Abstract
The association between factor XIII-A (FXIII-A) Val34Leu polymorphism and myocardial infarction (MI) risk remained controversial. We performed a meta-analysis. Online databases were searched. Twenty-eight studies were included. The FXIII-A Val34Leu polymorphism was significantly associated with MI risk (odds ratio (OR) = 0.83, 95% confidence interval [CI] 0.76-0.91; P < .0001). This result remained statistically significant when the adjusted ORs were combined (OR = 0.77, 95% CI 0.65-0.92; P = .004). When stratifying for race, this polymorphism showed decreased MI risk in Caucasians. In the subgroup analysis by age group, significant associations were observed in early-onset patients and in late-onset patients. In the subgroup analysis by gender, there was a significant association in women but not in men. In the subgroup analysis stratified by smoking status, MI risk was decreased in both smokers and nonsmokers. This study suggested that FXIIIA Val34Leu polymorphism was a protective factor for MI in caucasians.
Introduction
Myocardial infarction (MI) is the most disabling and deadly disease in western countries, causing about 15% of all deaths in the United States, according to the recent statistics of the American Heart Association. 1 Despite the available therapeutic approaches, MI is still associated with high rates of acute death and long-term complications. The MI is a complex disease influenced by modifiable risk factors as well as genetic susceptibility. 2 Recently, a lot of studies have focused on the associations between genetic variants and MI risk, 3 and the factor XIII (FXIII) gene has been extensively studied.
The FXIII is a protransglutaminase that circulates in the plasma in tetrameric form. 4 The potentially active A subunits (FXIII-A) are synthesized by cells of bone marrow origin (megakaryocytes and monocytes/macrophages); the carrier/inhibitory B subunits (FXIII-B) are produced by hepatocytes. Nahrendorf et al 5 found that FXIII tissue levels were decreased in patients with insufficient healing. They also showed that homozygous and heterozygous transgenic FXIII-deficient mice died of left ventricular cardiac rupture within 5 days, in contrast to nondeficient mice or deficient mice receiving FXIII supplementation. 6 Collectively, these results suggested FXIII may play a critical role in the pathogenesis of MI.
Factor XIII-A is a transglutaminase that cross-links peptide chains through ∊(γ-glutamyl)lysyl isopeptide bonds. Its main physiological function is to strengthen fibrin polymers and protect them from the prompt degradation by the fibrinolytic machinery. The gene coding for the A subunit of FXIII is localized on chromosome 6p24-25. Till now, many studies investigated the association between the FXIII-A gene polymorphisms and susceptibility of MI. 7 –33 Most of them focused on 1 single-nucleotide polymorphism: Val34Leu (rs5985). However, the results from these studies were inconsistent and inconclusive. Two meta-analyses regarding this association have been reported in 2007. 34,35 Both meta-analyses demonstrated an association between the FXIII-A 34Leu allele and a modest protective effect against MI. Since 2007, several articles have been published. Most of these studies did not confirm this protection. In addition, those 2 meta-analyses did not assess whether this protection was selective by age, gender, or smoking status. Thus, we performed an update meta-analysis of all eligible studies to derive more precise estimation of the association of FXIII-A Val34Leu polymorphism with MI risk. This was, to our knowledge, the most comprehensive meta-analysis of the association between FXIII-A Val34Leu polymorphism and MI susceptibility.
Methods
Publication Search
Published studies were identified through a computerized search of Pubmed, EMBASE, Chinese National Knowledge Infrastructure (CNKI), and Wanfang databases (last search was updated on May 2013). The search terms used are as follows: myocardial infarction or myocardial infarct, and Factor XIII or F13A1, and polymorphism or mutation or variant. The search was limited in human participants. We perused the reference lists of all retrieved articles and relevant reviews. There was no language restriction.
Inclusion and Exclusion Criteria
Studies included in the current meta-analysis should meet the following criteria: (1) evaluation of FXIII-A Val34Leu polymorphism and MI susceptibility, (2) using a case–control design, and (3) genotype distributions in both cases and controls should be available for estimating an odds ratio (OR) with 95% confidence interval (CI).
Studies were excluded if one of the following existed: (1) not relevant to FXIII-A Val34Leu or MI risk, (2) not designed as case–control studies, (3) genotype frequencies or number not offered, (4) nonclinical studies, (5) departure from Hardy-Weinberg equilibrium (HWE), and (6) editorials, reviews, or abstracts. In the case of overlapping studies, only the one with the largest sample numbers was included.
Data Extraction
The following information was collected from each study: first author’s name, year of publication, ethnicity, age of patients, gender, sample size, covariates, and genotype number in cases and controls. Any disagreement was resolved by consensus.
Statistical Analysis
The strength of the association between the FXIII-A Val34Leu polymorphism and MI risk was measured by ORs and 95% CIs. Because often there is no clear evidence of the genetic mode of inheritance in complex disease genes, we used a pooling method that did not a priori assume a genetic model and avoided the problem of multiple comparisons. 36 The OR1, OR2, and OR3 were calculated for the genotypes: Leu/Leu versus Val/Val (OR1), Val/Leu versus Val/Val (OR2), and Leu/Leu versus Val/Leu (OR3) for the FXIII-A Val34Leu polymorphism. These pairwise differences were used to indicate the most appropriate genetic model. 36 –39 Once the best genetic model was identified, this model was used to collapse the 3 genotypes into 2 groups (except in the case of a codominant model) and to pool the results. We used a random effects model to calculate the pooled ORs. The statistical significance of summary OR was determined using Z test. A P value <.05 was considered statistically significant.
