Abstract
Thromboelastography (TEG) is drawing more attention for clinical and laboratory studies of hemostasis. It has been applied to evaluate the effects of both psychological and physiological stress on whole blood coagulation from the onset of the coagulation cascade through clot formation, to the end with fibrinolysis. We conducted a comprehensive review on the applications of TEG for assessment of different stressors, ranging from physical exercise to emotional situations. The methodology is unique in terms of instrumentation, the methods to activate blood coagulation, the type of blood (citrated vs fresh blood), and study settings (in vitro vs in vivo vs clinical trials). Thromboelastography has most often been used to study the effects of physiological stress. The author’s own work and future directions are discussed as well. The review would facilitate future development of TEG for evaluating hemostasis and potential pathological pathways in response to various forms of stress.
Introduction
Physical and psychological stressors exert a broad range of biological effects on the human body. Some effects include cardiovascular changes and diseases 1 and induced neuroimmunological functions. 2 Moreover, stress is widely recognized as an important risk factor in the etiology of cardiovascular and immune-related diseases. 3 Importantly, stress has been shown to affect blood coagulation and fibrinolysis and to alter inflammatory responses. 4 For example, acute physical exertion generally increases platelet number and activity and activates coagulation and fibrinolytic processes. 5 Although acute exercise may change the hemostatic milieu in favor of increased coagulation, 6 its long-term effects on coagulation and fibrinolysis lead to a reduced risk of stress-related disease processes. 7 Hence, it is critical to understand and control the effects of psychophysiological stress on blood clotting, fibrinolysis, and thrombolysis.
Although well recognized, the true meaning of stress is often difficult to describe and pinpoint. Selye, who began his pioneering work in the early 20th century, defined stress as the “nonspecific response of the body to any demand” and a stressor is an event that threatens or is perceived to threaten homeostasis. 8 In general, any external factor affecting our normal bodily function can be considered as a stressor. Stressors can range from life-threatening acute injuries to chronic physical effects or psychological/mental disorders. 9 In the context of a military environment, the stressor is often termed as “operational stressors” and can be both physiological and psychological in nature. This review describes those reports without traumatic injuries and focuses on human studies unless human data are unavailable and in vitro or animal studies are deemed to be highly relevant.
Although a number of in vitro tests have been reported for assessing hemostasis under stress, these tests are mainly concerned with isolated components of the coagulation and fibrinolysis system, such as thrombus formation and platelet function, and thus cannot show overall effects and take into account the interactions of the clotting cascade and platelets in whole blood. It is, therefore, difficult to relate isolated findings from such tests to the overall effects in whole blood systems, as occurs in clinical settings.
Thromboelastography (TEG), invented by Hartet in 1948, 10 provides a “global” picture of hemostasis by quantitatively measuring the viscoelastic changes in whole blood during clotting, from the beginning of coagulation to the end with fibrinolysis. This includes the onset of clot formation, its progress, and maximum clot strength and clot stability, which provides important information about coagulation, fibrinolysis, and platelet function. 11 –13 It offers additional information compared to standard laboratory tests (eg, prothrombin time [PT], activated partial thromboplastin time [aPTT], and thrombin clotting time). 14 Thromboelastography may also identify the relative contributions of clotting factors, such as fibrinogen and platelets, to the overall coagulation process. Specific evaluation of a particular coagulation process can also be illustrated by modified TEG methods using partial blood components, for example, plasma or coagulation factor-deficient blood.
Thromboelastography has been widely used in clinical settings for monitoring blood coagulation system during surgical operation to determine the abnormal blood clotting patterns of patients and the requirement for the administration of life-saving hemostatic agents and blood products to correct defects in hemostasis. 15 In addition to clinical utilization, TEG has found applications in the studies of the coagulation effects of pharmacological agents, 16 vasoactive agents, 17 coagulation factors, 18,19 and resuscitation fluids. 20 However, its use as a research tool for evaluating effects of psychophysiological stress and for screening thrombogenicity risks has not been widely reported. Hence, a review was conducted to enable further development and application of the TEG technology for studying the effects of psychophysiological stress.
This article is structured into 3 major sections to review current usage of TEG for studying the effects of stress in various forms (eg, physical, psychological, emotional, fear) on blood coagulation and fibrinolysis. The first section discusses the existing TEG methodologies and techniques. The second section describes the use of TEG in studies using a variety of stressors. The topic is discussed according to the type and form of stressor, for example, physiological and psychological stressors, such as exercise, temperature extremes, hypobaric or hyperbaric conditions, mental, and emotional stress. These stresses possess both common and distinct biochemical mechanisms for alteration of the hemostatic system. For each type of stressor (eg, exercise), we will describe the typical physiological changes affecting coagulation and fibrinolysis under each stressor, followed by experimental findings and proposed mechanisms for the changes. The hemostatic effects of physical injuries, disease states, and other relevant factors are discussed briefly. The review also describes our efforts to applying the TEG method for evaluation of hyperbaric, physical exertion, and heat stress. The last section is devoted to the discussion of current limitations and possible future development of TEG.
Methodology
As TEG technology and utilization evolved, 2 types of systems have emerged: one manufactured by Haemoscope Co (Niles, Illinois) and called thromboelastogram and the other represented by Pentapharm GmbH (Munich, Germany) and called rotational thromboelastometry (ROTEM). 21 Both record the viscoelastic changes that occur during the whole coagulation process, but the primary hardware difference is that ROTEM has an immobile cup, wherein the pin/wire transduction system slowly oscillates an arc of 4°45′. In addition, each ROTEM system has 4 channels and a built-in computer to operate as opposed to 2 channels in the TEG system that requires a separate computer to operate. It is argued that the ROTEM system uses a ball-bearing system for power transduction, which makes it less susceptible to movement and vibration. 22
Figure 1A schematically shows how the TEG instrument works. The machine measures the viscoelastic properties of blood as it clots under a low shear. It has 2 channels; for each channel, a pin suspended by a torsion wire is immersed in whole blood or plasma in a plastic cup. The cup oscillates back and forth constantly at a set speed through an arc of 4°45′. The torque of the cup is transmitted to the pin, via the fibrin strands in the blood clots as coagulation proceeds, and to the torsion wire for conversion by a mechanical-electrical transducer to an electrical signal, which can be monitored by a computer. 12 Figure 1B shows the TEG 5000 hemostasis analyzer manufactured by Haemoscope Co used in our laboratory. The measurement is graphically represented as a characteristic shape profile over time (Figure 1C), from which the following parameters can be derived to provide main information about the coagulation and fibrinolysis: (1) reaction time “R” or time to first clot formation, which is related to plasma clotting factors and circulating inhibitory activity; (2) coagulation time (CT) “K” or time to a specific level of clot strength, which is associated with the activity of the intrinsic clotting factors, fibrinogen and platelets; (3) rate of clot polymerization “α angle” or rapidity of fibrin cross-linking, which is a main function of platelets, fibrinogen, and plasma components residing on the platelet surface; (4) maximum amplitude (MA) or maximum clot strength, which is a direct function of the maximum dynamic properties of fibrin and platelet number and function; (5) time to reach MA (TMA); and (6) fibrinolysis at 30 minutes “LY30” or the rate of amplitude reduction 30 minutes after MA, which relates to fibrinolysis.
