Abstract
Background
Factor XIII (FXIII) deficiency increases bleeding risk and delays wound healing, yet its recognition, diagnosis, and treatment remain challenging in the complex clinical routine.
Objective
To determine real-world diagnosis and management strategies of acquired FXIII deficiency.
Methods
Adult patients undergoing FXIII activity testing (diagnostic phase) and FXIII supplementation (treatment phase) for acquired FXIII deficiency were consecutively enrolled in this retrospective data collection. Treatment outcomes were analyzed in patients with postsurgical bleeding and wound healing disorders (WHD).
Results
A total of 255 patients were included in the diagnostic cohort, of whom 84% were diagnosed with clinically relevant FXIII deficiency and 77% received FXIII supplementation. Median FXIII activity in patients with a clinically relevant deficiency was 42.5%, compared to 74.0% in those without. Most patients with FXIII activity levels below 60% were considered to have a clinically relevant deficiency. The treatment cohort comprised 96 postsurgical bleeding cases and 78 WHD cases. Median FXIII activity levels were similarly reduced in both cohorts (44.0% vs 48.0%). Conventional coagulation tests remained mainly within reference ranges for most patients, highlighting the diagnostic gap. Persistent infection was the leading comorbidity, affecting 33% of patients in both cohorts.
Conclusion
In this real-world setting, FXIII supplementation was most frequently initiated at FXIII activity levels below 60%-70%. These findings suggest using this range as a clinical reference point for therapeutic intervention, while bearing in mind the exploratory nature of the data. Routine FXIII testing should be considered in patients with unexplained bleeding or impaired wound healing, particularly perioperatively, after major trauma, or with persistent infections.
Keywords
Background
Deficiency of Factor XIII (FXIII), a crucial coagulation factor for fibrin clot stabilization,1,2 can cause serious bleeding and impaired wound healing. 3 In contrast to congenital FXIII deficiency, which has established treatment guidelines, acquired FXIII deficiency is often underrecognized and underdiagnosed.4–6 This underdiagnosis is primarily attributable to a lack of awareness and the limitations of routine coagulation testing. Standard tests such as prothrombin time (PT), activated partial thromboplastin time (aPTT) and international normalized ratio (INR) assess the initiation of fibrin clot formation but do not evaluate fibrin cross-linking. Detecting FXIII deficiency requires specific FXIII activity assays, which are not routinely used.2,4
FXIII deficiency can be managed with a highly purified plasma-derived FXIII concentrate, which is well-established for routine prophylaxis and perioperative management of surgical bleeding in both adult and pediatric patients with congenital FXIII deficiency.7,8 Evidence from several studies and clinical experiences demonstrated that supplementing FXIII in patients with low FXIII plasma levels reduces postsurgical complications such as delayed wound healing or prolonged bleeding.9–11 It also has a favorable safety profile. 8 Despite this evidence, clinical guidelines for treating acquired FXIII deficiency remain limited and inconsistent across Europe.12–16 European clinical guidelines recommend FXIII monitoring and correcting FXIII deficiency in patients with prolonged bleeding unresponsive to multimodal coagulation therapy,12,16 trauma-related coagulopathy, 15 or impaired wound healing, 16 with German guidelines advising to maintain levels above 60% for managing polytrauma and peripartum hemorrhage.17,18 In contrast, the Scientific and Standardized Committee (SSC) of the International Society of Thrombosis and Hemostasis (ISTH) advises against the routine use of FXIII supplementation. 13
No consensus exists on the FXIII activity level indicating a clinically relevant deficiency or the threshold for initiating supplementation. Kleber et al proposed a cut-off value of less than 50%-70% of FXIII activity. 4 Additionally, clinical evidence suggests that acquired FXIII deficiency is particularly associated with certain risk scenarios, including major surgery, 9 trauma,4,19 increased consumption due to septic shock, 20 and synthesis disorders such as liver dysfunction. 6 However, large clinical trials with robust data on the diagnostic thresholds and specific risk factors for acquired FXIII deficiency remain scarce.
This multicenter registry aims to describe real-world diagnostic and treatment patterns for acquired FXIII deficiency, identify its clinical risk factors, and provide practical guidance for testing and supplementation.
Methods
Study Design
This multicenter, non-interventional, retrospective, registry data collection was conducted across nine expert centers in Germany between November 21st, 2022, and April 10th, 2024. The participating surgical specialties included orthopedics and traumatology, gynecology and obstetrics, neurosurgery, and cardiothoracic surgery. Adult patients undergoing FXIII activity testing (diagnostic phase) and FXIII supplementation (treatment phase) for acquired FXIII deficiency were consecutively enrolled. Patient data were collected using standardized case report forms.
Ethical approval was obtained from the Ethics Committee of the Medical Faculty of the University of Heidelberg before the study commenced (S-970/2021, Ethics Commission Heidelberg, dated February 4th, 2022; Trial registration: DRKS00030706). Data collection adhered to the principles of the Declaration of Helsinki.
Participants
The study population was analyzed as two distinct cohorts, with patient selection detailed in Figure 1. The diagnostic cohort included adult inpatients who underwent FXIII activity testing. The treatment cohort comprised adult patients who received FXIII supplementation. Patients with congenital FXIII deficiency were excluded from the study.