A chi-square test was used to determine whether observed frequency of genotype in control population conformed to HWE expectations. The homogeneity assumption was verified by chi-square-based Q test, which was considered to be significant for P <.10. I2 indicating the magnitude of between-study heterogeneity was also used, and I2 > 50% indicated severe heterogeneity. To explore the source of the heterogeneity, Galbraith plots were used. To evaluate the ethnicity-specific, age-specific, gender-specific, and smoking-specific effects, subgroup analyses were performed based on ethnicity, age, gender, and smoking status. The early-onset MI was defined as cardiac events occurring before the age of 50 years. To access the stability of the meta-analysis, 1-way sensitivity analysis was carried out. We also conducted cumulative meta-analysis by undertaking sequential random effects pooling, starting with the earliest studies. Publication bias was assessed by visual inspection of funnel plots, in which the standard error of log (OR) of each study was plotted against its log (OR). The Egger test was used to assess publication bias statistically. 40
All statistical tests were performed using the Revman 5.1 software (Nordic Cochrane Center, Copenhagen, Denmark) and STATA 11.0 software (Stata Corporation, College Station, Texas).
Results
Study Characteristics
The flow chart in Figure 1 summarizes this literature review process. In this current study, a total of 27 eligible studies met the inclusion criteria. 7 –33 Three articles reported 2 cohorts, 8,27,33 and each cohort was considered as a case–control study. There were 2 studies performed using Asians, 24 studies using caucasians, and 1 studies using mixed populations. A total of 13 studies were performed in patients younger than 50 years old and 14 in patients older than 50 years. In all, 2 studies included only male patients with MI, 3 studies included female patients with MI, and 4 studies included both male and female patients but data for these patients could be extracted separately. The characteristics of each study included in this meta-analysis are presented in Table 1. Genotype frequencies and HWE examination results are listed in Table 2.
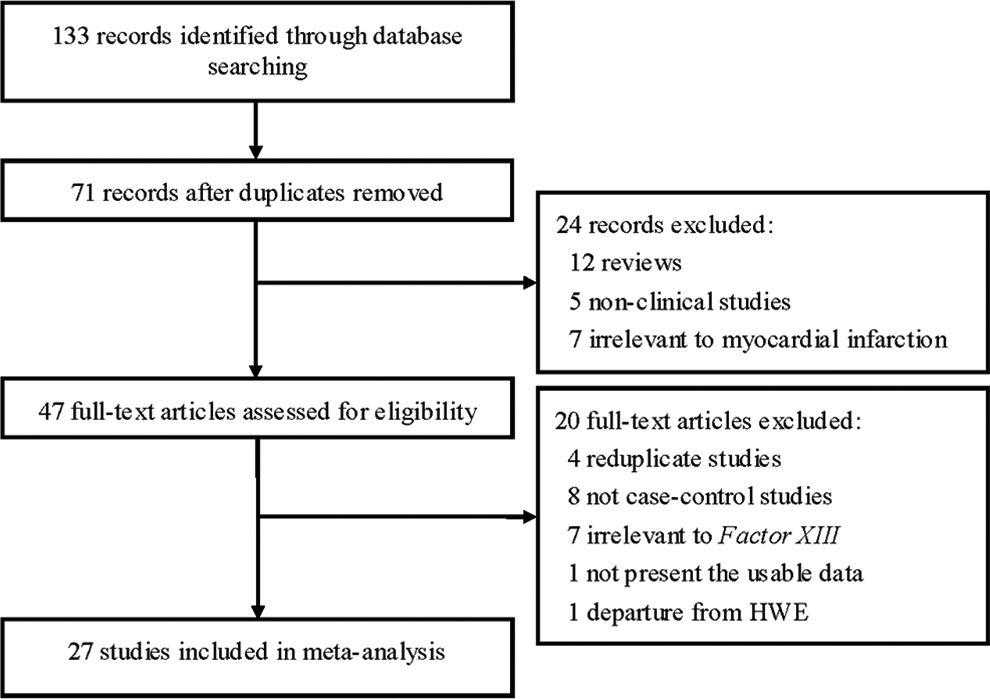
Flow of study identification, inclusion, and exclusion.
Characteristics of the Case–Control Studies Included in Meta-Analysis.
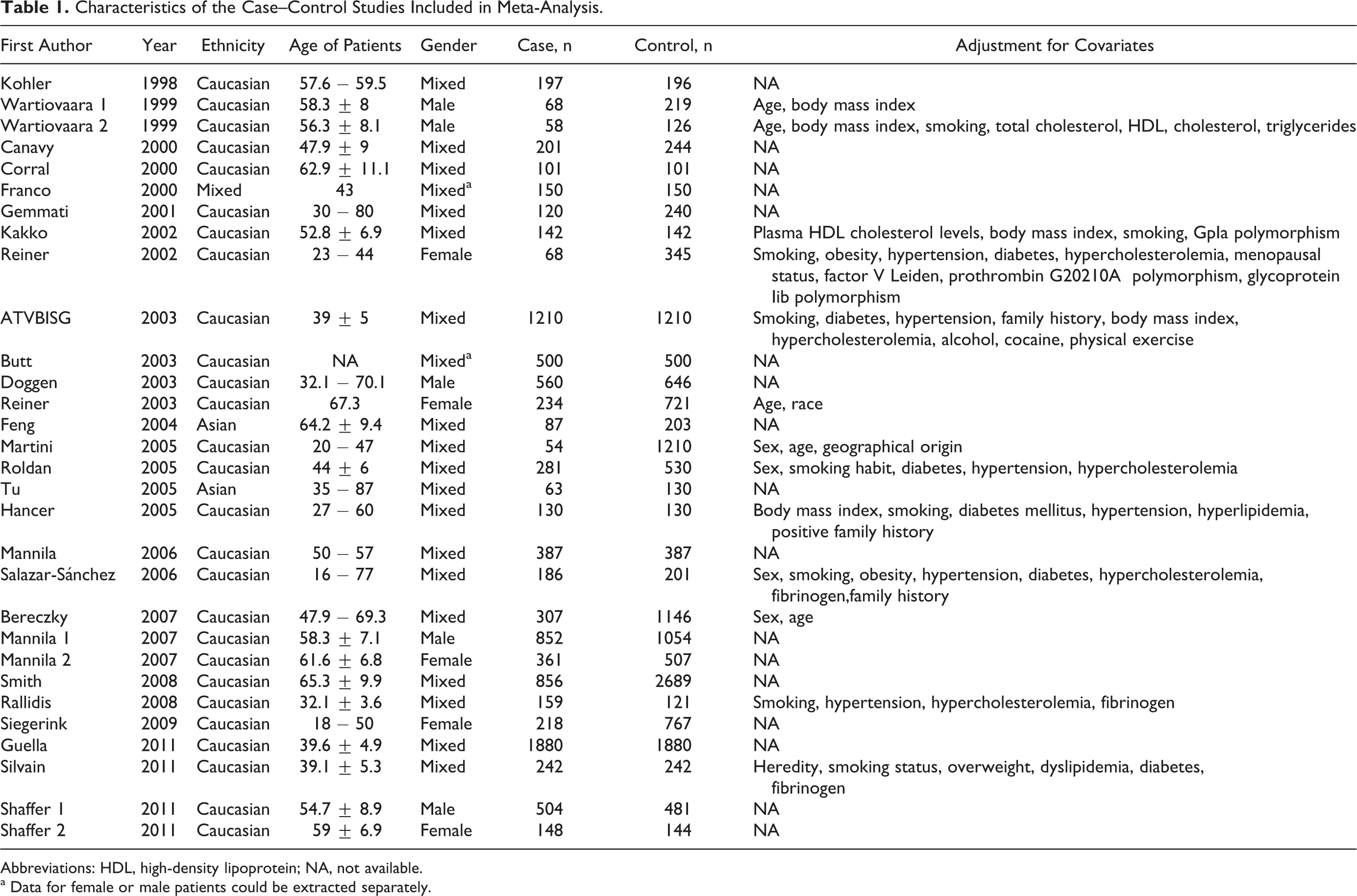
Abbreviations: HDL, high-density lipoprotein; NA, not available.
a Data for female or male patients could be extracted separately.
Distribution of Factor XIII Val34Leu Genotype Among Patients and Controls.
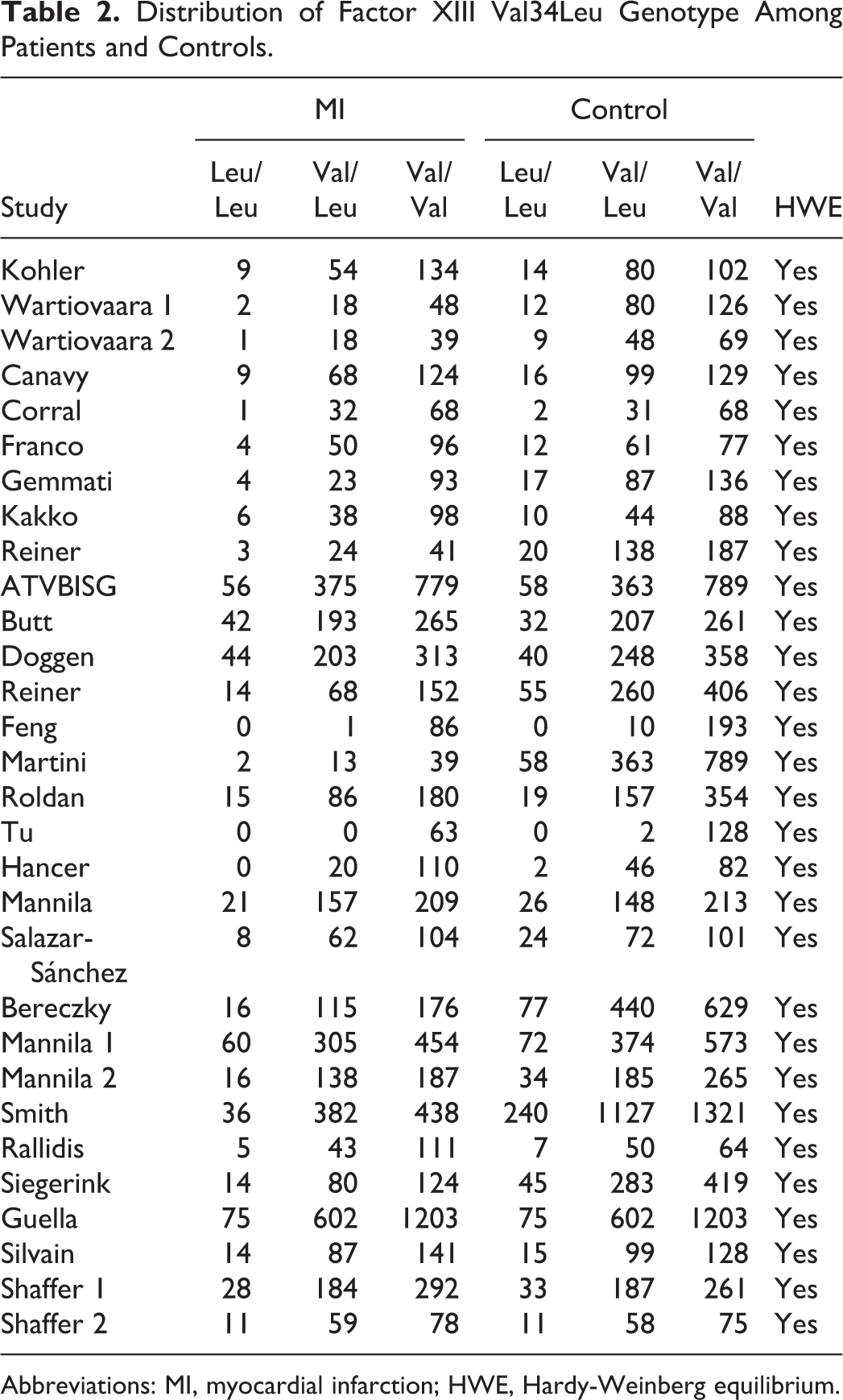
Abbreviations: MI, myocardial infarction; HWE, Hardy-Weinberg equilibrium.