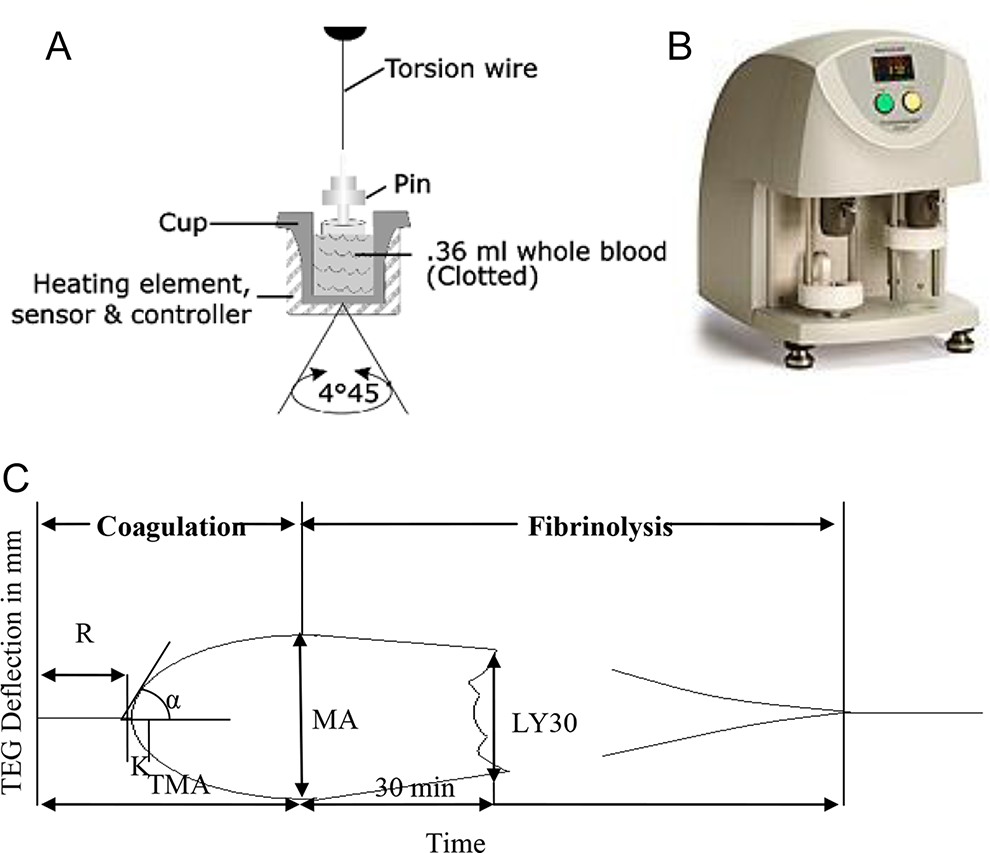
Schematic illustration of (A) the TEG mechanisms, (B) the TEG machine, and (C) a representative TEG tracing showing the relationship between the qualitative tracing and the quantitative parameters. TEG indicates hromboelastography.
Parameters similar to TEG (eg, CT, clot formation time [CFT], α angle, maximum clot firmness [MCF], clot lysis index CLI30) can be derived from ROTEM which are commonly used in Europe. 23 However, a study comparing TEG and ROTEM concluded that the significant differences between the 2 methodologies do not permit their results to be used interchangeably. 24 Both types of instrumentation are covered in the review. Unless specified, TEG is used as a general term for the elastic modulus-based assessment of hemostasis in the review. Thromboelastography refers to the system manufactured by Haemoscope Co, while ROTEM indicates the one manufactured by Pentapharm GmbH.
A PubMed search was completed for the TEG studies involving psychophysiological stress. The specific stress of interest (eg, exercise, heat stress) was also used as key words in the search. Abstracts were used to determine the relevance and when appropriate further review of the original articles was warranted. Additional publications were selected from the cross-references listed in the original papers and from the citing articles, and additional search was made through Medline, Scopus, and Institute of Scientific Information databases for those topics with limited findings from PubMed.
Table 1 summarizes different stress models and methods in thromboelastographic studies. Results are presented and discussed in the following sections. It shows that a variety of stressors, both physiological and psychological, have been studied using various TEG methods. The physiological stressors include acute exercise, 25 –28 extreme temperatures, 29 –36 hyperbaric, 37 and hypobaric conditions. 38,39 The psychological stressors include emotional stress. 40 Thromboelastography was also used in our recent study on multiple stressors.
Methods Used in Thromboelastographic Study of Psychophysiological Stress.

Abbreviations: CT, coagulation time; CFT, clot formation time; MCF, maximum clot firmness; ROTEM, rotational thromboelastometry; MA, maximum amplitude; TEG, thromboelastography.
Depending on the duration of stress (acute vs chronic), TEG was conducted in longitudinal or cross-sectional groups before, during, or after stress. Generally, whole blood samples were collected into citrate-anticoagulated vacuum tubes at baseline and different stress levels. Samples were then aliquoted into a TEG cup and measured immediately upon the addition of calcium chloride solution. The TEG was run to obtain the parameters of interest, typically, R, K, α, and MA in TEG and CT, CFT, α, and MCF in ROTEM. Different procedures have been developed at the discretion of the user/manufacturer as exemplified by whole and partial blood components and various activators to accelerate blood coagulation. Citrated whole blood is mostly used in TEG studies although native and processed blood such as fresh whole blood and plasma-rich blood has also been used. Measurement temperature was mostly set at 37°C but can be adjusted which is useful to study coagulation under hyper- and hypothermia. 41 In addition, modified TEG methods allow the evaluation of specific coagulation functions using the blood treated with different coagulation activators (eg, celite, kaolin, tissue factor, and thrombin) or anticoagulants (eg, heparin). For TEG, tissue factor and kaolin mixed with phospholipids and buffered stabilizer are supplemented with blood in most applications. 42 For ROTEM, the manufacturer recommends citrated blood recalcified with CaCl2 (star-TEM) and activation with tissue thromboplastin for extrinsic pathway (ex-TEM), activation with partial thromboplastin-phospholipid for monitoring the intrinsic pathway (in-TEM), addition of a thrombocyte inhibitor (cytochalasin D) for detection of fibrinogen deficiency and fibrin polymerization disorders (fib-TEM), addition of a fibrinolysis inhibitor (aprotinin) for confirmation of hyperfibrinolysis (ap-TEM), and addition of heparinize I for global analysis of coagulation without the influence of heparin (hep-TEM). There are a few studies where partial coagulation systems were used to understand the interactions with a specific blood component.
Most TEG applications were focused on blood coagulation system with few involving fibrinolytic system. Compared to the TEG methods, which evaluate blood coagulation and fibrinolysis based on a cell-based model for hemostasis, 43,44 there are more recent ROTEM studies involving both intrinsic and extrinsic coagulation pathways.
Physiological Stress
A diverse group of stressors are considered under this category, such as exercise, exposure to extreme temperatures, hypobaric and hyperbaric conditions, and their associated changes in blood coagulation and fibrinolysis have been documented.
Exercise/Physical Exertion
Exercise or strenuous physical activity induces profound physiological perturbations. Exercise can be acute or chronic. The former can be further defined as light, moderate, or heavy activity as based on heart rate and oxygen uptake measurements. 45 Chronic exercise, sometimes referred to as exercise training, has been defined as acute exercise repeated over time (>3 times/week). 46
Among the physiological changes associated with acute and chronic exercise, 46,47 immediate cardiovascular responses and long-term risk of development/progression of cardiovascular disease (CVD) have been well documented. 6,45 Blood hemostasis in exercise and training has been reviewed. 5,48 Several thromboelastographic studies were conducted to assess the effects of different acute exercise conditions on coagulation and fibrinolysis. Blood samples were collected before, during, and after exercise, and TEG was conducted according to different procedures. Comparatively little information is available for the effects of training.
The seminal TEG study examining the effects of acute exercise on blood coagulation dates back over almost 4 decades and demonstrated enhanced coagulation at a high exercise workload (ie, 200 W) and at exhaustion. 25 The procoagulant effects were attributed to the increase in factor VIII via adrenergic stimulation as circulating catecholamine levels were elevated. In addition, fibrinolysis was activated in direct relation to the exercise intensity and release of plasminogen activator from endothelial cells as the results of changes in vascular diameter and activation via epinephrine. On the other hand, recent studies have suggested that the increased concentration of circulating plasminogen activator was mainly due to its decreased hepatic clearance with proportional decreases in liver blood flow during submaximal exercise. 49 This is in agreement with 2 earlier studies that elucidated hypercoagulable changes in the clotting system as indicated by a decrease of 38% in R and 31% in K values, and an increase of 26% in MA immediately after a 400-m sprint 50 and by shortened R time after 6 minutes of heavy exertion (205 W) on a cycle ergometer. 51 Although modest hemoconcentration was evident by a rise of 5.2% in hematocrit, this was not responsible for the observed exercise-induced hypercoagulability. 50 A rapidly emerging area of research suggests that exercise increases circulating leukocyte and platelet-derived procoagulant microparticles, 52,53 which may be at least partly responsible for the hypercoagulability immediately following exercise. However, very few studies have directly compared the impact of acute exercise on microparticle generation and TEG.
