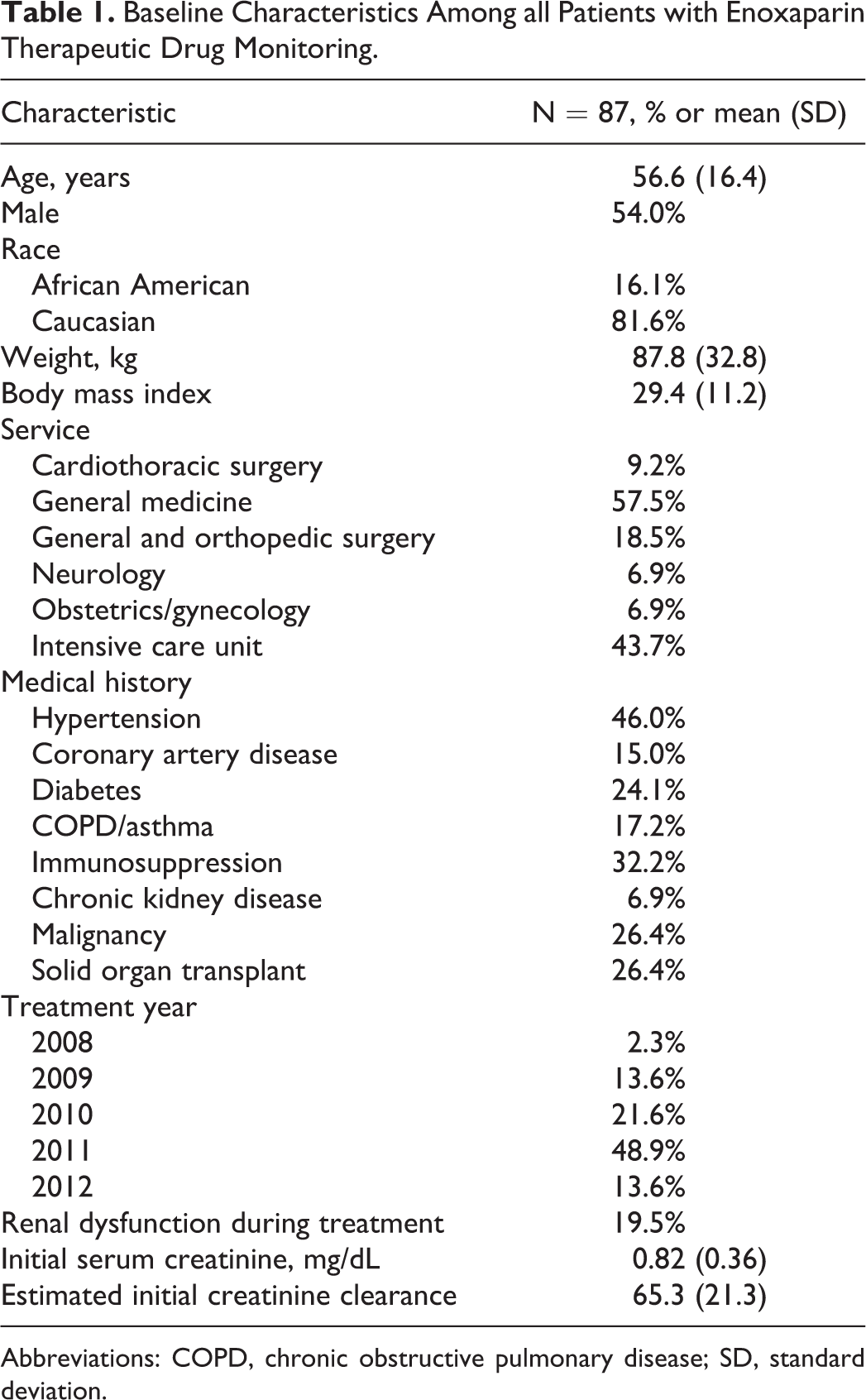
Abbreviations: COPD, chronic obstructive pulmonary disease; SD, standard deviation.
Treatment-Related Characteristics Among Patients With Therapeutic Drug Monitoring of Enoxaparin.
