Abstract
Factor VIII (FVIII) activity, antigen, and its gene mutation in patients may give us some insight into structure–activity relationship and probable reason for its low level in a particular patient. A total of 53 cases with hemophilia A and 17 normal individuals were analyzed for FVIII:C and FVIII:Ag levels along with their blood groups to determine their cross-reacting material (CRM) status. In all, 37 cases (18 moderate and 19 mild) were found to have discordant CRM status. Missense mutations (Ala723Thr and Lys439Ser) and protein truncating changes (Trp1854* and Arg2326*) were observed in 2 each of these cases. Although 37 (70%) of the 53 had discordant antigen–activity ratio, majority of those mutations produced FVIII with low FVIII-specific activity. However, 4 (7.5%) of the 53 mutations produced higher specific activity of FVIII. It is possible that these mutations either produce a secretory defect or an increased metabolic turnover to account for the low levels of FVIII with these mutations.
Introduction
Hemophilia A (HA) is an X-linked recessive disorder caused by faulty factor VIII (FVIII) gene (F8), affecting 1 in 5000 live males. It is one of the largest genes located at Xq28 position at the telomeric end of X-chromosome spanning 186 kb, comprising of 26 exons encoding 2332 amino acids. A high GC content makes it hypermutable, with approximately 30% of the mutations arising de novo.
In case of predicted single amino acid substitutions, 2 broad categories of phenotypes are found: (1) cross-reacting material (CRM) reduced or CRM−ve, in which plasma FVIII:Ag is reduced concomitantly or discordantly with the activity, and (2) CRM positive or CRM+ve, with absent FVIII:C activity. Approximately 5% of cases with HA turn out to be CRM positive, whereas almost 45% of patients have plasma FVIII protein reduced to an extent roughly comparable to the level of FVIII activity and are designated CRM reduced. 1 The CRM-positive patients generally have 30% of the normal FVIII protein in their plasma, but alas it is nonfunctional, due to which the FVIII activity is much less than the FVIII protein level. In the CRM−ve cases, FVIII antigen is almost undetectable. Patients with CRM-reduced hemophilia A have reduced plasma FVIII antigen levels regardless of activity levels. Mutations at or after critical arginine residues at thrombin cleavage sites (residues 391/392 and 1708/1709) have been shown to render the molecule resistant to thrombin activation resulting in reduced coagulant activity. 2,3 Intracellular accumulation and subsequent degradation of FVIII might be the reason for reduced levels of both FVIII:C and FVIII:Ag in plasma of patients with mild HA having corresponding genetic defects. 4 ABO histoblood group is a major determinant of plasma FVIII levels. Although the underlying mechanism is unknown, there is an association between the ABO blood group and FVIII–von Willebrand factor (vWF) levels in the plasma. Factor VIII activity, antigen, and its gene mutation in patients may give us some insight into structure–activity relationship and probable reason for its low level in a particular patient.
Patients, Materials, and Methods
A total of 53 intron 22 and intron 1 inversion negative patients with HA (25 moderate and 28 mild) were analyzed for characterizing the causative mutations of HA. The study was approved by the institutional ethics committee (IEC/IIH/15-2007).
After obtaining informed consent, 9 mL venous blood was collected in 3.2% trisodium citrate in the ratio 1:9 (anticoagulant:blood). It was spun at 4000 rpm at 4°C for 15 minutes. The supernatant containing the platelet poor plasma was separated and used for phenotypic analysis. The cell pellet was used for DNA extraction, which was done by using commercial kits (Invitrogen, California).
Phenotypic Assessment
Measurement of the prothrombin time, activated partial thromboplastin time, and thrombin time was done using commercial reagents (Dade Behring, Marburg, Germany). Mixing studies at 0, 1, and 2 hours were performed in all cases to rule out the presence of inhibitors against FVIII. FVIII:C was measured by 1-stage assay in duplicates using commercial FVIII-deficient plasma (Diagnostica Stago, Asnieres, France) using a semiautomated coagulometer (ST Art; Diagnostica Stago). FVIII:Ag was assayed by enzyme-linked immunosorbent assay using commercial kits (Asserachrom FVIII:Ag; Diagnostica Stago). Healthy normal volunteers of different blood groups such as A, B, and O also had the same parameters measured in duplicates, and mean value was used for measurement. F8 mutations in the cases with HA were identified by using a combination of conformation-sensitive gel electrophoresis and DNA sequencing or only direct DNA sequencing.
