Abstract
Both plasma- and recombinant activated factor VII (rFVIIa)-based algorithms can be used to correct coagulopathy in preliver transplant patients with acute liver failure requiring intracranial pressure monitor (ICPM) placement. A decision model was created to compare the cost-effectiveness of these methods. A 70-kg patient could receive either 1 round of plasma followed by coagulation testing or 2 units of plasma and 40 μg/kg rFVIIa. Intracranial pressure monitor is placed without coagulation testing after rFVIIa administration. In the plasma algorithm, the probability of ICPM placement was estimated based on expected international normalized ratio (INR) after plasma administration. Risks of rFVIIa thrombosis and transfusion reactions were also included. The model was run for patients with INRs ranging from 2 to 6 with concomitant adjustments to model parameters. The model supported the initial use of rFVIIa for ICPM placement as a cost-effective treatment when INR ≥2 (with incremental cost-effectiveness ratio of at most US$7088.02).
Keywords
Introduction
Patients with acute liver failure (ALF) have high mortality, even with improved medical management in today’s critical care settings. 1,2 They could develop hepatic encephalopathy, which can lead to cerebral edema and intracranial hypertension (ICH). 3,4 The exact pathogenesis of cerebral edema and encephalopathy is not well understood; however, it has been suggested that systemic and local inflammation as well as neurotoxins, such as ammonia, play critical roles. 4 –6 The risk of ICH is low in grade I and II hepatic encephalopathy but it increases up to 75% in patients with grade III and IV hepatic encephalopathy. 3 Persistent ICH can lead to brain herniation and ischemic brain injury, which might result in poor neurological function recovery after transplantation. 3,7 Therefore, monitoring the intracranial pressure (ICP) in patients with ALF could be beneficial because it allows early detection and management of an elevated ICP as well as guides the decision to transplant emergently. 7 Although it is unclear at this time whether the outcomes in patients with ALF improved with the use of intracranial pressure monitor (ICPM), the use of ICPM in this setting continues to grow, 3 and currently, placement of ICPM in patients with grade III/IV hepatic encephalopathy is part of the optimal management recommended by most members of the Acute Liver Failure Study Group (ALFSG). 8
Many patients with ALF have severe coagulopathy 9 and acute hemorrhage could be a clinical concern in these patients. 10 In addition, it is widely believed that the presence of coagulopathy increases the risk of procedure-related bleeding. 11 Therefore, most neurosurgeons are hesitant to perform this procedure without correction of the international normalized ratio (INR) to approximately 1.6. 12 Traditionally, plasma has been used to temporarily reverse the coagulopathy prior to procedures. However, it is often unsuccessful, time intensive, and could lead to volume overload, anasarca, and further increase in ICP. 11,13 –15 Recombinant activated factor VII (rFVIIa; NovoSeven, Novo Nordisk, Bagsvaerd, Denmark) is approved in the United States to treat bleeding episodes in patients with hemophilia A or B having inhibitors to factor VIII or factor IX, in patients with acquired hemophilia, and in patients with congenital factor VII deficiency. 16 Recently, it has been used off label in 2 small case series to correct coagulopathy in patients with ALF allowing them to undergo ICPM placement successfully without thrombotic or hemorrhagic complications. 11,17 However, there is very limited data regarding when plasma should be attempted first or when rFVIIa should be given upfront for ICPM placement in pretransplant patients with ALF. Thus, the goal of this study is to develop a decision analytic model to examine the cost-effectiveness of using plasma versus rFVIIa in pretransplant patients with ALF and varying coagulopathy who need ICPM placement.
Materials and Methods
Study Design
A decision analytic model was designed to model the probability that a pretransplant patient with coagulopathy having ALF would be able to undergo an ICPM placement at the New York-Presbyterian Hospital—Columbia University Medical Center (CUMC). The costs, time for various transfusions and procedures, and the probabilities estimated for various decision points in the model were derived from the medical literature and theoretical calculations as well as our institutional experiences.
