Abstract
Propylene glycol alginate sodium sulfate (PSS) is the world’s first oral heparinoid approved by Chinese Food and Drug Administration in 1987. Propylene glycol alginate sodium sulfate is produced by modifying partially hydrolyzed alginate, one of the most abundant marine polysaccharides isolated from brown algae, by epoxypropane esterification and by chemical sulfation. It is used for treating and preventing cardiovascular-related diseases. The low cost (US$1.29/100 tablets, ∼4 tablets/day), remarkable clinical effects, and convenient oral administration make PSS an ideal long-term prevention drug. Propylene glycol alginate sodium sulfate is available in most drug stores in China, and millions of patients take PSS routinely during the past 27 years. The 22 784 reported clinical cases as well as the structure, preparation, clinical efficacy, adverse reactions, pharmacokinetics, pharmacodynamics, and future perspectives of PSS based on the results of peer-reviewed publications will be discussed. This review should bring the knowledge of PSS gained in China to the world to stimulate in depth academic and clinical studies of PSS.
Keywords
Introduction
Cardiovascular diseases include any disease that affects the heart or circulatory vessels including myocardial infarction (heart attack), coronary artery disease, arrhythmia, hypertension, angina, atherosclerosis, cerebrovascular diseases including stroke, and peripheral circulatory diseases such as obstructions of arteries or veins in the hips, arms, and legs. The diseases are the leading causes of deaths in the world. However, the complications can be treated with anticoagulant drugs. The life-saving drug heparin, a polysaccharide or glycan purified from animal tissues, remains to be a major and essential anticoagulant in modern medicine after 79 years of hospital use. During the past 2 decades, low-molecular-weight heparins (LMWHs), that is, chemically or enzymatically cleaved heparin with reduced chain length, have progressed heparin from a hospital-administered drug into preventive subcutaneous injecting anticoagulant drugs with an annual sale over US$7 billion dollars. 1 Unfortunately, both heparin and LMWHs cannot be delivered by oral administration, a highly desirable feature for a disease-prevention drug. Although there are several clinical trials using alternative polysaccharides or special carrier to solve the oral delivering issue for heparinoid-based drugs, the first Chinese Food and Drug Administration (SFDA)-approved anticoagulant, propylene glycol alginate sodium sulfate (PSS), has been used both orally and intravenously for treating cardiovascular diseases in China since 1987.
Propylene glycol alginate sodium sulfate was developed by a team led by professor Huashi Guan at Ocean University of China. In 1960s, professor Guan participated in a national project involved in industry-scaled iodine extraction from brown algae to meet iodine demands in China. This project generated huge amount of brown algae polysaccharides as a by-product. The team took part in the by-product exploitation project in 1970s and subsequently developed several products including an algae polysaccharide-modified barium meal that is still used in China. Professor Guan discovered that the algae polysaccharides significantly reduced viscosity of the liquid suspensions of barium sulfate. He then added the algae polysaccharides to a hospital used barium meal. The new meal is thinner, smoother, and easy to swallow and produced better x-ray results for patients. Based on this observation, it was speculated that brown algae polysaccharide might be able to decrease the viscosity of blood, a prevalent medical problem for patients with cardiovascular diseases. Through trial and error, the team finally made the heparinoid PSS through epoxypropane esterification and chemical sulfation. Subsequent biological and clinical studies of PSS showed that it also has anticoagulant, 2 thrombolytic, 3 hypoglycemic, 4 and hypolipidemic properties documented for heparin. 5 In this review, the 22 784 reported clinical cases as well as the structure, preparation, clinical efficacy, adverse reactions, pharmacokinetics, pharmacodynamics, and future perspective of PSS based on the results of peer-reviewed publications will be summarized.
Preparation, Chemical Structure, and Clinical Doses
Alginate is one of the most abundant polysaccharides or glycans distributed in the cell walls of brown algae. It is a water-soluble linear glycan composed of guluronic acid (G) and mannuronic acid (M) with G α (1-4) and M β (1-4) linkages in the form of homopolymeric (MM or GG blocks) and heteropolymeric sequences (MG or GM blocks; Figure 1A). Alginate sodium prepared from brown algae usually has an average molecular weight over 900 kDa and is capable of absorbing 200 to 300 times its own weight in water, which makes the solution very viscous. To make PSS resembling the molecular weight of heparin, the 900 kDa alginate is partially hydrolyzed to obtain the ∼11 kDa fragment. The fragment is then chemically modified by esterification using epoxypropane to mimic N-acetylglucosamine residues in heparin and by sulfation using chlorosulfuric acid to mimic highly sulfated heparin (Figure 1B). Propylene glycol alginate sodium sulfate has ∼1.5 sulfates per disaccharide compared to 2 sulfates per disaccharide in heparin. Propylene glycol alginate sodium sulfate has an M to G ratio of 7:3 and a peak molecular weight at 11 kDa with a distribution width 4,6 of 1.6 as shown in Figure 1A.