Quantitative Data Synthesis
The frequency of the 34Leu allele among caucasians is about 25%; in Africans, its frequency is significantly lower, and in Asians, it is extremely rare. 41 Thus, we did not include 2 studies from Asians in meta-analysis. 19,22 Finally, 28 case–control studies determined the association between FXIII-A Val34Leu polymorphism and MI risk. The sample sizes for case and control groups were 10 109 and 16 245, respectively. The estimated OR1, OR2, and OR3 were 0.77 (0.65-0.91), 0.86 (0.78-0.94), and 0.87 (0.76-1.01; Table 3). These estimates suggested a dominant genetic model, therefore Leu/Leu and Val/Leu were combined and compared with Val/Val. The pooled OR was 0.83 (95% CI 0.76-0.91, P < .0001; Figure 2). Thirteen studies reported adjusted ORs. 8,13 –15,18,20,21,23,25,26,29,32 The combination of adjusted ORs for MI was 0.77 (95% CI 0.65-0.92; P = .004).
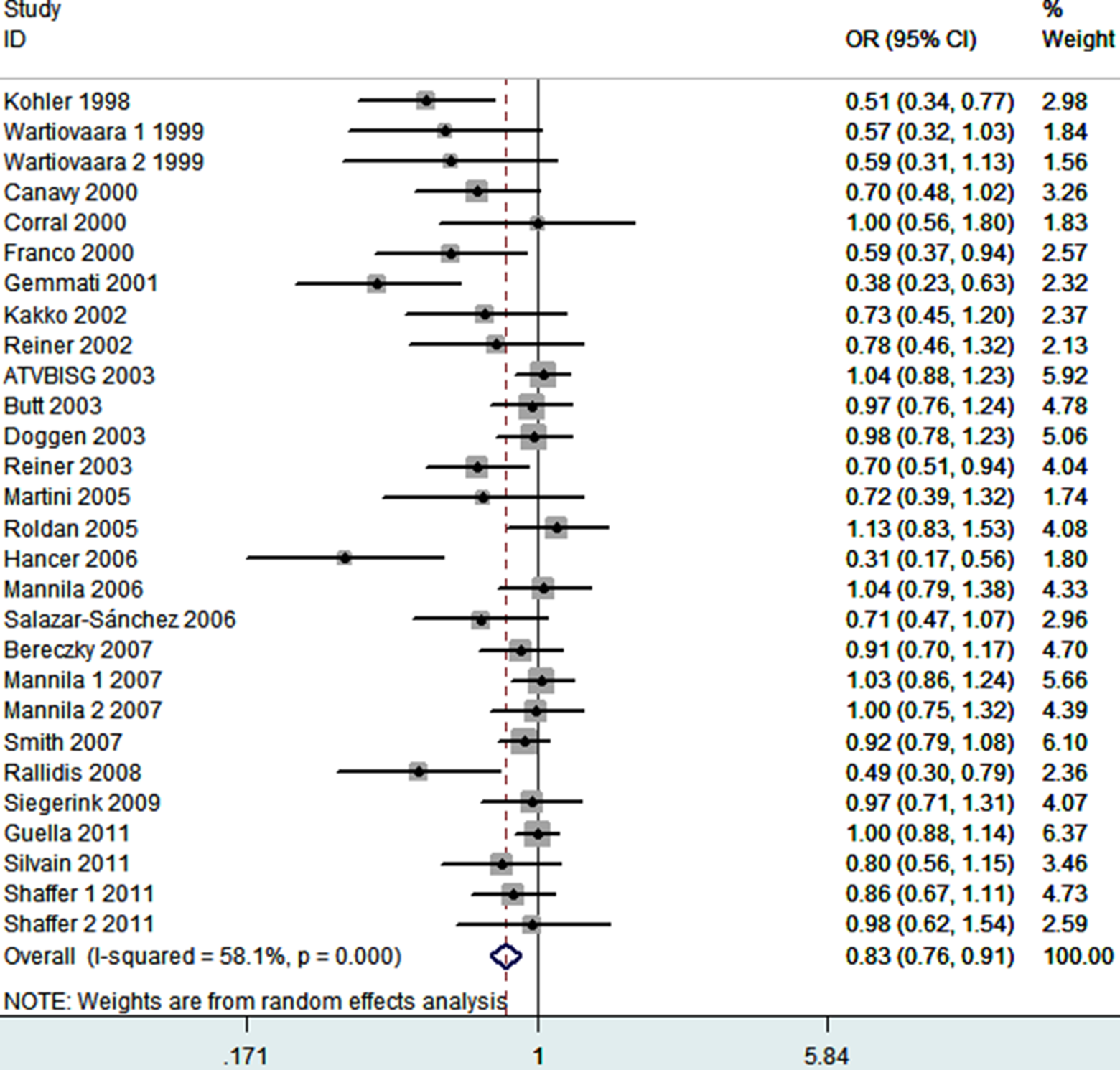
Meta-analysis for the association between MI risk and the FXIII-A Val34Leu polymorphism. FXIII-A indicates factor XIII A; MI, myocardial infarction.
Determination of the Genetic Effect of Factor XIII Val34Leu Polymorphism on Myocardial Infarction.