Decision model
A decision analytic framework was designed using TreeAge Pro 2012 (TreeAge software, Williamstown, Massachusetts) to examine the effectiveness of using plasma versus rFVIIa to correct coagulopathy allowing a pretransplant patient with ALF to undergo ICPM placement (Figure 1). In this model, a 70-kg patient with coagulopathy with estimated blood volume of 5000 mL and plasma volume of 3000 mL and with no other medical problems (including cardiovascular diseases or history of thrombosis) other than ALF entered the model. The patient was assumed to have a fibrinogen level ≥100 mg/dL and platelet count ≥100 × 109/L (or they are transfused with either cryoprecipitate and/or platelets to meet the thresholds prior to entering the model) so that coagulopathy was the only limiting hemostatic factor to ICPM placement. This patient could receive either 1 round of plasma (6 units, ∼20 mL plasma/kg—the traditional algorithm) or 2 units of plasma plus 3000 μg rFVIIa (∼40 μg/kg of rFVIIa—the alternative algorithm). If only plasma was administered to the patient, then posttransfusion prothrombin time (PT) and INR were required to decide the next step of management (ie, undergoing the procedure vs requesting a second round of plasma vs administering rFVIIa). If the patient received rFVIIa, then the ICPM procedure would take place immediately after administering rFVIIa without checking the PT/INR. This sequence of events was based on the algorithms applied in 2 published case series 11,17 as well as the recommendation by the ALFSG. 8 Shami et al 11 demonstrated that at this dose of rFVIIa, all patients normalized their PTs/INRs, allowing ICPM placement. Furthermore, Le et al 17 showed that with an average dose of rFVIIa, all patients were able to undergo the procedure without the need for repeat coagulation testing and none of these patients had postprocedure hemorrhagic complications. Detailed algorithm is provided in the supplemental material.
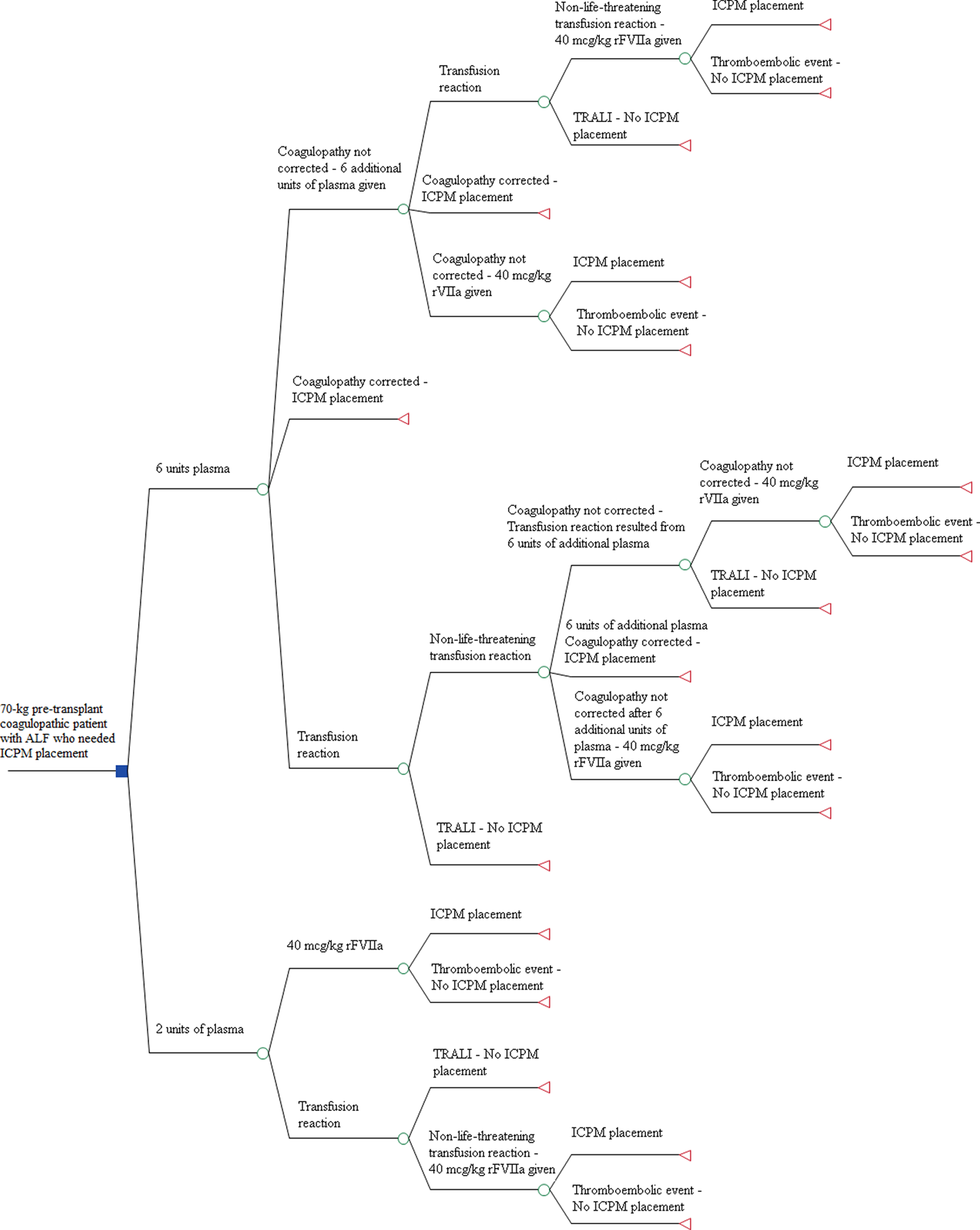
Decision algorithm.