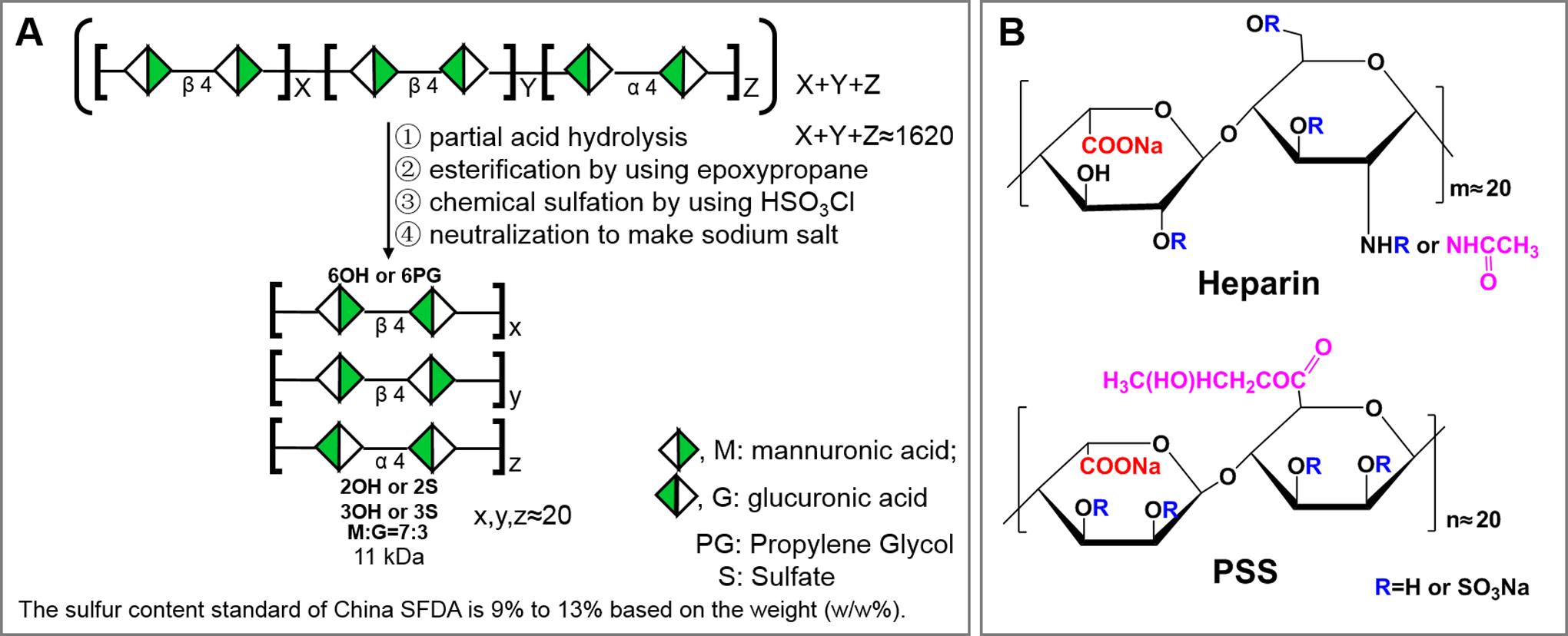
Preparation and structure of PSS. A, Preparation: to make PSS having the molecular weight of heparin, the 900 kDa alginate is partially hydrolyzed to obtain the ∼11 kDa fragment. The fragment is then chemically modified by esterification using epoxypropane to mimic N-acetylglucosamine residues in heparin and by sulfation using chlorosulfuric acid to mimic highly sulfated heparin. Propylene glycol alginate sodium sulfate has ∼1.5 sulfates per disaccharide compared to 2 sulfates per disaccharide in heparin. Propylene glycol alginate sodium sulfate has an M to G ratio of 7:3 and a peak molecular weight at 11 kDa with a distribution width of 1.6. The SFDA specifies that organic sulfur in PSS must be within 9% to 13% (w/w%). B, Repeating disaccharide comparison between PSS and heparin: epoxypropane esterification in PSS is used to mimic N-acetylglucosamine residue, and sulfation is used to mimic highly sulfated repeating disaccharides in heparin. However, the presence or the amount of propylene glycol in PSS has not been established in published literatures or specified by the SFDA as a drug. PSS indicates propylene glycol alginate sodium sulfate; SFDA, Chinese Food and Drug Administration.
As a drug, PSS can be taken both orally and intravenously. Around 50 to 100 mg of PSS is recommended by SFDA for intravenous infusion once a day, whereas 50 to 100 mg of PSS, 2 to 3 times a day, is recommended if taken orally.
Clinical Uses and Efficacy
Propylene glycol alginate sodium sulfate was approved by SFDA as a heparinoid anticoagulant to treat atrial fibrillation, pulmonary embolism, deep vein thrombosis, myocardial infarction, venous thromboembolism, stroke, congestive heart failure, and genetic or acquired hypercoagulability in 1987. Since no patent was filed for PSS, SFDA subsequently granted 294 drug manufacturers the rights for PSS-related drug production, which include 241 tablet and 53 injection formulations. 7 Over the years, PSS are trialed and progressed to treat other diseases as well (Figure 2).
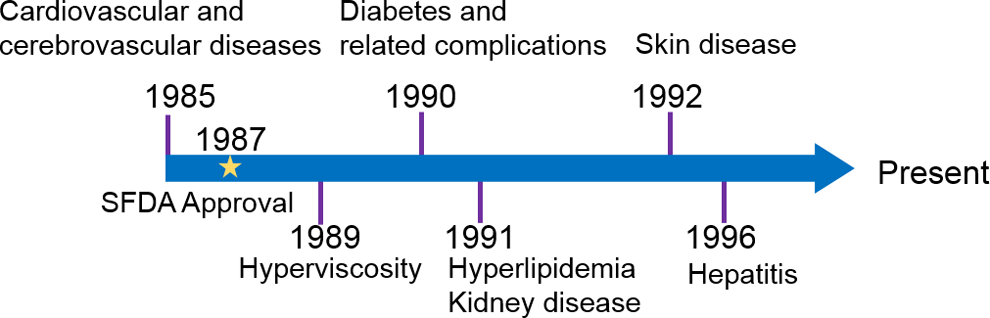
Extended clinical uses of PSS since its official approval in 1987. Propylene glycol alginate sodium sulfate was approved by Chinese Food and Drug Administration (SFDA) as a heparinoid anticoagulant to treat atrial fibrillation, pulmonary embolism, deep vein thrombosis, myocardial infarction, venous thromboembolism, stroke, congestive heart failure, and genetic or acquired hypercoagulability in 1987. However, PSS was subsequently used for treating hyperviscosity, diabetes and related complications, hyperlipidemia, kidney disease, skin disease, and hepatitis over the years based on peer-reviewed publications. PSS indicates propylene glycol alginate sodium sulfate.
