Abstract
In order to establish the efficacy and biosimilar nature of AryoSeven to NovoSeven in the treatment of congenital factor VII (FVII) deficiency, patients received either agent at 30 μg/kg, intravenously per week for 4 weeks, in a randomized fashion. The primary aim was to compare FVII:coagulation activity (FVII:C), 20 minutes after recombinant activated FVII (rFVIIa) injection, in the 2 groups. A secondary measure was self-reported bleeding. The median interquartile baseline range of the plasma level of activated FVII (FVIIa) activity in the 2 groups was 1.6 (1.1-14.0) IU/dL and 5.0 (1.1-25.5) IU/dL. All patients achieved levels of FVIIa (FVII:C) >30 IU/dL, 20 minutes after the injection of rFVIIa. Bleeding was similar between the 2 groups, with a comparable decrease in severity and frequency compared to the last month prior to treatment. AryoSeven is similar to NovoSeven in increasing postinjection FVIIa activity as well as in clinical safety and efficacy.
Introduction
The availability of recombinant activated factor VII (rFVIIa) has changed the management of bleeding in patients with congenital disorders, such as congenital factor VII (FVII) deficiency, hemophilia A/B with an inhibitor, and Glanzmann thrombasthenia. 1 –4 Coagulation FVII deficiency is a rare autosomal recessive hereditary disease caused by mutations in the FVII gene on chromosome 13, affecting both males and females. 2 –8 It has wide phenotypic and genotypic variability similar to other hereditary bleeding disorders, for example, factor XI and factor VIII deficiency. 9 –13 The incidence of the disease is about 1 of every 500 000 people worldwide but is significantly higher in countries such as Iran where interfamily marriages are more common. 8,14 According to the transplant and special diseases office in Iran, and also Iranian comprehensive Hemophilia care center (ICHCC) data registry, 7128 patients are registered with hereditary bleeding disorder and 232 patients have a congenital FVII deficiency. 15
Coagulation FVII is naturally produced in the liver, with the plasma level approximately 0.5 μg/mL in healthy individuals, 2,16 and only 1% of total FVII is in the activated form in a normal person (FVIIa, 10-100 pmol/L). 3,17 Since the binding of FVII to tissue factor is required to trigger the extrinsic coagulation cascade, the lack of this factor reduces the generation of thrombin that is required to activate platelets, this results in inadequate production of fibrin. In patients with severe FVII deficiency, lethal bleeding occurs within a short time after birth and neonatal death may occur. 18 There is poor correlation between phenotypic manifestation and genotype and also between the plasma FVII:coagulation activity (FVII:C) level and bleeding severity. 7 –14 Clinical manifestations can range from mild, such as mucosal bleeding, to severe such as cerebral hemorrhage and abnormal perioperative bleeding. 13 , 14,16 –19 Bleeding in infants usually affects the central nervous system and is associated with adverse effects. 18
Recombinant FVIIa is a replacement therapy in patients with congenital FVII deficiency and is used for the treatment of bleeding episodes and prevention of bleeding during surgery and other invasive procedures in these patients. 14,16 –19 Since FVII deficiency is a rare bleeding disorder, there are few guidelines for management of bleeding episodes and prevention of hemorrhagic events. 18 –20
In July 2005, NovoSeven received Food and Drug Administration approval for use in congenital FVII deficiency bleeding episodes. 20,21
AryoSeven is designed as a biosimilar molecule of NovoSeven. In this study, we compared therapeutic effects, side effects, and safety between rFVIIa product AryoSeven and NovoSeven in patients with congenital FVII deficiency.
Patients and Methods
Trial Design
This trial was a double-blinded, randomized, multi-center study in 8 comprehensive hemophilia care centers around Iran. Eligible patients who met inclusion criteria were randomized between 2 treatment groups (group A: Aryoseven as the intervention group, group B: Novoseven as the control group). Randomization was based on consecutive balanced block randomization tables that were assigned separately for each study site. We designed a form to collect patients’ data including demographic information, medical history, physical examination, and laboratory results for the study. The randomized patients received 1 single dose of rFVIIa (Aryoseven in group A or Novoseven in group B) 30 µg/kg, intravenously, once a week for 4 consecutive weeks as prophylaxis. 22 –24 A total of 66 patients were enrolled in the trial and there was no withdrawal during the study (group A: 35 patients and group B: 31 patients).