The model was run for a patient with an initial INR (INRinitial) of 2, 3, 4, 5, and 6. The outcomes modeled were cumulative cost and time-to-ICPM placement. The effectiveness outcome was defined to reflect the probability of a surgeon inserting ICP monitor in the shortest time. Finally, the cost-effectiveness outcome, which was represented by the incremental cost-effectiveness ratio (ICER), was investigated for the model.
Data for Decision Tree Analyses
Time-to-procedure estimates
In this model, all times were based on ideal time derived from standard operating procedures (SOP) at CUMC with assumption of no delay regarding to testing and transfusion. Neurosurgeon preparation time was not assessed or included; we assumed that the ICPM procedure would occur immediately after transfusion and/or coagulation test completion. We estimated that it would take 1 hour to thaw and prepare 1 round of plasma for transfusion. At CUMC, plasma cannot be transfused at a rate >2 units/h; thus, it would take approximately 3 hours to transfuse 1 round of plasma in the traditional algorithm and 1 hour to finish plasma transfusion in the alternative algorithm. We did not take into account the nurse’s time for preparation and checking plasma units prior to transfusion in this model. If a nonlife-threatening reaction in plasma transfusion occurred, then the average workup involving clerical check, posttransfusion sample analysis, and transfusion medicine service consultation would take about 1 hour before further blood products could be issued in an ideal situation. The fastest time a PT/INR result could be released from the laboratory from collection to result is 30 minutes based on our assay methodology (Stago’s STA-R Evolution [Diagnostica Stago, Parsippany, New Jersey]) and our SOP (Table 1, panel a).
Model Parameter Estimates.
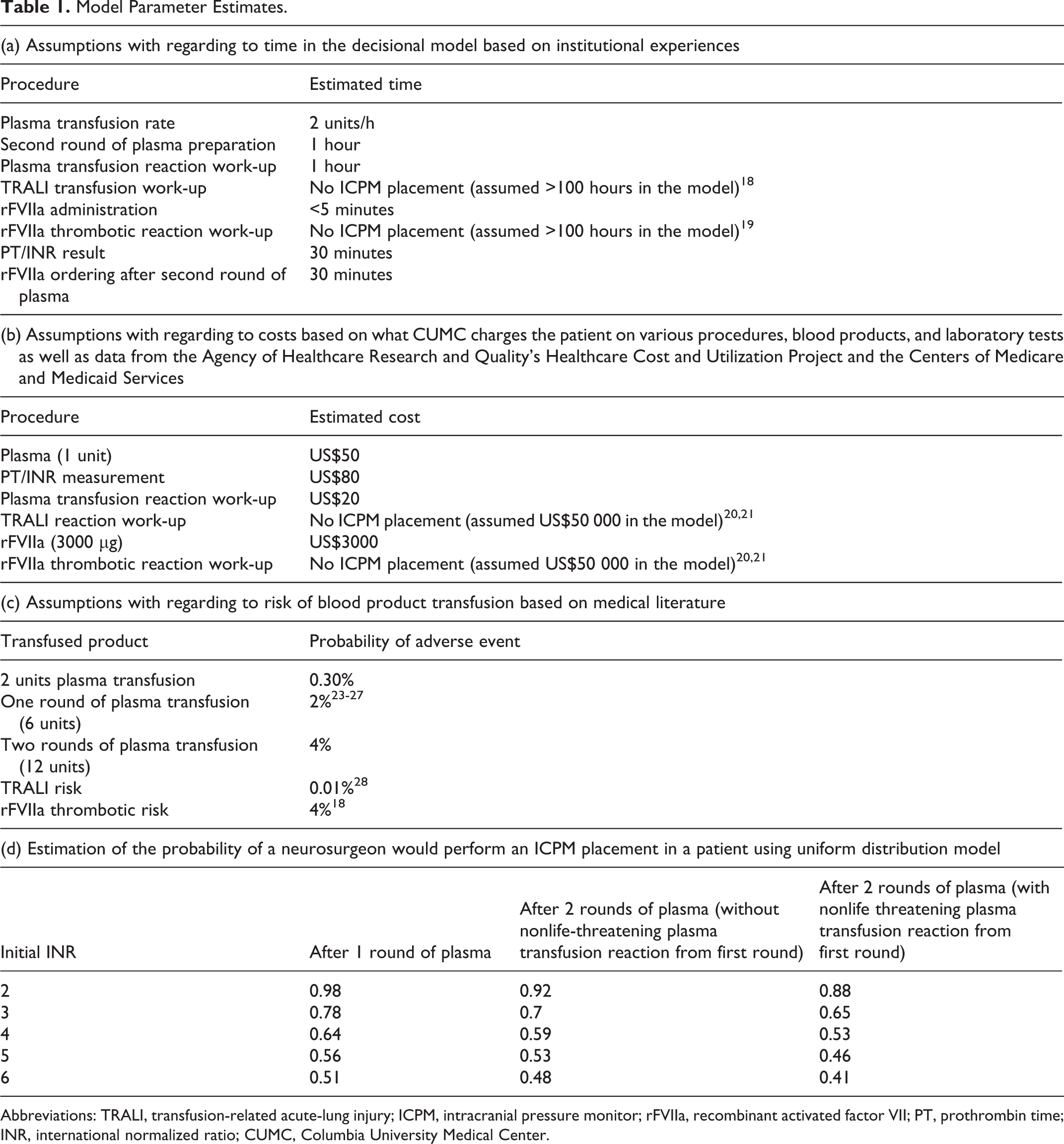