There are 22 784 clinical cases related to PSS treatment, which is summarized in Figure 3 according to the percentages of cases in each disease including reported adverse events (Figure 3). As illustrated, 77% of reported treatment cases are patients with cardiovascular, cerebrovascular, 8 hyperlipidemic, and hyperviscositic diseases. 9 Propylene glycol alginate sodium sulfate is also used for treating diabetes and diabetes-associated complications, 10 kidney disease, 11 skin diseases, 12 and hepatitis. 13 There are rare cases where PSS is used for treating pneumonia (261 cases), 14 –16 emphysema (116 cases), 17 –19 gout (68 cases), 20 restless leg syndrome (3 cases), 21 sudden deafness (67 cases), 22,23 systemic lupus erythematosus (32 cases), 24,25 retinopathy (30 cases), 26 and acute nonlymphocytic leukemia (43 cases). 27
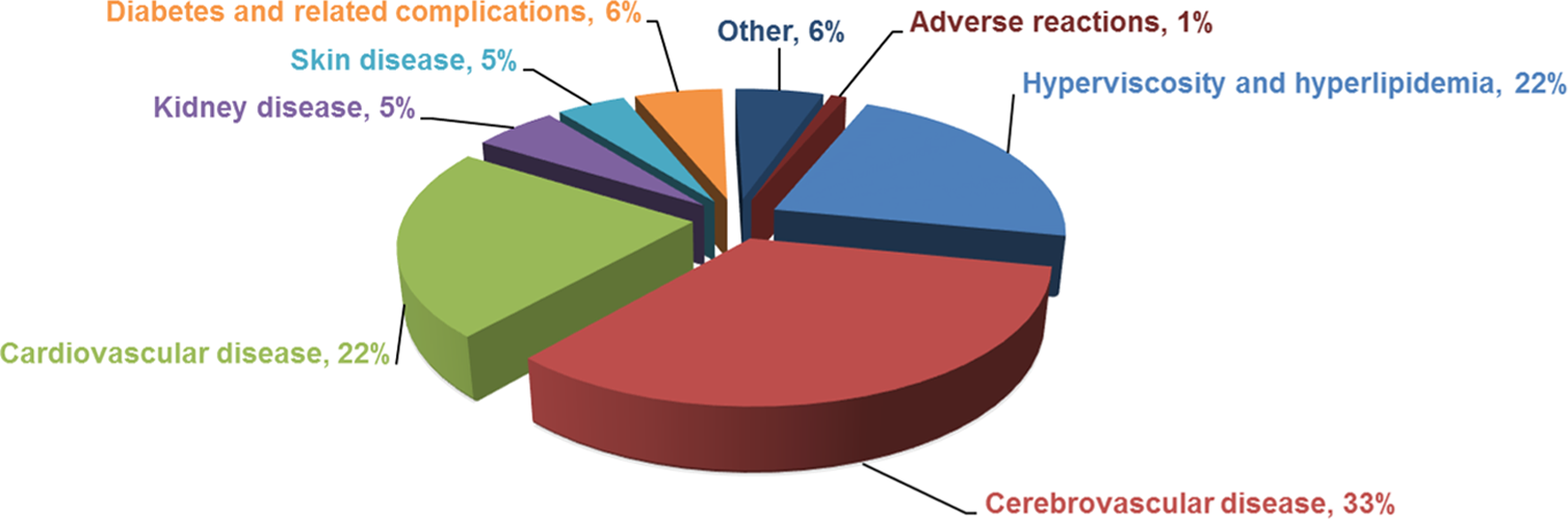
A summary of 22 784 reported clinical cases where patients with different diseases are treated with PSS. As illustrated, 77% of reported treatment cases are patients with cardiovascular, cerebrovascular, hyperlipidemic, and hyperviscosity diseases. Propylene glycol alginate sodium sulfate is also used for treating diabetes and diabetes-associated complications, kidney disease, skin diseases, and hepatitis. There are rare cases where PSS is used for treating pneumonia (261 cases), emphysema (116 cases), gout (68 cases), restless leg syndrome (3 cases), sudden deafness (67 cases), systemic lupus erythematosus (32 cases), retinopathy (30 cases), and acute nonlymphocytic leukemia (43 cases). PSS indicates propylene glycol alginate sodium sulfate.
The reported overall treatment efficacy is between 72.9% and 98.3% as summarized in Table 1. The efficacy includes cases where PSS is used in combination therapy, the detail is described in Table 2. In addition, novel combined clinical applications of PSS with other approved drugs are constantly developing, such as PSS plus hyaluronate sodium 28 in the prevention of postoperative adhesions (71 cases are reported in 2012).