In the recent studies, ROTEM has been applied to study physiological stress. In a report by Sumann et al, 26 ROTEM was used together with plasma markers to investigate blood coagulation and fibrinolysis during a downhill marathon run. Rotational thromboelastometry measurements made on citrated whole blood collected 3 hours before the event and within 30 minutes after the marathon showed a shortening of in-TEM clotting time and CFT from 215 to 143 seconds and from 72 to 57 seconds, respectively, and an increase in in-TEM α from 76° to 79° and MCF from 60 to 61.5 mm. The ROTEM results are in agreement with the measurements by standard laboratory coagulation tests (eg, PT, aPTT, thrombin generation assay) and specific parameters for coagulation (eg, platelet count, fibrinogen concentration). 26 Furthermore, molecular plasma markers for fibrinolysis (eg, tissue-type plasminogen activator, plasminogen activator inhibitor) indicated activation of fibrinolysis. However, it would be more informative if the authors had also included fib-TEM and ap-TEM for fibrinogen activity and fibrinolysis in the study.
As opposed to the application of strong activators of the intrinsic or extrinsic pathway, ROTEM has also been carried out with citrated whole blood initiated only by recalcification in order not to mask distinct effects in another study of marathoners. 27 The procoagulant effects were analyzed by TEG using the ROTEM 4-channel system (Pentapharm, Munich, Germany). Immediately before and after the run, CT, CFT, α angle, and MCF were obtained with the blood collected. All samples were analyzed within 3 hours after collection. It was demonstrated that CT was shortened from 662.9 to 505.6 seconds and MCF was increased from 48.4 to 51.5 mm after the marathon, but there were no significant changes in CFT and α angle. Interestingly, some participants showed prolonged CT and compromised MCF, which might be explained by different training levels and individual fitness. No other clotting tests were performed but were suggested for the future study. It is worth to note that compared with the report by Sumann et al, 26 the CT and CFT are longer and α angle and MCF are smaller as no activators of the intrinsic and extrinsic pathway were used, while less changes were observed perhaps due to the longer time (30 minutes vs 3 hours) after the marathon.
Rotational thromboelastometry was also conducted to study and compare the effects of different types of exercise (ie, marathon, triathlon and long distance cycling) on whole blood coagulation. 28 Two ROTEM assays (in-TEM and fib-TEM) were carried out to measure CT, MCF, and fibrin polymerization, respectively, before and after the exercise. It was found that CT was decreased by 7.4% to 9.9% and MCF was increased by 6.1% to 8.3%, fibrin polymerization was also increased by 6.1% to 14.7% with marathon and triathlon (2.5 km swimming, 90 km cycling, and 21 km running) exhibiting most and least significant effects, respectively. These results were consistent with those obtained by conventional clinical coagulation assays including platelet activation and similar to coagulation profiles of the whole blood as indicated by the TEG parameters R, K, α, and MA before and after exercise. Overall, these results warrant further study of the risk of thromboembolic incidents in prolonged running.
In summary, most studies show hypercoagulable responses to exercise on blood coagulation, despite activation of fibrinolysis as the intensity of exercise increases. It appears that even with differences in experimental protocols, reaction time R in TEG or CT in ROTEM was the most markedly affected variable, while maximum clot strength or firmness was also modified. Increases in factor VIII: C activity and fibrinogen plasma level are most plausible explanation for reduced CT. 27 These changes cannot be attributed to simple hemoconcentration and more likely reflect the complex interactions of the physical stress upon the coagulation system. Endothelial cell activation due to mechanical factors 54 and enhanced platelet function caused by activation 55 along with catecholamine-mediated mobilization of platelets 56 may also contribute to these effects. Increased release from endothelium and reduced hepatic clearance may contribute to the increase in plasma levels of tissue-type plasminogen activator and consequently enhanced fibrinolysis. 49 In future studies, fibrinolysis-related parameters such as LY30 in TEG and LI30 in ROTEM should be measured since activation of fibrinolysis is a well-known effect of exercise. In addition to exercise intensity, the type of exercise and duration of activity may affect coagulation and fibrinolysis. For example, the exercise with more running fractions led to a higher extent of platelet activation. 28 Prolonged running exercise (eg, marathon) tended to have a procoagulant effect even the next day. 26 Moreover, the majority of studies have demonstrated that sedentary individuals exhibit marked coagulopathy due to attenuated fibrinolytic response and exaggerated changes in coagulation and platelet variables compared to trained or individuals of a high level of physical activity. 6
Temperature Extremes
Another common stressor shown to affect blood coagulation and fibrinolysis is exposure to extreme hot 57 or cold temperatures. 58 This could occur during seasonal or regional climactic changes, 59,60 medical treatment-induced hyperthermia, 57 hypothermia, 61 or recreational activities like hot bathing. 62 –65 Hyperthermia occurs when body core temperature increases above 37.5 to 38.3°C, 66 while hypothermia occurs when body core temperature drops below 35°C. 61 The former could develop through exposure to tropical/desert climates, while undergoing chemotherapy, and by participation in intense exercise or recreational activities (eg, sauna). Passive increases in body temperature may change hemodynamics, plasma volume, and blood flow distribution in a manner similar to exercise. Moreover, excessive activation of coagulation is thought to contribute to the progression of multiple organ dysfunction and death accompanying both severe exertional or classic heat stroke. 67 Extreme cold exposure could occur during medical treatment, for example, cardiopulmonary bypass (CPB), plastic surgery, 68 and cerebral aneurysm clipping and during exposure to Arctic or Antarctic environments. Hypothermia causes an initial increase followed by a decrease in heart rate and inhibition of platelet functions. 69 The cardiovascular and hemodynamic effects of cold may lead to reduced oxygen consumption and changing enzyme kinetics, resulting in impaired coagulation and fibrinolysis. Temperature itself may also affect these biochemical processes. 41 For example, it is expected that the speed at which a series of enzymatic reactions occur in the coagulation cascade would vary with temperature. 70 In addition, as another way to assess the temperature effects for a long period of time, seasonal changes in blood coagulation and fibrinolysis have been investigated indicating a greater tendency to clot in circulatory system in cold weather with a mean temperature <20°C than in warm weather (>20°C) due to the activated coagulation system. 71 The long-term impacts of climatic extremes on human health and medications have been reviewed as well. 72
As summarized in Table 1, TEG studies have been conducted under various hyperthermic and hypothermic conditions lasting from hours to a few days. Blood was normally taken from human exposed to extreme temperature or undergoing medically induced temperature changes and was analyzed at the specific body temperature of each individual or at 37°C as previously described.
An early thromboelastographic investigation of coagulation and fibrinolytic response to heat was conducted in early 1970s. 30 Citrated whole blood samples, taken from volunteers in a sauna bath, were thromboelastographically analyzed. Compared with the control before sauna, the coagulation profiles indicated by R and K were not significantly altered by the heat exposure for 10 minutes. A decrease in R was noticed after 15 minutes in the sauna, while the K value was reduced less. Augmented blood coagulation was ascribed to the activation of factor VIII by adrenaline as evidenced by its concomitant increase in plasma after the sauna. However, the fibrinolytic activity was not measured by the thromboelastograph.
Pivalizza et al conducted a series of thromboelastographic studies on coagulation and fibrinolysis in patients with cancer undergoing whole-body hyperthermia–carboplatin therapy. 29 Thromboelastography was carried out before, during, and after the treatment. During the treatment, body temperatures were raised above 37°C for 6 hours and maintained at a maximum core temperature of 41.8°C for 1 hour followed by sequent cooling to 37°C. The TEG results showed that whole-body hyperthermia led to a 30% to 40% decrease in R time. There was also a decrease in K time and an increase in percentage of fibrinolysis at 41°C compared to the untreated. However, there were no differences between measurements made at 37°C and actual patient temperatures. Thromboelastography may be more clinically relevant for further investigation of potential thromboembolic complications in patients with whole-body hyperthermia. In addition to healthy controls, TEG measurements were made in clinical blood samples obtained from patients with trauma at 38°, 36°, 34°, and 32°C. 73 The data showed increased R time in both control and trauma groups as temperature decreased. Clot strength MA was unaffected except for an increase at 32°C compared with other temperatures in the control group. No temperature effects were seen on clot lysis in either group. In addition, the patients were more hypercoagulable than the healthy control.