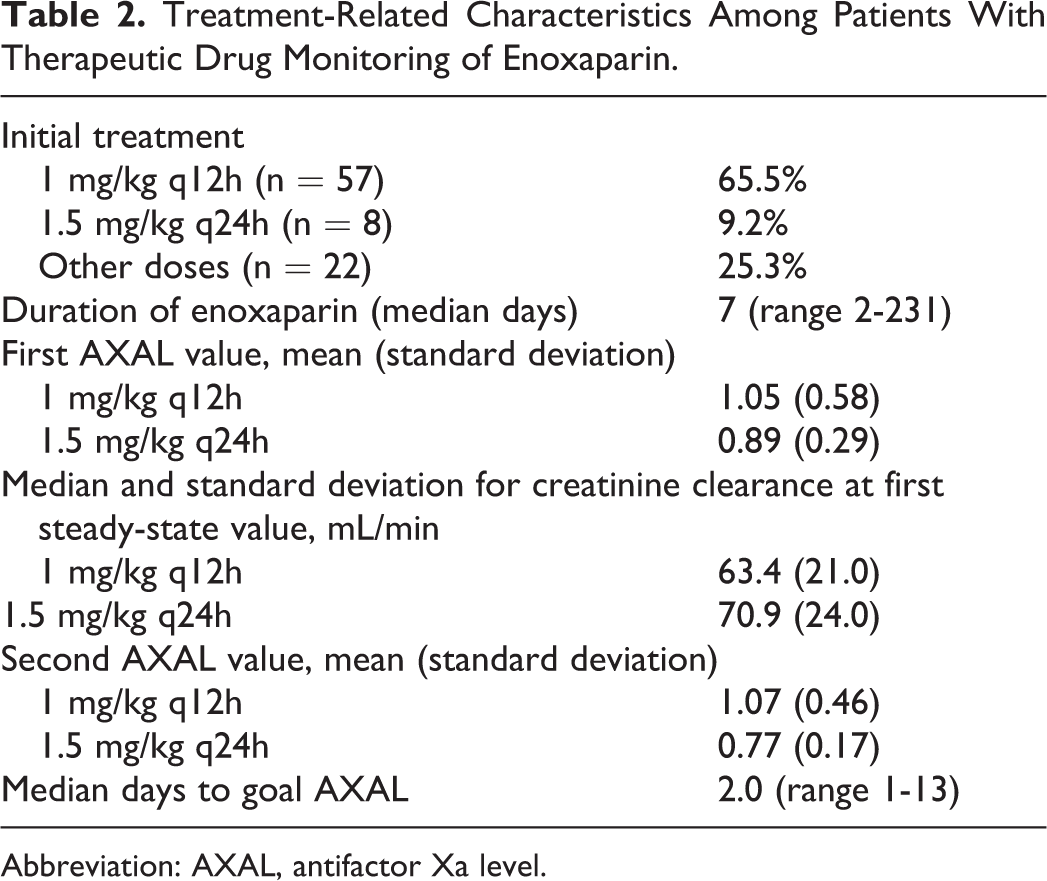
Abbreviation: AXAL, antifactor Xa level.
Among the 87 patients, 19 did not have a subsequent AXAL drawn at a time appropriate for evaluation of dose titration, thus resulting in a final cohort of 68 patients. The effect of initial and secondary enoxaparin dose titration on subsequent AXAL is summarized in Figure 1. Ultimately 54 (79.4%) of the 68 patients achieved a therapeutic AXAL during the study with a median number of dose titrations of 1.5. The initial AXAL was at goal in 27 (30.7%) of the 87 patients, and 20 of these 27 patients had subsequent evaluable AXAL.
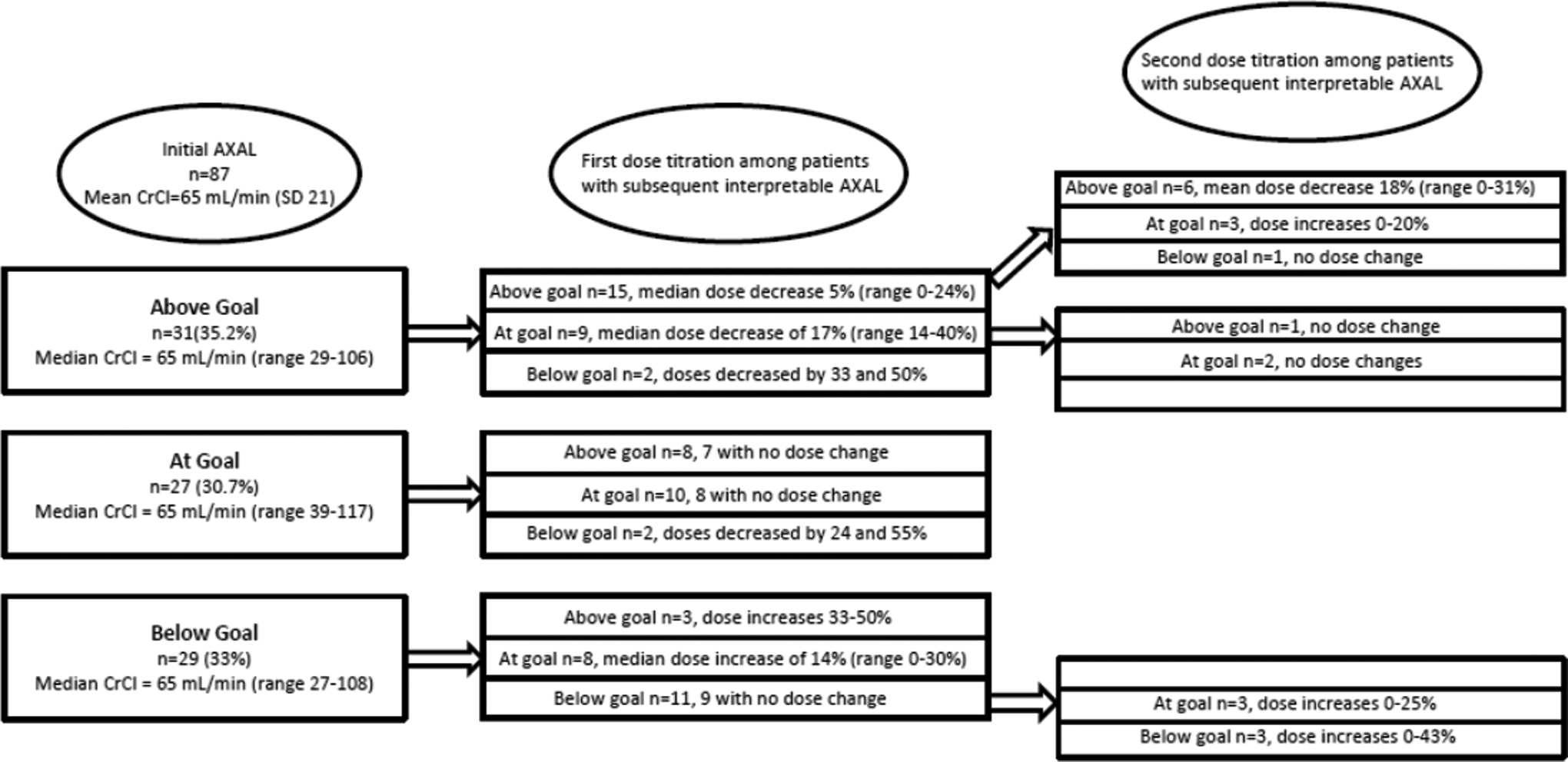
Effect of initial and second enoxaparin dose titration on subsequent AXAL. AXAL indicates antifactor Xa level; CrCl, creatinine clearance.
Additionally, 31 (35%) of the 87 patients had an initial AXAL above goal, and 26 (83.9%) of the 31 had subsequent evaluable AXAL. Of the 26 patients, 9 (34.6%) achieved goal AXAL with first-dose titration and an additional 4 (13 of 26; 50%) achieved goal AXAL with additional dose titrations, for a total median dose decrease of 24%.
Twenty-nine patients (29 of 87= 33%) had an initial AXAL below goal and 22 of the 29 had subsequent evaluable AXAL (75.9%). Of the 22 patients, 8 (36.4%) achieved goal AXAL with the first-dose titration and an additional 3 (11 of 22; 50%) achieved goal AXAL with additional dose titration, for a total median dose increase of 16%.
Conclusions