DNA Analysis
The coding region, intron/exon boundaries, and the untranslated regions of the F8 were amplified in multiplex polymerase chain reactions using specific primers (Sigma Aldrich, Missouri). 5 The novel missense mutations were screened for their deleteriousness using the various prediction softwares—PolyPhen 6 and PANTHER. 7
Results
In all, 37 (36 unrelated and 1 familial) cases (70%) with mild to moderate HA showed discordant CRM status. The ratio of FVIII:C to FVIII:Ag showed 4 cases (3 unrelated and 1 familial) with a higher specific activity as compared to the antigenic activity (Table 1). Of these, 2 cases had missense mutations (Ala723Thr and Lys439Ser), whereas the other 2 had protein truncating mutations (Trp1854* and Arg2326*). All the severe cases exhibited a concordant ratio of FVIII:C and FVIII:Ag activities. Blood group “A” shows a lower standard deviation indicating the expected value, whereas blood group “O” exhibited a higher standard deviation. This is due to many CRM discordant levels found in the patients with this blood group (Figure 1). Maximum CRM+ve cases in this study were found in patients with blood group “O.” The analysis of FVIII:C/FVIII:Ag in normal individuals belonging to the different ABO blood group revealed values with lower standard deviation, pointing toward the expected levels (Figure 2 and Table 2).
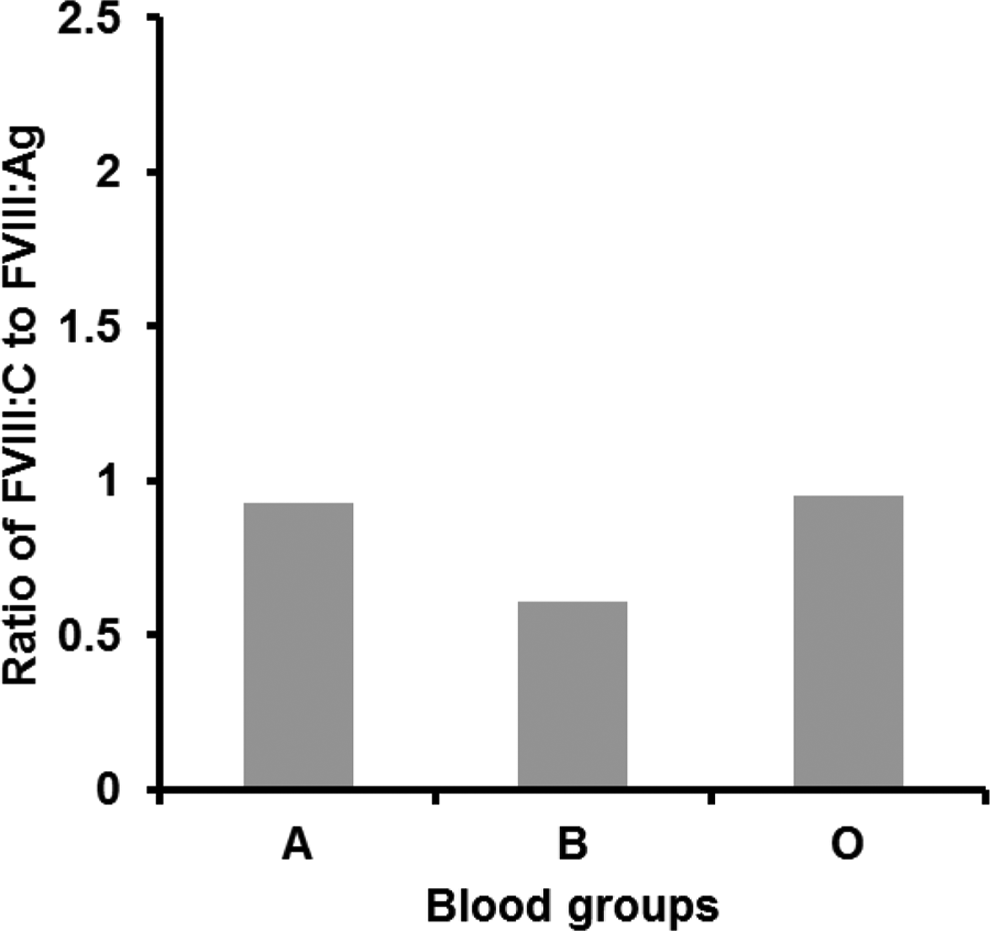
FVIII:C/FVIII:Ag among cases with mild/moderate hemophilia A (HA) with respect to blood groups in the present study.