Thromboelastography was also used to study the clinical effects of hypothermia in anesthetised patients who were cooled to a core body temperature of 32°C before elective intracranial surgery (see Table 1 for details). 33 Thromboelastographic results suggested significantly higher R and K values, and a lower α angle as a result of cooling, but no changes in MA. Compared with plasmatic coagulation tests showing less reduction in PT and unchanged aPTT, this study also demonstrated the high sensitivity of TEG to changes in temperature. This may be because TEG incorporates more enzymatic reactions, interactions of platelets with coagulation proteins, and platelet functions in the coagulation cascade reflecting the cumulative effects of temperature. A limitation of the study is the relatively short duration (4 hours) of the hypothermic effects. Longer durations may be clinically relevant in patients with preexisting coagulopathies.
Standard and modified TEG has been used in patients undergoing CPB under mild hypothermia. 35 The findings from heparinase-modified TEG measurements suggested prolonged R by 50%, K by 70%, and reduced α angle by approximately 30% as obtained at body temperature of 33.3°C during CPB compared with those measured at 37°C, but no effects on MA, supporting the temperature effects reported by other studies. 33 Further study of the enzymatic activity of coagulation proteins and platelet activity under hypothermia would determine contributions of different mechanisms to the global hypothermia effects. Blood-contacting surgical devices may damage both the cellular and the humoral components of the coagulation system (eg, platelet activation and protein denaturation), which can be complicated by the patient’s disease state.
A similar study was conducted to monitor coagulation in patients undergoing orthotopic liver transplantation, where the patient’s temperature ranged from 36.9°C to 32°C during the course of the operation. 36 Four clotting TEG parameters R, K, α, and MA were measured as different temperature groups as ≤33.9°C, 34.0°C to 34.9°C, 35.0°C to 35.9°C, 36.0°C to 36.9°C on the basis of the patient body temperature at the time of blood sampling. Prolonged R, K, and compromised α angle were revealed compared to control variables obtained at 37°C, illustrating consistency with the in vitro study and the need for TEG measurement at actual body temperature. On the other hand, there were no differences in the coagulation effects of the hypothermia between different temperature groups.
To investigate how mild therapeutic hypothermia affects the coagulation system of patients with cardiac arrest, heparinase-modified ROTEM was performed after a bolus infusion of cold crystalloids. 74 Rotational thromboelastometry parameters (CT, CFT, MCF) and maximum lysis were measured at 37°C at 0 hour (before hypothermia), and 1, 6, and 24 hours when mean patient temperature was 34.3°C and close to 33°C. To simulate physiological conditions, while assessing the intrinsic changes in coagulation during 24 hours of hypothermia, no additional activators were added to the test blood. In contrast to a 2.7-fold increase in aPTT time at 24 hours, thromboelastographic values did not significantly change except for a 20% increase in CT at 1 hour after induction of mild hypothermia. This could be due to the thromboelastographic measurement at 37°C rather than at patient’s temperature during hypothermia, overestimating the speed of clot formation and therefore underestimating in vivo coagulation impairment. Alternatively, hemodilution by a total volume of 2528 mL crystalloid fluid within half an hour to induce hypothermia may impair blood coagulation.
In summary, the hemostatic changes were highly dependent on the specific heating/cooling methods, duration, and target temperature. It was found that TEG studies are generally in agreement with plasma coagulation tests. Taken together with other biological evaluation such as PT and aPTT, 70 acute hypothermia compromised blood coagulation mostly on clotting initiation and propagation. This is in contrast with the effects of climatic temperature, which resulted in a more prothrombotic effect with exposure to cold environments. The class of changes was considered potential risks for various cardiovascular conditions in clinic in patients with existing diseases.
The hemostatic activity of extreme temperature was ascribed to its acceleration or inhibition of the enzymatic reactions of the coagulation cascade, its interactions with platelets and vascular cells. The temperature effects on vascular system cannot be accounted in in vitro studies. For example, in vivo, vasodilatation and consequent exposure of endothelium to altered blood flow may occur under physiological conditions.
Hyperbaric/Hyperoxic Stress
Hyperbaric and hyperoxic stressors as exemplified by diving and decompression, which could trigger physiological changes in hemodynamic properties (blood volume and flow rate), respiration, homeostasis, and central nervous system activation. 75 Compared to other stressors, hyperoxia exposure in human experiments is relatively short, lasting for no more than 2 hours. Theoretically, human coagulation and fibrinolysis may be modified as a result of several factors: (1) a reduction in the number of circulating platelets, activation, and aggregation of platelets 76 ; (2) decreased concentrations of molecular hemostatic mediators, such as coagulation factors I, X, and XII 77 and plasminogen activator inhibitor-178; and (3) elevated concentrations of plasmin-antiplasmin complex. 77,78 These changes may exert mutually opposing effects on extrinsic and intrinsic blood coagulation and fibrinolysis pathways. The clinical implication of the altered coagulation system may be either bleeding risk or thrombosis. On the other hand, some studies found no changes in the concentration and activity of other molecular hemostatic cofactors such as thrombin activatable fibrinolysis inhibitor. 79
Although the effects of hyperbaria and decompression on different coagulation and fibrinolytic pathways have been studied, 77,80 their global effects on blood coagulation are unclear. Most assays have tended to focus on different parts of the coagulation system, such as platelet counts and functions, and do not account for the interactions in the clotting cascades and platelets in whole blood. Thus, it is difficult to relate isolated findings from such tests to overall effects on whole blood, as to what actually occurs in clinical settings.
Very few studies have examined hyperbaria-induced changes in blood clotting using thromboelastographic techniques. A single report used TEG to investigate the effects of hyperbaric oxygen in experimental animals. 37 Thromboelastograms were obtained for citrated plasma from normal control and rats exposed to hyperbaric oxygen (see Table 1 for detailed TEG procedures) and revealed that the hyperbaric oxygen resulted in a prolonged K and decreased MA. Such modifications in TEG tracings were more evident in the group with a longer exposure to the stress. It was speculated that the liver damage induced by hyperbaric oxygen might account for reduced production of clotting factors and thus compromised blood coagulation, apart from direct alterations by oxygen at the various stages of coagulation cascade. However, supporting evidence from human studies is lacking.
As the technology has advanced in recent years, we conducted a series of TEG studies on healthy volunteers to better understand the global effects of exposure of hyperbaric and decompression stress on blood coagulation and fibrinolysis. In these studies, a TEG method was successfully applied to test different blood samples taken before and at 20 (P20) and 60 minutes (P60) postacute hyperbaric exposure to 180 kPa for 80 minutes. Specifically, citrated whole blood was stored at room temperature for at least 30 minutes prior to TEG analysis, as it has been consistently documented that blood stored for less than 30 minutes is not stable. 81 –83 Thromboelastographic measurements were carried out using a computerized TEG Hemostasis System 5000 (Figure 1B; Haemoscope Co). After the system had passed the electronics testing and quality control according to manufacturer’s protocol, 1 mL of citrated blood was transferred to a vial with buffered stabilizers and Kaolin (Haemoscope Co). The sample was mixed by inverting 5 times and 340 μL was transferred into a 37ºC prewarmed disposable cup containing 20 μL of 0.2 mol L− 1 calcium chloride. The measurement was started immediately and run until all parameters of interest (ie, R, K, α, MA, TMA, and LY30) were finalized.
Figure 2 shows the TEG tracings of a heat-acclimated healthy volunteer, sampled predive and at 20, 60 minutes postdive. These data indicate that the combined hyperbaric and decompression stress led to an earlier onset of clot formation (ie, R time, as indicated in Figure 2), and a stronger clot (as measured by the maximal amplitude MA). The hypercoagulable state was continuous and the coagulatory response increased with time up to 60 minutes after the exposure.