The patients’ visits included 6 visits for each patient that consisted of a screening visit (visit 1), 4 weekly visits (visits 2-5), and 1 follow-up visit (visit 6).
Bleeding Severity Assessment
The patients were assessed for bleeding severity which was classified from mild to severe at each center by 1 physician with prepared form. Mild bleeding was defined as bruising, hemorrhoid bleeding, microscopic hematuria, and situations considered being part of everyday life which did not interfere with daily activities nor requires medical attention. Moderate bleeding was clinically overt but not meeting criteria for severe bleeding. Severe bleeding was defined as clinically overt bleeding associated with a drop in hemoglobin more than 2 g/dL or requiring blood transfusion more than 2 units of red blood cells, retroperitoneal or intracranial bleeding. 25,26
Ethical Consideration
This study was approved by the ethics committee of the Iranian Blood Transfusion Organization and the Ministry of Health in agreement with Declaration of Helsinki and good clinical practice. The trial was registered on the IRCT Web site with registration number: 201104266302N1.
Patient Eligibility
Inclusion criteria consisted of patients with congenital FVII deficiency and more than 1 episode of bleeding per month and age >2 years. All patients included in study had lower than 30 IU/dL of FVII:C. Exclusion criteria included any other coagulation disease, previous prophylaxis therapy with rFVIIa during the past month, platelet count <50 000, presence of a neutralizing anti-FVII antibody, and a history of severe atherosclerosis.
Patients met inclusion criteria by scientific committee of clinical trial team eligible for study. Written informed consent was signed by all patients or their legal guardians.
Laboratory Methods
FVII coagulation activity was measured by 1-stage clotting method using recombinant-tissue factor preparation (STA-Deficient and STA-Neoplastin; Diagnostica Stago, France). Factor VII inhibitor was measured by classic “Bethesda assay” and the inhibitor titer reported in “Bethesda Unit.”
Outcomes and Safety
The primary outcome was the level of plasma FVII:C activity 20 minutes after rFVIIa injection, with a level of FVII: C <30 IU/dL considered a treatment failure. Secondary outcome measures were self-reported incidence of bleeding or severity and frequency of side effects and adverse events. Treatment safety was analyzed by evaluation of adverse events reported throughout the study period until 3 months after the fourth injection.
Statistical Analysis
Participants’ data were collected in the Case Report Forms (CRF) during the study and their accuracy was monitored by study supervisor to ensure their adherence to study protocol.
Data were analyzed by SPSS software (version 16.0, SPSS Co, Chicago, Illinois) and were described based on their statistical analysis. Quantitative data were expressed as mean ± standard deviation and qualitative data were expressed as frequency and proportion (percentage). The FVII plasma levels (FVII:C) did not exhibit a normal distribution, therefore the variation was presented as median and interquartile range (IQR). Quantitative data comparing the study group and the control group were analyzed using a parametric t test for data with normal distribution and nonparametric Mann-Whitney U test for data with an abnormal distribution (FVII:C plasma levels and rates of change during treatment). Independent qualitative data were analyzed by chi-square test and if it was necessary the Fisher exact test and dependent qualitative data were analyzed by McNemar test. The standard level of statistical significance was considered to be P ≤ .05.
Results
The mean age of the patients in the study was 21.8 ± 15.7 years (range: 2-65 years) and a total of 66 patients (33 male and 33 female) were enrolled in this study. The comparison of baseline data between 2 groups regarding age, sex, and FVII:C plasma levels did not show any significant difference (Table 1). Other factors, such as vital signs (respiratory rate, heart rate, blood pressure, and body temperature), coagulation tests (prothrombin time and partial thromboplastin time), liver and renal function tests, and blood count were not significantly different between the 2 groups. Median IQR plasma levels of FVIIa activity (FVII:C) in groups A and B were 1.6 (1.1-14.0) IU/dL and 5.0 (1.1-25.5) IU/dL, respectively, before injection (Table 1). All patients achieved target levels of FVII:C. All patients in both the groups had levels of FVII:C >30 IU/dL, 20 minutes after rFVIIa injection; therefore, there were no treatment failures. The increased levels of FVII:C were comparable between the 2 groups. There was no difference in levels of FVII:C 20 minutes after rFVIIa injection between the 2 groups in each of the 4 weeks of treatment (Table 2).