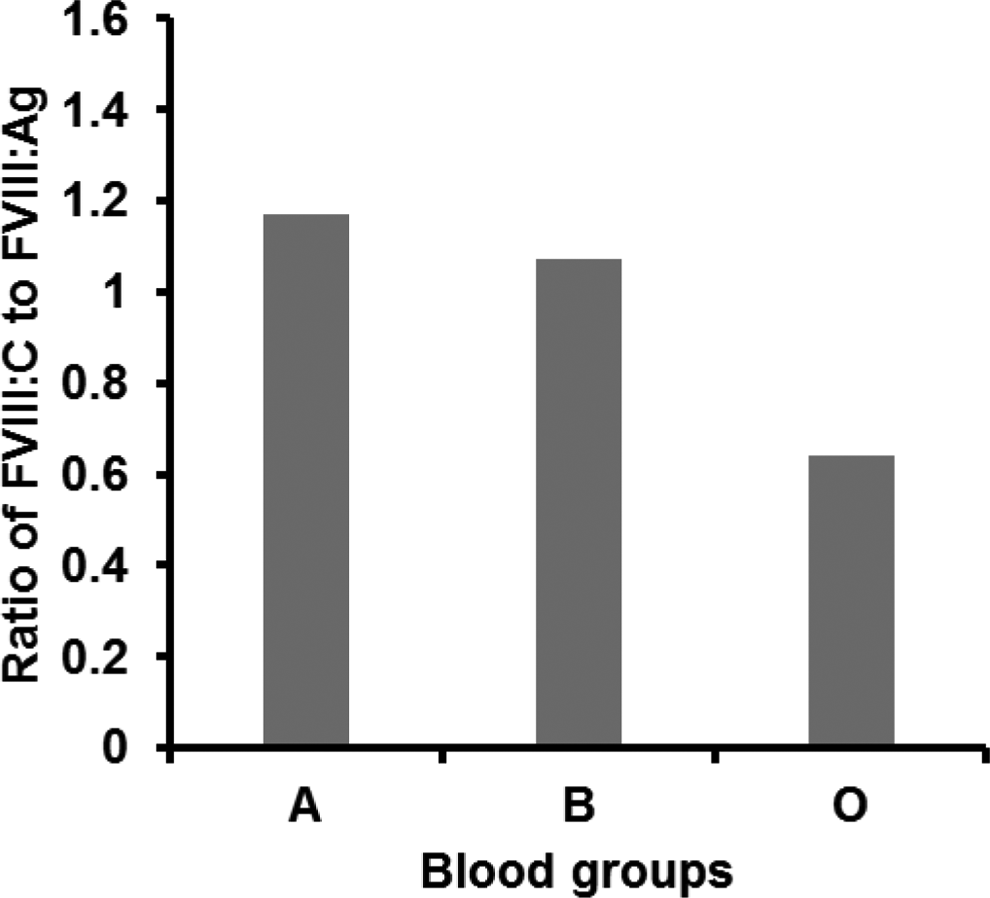
FVIII:C/FVIII:Ag of normal individuals with respect to blood groups.
Discordant CRM Status in Patients With Mild/Moderate Hemophilia A in the Present Study.