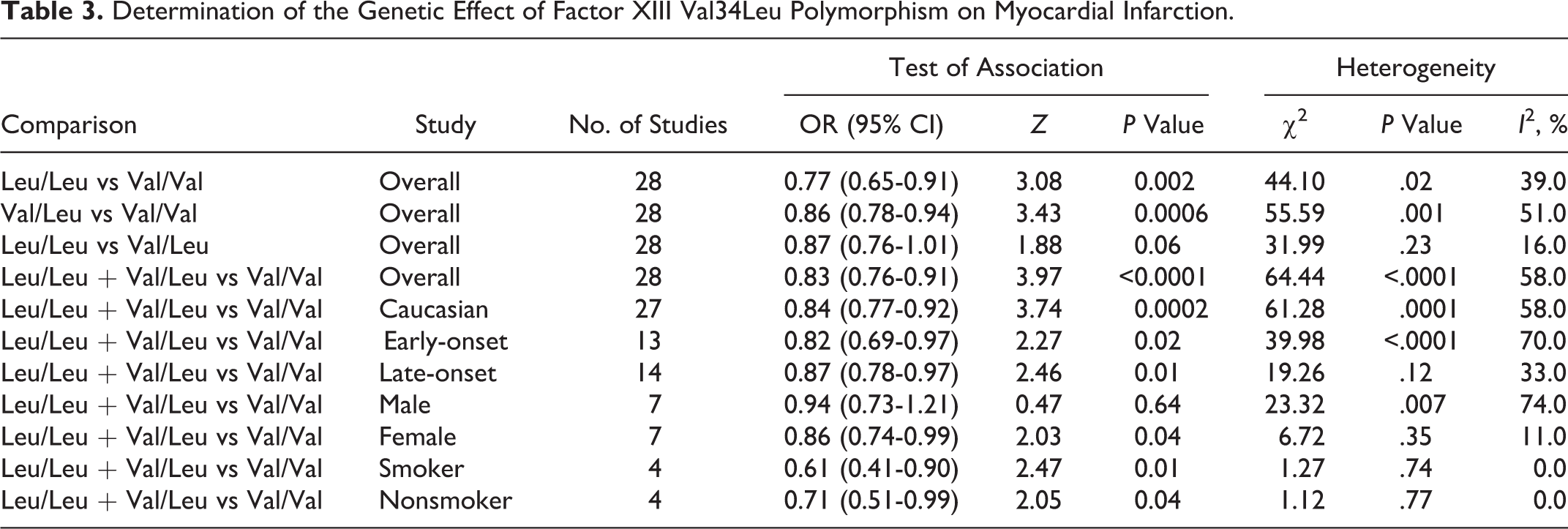
In the stratified analysis by ethnicity, a statistically significant association was found for studies with caucasians (OR = 0.84, 95% CI 0.77-0.92; P = .0002). In the subgroup analysis by age, the FXIII-A Val34Leu polymorphism was significantly associated with early-onset MI risk (OR = 0.82, 95% CI 0.69-0.97, P = .02) and with late-onset MI risk (OR = 0.87, 95% CI 0.78-0.97, P = .01). In the gender subgroup analyses, a statistically significant association was found in female patients with MI (OR = 0.86, 95% CI 0.74-0.99, P = .04) but not with male patients with MI (OR = 0.94, 95% CI 0.73-1.21, P = .64). Stratification by smoking status showed that both smokers and nonsmokers carrying 34Leu allele were associated with decreased MI risks (OR = 0.61, 95% CI 0.41-0.90, P = .01; OR = 0.71, 95% CI 0.51-0.99, P = .04).
Cumulative Meta-Analysis and Sensitivity Analysis
As shown in Figure 3, significant associations were evident with each addition of more data over time. The results showed that the pooled ORs tended to be stable. To evaluate the stability of the results of the meta-analysis, sensitivity analysis was performed through sequentially omitted individual studies. None of the results were materially changed, which suggested the robustness of our results (Figure 4).
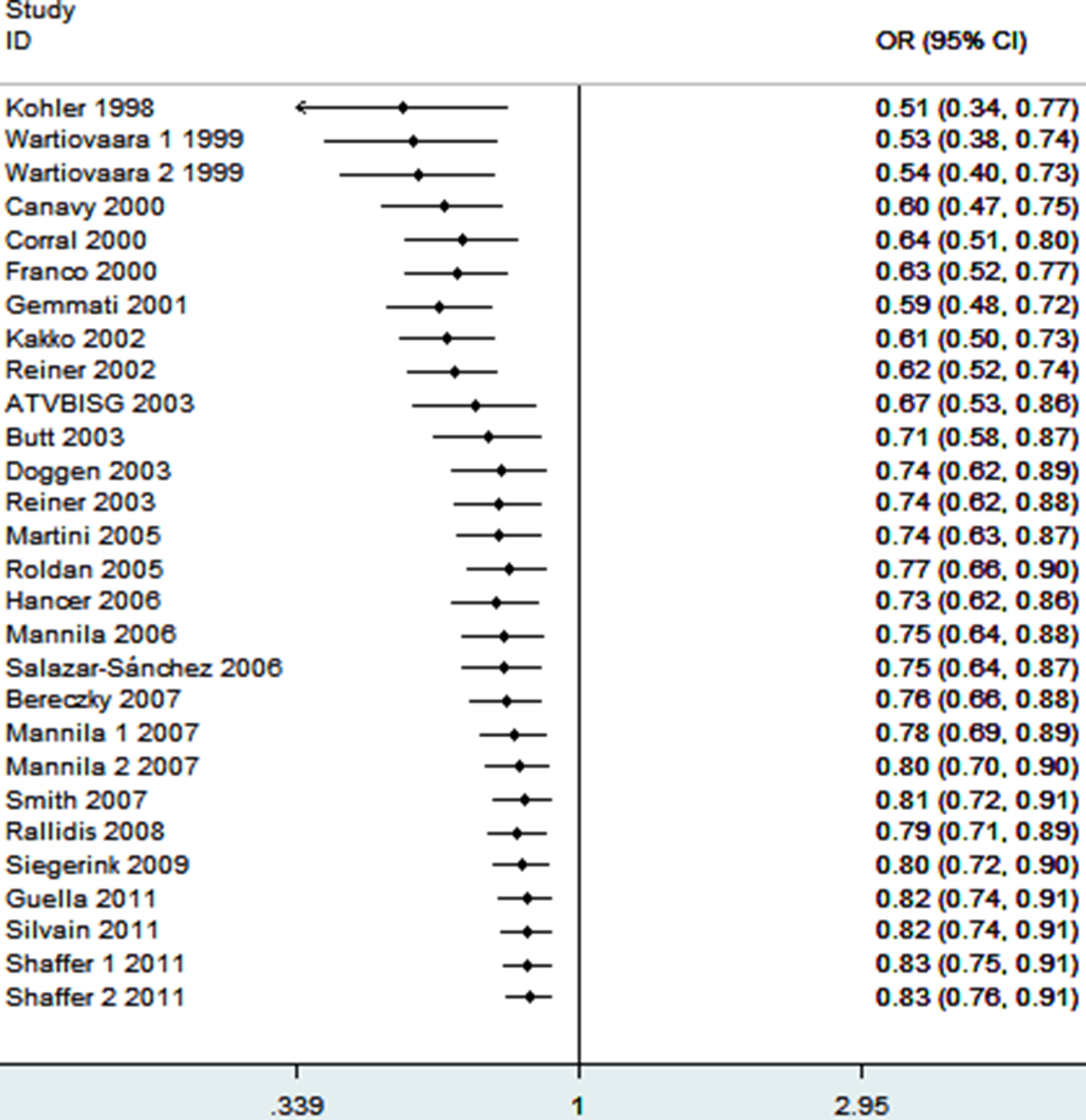
Cumulative meta-analysis of associations between the FXIII-A Val34Leu polymorphism and MI risk. FXIII-A indicates factor XIII A; MI, myocardial infarction.