Abbreviations: TRALI, transfusion-related acute-lung injury; ICPM, intracranial pressure monitor; rFVIIa, recombinant activated factor VII; PT, prothrombin time; INR, international normalized ratio; CUMC, Columbia University Medical Center.
Regarding rFVIIa transfusion, transfusion medicine residents and/or attending prospectively audit all the rFVIIa requests at CUMC. As a result, it takes approximately 30 minutes from order to release of rFVIIa. We assumed that administration of this medication by intravenous bolus would take a negligible amount of time (less than 5 minutes). We also assumed that all rFVIIa reactions would be thrombotic with a complex workup that would prevent the patient from undergoing the procedure. Recombinant activated factor VII can cause both arterial and venous thrombosis and many can be clinically significant. 19 Thus, in the model, we used 100 hours as an approximation of the time to complete the thrombotic workup secondary to rFVIIa. The impact of this estimate on the model was tested in the sensitivity analysis (with range tested from 1 to 400 hours to cover the wide range of thrombotic event, from clinically nonsignificant thrombosis to an overt stroke or myocardial infarction). A similar approach was taken to model life-threatening transfusion reactions (transfusion-related acute-lung injury; TRALI in this model) which ICPM placement would likely not occur after the reaction in a timely manner because for TRALI, 80% of patients will improve within 96 hours but 20% will have protracted clinical course. 18 In this model, we also used 100 hours as an approximation of the time to complete the evaluation of TRALI. We also tested the impact of this estimate on the model in the sensitivity analysis (with range tested from 1-400 hours to cover the wide range of the time requiring for the workup and management of TRALI; Table 1, panel a).