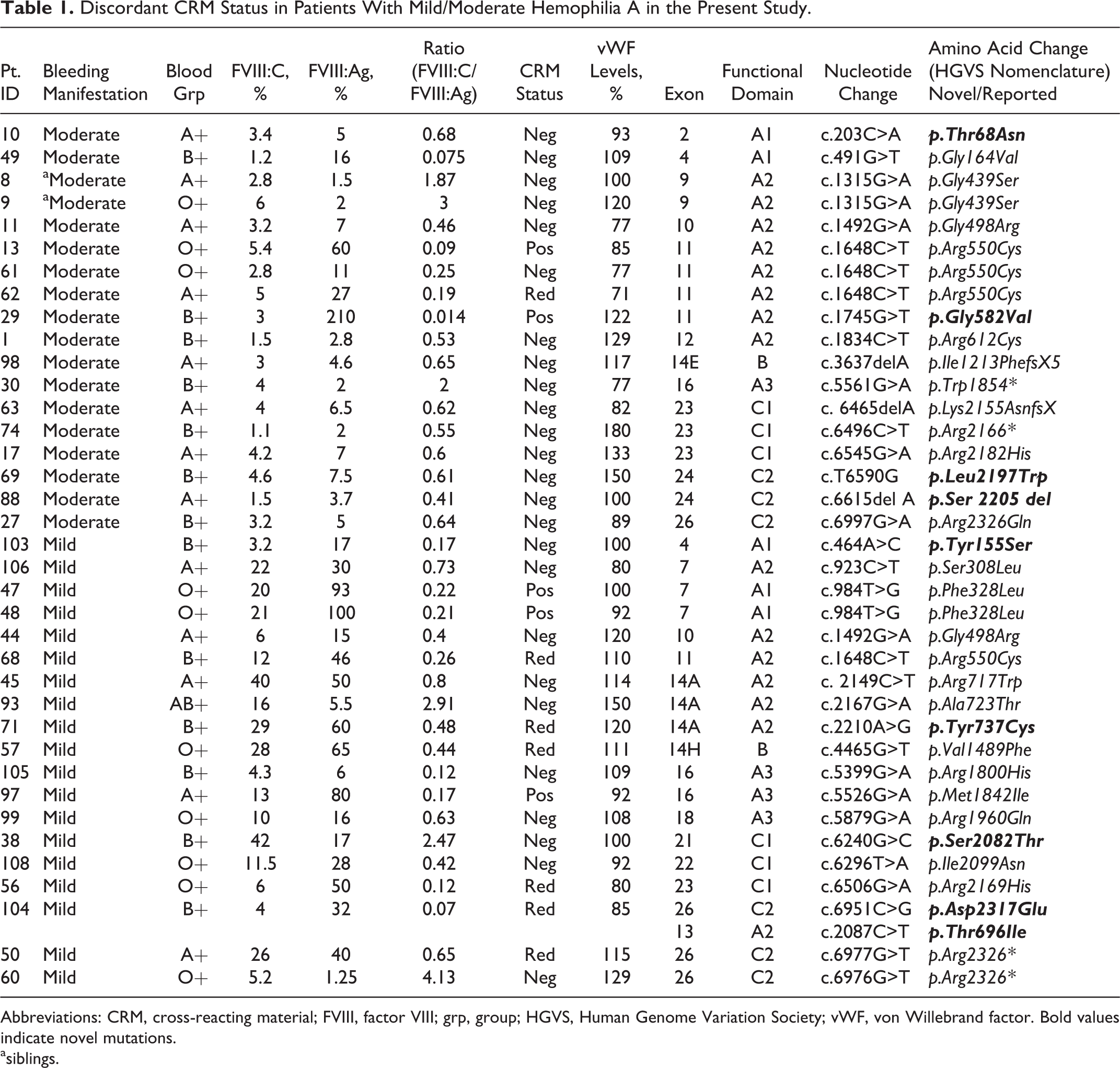
Abbreviations: CRM, cross-reacting material; FVIII, factor VIII; grp, group; HGVS, Human Genome Variation Society; vWF, von Willebrand factor. Bold values indicate novel mutations.
asiblings.
FVIII:C and FVIII:Ag in Normal Individuals With Respect to Their Blood Groups.