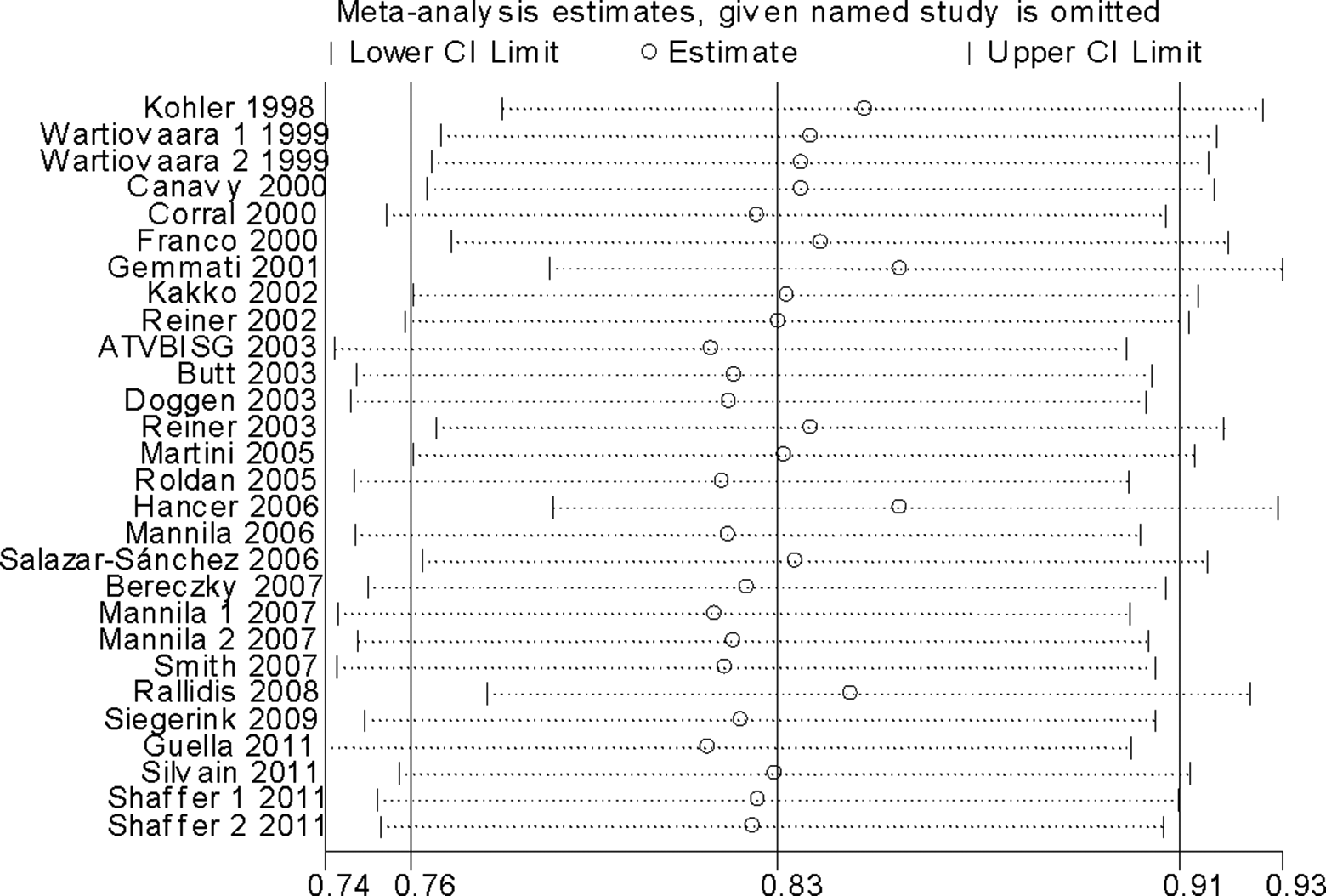
Sensitivity analysis for the FXIII-A Val34Leu polymorphism with MI risk. FXIII-A indicates factor XIII A; MI, myocardial infarction.
Heterogeneity Analysis
For MI risk, there was significant heterogeneity (I2 = 58%, P < .0001) in the dominant genetic model. The Galbraith plot was used to find the source of the heterogeneity. As shown in Figure 5, 4 studies were the outliers. 7,12,23,29 After excluding these studies, the between-study heterogeneity effectively decreased and there was no obvious heterogeneity among the 24 remaining studies (I2 = 10%, P = .33). Besides, the result was still statistically significant (OR = 0.93, 95% CI 0.87-0.98, P = .01).