Cost estimates
Costs in the model were approximated from average costs at CUMC charged for each patient for various procedures, blood products, and laboratory tests. Only direct institutional costs were included in the model. Workup and treatment for thrombotic reactions from rFVIIa and life-threatening transfusion reaction (TRALI in this model), as explained previously, were assumed to be extremely complex and very expensive, and these costs were estimated based on data from the Agency of Healthcare Research and Quality’s Healthcare Cost and Utilization Project and the Centers of Medicare and Medicaid Services (CMS). 20,21 Furthermore, the management of thrombotic reaction from rFVIIa and TRALI is complicated and may likely involve multiple procedures, and thus, the exact cost is difficult to discern. In the model, we used US$50 000 (an approximation of the cost for managing a myocardial infarction) as an estimation of the cost to complete these workups and management of rFVIIa thrombotic reaction. Similar estimation was performed for TRALI reaction. The impact of these estimates for the cost of working up rFVIIa thrombotic reaction and TRALI reaction on the model was tested in the sensitivity analysis (with range from US$1-US$1 000 000 to cover the wide possibilities of clinical presentation, evaluation, and impact of these reactions as discussed previously; Table 1, panel b).
Adverse reaction risk estimates
Blood transfusion is not without risks, and statistically, the greatest risk to the patient lies in either the bacterial contamination of the transfused unit or noninfectious complication from transfusion. 22 Bacterial contamination is uncommon in plasma transfusion due to the manufacturing and storing process used. Thus, adverse plasma transfusion reactions are more likely to be from noninfectious causes and most likely due to allergic reaction to exogenous plasma protein or transfusion-associated circulatory overload (TACO). 22 For all blood products, the incidence for allergic reaction is 1% to 3%, 22 and it has been estimated that the incidence for TACO is 1% to 8%. 23 –27 Using the incidence of 1 reaction for every 360 plasma transfusion, including TACO, 26 a risk of 2% for each dose of plasma transfusion (6 units) was assigned to the patient in this model. Because the reactions were assumed to be nonlife threatening, the patient could receive further blood transfusion therapy and undergo the ICPM placement after workup. In addition, in order to simplify the model, we assumed that the reaction would occur at the end of each round of plasma transfusion. In the model, TRALI was used to represent the life-threatening risk of transfusion due to the complex management of the patient with this kind of transfusion reaction. Toy et al 28 estimated the incidence of TRALI to be decreased from 1:4000 to 1:12000 plasma units transfused after using only plasma from male donors and/or never pregnant female donors. In this model, we assigned the probability of TRALI after plasma transfusion to be 0.01% because only male-derived plasma is used for transfusion at CUMC (Table 1, panel c).
Regarding the thrombotic risk of rFVIIa, there have been reports of thromboembolic complications from off-label usage of rFVIIa 16,19,29 although in this specific group of patients with ALF, studies by Le et al 17 and Shami et al 11 as well as our 8-year institutional experience did not find any thromboembolic events associated with rFVIIa. 30 In this model, because our patient is young (between 18 and 64 years old) and has no previous history of cardiovascular disease, 4 we assumed a thrombotic risk of 4% (Table 1, panel c). 19
Probability estimates
Assuming that there was no significant change in the synthesis or destruction of clotting factors in the patient other than the transfusion and that there was no third-space shifting, estimates of INR post-1 and post-2 doses of plasma at varying initial INRs were calculated based on estimates of coagulation factor activity after transfusion (accounting for transfusion volume and time for plasma preparation, transfusion, and reaction workup if applicable). We also compared our estimates with published data in the medical literature. Additionally, the willingness of the neurosurgeon to place the ICPM after transfusion was modeled using a uniform probability distribution that incorporated the patient’s initial INR and his or her predicted posttransfusion INR (Table 1, panel d). Detailed mathematical calculations can be found in the appended supplemental material.
Sensitivity Analysis
A sensitivity analysis was performed to assess the robustness of our model to select assumptions using TreeAge Pro 2012. We chose to limit the main sensitivity analysis to only the cost-effectiveness outcome of ICPM placement.
Results
Decision Analysis Model
The model was analyzed for a hypothetical pretransplant patient with INRinitial of 2, 3, 4, 5, and 6, each in ALF and requiring ICPM placement using the assumptions described previously regarding time, cost, risk of reaction to various blood products and rFVIIa, and the estimated probability that a neurosurgeon would place the ICPM. When cost was the primary outcome, plasma was less expensive than rFVIIa regardless of INRinitial. However, when only time-to-ICPM-placement was considered, plasma was superior to rFVIIa only if the INRinitial was ≤3. Furthermore, rFVIIa achieved better effectiveness outcome (defining as the ratio of probability of neurosurgeon inserting ICP monitor to time-to-procedure) when the INRinitial was ≥2. When the cost-effectiveness outcome was considered, using the conventional ICER cutoff of US$50 000 as being a cost-effective intervention, the model supported the initial use of rFVIIa for successful placement of ICPM when INR ≥2 (with ICER of at most US$7088.02). In addition, the use of rFVIIa upfront is more cost-effective as INRinitial increased (Table 2).