Comparison of Baseline Characteristics in the 2 Treatment Groups.
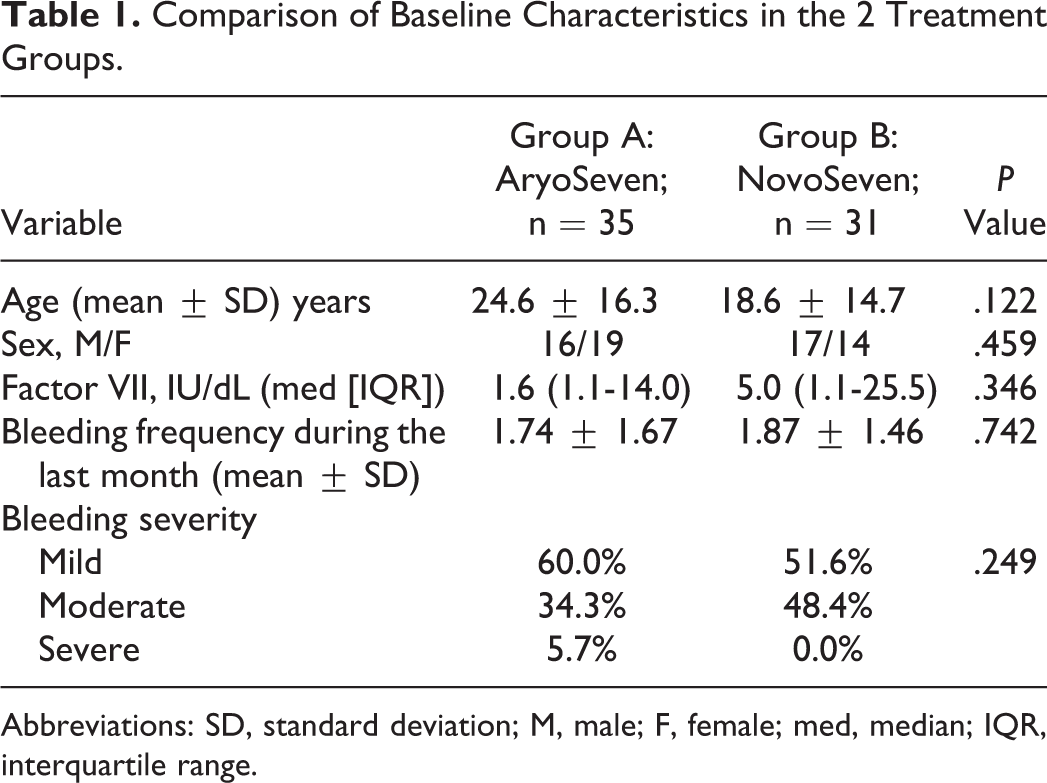
Abbreviations: SD, standard deviation; M, male; F, female; med, median; IQR, interquartile range.
Comparison of Changes in Factor VII Plasma Level (FVII:C) From Before to After rFVIIa Injection Between the 2 Groups.a,b
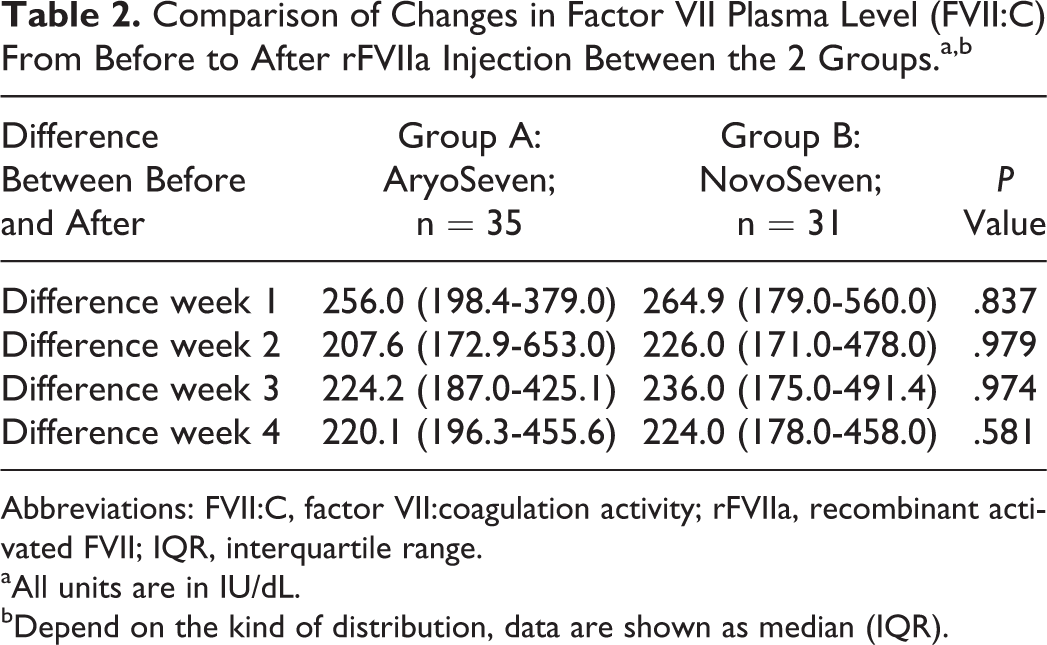
Abbreviations: FVII:C, factor VII:coagulation activity; rFVIIa, recombinant activated FVII; IQR, interquartile range.