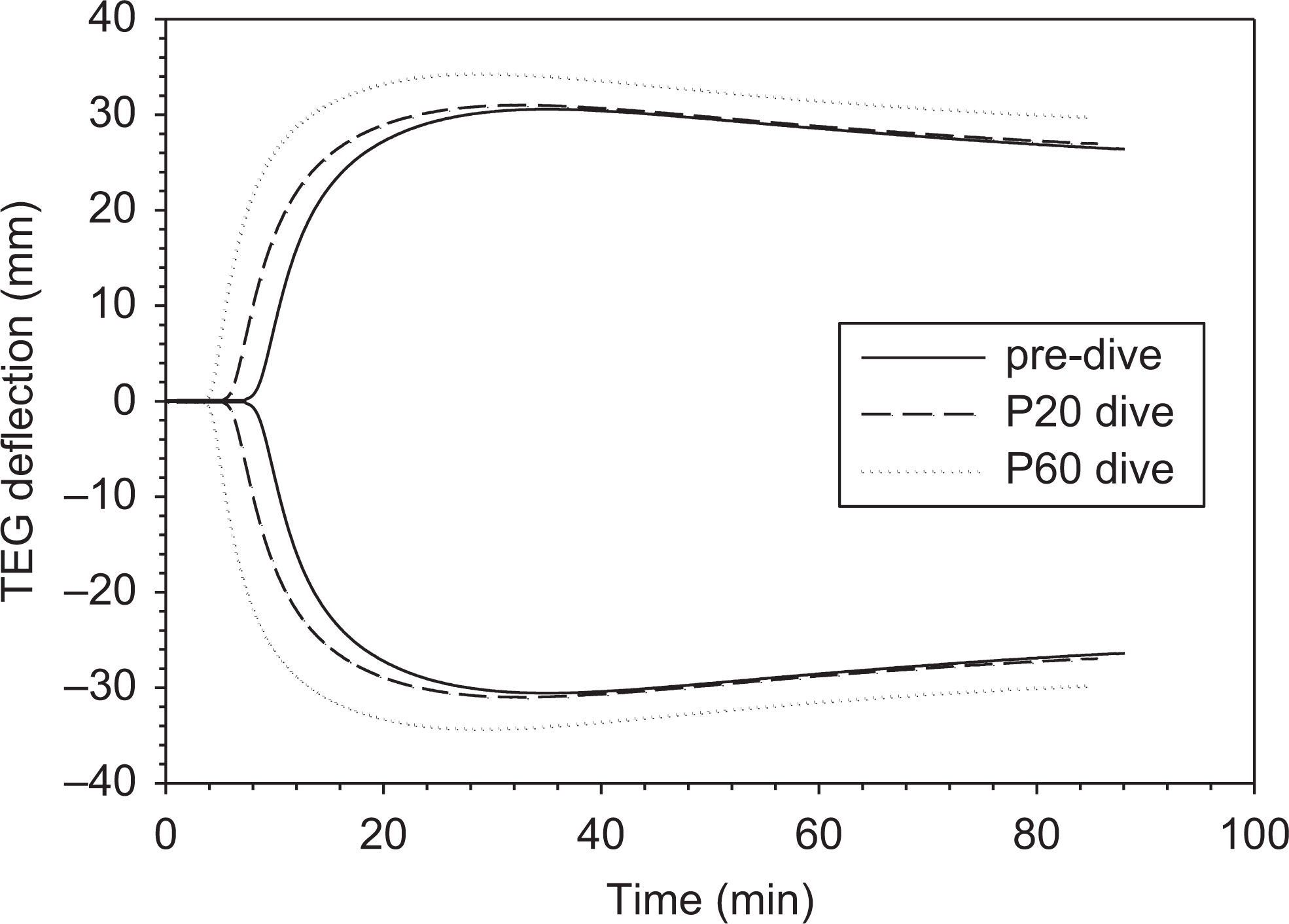
Typical thromboelastography (TEG) tracings of blood from the same healthy volunteer before and after exposure to diving and decompression.
Figure 3 shows the R, K, α angle, MA, and LY30 values of blood at predive, P20, and P60 before the stress. Compared to predive values, R, K, and LY30 tended to decrease following dive, especially at P60, but did not achieve statistical significance. This is most evident for R value. We found α and MA were similar at all pre- and postdive time points. The insignificant changes may be due to small sample size (n = 9) and large individual variations. It should be noted that some individuals showed opposing effects. On the other hand, the stress level in our study (80 minutes of exposure to 180 kPa) may have been insufficient compared to that in other studies (eg, 280 kPa) where significant activation of platelets and fibrinolysis were evident. 80 Future analyses of the stress level and response in each individual (eg, bubble scores, measures of coagulation factors, or inflammatory mediators) may help to explain the interindividual differences in stress-induced hemostatic alterations.
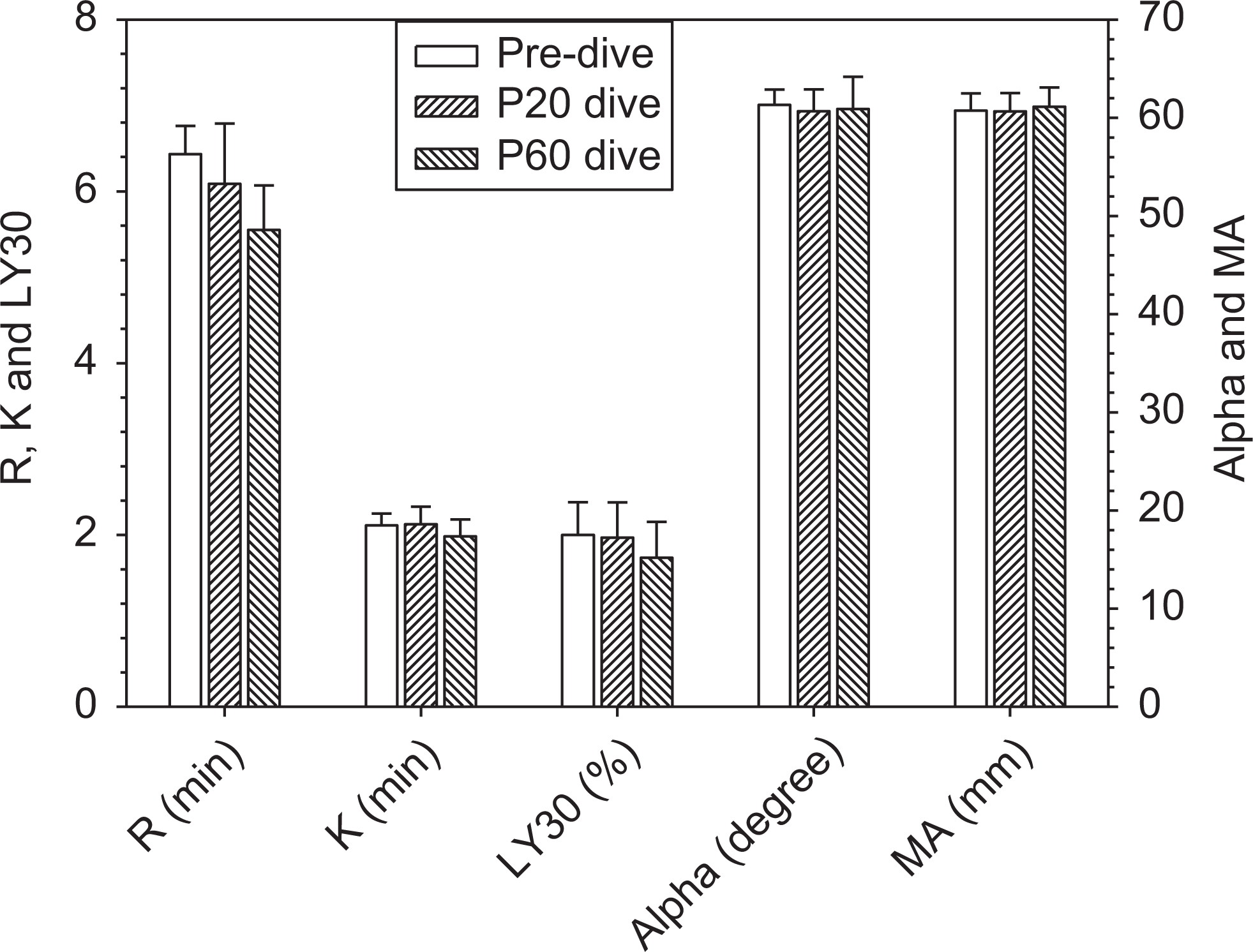
Thromboelastography parameters of blood sampled from healthy volunteers (n = 9) at predive, 20-, and 60-minute postdive and decompression. Data represent mean ± standard error of the mean (SEM).