Cost-Effectiveness Analysis for Traditional (Plasma Arm) Versus Alternative Algorithm (rFVIIa Arm).
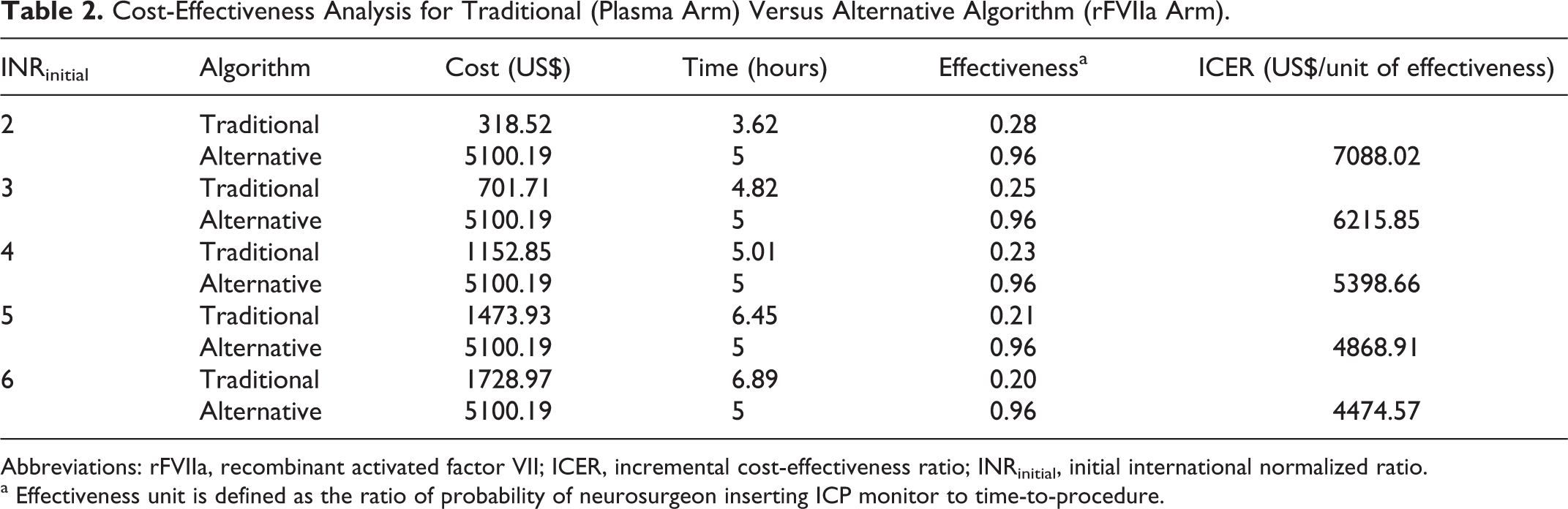
Abbreviations: rFVIIa, recombinant activated factor VII; ICER, incremental cost-effectiveness ratio; INRinitial, initial international normalized ratio.
a Effectiveness unit is defined as the ratio of probability of neurosurgeon inserting ICP monitor to time-to-procedure.
Sensitivity Analysis
Sensitivity analysis was performed for the cost-effectiveness outcome of ICPM placement to assess the robustness of the model (Table 3). We decided to perform sensitivity analyses only at INRinitial = 2 because it is the model with the highest ICER. Therefore, if the model with the least cost-effectiveness (ie, highest ICER) is robust, then other models with parameters yielding higher cost-effectiveness (ie, had lower ICER) will be robust as well. Results from sensitivity analyses showed that the model was robust on all estimated parameters except for the cost of the rFVIIa thrombotic workup. Using the conventional ICER cutoff of US$50 000 as being a cost-effective intervention, the alternative algorithm (rFVIIa arm) is more cost-effectiveness than the traditional algorithm (plasma arm) when the cost of working up an rFVIIa thrombotic event is less than US$750 000.
Sensitivity Analysis for Cost-Effectiveness Outcome. All Sensitivity Analyses Were Performed Using the Parameters From the Model With INRinitial = 2.