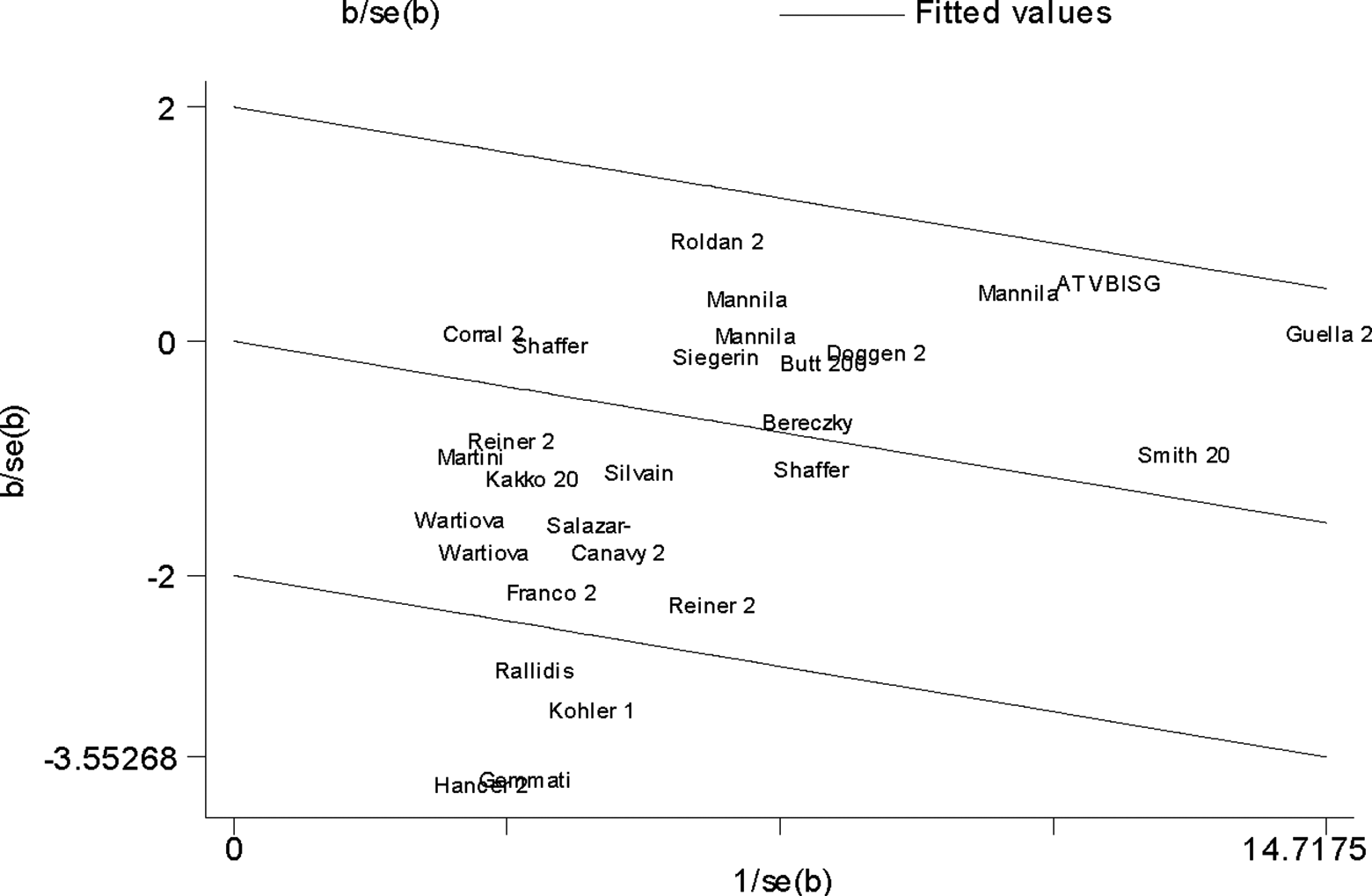
Galbraith plot of FXIII-A Val34Leu polymorphism and MI risk. FXIII-A indicates factor XIII A; MI, myocardial infarction.
Publication Bias
Publication bias was examined by the funnel plot. The shape of the funnel plot was symmetrical (Figure 6). There are 14 studies on the left side and 14 studies on the right side. However, Egger test indicated significant publication bias (P = .000).
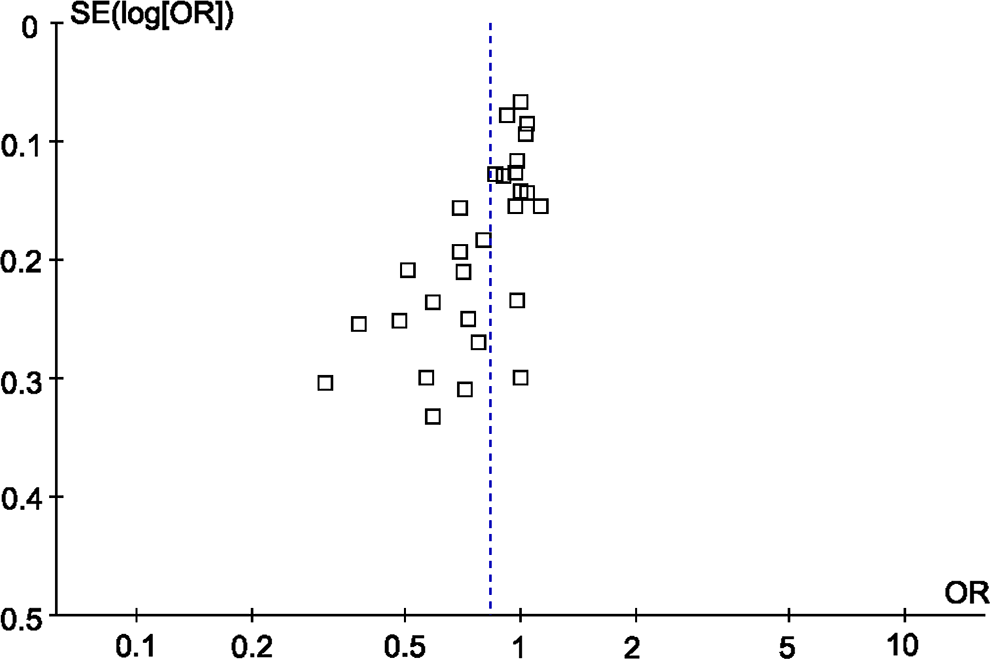
Funnel plot for MI risk and the FXIII-A Val34Leu polymorphism. FXIII-A indicates factor XIII A; MI, myocardial infarction.