aAll units are in IU/dL.
bDepend on the kind of distribution, data are shown as median (IQR).
The results also showed that the distribution of changes in FVII:C plasma levels after rFVIIa injection at the critical cutoff point (percentile 20) was similar between the 2 groups (Table 3). The 20th percentile was chosen because 80% of patients had increase over the amount of the serum levels of FVII in each treatment group and each week. So the comparison between the cutoff points help us to the better understand their performance relative to each other.
Comparison of Distribution of Changes in Factor VII Level Between Before and After rFVIIa Injection at the Critical Cut of Point (Percentile 20) in the 2 Groups.
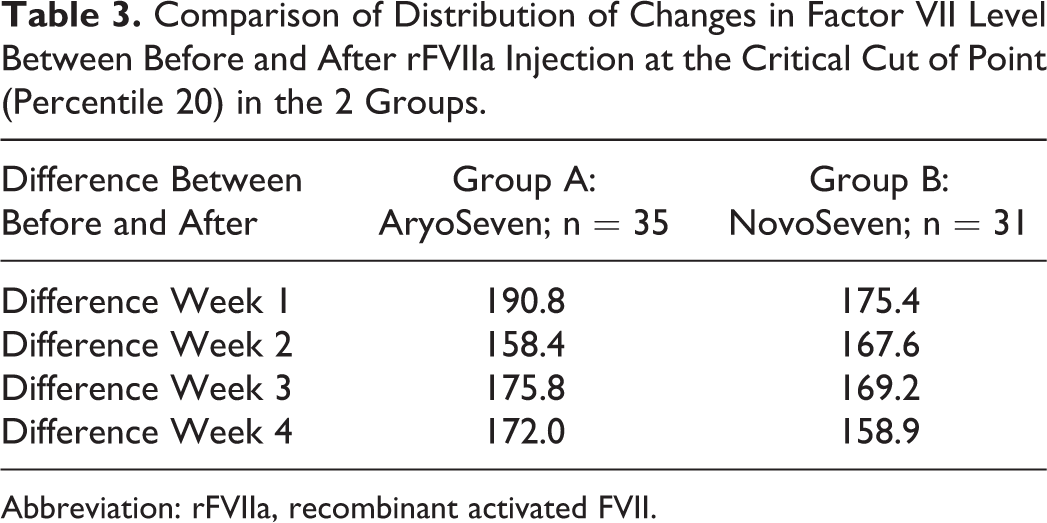
Abbreviation: rFVIIa, recombinant activated FVII.
The change in severity of bleeding was similar between the 2 groups, with a similar reduction in bleeding observed after treatment compared to before treatment of rFVIIa (Table 4).
Comparison of Changes of Bleeding Severity and Frequency Between Before and After Treatment Period in the 2 Groups.