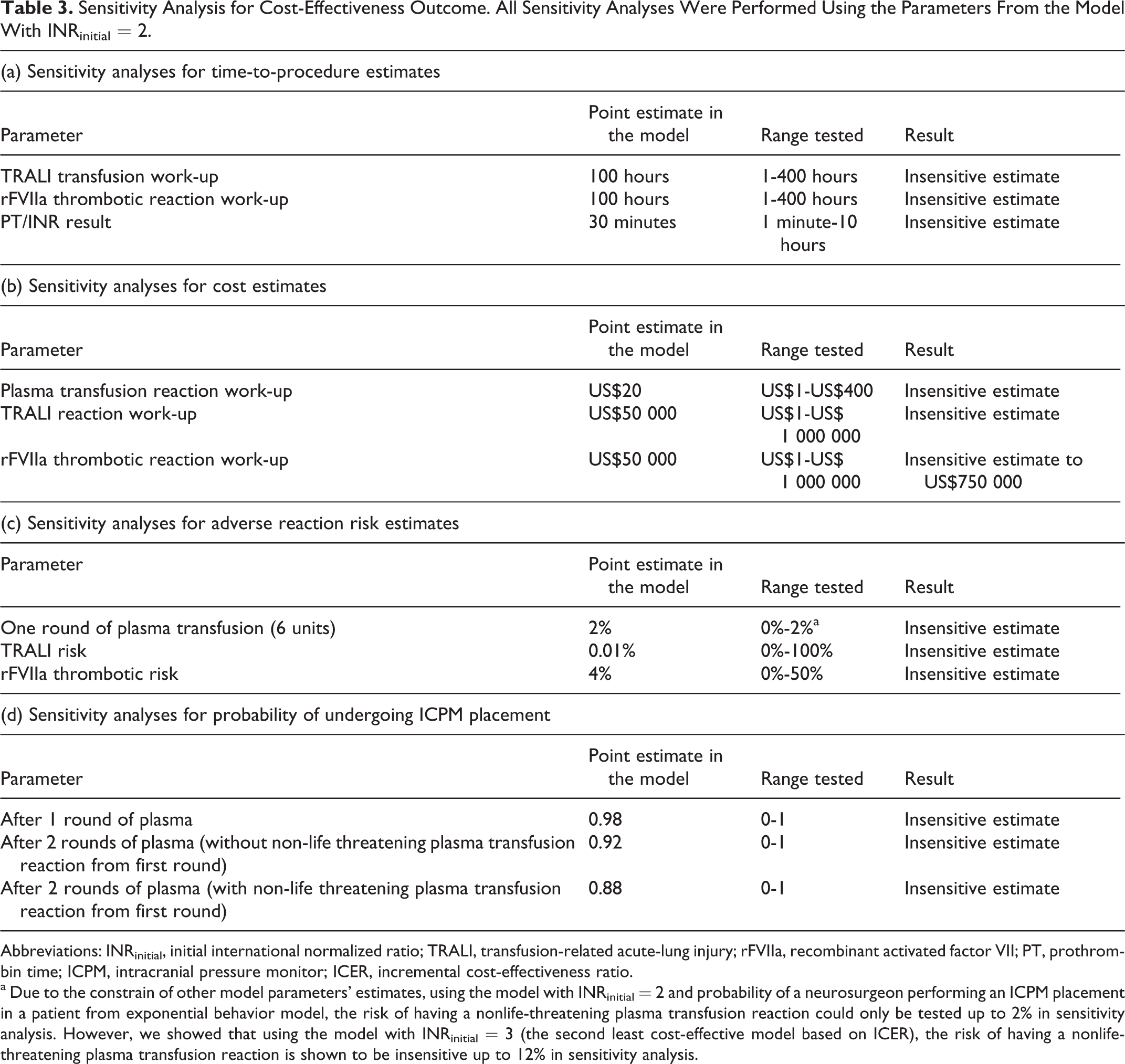
Abbreviations: INRinitial, initial international normalized ratio; TRALI, transfusion-related acute-lung injury; rFVIIa, recombinant activated factor VII; PT, prothrombin time; ICPM, intracranial pressure monitor; ICER, incremental cost-effectiveness ratio.
a Due to the constrain of other model parameters’ estimates, using the model with INRinitial = 2 and probability of a neurosurgeon performing an ICPM placement in a patient from exponential behavior model, the risk of having a nonlife-threatening plasma transfusion reaction could only be tested up to 2% in sensitivity analysis. However, we showed that using the model with INRinitial = 3 (the second least cost-effective model based on ICER), the risk of having a nonlife-threatening plasma transfusion reaction is shown to be insensitive up to 12% in sensitivity analysis.