Discussion
In this meta-analysis, we investigated the association between the FXIII-A Val34Leu polymorphism and MI risk, including 10 109 cases and 16 245 controls. We found that individuals with the 34Leu (Leu/Leu or Val/Leu) showed a decreased risk of MI in the overall population. The result from our meta-analysis suggested that carriers of the Leu/Leu or Val/Leu genotype had 17% decreased MI risk compared to those individuals with the Val/Val genotype. The FXIII-A Val34Leu polymorphism is common in caucasian population, with a frequency of approximately 0.25 to 0.30. 42,43 However, the frequency varies among ethnic groups, with the lowest (0.01) in Japanese and the highest (0.40) in Pima Indians. 44 In the stratified analysis by ethnicity, the significant association was observed in caucasians. Only 2 studies conducted in Chinese population were searched in our meta-analysis. Thus, meta-analysis in this population was not performed. In addition, more studies with other ethnic groups are still needed to address the role of FXIII-A Val34Leu polymorphism in MI risk. In the subgroup analysis by age, we found FXIII-A Val34Leu polymorphism exhibited decreased early-onset MI risk and late-onset MI risk. Actually, when we limited the meta-analysis to studies that controlled for age, a significant association between FXIII-A Val34Leu polymorphism and MI risk remained (OR = 0.76, 95% CI 0.63-0.90, P = .002). This result indicated that the protective role of FXIII-A Val34Leu polymorphism was not selective by age. The subgroup analysis based on sex found that this polymorphism showed decreased MI risk in female patients but not in male patients. This meta-analysis included only 7 studies using male patients, the positive association between male patients and MI could not be ruled out because studies with small sample size may have insufficient statistical power to detect a slight effect. In the future, more studies should be performed focusing on male patients with MI. When subgroup analysis was performed according to smoking status, significant associations were shown in smokers and nonsmokers. This result suggested that smoking status did not change the protective role of FXIII-A Val34Leu polymorphism in MI. Since the number of studies included in this subgroup analysis was small, the results lacked sufficient reliability to confirm or refute an association in a definitive manner. In the future, more studies should be designed to analyze these associations.
Although the mechanisms underlying the association between FXIII-A Val34Leu polymorphism and the decreased risk of MI are not clear, 2 possibilities have been suggested. First, FXIII-A 34Leu was cleaved by thrombin more rapidly and by lower doses than 34Val. 45 Faster activation of FXIII resulted in accelerated fibrin cross-linking and in a higher rate of α2 plasmin inhibitor incorporation into fibrin. 45 –48 Second, possibility was the downregulation of the adhesion of platelets to the fibrin network by cross-linking fibrin. 49
Our result was consistent with previous 2 meta-analyses. 34,35 We also found a significant association between FXIII-A Val34Leu polymorphism and MI risk. However, our study had some advantages. First, it was the first one studying the age, sex, and smoking status specificities and FXIII-A Val34Leu polymorphism interactions. Second, the methodological issues for meta-analysis, such as 1-way sensitivity analysis and cumulative meta-analysis, were well investigated. Third, the main result remained statistically significant when the adjusted ORs were combined.
Results from 1-way sensitivity analysis and cumulative meta-analysis suggested high stability and reliability of our results. Besides, we had to mention the importance of heterogeneity and publication bias, which might influence the results of meta-analysis. In our study, significant heterogeneity was observed. We used Galbraith plots to explore the sources of heterogeneity. We found that I2 value was decreased after excluding the outliers. The results suggested that the 4 outlying studies 7,12,23,29 might be the major source of the heterogeneity. However, heterogeneity did not seem to influence the results because the significance of the result was not altered after excluding the outliers. Additionally, funnel plots and Egger tests were used to find potential publication bias. The results indicated that there was significant publication bias. Thus, our result should be interpreted with caution, and more studies are still needed to confirm the effect of FXIII-A Val34Leu polymorphism on MI risk.
Our meta-analysis had some limitations that might affect the interpretation of the results. First, the numbers of published studies were not sufficient for a comprehensive analysis, particularly for Asians and Africans. Second, other than Val34Leu polymorphism, there are Tyr204Phe (rs3024477) and Pro564Leu (rs5982) variants in the FXIII-A gene. 30 We did not carry out meta-analysis on these 2 polymorphisms due to limited data. Third, lacking of the original data of the eligible studies limited the evaluation of the effects of the gene–gene interactions in MI.
Conclusions
In conclusion, this meta-analysis suggested that FXIII-A Val34Leu polymorphism may be associated with the risk of MI. Well-designed studies with larger sample size and more ethnic groups should be considered to further confirm this association. Future studies should address the potential effect modification by gender.
Footnotes
Acknowledgments
We thank John R. Shaffer, PhD (Department of Human Genetics, Graduate School of Public Health, University of Pittsburgh) and Richard P. Donahue, PhD, MPH (Department of Social & Preventive Medicine, School of Public Health and Health Professions, State University of New York) for providing relevant information.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