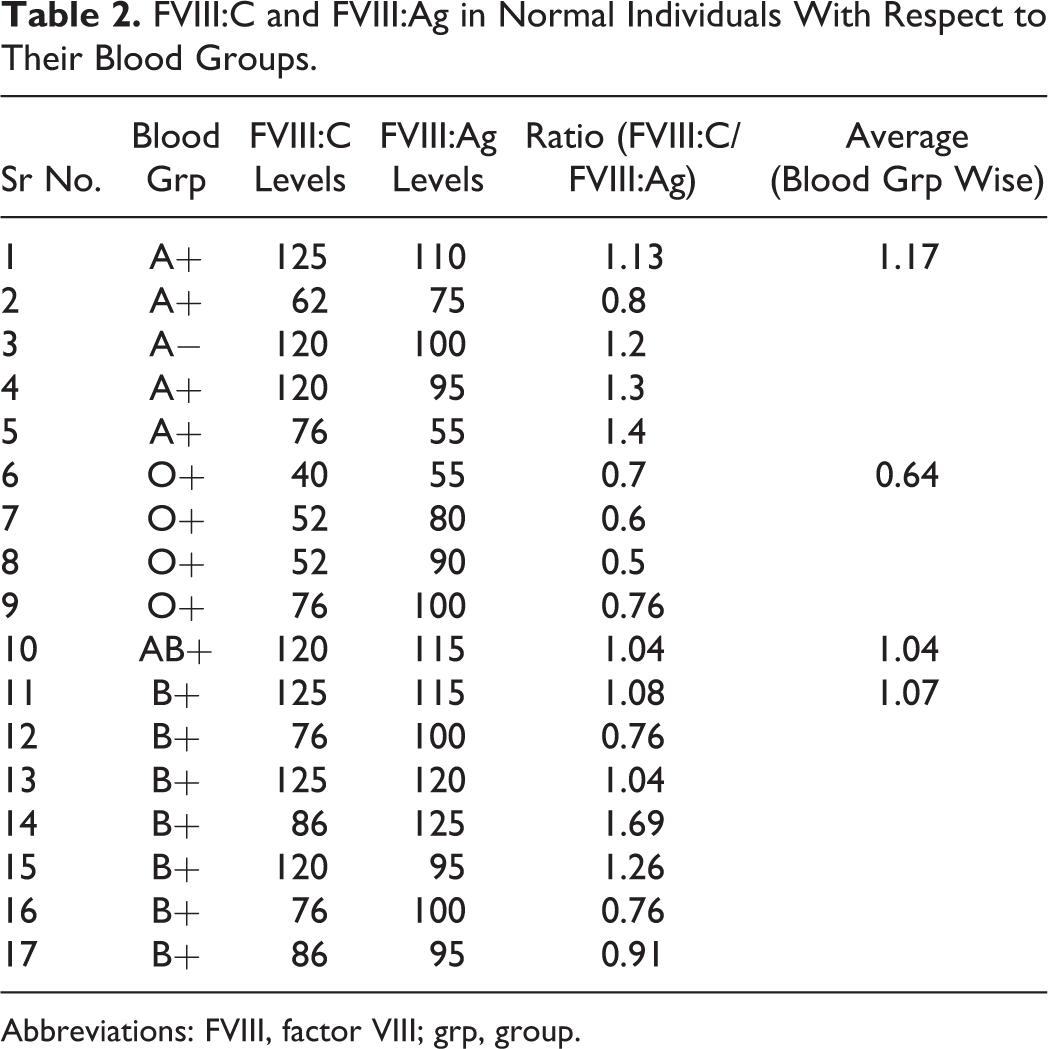
Abbreviations: FVIII, factor VIII; grp, group.