Hypobaric and Hypoxia Stress
Hypobaric and/or hypoxic stress commonly occurs at high altitude and during long-haul flights. Interest in the effects of this type of stress on human coagulation lies in possible links with peripheral thromboembolic events. 84 Moderate altitude exposure has been identified as a potential risk factor for venous thrombosis. 85 At high altitudes, there are increases in the hematocrit (number of erythrocytes), cardiac output, blood flow to the brain, 86,87 and decreases in oxygen and vasoconstrictor response. 88 In addition, blood coagulation may be affected under hypoxic conditions, due to a change in the activity of oxygen-dependent enzymes. These physiological sequelae could last from hours to months depending upon the duration of the stress. However, there are discrepancies in the changes in enzyme activities, which may be due to the differences in the severity and duration of hypoxia experienced.
A number of studies have shown changes in coagulation markers at high altitudes after acute (hours to days)
89
and chronic (weeks to months)
90
exposition. The hypobaric stress was generally found to impair coagulation as evident by significantly elevated
Only 1 recent study was found involving thromboelastometry measurements at an intermediate altitude (3300 and 5400 m). 38 In comparison with variables of in-TEM assay measured at sea level, CT and CFT were extended, the α angle was reduced, while MCF was not altered after acute exposure to an altitude of 3300 and 5400 m for 1 to 2 days. Interestingly, after prolonged exposure to the altitude of 5400 m for more than a week, the variables reverted to the baseline values with the exception of MCF, which increased significantly above the acute exposure levels. Furthermore, the coagulation parameters were associated with a significant increase in plasma norepinephrine. This study demonstrated the benefit of ROTEM as a field-deployable point-of-care test, which can be to overcome all logistic difficulties in performing a large battery of single coagulation tests within the frame of high altitude expedition.
Whether hypoxia can influence the hemostatic system is still the subject of debate. 92 Studies simulating the conditions of reduced cabin pressure during commercial air travel have found no changes in hemostatic parameters (eg, international normalized ratio, PT, aPTT, platelet counts functions, prothrombin fragments 1 and 2, thrombin-antithrombin III complexes, α2-antiplasmin, tissue plasminogen activator, D-dimer) under hypobaric hypoxia for 8 hours, 93 normobaric hypoxia for 7 minutes 94 and 2 hours, 95 and isocapnic hypoxia. 96 Although others report transient increases in the concentrations of prothrombin fragments 1 and 2 by 2.5-fold, thrombin–antithrombin complex by 8.2-fold, and factor VII activity by 17% after 8 hours of exposure to 76 kPa air pressure in a hypobaric chamber. 97 The discrepancies may be due to the differences in the degree and duration of hypoxia, normobaric versus hypobaric conditions, and other potentially confounding factors, such as sedentarism, dehydration, and even mental stress. 98 Most hypoxic environments were created for a short duration simulating a low altitude of 2400 m using hypobaric chambers rather than measurements during actual flights. The clinical significance of any laboratory findings for perturbations in coagulation remains to be explored. 92
The utility of TEG has been demonstrated in evaluating coagulation changes in an aircraft during and after a round-trip flight (∼8 hours each way). 22 Both in-TEM and ex-TEM measurements revealed an accelerated clot initiation (CF), clot propagation (CFT, α angle), and increased clot firmness (MCF) values that were within the upper reference limit. These changes peaked immediately after the flight, but MCF remained elevated to a lesser extent 3 days after the flight. In addition, the activation of coagulation and fibrinolysis was confirmed by reduced aPTT and increased activities of certain plasma markers (eg, factor VII and plasminogen activator inhibitor). This is in contrast with another study simulating normobaric hypoxic conditions in a special chamber corresponding to altitudes between 2000 and 2400 m, where patients remained in a sitting position in the aircraft for 10 hours. 39 The in-TEM measurements showed no thromboelastographic changes throughout the study, which is supported by either no changes or minor variations in a number of plasma markers (eg, fibrinogen, plasminogen activator, PT). To differentiate the influence of moderate hypoxia from other factors (eg, prolonged sitting), ROTEM was performed with other coagulation tests on the effects of 10-hour bus travel without hypoxic stress. 99 All measured parameters were altered, suggesting an accelerated clot initiation (in-TEM CT), clot propagation (in-TEM CFT and α angle), and clot firmness (in-TEM MCF). Similar results were noted for the ex-TEM, with the exception of MCF which was unchanged. The results of this study imply that additional factors other than hypoxia such as postural constraints are involved in actual conditions of a long-haul flight that may play a role in coagulation changes.
Furthermore, the thromboelastographic technology has been widely used to monitor changes in coagulation status in clinical settings following acute physical trauma and critical illness, such as thermal/burn injuries, 100 multiple trauma, 101 cardiac surgeries and liver transplantations, 102,103 sepsis, and septic shock, 104 all of which affect coagulation and fibrinolysis to various extents. In particular, injury-related hypothermia and acidosis affect the degree of acute coagulopathy of trauma. 105 Patients with trauma display different coagulopathies according to TEG; endogenous acute traumatic coagulopathy is associated with shock and hypoperfusion, whereas the exogenous effects of dilution from fluid resuscitation and/or consumption of coagulation factors through bleeding and blood loss further add to trauma-induced coagulopathy. 10 Overall, there is evidence to support the use of TEG as a point-of-care test for assessing patients with critical injuries 106,107 and potentially as a guide for transfusion practice after trauma. 102,108
Psychological Stress
Although psychological effects on coagulation and fibrinolysis are known, 109 there are limited studies using TEG to assess these changes. Most studies have focused on soluble molecular markers and standard laboratory tests, such as fibrin D-dimer, fibrinogen, factors VII, VIII, XII, and PT, activated partial prothrombin, associated with acute mental stress, 110 –112 anxiety, 113 vital exhaustion, 114 depressive symptoms, 115 overcommitment 116 and posttraumatic stress disorder (PTSD). 117 Furthermore, psychological functioning, exhaustion, affectivity, depression, anxiety, worrying, vigor, and social support were assessed for their associations with stress-induced procoagulant reactivity. 118 Moreover, psychological stress and mental disorders have long been linked to cardiovascular disorders. 119 In general, prothrombotic changes have been detected under stress through alterations in blood rheology and hemostasis with acute stress being extensively reviewed and strongly associated with the pathogenesis of CVD and acute coronary syndromes. 4 Importantly, recent clinical studies show a conclusive link between PTSD and the presence and/or severity of coronary atherosclerosis and suggest that PTSD is an independent risk factor for mortality. 120 The biological mechanisms underlying the alterations in coagulation markers have been well addressed but are less conclusive for changes in fibrinolysis due to the limited number of studies and the inconsistency of the findings. Nevertheless, there are a few published reports involving TEG (see Table 1 for details).
An early thromboelastographic study of psychological stress was made of healthy humans under emotion stress. 40 The stress was simulated in a standardized situation involving intellectual activity with an imposed rapid rate of work and simultaneous interaction between 2 persons on the “Gomeostat” apparatus. The effects on blood coagulation were characterized by thromboelastograms recorded on Thromb-1 thromboelastograph. In addition to reaction time R, time to clot formation K, and maximum clot strength MA, maximum elasticity of thrombus, total blood coagulation constant T, and Serradimigni index were determined demonstrating mixed TEG changes among 35 patients before and after the stress. In particular, 9 of 35 volunteers were categorized as functionally hypercoagulable, 15 were hypocoagulable, and no changes were detected in the remaining patients. The opposite results were found using a model situation of emotional stress involving group interactions. Further studies would provide a better understanding of the regulatory role of the cerebral cortex on the hemostasis system. Yet, the clinical relevance of the changes in TEG remains to be examined in vivo.