Our study illustrates that AXAL monitoring is at times utilized in clinical practice even in patients with normal body weight and renal function. At our center, AXAL was used to guide enoxaparin titration among patients without initial AXAL at goal. Fifty percent of evaluable patients with initial AXAL above goal subsequently achieved a goal AXAL with a median dose reduction of 24%. In addition, 50% of patients with initial AXAL below goal subsequently achieved a goal AXAL with a median dose increase of 16%. Therefore, we recommend a dose modification of approximately 20% (range 16%-24%) in patients with either above or below goal AXAL, respectively. Remarkably, only 31% of patients had an initial goal AXAL and only half of the dose titrations led to achievement of goal AXAL, suggesting that enoxaparin pharmacokinetics may not be as predictable as previously thought.
Our study has several important limitations including its retrospective nature and the fact that treatment decisions to adjust doses were left at the discretion of the prescriber during the study period. Additional factors that affect anti-Xa levels including the presence of hyperbilirubinemia and hypertriglyceridemia were not evaluated in this limited review. Further areas of research include the development of a larger, prospective study to design a dosing nomogram for clinicians desiring to use LMWH in conjunction with AXAL monitoring. The patient’s weight and renal function should be taken into account as a low creatinine clearance and high body weight have been shown in this study and others to correlate with lower enoxaparin dosing requirements. Future studies should also focus on the impact of AXAL monitoring on clinical outcomes such as recurrent VTE and bleeding.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