Reported Clinical Cases and Efficacy of PSS.a
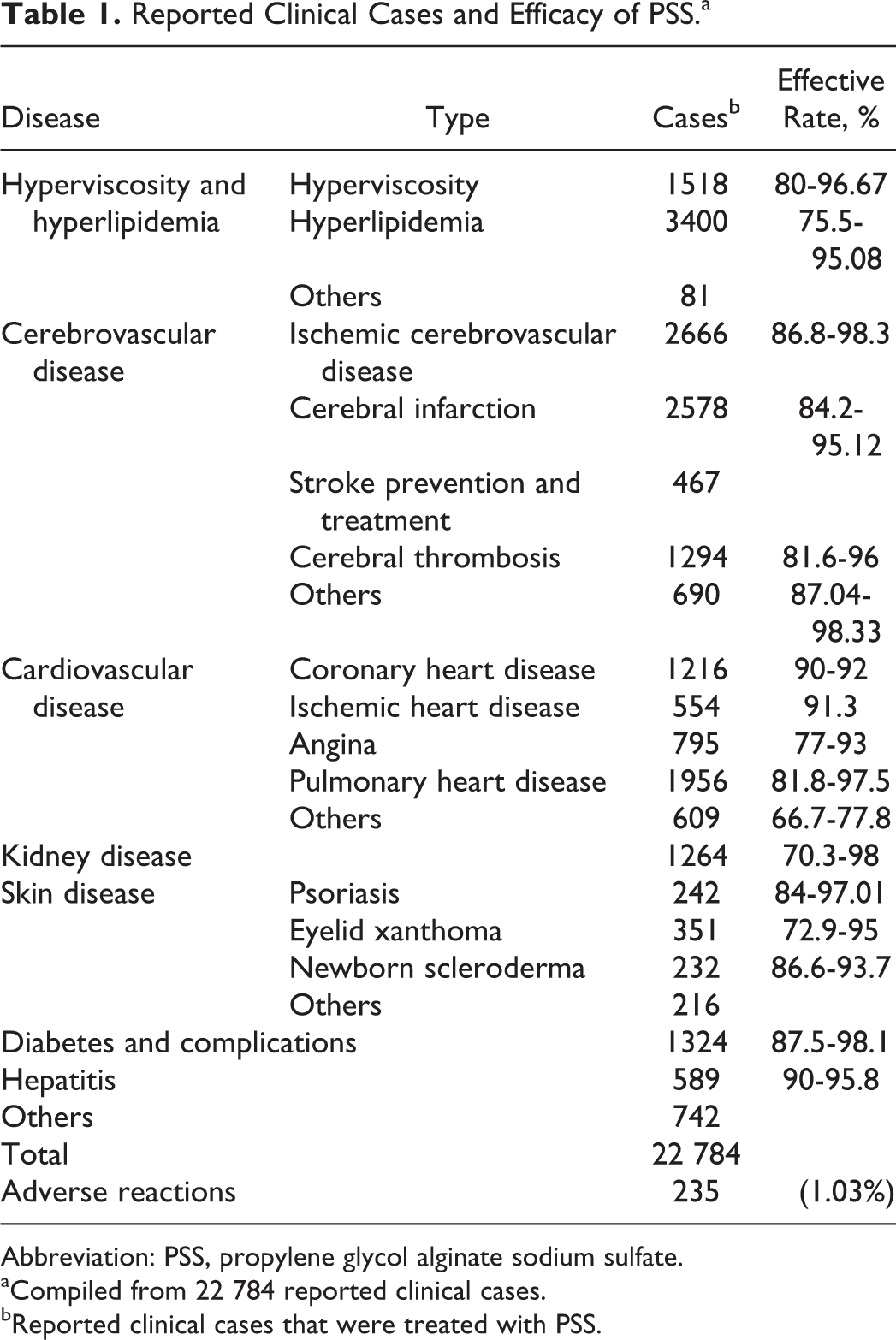
Abbreviation: PSS, propylene glycol alginate sodium sulfate.
aCompiled from 22 784 reported clinical cases. bReported clinical cases that were treated with PSS.
Propylene glycol alginate sodium sulfate Used in Combination Therapy.

Adverse Reactions
In the reported 22 784 PSS treatment cases, there are 235 cases of reported adverse events (1.03% in all reported cases as shown in Figure 2). The adverse events are described in 92 peer-reviewed publications. Strikingly, most reported cases (228 of 235 cases) of adverse reactions are associated with intravenous infusion. Only 7 cases are associated with oral administration.
The adverse reactions (Figure 4) include edema, leukopenia, allergies, bleeding, alopecia, anaphylactic shock, hypotension, hepatic dysfunction, muscle pain, abnormal penis erectile, atrioventricular block, and others. To avoid adverse reactions, intravenous infusion of PSS follows almost identical instruction recommended for heparin. To infuse, PSS is diluted with 500 to 1000 mL of saline ± glucose and then infused slowly once per day, 10 to 14 days per course. Patients are not recommended to use PSS if they are allergic to PSS or have thrombocytopenia, bleeding/clotting disorders, such as hemophilia, cerebral hemorrhage, severe liver, and kidney dysfunctions. Propylene glycol alginate sodium sulfate cannot be injected either intravenously or intramuscularly. Propylene glycol alginate sodium sulfate is not recommended to be coinfused with amikacin sulfate, clindamycin, lomefloxacin, and ciprofloxacin to avoid complications.