Multiple Stress
To date, few studies have evaluated the combined effects of multiple physical or mental stressors on coagulation (eg, exercise and hypoxia 95 as exemplified by exercise at high altitude during climbing mountains). DeLoughery et al conducted a study where patients exercised both at room air and at 12% oxygen and found that hypoxia may exert an antithrombotic effect by suppressing exercise-induced procoagulant changes and stimulating fibrinolysis, 121 as opposed to their synergetic effects since each stress tends to promote blood coagulation.
Military operations routinely expose personnel to a multitude of physical and psychological stressors that can cause deterioration in performance and lead to negative health consequences in soldiers. 122 The increased frequency and pace of deployments in both active-duty and reserve populations enhances the potential for stress-related coagulation abnormalities and associated cardiovascular risk factors. 123 The impact of operational stress on hemostasis was examined in military fighter pilots, showing a hypercoagulable state after a standardized training flight mission, attributed to the psychophysical stress associated with operational jet flight and exposure to G forces, 124 but TEG/ROTEM was not performed. An earlier report using TEG showed a relatively low prevalence (∼3%) of hemostatic disorders in air force personnel, with no correlation to flight history. 125
Taking advantage of the latest advances in the TEG technology, we have recently conducted an extensive thromboelastographic study to evaluate the effects of 9 days of repeated exertional heat stress (EHS) on blood coagulation and fibrinolysis. Briefly, healthy volunteers walked on a treadmill (4.5 km·h− 1, 2% incline) in a hot–dry environment (40°C, 30% relative humidity) to exhaustion or an ethical maximum core body temperature of 40°C. During acute EHS exposures, Canadian military nuclear, biological, and chemical protective clothing was worn producing an uncompensable exercise-heat stress condition. 126
Venous blood was collected at baseline (pre-EHS) and exhaustion on days 1, 6, and 9 of the 9-day repeated EHS exposures through an indwelling venous catheter into citrated tubes. Thromboelastographic measurements were carried out using a computerized TEG Hemostasis System 5000 following the same procedure as described in our aforementioned hyperbaric study.
Figure 4 presents representative thromboelastograms for citrated whole human blood from the same individual at the baseline and exhaustion to elucidate the effects on blood coagulation on day 1. The TEG tracings show that the EHS increased blood clottability. As far as blood coagulation is concerned, the stress led to the earlier onset of clot formation (ie, R time, as indicated in Figure 4) and the stronger clot (as measured by the maximal amplitude MA). Similar thromboelastographic profiles were noticed on day 6 and 9. The observed effects could not be attributed to hemoconcentration, as there were no significant shifts in plasma volume during the acute EHS exposure.
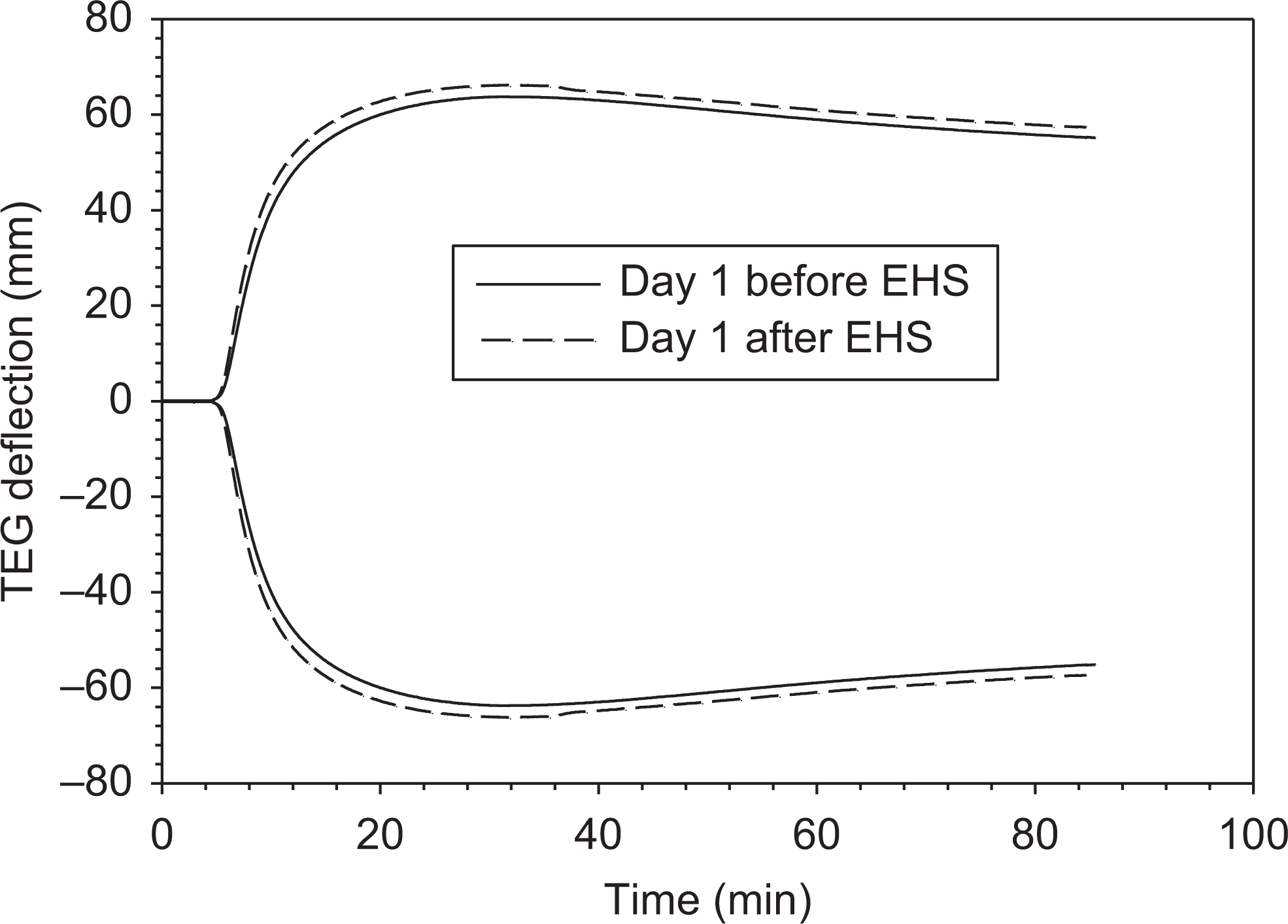
Typical thromboelastography (TEG) tracings of blood from the same healthy volunteer before and after exposure to exertional heat stress (EHS).
Figure 5 depicts averaged procoagulant effects and fibrinolytic activation for all patients. The differences in most TEG parameters between the pre- and post-EHS exposure were not profound. This may be due in part to the large individual variation. Another possibility is that the stress not only initiates the normal cascade but also its mode of action relies on activation to blood vessel, which cannot be measured in our current TEG method. On the other hand, markedly procoagulant effects of the stress on days 1 and 9 were indicated by an approximately 20% reduction in K and 5% increase in MA, confirming significant activation of the coagulation cascade and possible platelet function. In contrast, the effect on MA is less, which is in accordance with the reported effects of the stress on the maximum clot strength in the literature. 50 It should be noted that while there is ample evidence that exercise and heat play a crucial role in thrombosis and hemostasis, respectively, as discussed earlier, it is promising for typical use of TEG to characterize overall synergistic effects of combined and/or multiple stressors on the coagulation and fibrinolytic activities.