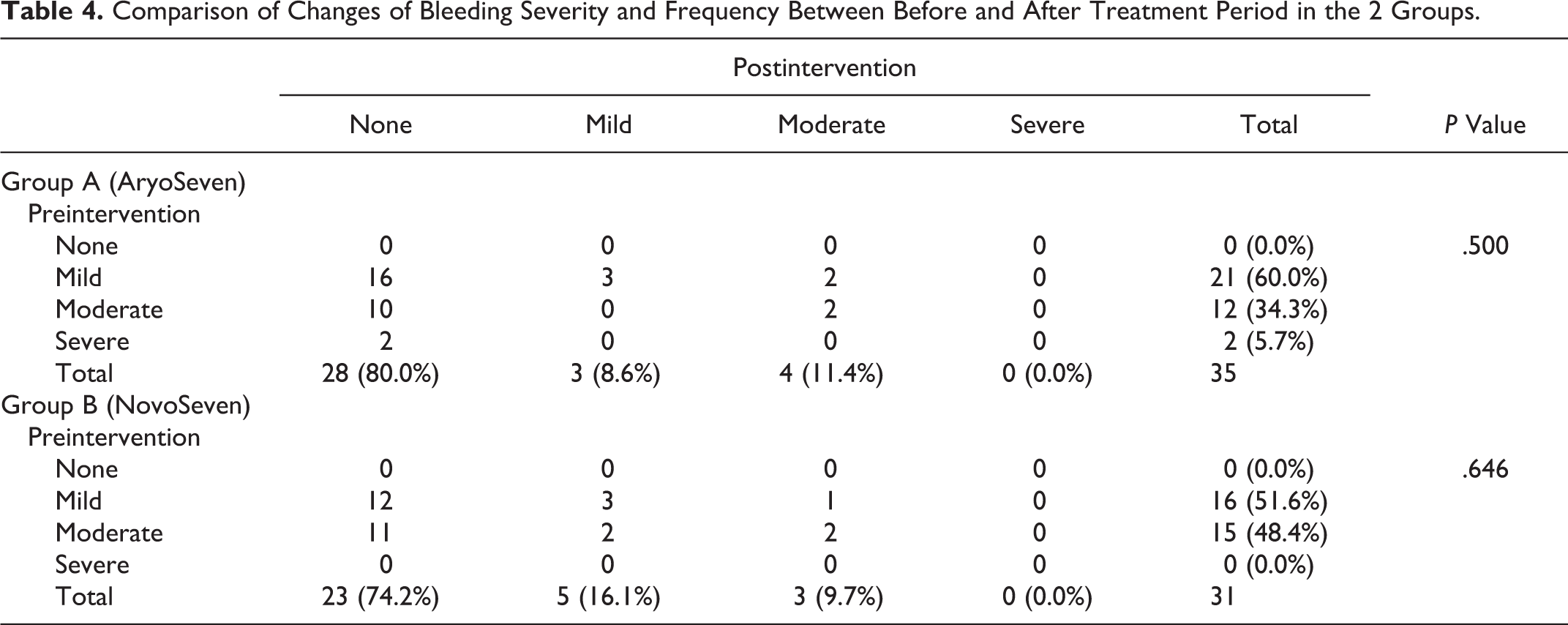
The change in frequency of monthly bleeding episodes was also similar between the 2 groups, with a similar reduction compared to the baseline (reduction in median of bleeding frequency during the month of treatment in group A and group B was 1 [IQR: 1-2] per month, P = .626; Table 4).
Reported side effects were minor (headache, nausea) and occurred at a similar frequency in both the groups (group A: 8 [22.9%] and group B: 3 [9.7%]). The frequency of side effects is presented in Table 5. No severe side effects were observed during the study period and up until 3 months after the fourth rFVIIa injection
Side Effects Observed During Treatment of FVIIa.