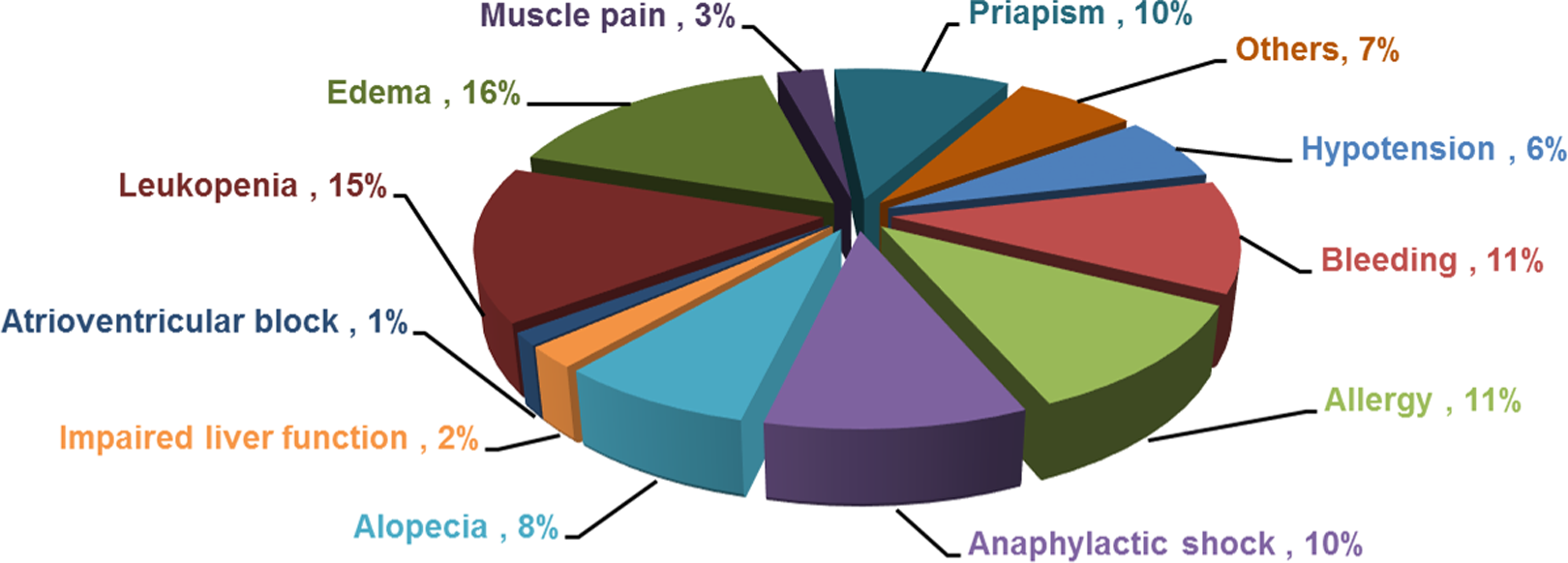
Reported adverse reactions of PSS. In the reported 22 784 propylene glycol alginate sodium sulfate (PSS) treatment cases, there are 235 cases of reported adverse events (1.03% in all reported cases as shown in Figure 2). The adverse events are described in 92 peer-reviewed publications. Strikingly, most reported cases (228 of 235 cases) of adverse reactions are associated with intravenous infusion.
Pharmacodynamics
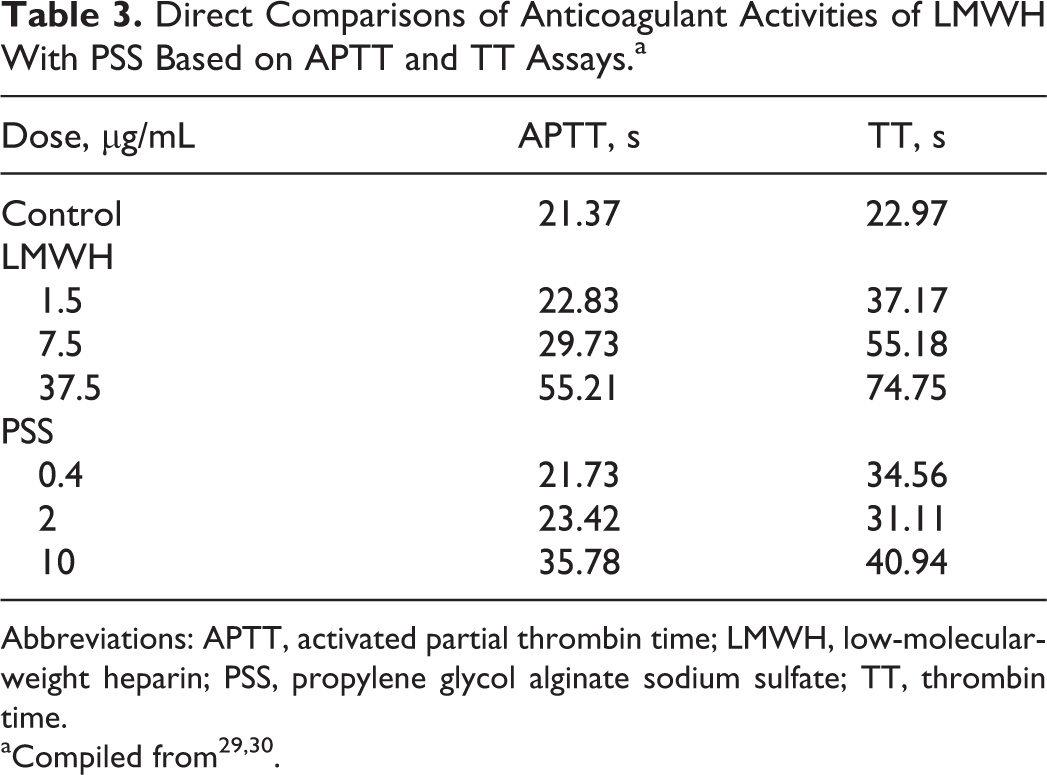
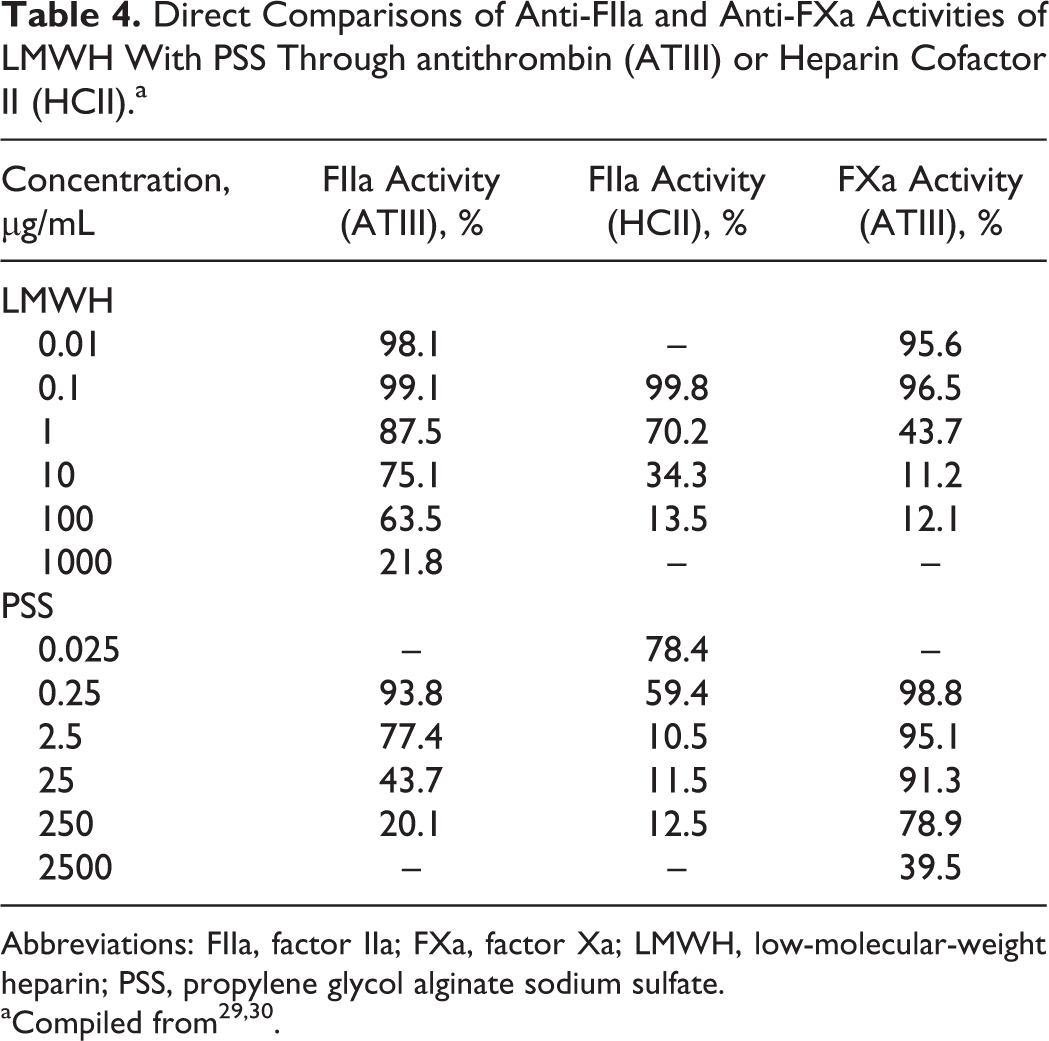
As a heparinoid, the anticoagulant activities of PSS are 1/3 to 1/2 to that of heparin by weight. However, its anticoagulant activities are similar or higher compared to all clinically approved LMWHs. 7 Table 3 shows the anticoagulant activity data of LMWH (Enoxeparin) versus PSS at different concentrations based on activated partial thrombin time (TT) and TT assays, 29,30 which support the idea that PSS has similar anticoagulant activities to that of LMWH based on these 2 assays. To understand the anticoagulant mechanisms, direct comparisons of antifactor IIa (anti-FIIa) and antifactor Xa (anti-FXa) activities of LMWH with PSS through antithrombin (ATIII) or heparin cofactor II (HCII) were also conducted, 29,30 which is summarized in Table 4. Interestingly, PSS has better anti-FIIa activities through HCII than that of LMWH and has similar anti-FIIa activities through ATIII to that of LMWH, but LMWH has better anti-FXa activities through ATIII than that of PSS. Thus, it has been proposed that the pharmacological effects of PSS is mainly achieved by reducing thrombin (FIIa) 29,31 activities. Propylene glycol alginate sodium sulfate also releases von willebrand factor, 32 constrains plasminogen activator inhibitor level, and promotes the activities of both tissue plasminogen activator 33 and thrombomodulin 34 (Figure 5). The antithrombotic effect of PSS is explained by its ability in lowering endothelin 1, platelet activating factor, and P-selectin levels 34 (Figure 5). Recent studies also found that PSS plays a protective role in nerve cells after cerebral ischemia and reperfusion in mice by inhibiting the express of Bax and P53 (Figure 5). 35 –42
Direct Comparisons of Anticoagulant Activities of LMWH With PSS Based on APTT and TT Assays.a
Abbreviations: APTT, activated partial thrombin time; LMWH, low-molecular-weight heparin; PSS, propylene glycol alginate sodium sulfate; TT, thrombin time.