Discussion
Approximately 5% of the patients with HA have normal levels of dysfunctional FVIII protein and are termed as CRM+ve. Such patients generally have 30% of the normal FVIII protein in their plasma, but alas it is nonfunctional, due to which the FVIII activity is much less than the FVIII protein level. In the CRM−ve cases, FVIII antigen is almost undetectable. Patients with CRM-reduced hemophilia A have reduced plasma FVIII antigen levels regardless of activity levels. Some CRM-reduced patients also display lower activity values compared with the plasma antigen levels. 8 Almost 40% of patients with CRM+ve HA and CRM-reduced HA exhibit missense mutations in the A2 domain. 1 The A2 domain of F8 encodes for 330 amino acids and consists of 15% of the entire amino acid sequence of the gene. This region is important for FVIII procoagulant activity. The CRM reduced cases can arise due to mutations in sulfated tyrosine residues, loss of high-affinity binding to vWF at the binding site, and inability of the new residue to be sulfated. The other possibilities are mutations abolishing existing cysteine residues known to be involved in disulfide bridge formation, introducing new cysteine that may promote novel unnecessary disulfide bond formation/mutations at or near sites of binding to vWF can result in defective association with plasma carrier of F8. Wherein the FVIII:Ag levels are essentially normal/only mildly reduced while activity is grossly reduced or undetectable, mutation causes HA by generation of functionally inactive molecule that circulates normally. 9
ABO histoblood group is a major determinant of plasma FVIII levels. Individuals with blood group “O” have significantly (approximately 25%) lower levels of both plasma glycoproteins FVIII and vWF. Although the underlying mechanism is unknown, there is an association between the ABO blood group and FVIII–vWF levels in the plasma. It has been noted that the ABO effect is mostly mediated through a direct functional effect of ABO locus on plasma vWF levels. 10 The vWF clearance rate also known to be dependent upon the ABO type. Moreover, the N-linked oligosaccharides may also be responsible for the effect of ABO blood group on the plasma vWF levels which in turn can affect the FVIII levels of the individuals. The vWF levels in the present study did not show a significant variation across the ABO blood group, except blood group “B” exhibiting a slightly higher vWF levels that the other 2. However, the standard deviation for the FVIII:C/FVIII:Ag levels showed a significant variation across the blood groups, with blood group “O” showing more CRM+ve cases compared to the other 2 blood groups.
In the present study, we came across 4 unrelated and 1 familial cases with mild to moderate HA (ID-8, 9, 30, 60, and 93) showing an increased FVIII:C activity compared to the antigenic levels. Two of these cases (ID-30 and 60) showed protein truncating mutations in A3 and C2 domains, respectively. The change Arg2326* was observed in a mild case in the vWF/phospholipid binding region of F8, whereas the change Thr1854* was observed to be located near to a potential N-glycosylation site. 11 Both these regions are functionally important for FVIII activity. However, these cases produced 4% to 5% functional FVIII activity, inspite of having protein truncating changes. A possible explanation for such a phenomenon would be occurrence of nonsense mutation suppression. At times, in cases with protein truncating codons, a nonsense-mediated decay (NMD), which is an evolutionary surveillance mechanism, gets activated which controls the quality of messenger RNA (mRNA) by eliminating aberrant mRNA. 12 Although NMD functions to eliminate premature termination codon-bearing mRNAs to minimize the production of mutant proteins, this elimination process is not 100% efficient and often results in the generation of low levels of truncated proteins produced from residual aberrant transcripts. At Arg2326 which is in the vWF/phospholipid binding region, F8 has the possibility of behaving in similar fashion like vWD type 2 Normandy.
The 2 missense changes, that is, p.Gly439Ser and p.Ala723Thr, were also noted in this study to give rise to discordant levels. The hydrogen chain of glycine contributes to the flexibility, and a change to serine can affect the secondary structure of the protein leading to instability, 13 whereas the change Ala723Thr is near to a sulfotyrosine residue in the A2 domain.
In conclusion, although a majority of patients with mild/moderate HA showed discordant antigen:activity ratio, most of these mutations produced FVIII with lower specific activity. However, 7.5% of the mutations, which included 2 missense mutations p.Lys439Ser and p.Ala723Thr and 2 nonsense mutations p.Trp1854* and p.Arg2326*, produced higher specific activity of FVIII. We hypothesize that these mutations either produce a secretory defect or an increased metabolic turnover to account for the low levels of FVIII.
Footnotes
Acknowledgments
Authors are grateful to Department of Science and Technology, Government of India (DST), for funding this study and also the West Bengal University of Health Sciences for including this study in their PhD program.
Authors’ Note
Authors do not have a financial relationship with the organization (DST) that sponsored the research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Authors are grateful to Department of Science and Technology, Government of India (DST), for funding this study.