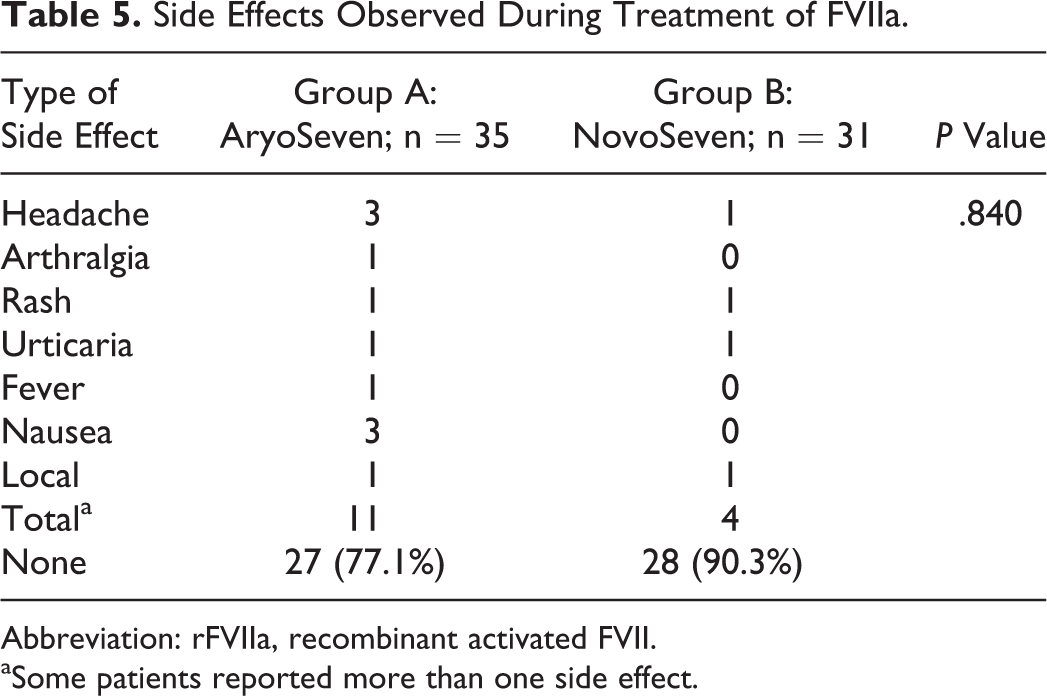
Abbreviation: rFVIIa, recombinant activated FVII.
aSome patients reported more than one side effect.
Discussion
A comparative clinical trial was undertaken to establish the biosimilar effects of rFVIIa (AryoSeven) to NovoSeven in terms of quality and clinical efficacy. The present study intended to elucidate the noninferiority of AryoSeven in comparison to NovoSeven in increasing the FVII level (FVII:C) and its clinical efficacy in prevention of bleeding events. Some similarities exist between our findings and the previous clinical trials designed for Novoseven. Baseline characteristics were similar to other studies considering the range of age, sex, bleeding severity, and FVII levels. In other studies, the age range was between 1 and 45 years and 52% of patients were female. 2,27 The prophylactic dosages of rFVIIa in those studies ranged from 30 to 90 μg/kg per week. In this study, a dosage of 30 µg/kg per week was administered. 21 –24 The data obtained in this study demonstrated a reduction in the number of bleeding episodes and also bleeding severity in both treatment groups when using the recommended dosage. The mean level of FVII before intervention in other studies was between 1 and 20 IU/dL, which is similar to our findings. 2 –9 Factor VII levels were increased to at least 50 IU/dL in the current study, which was similar to previous trials. 22 –28 Thrombosis was reported in some cases after injection of rFVIIa in other studies but we did not encounter any thrombosis among our patients. 28 Also similar to other studies, study patients did not demonstrate any serious side effects and there was no significant difference between frequency of side effects. 5,29
Based on the present data, this study demonstrates noninferiority of AryoSeven compared to NovoSeven in controlling and preventing bleeding episodes. The incidence of side effects showed no significant differences between 2 groups.
Although the therapeutic advances in control of bleeding episodes have led to a decreased morbidity and a better quality of life, the expense limits its access in some countries. 15,28 Biosimilar rFVIIa (AryoSeven) has similar activity with significantly lower cost compared to NovoSeven. Access and availability of treatment vary widely around the world. 21 The noninferiority of this drug at a significantly lower cost would fulfill the World Federation of Hemophilia mission to find efficient recombinant coagulation products at a reasonable price to close the gap in care and achieve treatment for all. 20,21
Conclusion
AryoSeven is similar to NovoSeven in postinjection FVIIa activity as well as in clinical efficacy. The frequency of side effects was also similar; however, the study was underpowered to detect differences in rare complications between the 2 compounds. The results of this study warrant additional clinical investigations to demonstrate the similar clinical and safety profile of AryoSeven in comparison to the branded NovoSeven.
Footnotes
Acknowledgments
The authors thank the patients with hemophilia, Aryogen Biopharma Company, Iranian Comprehensive Hemophilia Care centers and Iranian Blood Transfusion Organization for their help in conducting the present study. We are also thankful to Dr Rakesh Wahi for his review and advice in preparing this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was supported by a grant from Aryogen Biopharma Company.