Direct Comparisons of Anti-FIIa and Anti-FXa Activities of LMWH With PSS Through antithrombin (ATIII) or Heparin Cofactor II (HCII).a
Abbreviations: FIIa, factor IIa; FXa, factor Xa; LMWH, low-molecular-weight heparin; PSS, propylene glycol alginate sodium sulfate.
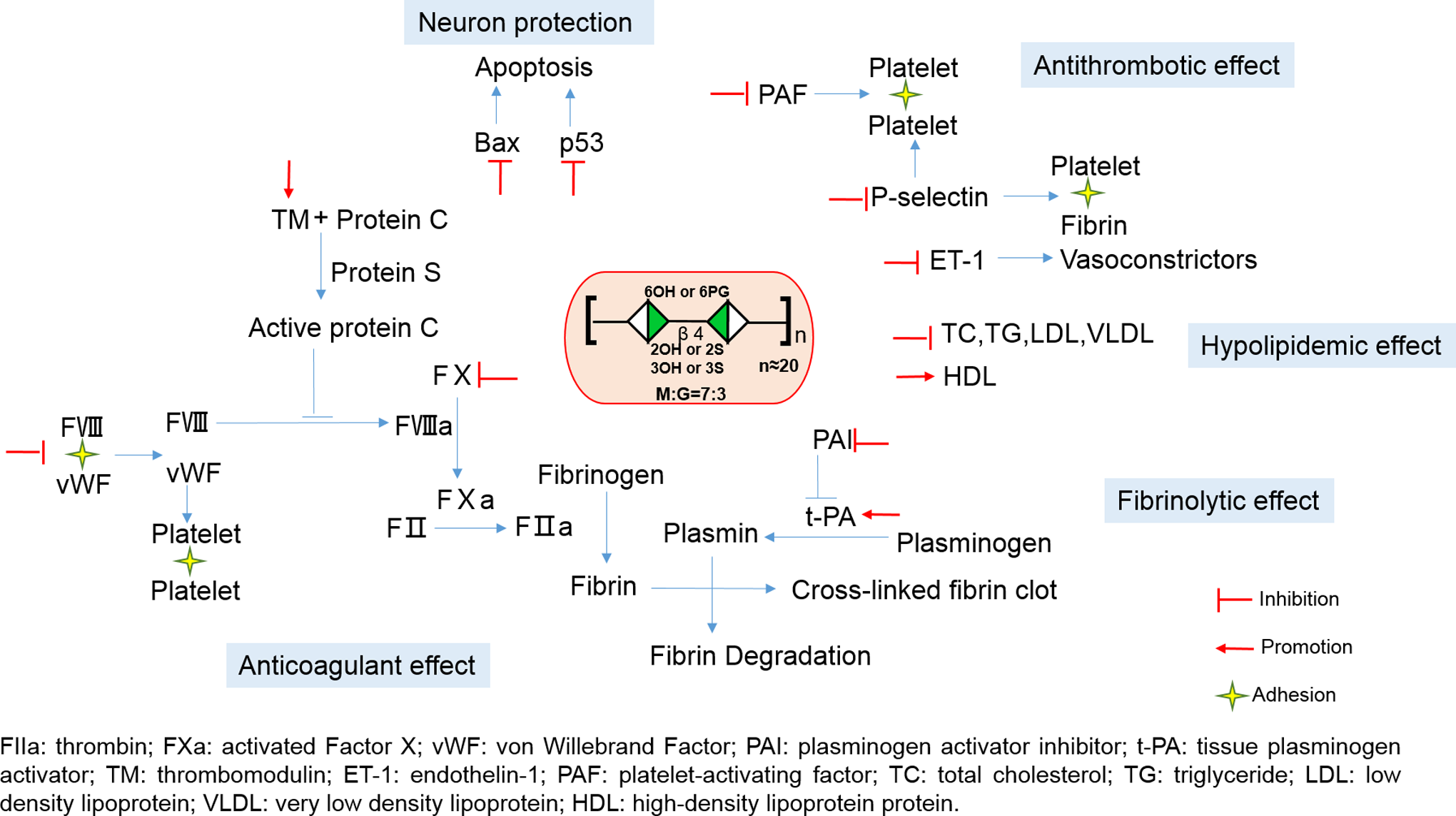
Molecular targets of PSS. The pharmacological effects of PSS is achieved by reducing thrombin (FIIa) and the factor Xa (FXa) activities. Propylene glycol alginate sodium sulfate also releases von willebrand factor (vWF), constrains plasminogen activator inhibitor (PAI) level, and promotes the activities of both tissue plasminogen activator (t-PA) and thrombomodulin (TM). The antithrombotic effect of PSS is explained by its ability in lowering endothelin 1 (ET-1), platelet activating factor (PAF), and P-selectin levels. Recent studies also found that PSS plays a protective role in nerve cells after cerebral ischemia and reperfusion in mice by inhibiting the express of Bax and P53. FIIa indicates antifactor IIa; PSS, propylene glycol alginate sodium sulfate.