Discussion
We have developed a cost-effectiveness decision analytical model to rigorously predict the ideal management of pretransplant patients with ALF in need of ICPM placement, based on presenting INR. In today’s health care environment, such models taking into account cost, outcomes, and time to outcome will be increasingly important in medical decision making. Our model used calculated, institutional and literature-based assumptions and we demonstrated the utility of such modeling approaches to complex medical decision making.
Sensitivity analysis for the cost-effective outcome showed that our model is robust to dramatic variation in most estimates except for the cost of the rFVIIa thrombotic workup. In this model, we assumed that the cost of evaluation of rFVIIa thrombotic reaction was US$50 000 and we tested this estimate in the sensitivity analysis (with range from US$1-US$1 000 000). If the cost of rFVIIa thrombotic workup is >US$750 000, then the ICER for our model would exceed US$50 000, and thus, the alternative algorithm is not cost-effectiveness. However, this threshold for this estimate is realistic unlikely, which allows the results of our cost-effectiveness analysis to be valid.
There are some limitations to using theoretical modeling approaches, including this one, in medical decision making. First, although many model parameters were derived from medical literature and institutional experiences and were shown to be robust in the sensitivity analyses, a few of these probabilities were assigned based on theoretical calculations, such as the probability of a neurosurgeon performing the ICPM placement procedure at varying INRs. Second, models assumed ideal clinical scenarios with the optimal turnabout time and resource availability. Indeed, in this model, we assumed no delay in product availability for transfusion, coagulation testing, and surgeon availability to perform the procedure. In an actual clinical setting, delays in obtaining laboratory results, neurosurgeon schedules, and the ability of patients to handle rapid transfusion would introduce additional delays that were not included in this model. This would result in longer time between transfusion and ICPM placement, especially at higher INRinitial, resulting in the alternative algorithm being more cost-effective than predicted in the model. Third, models often simplify the patient’s clinical picture in order to allow for clearer modeling. In our model, because the majority of patients with ALF are previously healthy adults in theirs 30s, 4 we assumed that other than coagulopathy secondary to underlying liver disease, this hypothetical patient did not have any other comorbidities. In actual practice, the patient could and would likely have other medical conditions, such as cardiovascular and renal diseases. These medical conditions would affect the choice of algorithm (plasma or rFVIIa) as they might alter the thrombotic probability of rFVIIa and/or the ability of the patient to handle large-volume plasma transfusion, and thus, medical judgment should be made in these situations regarding what product is best for the patients. In a similar simplifying vein, in the model, we assumed that coagulopathy was the only limiting factor that predicted successful ICPM placement. We also did not take into account the skill or seniority of the neurosurgeon, a critical factor for the success of the procedure as well as the possibility of the INR threshold difference to perform the procedure between a senior neurosurgeon and a junior surgeon.
Finally, in the model, we assumed that in vitro correction of coagulation abnormalities is equal to in vivo hemostasis, which is the assumption made by many neurosurgeons currently. Furthermore, it has been suggested that routine coagulation testing, such as PT/INR, does not reflect the bleeding risk in a patient with liver disease because both prohemostatic and antihemostatic factors are decreased, and routine tests only measure prohemostatic factors. 31 This reasoning implies that mild elevations of PT/INRinitial might not necessarily have to be corrected in this patient group prior to undergoing procedures. 31 Despite the possibility of this logical theory, in this model, we assumed that a neurosurgeon is less likely to operate with an INRinitial > 1.6, which is supported by many neurosurgery textbooks 12 and in-line with our institutional experience regarding the current clinical practice of neurosurgeon in this population.
In conclusion, we have developed a theoretical model based on calculation, literature, and institutional experience to rigorously aid in medical decision making. In our model, we predict that rFVIIa should be considered upfront in correcting the coagulopathy in pretransplant patients who have ALF with an INRinitial ≥2. This model can be used to inform clinical studies to refine and validate our model parameters. For example, a validated questionnaire could be administered to neurosurgeons in order to better characterize the probability of performing the procedure at varying INRs. Most importantly, similar decision analytic models with rigorous sensitivity analyses should be developed and applied to other difficult clinical decisions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