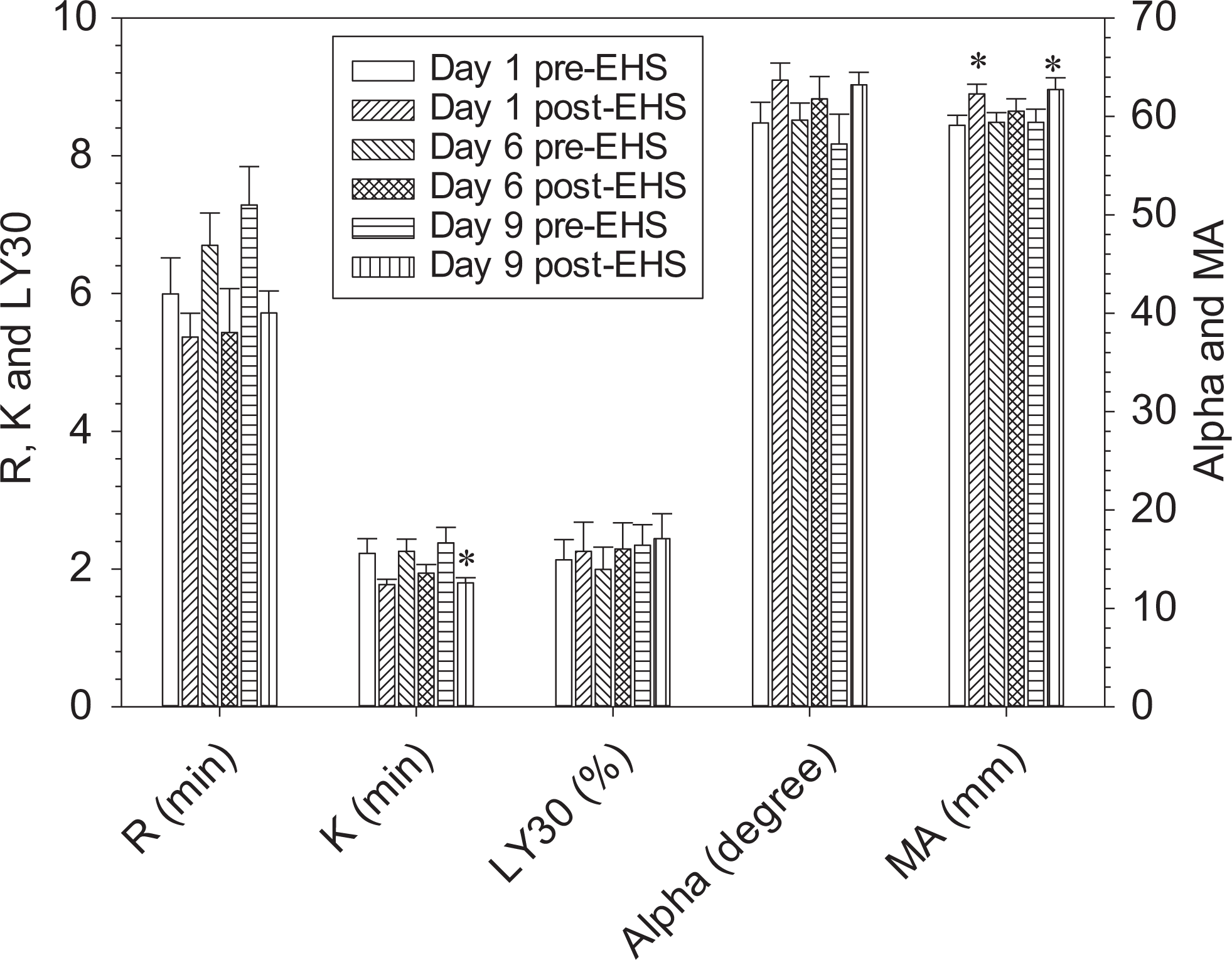
Coagulation changes in healthy participants in response to exertional heat stress (EHS) at different times. Data represent mean ± standard error of the mean (SEM; n = 9). *Significant difference from the pre-EHS (P < .05) based on 1-way repeated measures analysis of variance.
Our findings for the hemostatic impact of the dual stressors is consistent with previous TEG results, 25,30 thereby validating the use of TEG as a tool for assessing the potential effects of other unknown stress.
Remarks and Conclusions
Since its invention over 60 years ago, TEG has undergone continuous technological refinements to improve its reproducibility, ease of use, and interpretation in clinical and scientific applications. In theory, TEG has many of the properties of an ideal modern coagulation test for direct monitoring of stress-related hemostatic changes. It provides a comprehensive assessment of the complex hemostatic system in response to both physiological and psychological stress rather than evaluating only discrete portions of the coagulation cascade as with conventional laboratory tests. It is an inexpensive method that can provide immediate results, is simple to perform, easy to interpret, and can account for the effects of body temperature if needed. Yet, very few studies have been devoted solely to the evaluation of TEG in stress research and none has explored its potential to guide rational design for studying the implications of stress on CVD mechanisms.
There are several reasons why TEG has not been widely adopted for experimental studies of human stress physiology. One reason is its excessive reliance on operators and manual procedures compared to other automated coagulation testing devices, a source of criticism by laboratorial societies. 82,83 A major virtue of TEG is its remarkable versatility. It can be performed with the same instrument using fresh or anticoagulated whole blood or plasma. The application of specific activators, such as kaolin, tissue factor, and celite, provide the opportunity to investigate the role of contact proteins or tissue factor in initiating the clotting process. However, such versatility also results in TEG being done in many different ways, which leads to difficulty in establishing standard reference values and comparing experimental results between studies. In addition, different protocols in terms of sampling tubes and sample storage have been used. For example, the clotting time of citrated blood is strongly affected by the types of tubes used to collect blood (eg, Vacutainer from Becton Dickinson, vs Monovette from Sarstedt, Germany). 127 It has also been consistently documented that widely used citrated blood, stored for less than 30 minutes, is not stable. 82,83 Collectively, these factors likely have contributed to the current lack of application of TEG in stress research.
The current lack of universal recognition of TEG as a reliable routine laboratory test may be changed due to recent progress in TEG technology 128,129 leading to standardization of the method and improvement in reproducibility. For example, the recent development of computerized interfaces has led to a renewed interest in TEG, particularly to guide blood transfusion practices. 130 In addition to the use of normal and abnormal biologic controls as currently recommended quality assurance by the manufacturer, the accuracy of the device may be further calibrated with clinically relevant substances with known viscosities. 131
As a point-of-care system, TEG measurements have been performed in the field, such as onboard an aircraft. 22 In most studies using TEG, other assays (eg, PT, aPTT, platelet activation and adhesion tests) have been used in parallel to fully understand various hematological effects of stress. In addition to the most commonly measured 4 parameters (ie, R, K, α, and MA in TEG or CT, CFT, α, and MCF in ROTEM), additional parameters, such as LY30 which can be analyzed for fibrinolysis, have not been used as often for assessing the fibrinolytic effects of stress. Currently, TEG has been mainly used to measure coagulation properties, but should be further explored for understanding the complex mechanisms underlying stress–blood interactions.
Stress may alter hemostasis through its effects on neuroendocrine responses, such as enhanced sympathetic-mediated catecholamine release, 30,132 activated hypothalamic–pituitary–adrenal axis-induced cortisol release, 133 immunoinflammatory effects on leukocyte activation/mobilization, mechanical or physical stress on hemolysis of red blood cells 28 and endothelial cells, 134 and hemoconcentration. 135 Considerable interindividual differences in the response of the hemostatic system to psychophysiological stress have been observed. This could be ascribed to training status in the case of exercise. 27 Although aberrant coagulation has been associated with an increased incidence of CVD, the clinical significance of stress-induced changes in coagulation remains incomplete.
It should be noted that most studies were focused on the effects of a single stressor. Moving forward, it would be important to investigate the effects of multiple stressors, similar to those experienced during military operations, from both a physiological and psychological perspective.
In conclusion, both qualitative studies based on thromboelastograms in early applications and quantitative studies using many variables in recent applications have been reviewed. Different methodologies have been used that need to be considered when comparing results across the current literature. It is noted that exercise has an impact on a broad range of biological pathways including blood coagulation and fibrinolysis, compared to the other stressors. Although each stressor may modify physiological conditions through a common pathway (eg, activation of the sympathetic nervous system), other mechanisms such as temperature effects on plasma protein properties may exist that are ascribed to different effects and need to be considered when investigating the physiological changes under stressful conditions. In addition, it should be noted that stress which influences the physiology of hemostasis through interactions with the vascular endothelium or effects on endothelial-platelet interactions may be evaluated using TEG methods incorporating adherent endothelial cells. 136 One limitation of the TEG method is that once the process has started, it is not possible to manipulate it further. With advances in the instrument, development and standardization of coagulation assays, TEG can be a valuable tool to study hemostasis and potential pathological pathways in response to stress.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Defence Research and Development Canada.