In the prescribing booklet for both tablet or injecting PSS, the pharmacodynamics of PSS in treating different diseases is mainly explained by a model summarized in Figure 6. In this model, the polyanions of PSS endorse lipid droplets and cells in blood circulation as well as vascular walls with more negative charges. The increased negative charges prevent the particles and cells from interactions with each other. The deceased interactions caused by the polyanion PSS thus reduce the blood viscosity and hypercoagulability. 43 –45 It further reasoned that the repulsion between lipid droplets and endothelial cells reduces the deposition of cholesterol and triglycerides (TGs) in the blood vessel wall, thereby prompts cholesterol degradation in the liver and its excretion. 46 –49 In supporting this model, several studies showed that PSS has hypolipidemic activities 46 by lowering the level of plasma total cholesterol, TG, low-density lipoprotein, and very low-density lipoprotein and elevating the level of serum high-density lipoprotein protein through inhibiting arterial wall lipid deposition and connective tissue proliferation. 46 –49
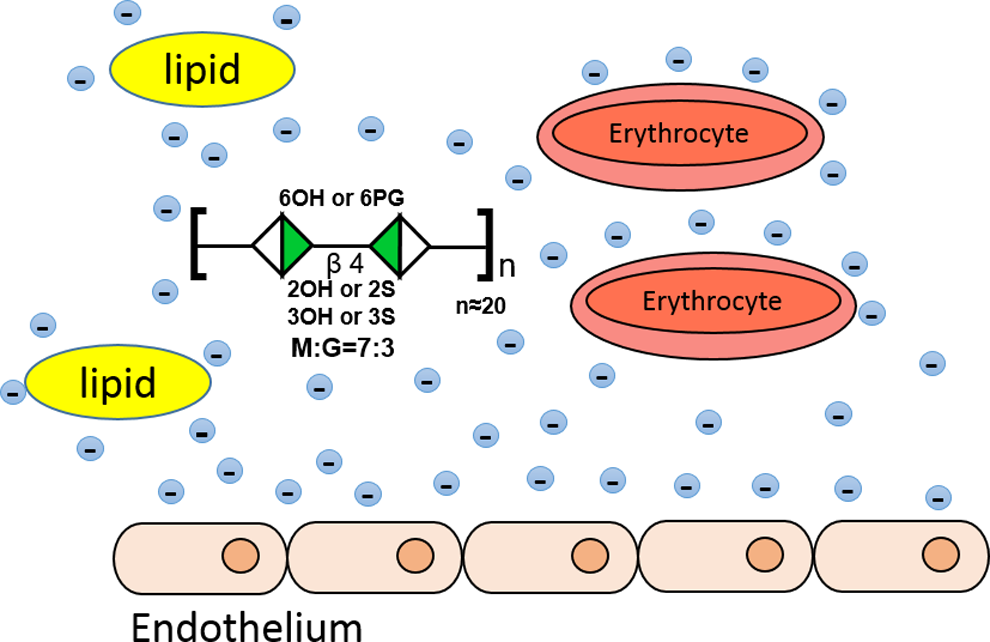
Pharmacodynamic model proposed for PSS in its prescribing guidelines. In this model, the polyanions of PSS endorse lipid droplets and cells in blood circulation as well as vascular walls with more negative charges. The increased negative charges prevent the lipid particles and cells from interactions with each other. The deceased interactions reduce the blood viscosity and hypercoagulability thus make PSS an anticoagulant. PSS indicates propylene glycol alginate sodium sulfate.
Pharmacokinetics
Like heparin, PSS has no chromogenic groups, which makes it impossible to follow its pharmacokinetics directly without tagging with radioactive or fluorescent groups. In addition, PSS interacts with many plasma proteins and interferes blood coagulation by multiple mechanisms as described earlier. Therefore, indirect anticoagulation assays used for pharmacokinetic studies for heparin, LMWHs, and other anticoagulants have never been reported for PSS, either. In 1992, Liu et al 50 studied pharmacokinetics of PSS in mice and rats by administrating different doses of radioisotope [3H]-labeled PSS either orally or intravenously. This study showed that most of [3H] PSS are distributed in the liver and kidney with lesser amounts found in brain, fat, and bone 12 hours after intravenous administration. In mice, 60% of [3H]-labeled PSS is excreted in urinary and 20% of [3H]-labeled PSS is excreted in feces 72 hours after intravenous injection. It was also reported that oral suspension of [3H]-labeled PSS in 0.8% carboxy methylated cellulose is absorbed and peaked in blood in less than 1 hour and up to 76% [3H]-labeled PSS is detected in the blood circulation and plasma radioactivity declined with mean half-lives of 30 hours.
Future Perspectives
Propylene glycol alginate sodium sulfate has multiple protein targets and multiple ways to work as an anticoagulant in vivo (Tables 3 and 4, Figures 4 and 5). Since multiple targets or system approach in treating diseases is a well-accepted concept in traditional Chinese medicine, the polydispersity and polymolecular targets of PSS did not prevent it from getting SFDA approval in 1980s. However, there are multiple issues need to be addressed before PSS, and other glycan-based drugs are accepted by governments and clinicians worldwide, such as how to understand the pharmacodynamics of glycan-based drugs at molecular level, how to control the quality of the highly dispersed drugs, how to perform reliable pharmacokinetic studies, and how to predict and prevent adverse events associated with such drugs, and so on.
Polysaccharides or glycans are as essential for life as DNAs, RNAs, proteins, lipids, and metabolites of the biological system. Up to date, over 20 different kinds of heparin- and other glycan-based drugs have proceeded through clinical trials and are used world wide clinically. The impressive clinical efficacy of PSS and unreplaceable status of heparin indicate the glycan-based drugs are largely untapped natural resources of modern medicines and may in many cases supersede more traditional approaches to rational drug design. Understanding structure, biological activities, and pharmacology of glycans in microorganisms, plants, and animals from both land and ocean would allow rapid developing glycan-based drugs.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Natural Science Foundation of China (Grant no. 91129706).