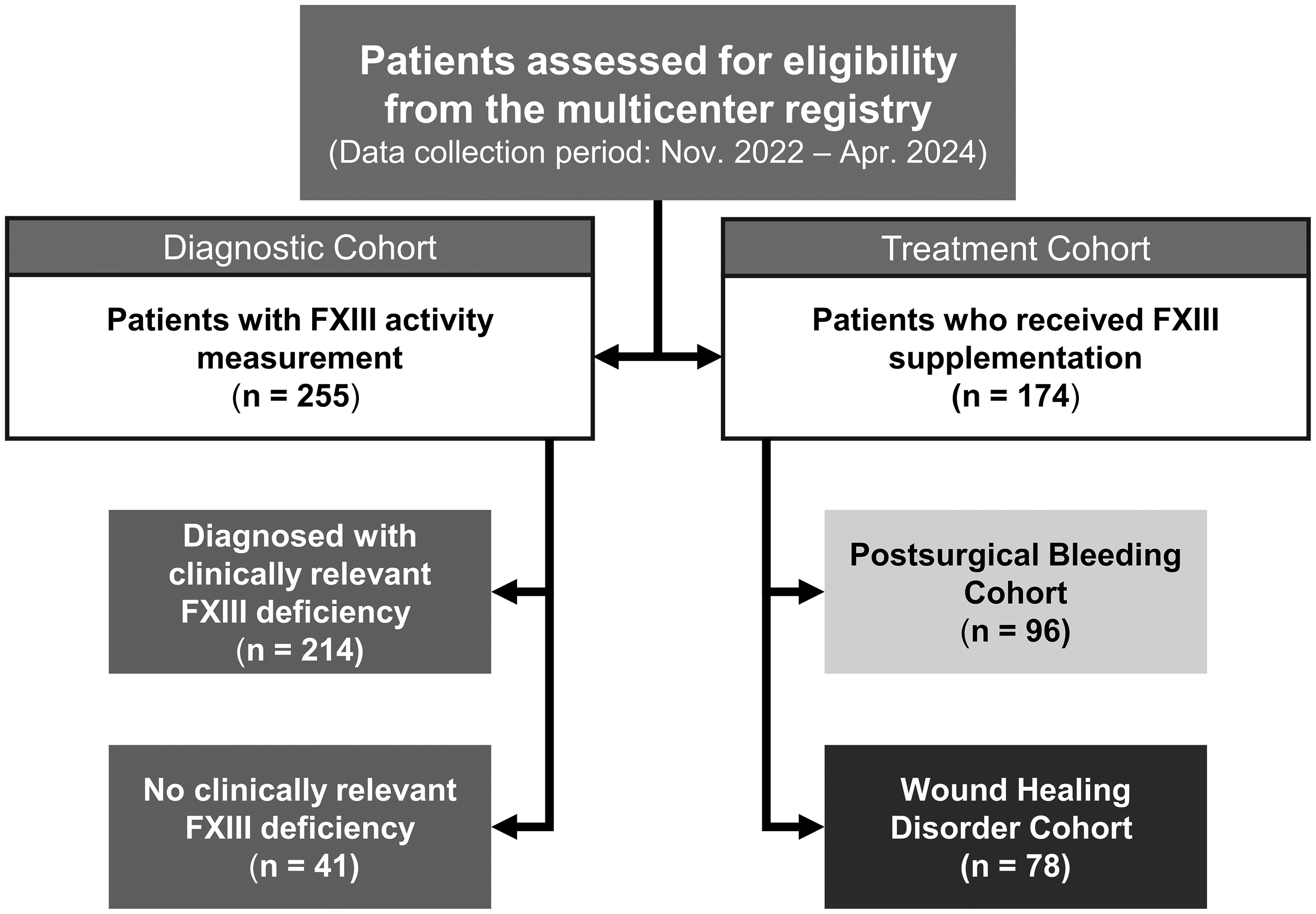
Flow diagram of patient selection: the diagram illustrates the selection of patients for two distinct analyses from the multicenter registry. The diagnostic cohort includes all patients prospectively screened for FXIII deficiency within a defined time frame. The treatment cohort includes all patients retrospectively identified as having received FXIII supplementation for specific indications. Abbreviation: FXIII, Factor XIII.
Assessment Using Standardized Forms
Diagnostic Phase
A standardized questionnaire was used to document the real-world management of suspected FXIII deficiency.
Physicians documented data from consecutive patients with FXIII laboratory results. The following parameters were recorded: baseline characteristics (eg age, sex), date of first FXIII measurement, frequency and reason for FXIII testing, lowest measured FXIII activity level, and potential causes of FXIII deficiency. Additionally, clinicians reported whether they suspected clinically relevant FXIII deficiency and whether the patient received FXIII supplementation.
Investigators assessed a “clinically relevant FXIII deficiency” based on a synthesis of laboratory evidence (low FXIII activity) and clinical signs (eg presence of active bleeding, blood loss, hemoglobin drop, or transfusion requirements) or impaired wound healing that could not be explained by other coagulopathies.
Treatment Phase
Separate questionnaires were applied to document treatment modalities and outcomes in patients receiving FXIII supplementation for postsurgical bleeding or wound healing disorders (WHD). Data collection included baseline demographics (eg age, sex, weight, height), relevant medical history, and coagulation-related blood parameters (FXIII activity, INR, PTT, fibrinogen, and platelet count) before and after FXIII supplementation. Comorbidities assessed were diabetes, hepatic insufficiency, renal insufficiency, and infections. Documented infections included Staphylococcus, Streptococcus, Enterococcus, Bacillus cereus, Citrobacter freundii, Candida, Helicobacter, hepatitis B or C, sepsis, bacterial pneumonia, and Fournier's gangrene.
For WHD patients, additional parameters were recorded, including bleeding history prior to WHD, the cause of the wound, and any treatments with procoagulants prior to FXIII supplementation. Laboratory evaluations included alanine aminotransferase (ALAT), aspartate aminotransferase (ASAT), creatinine (KREA), hemoglobin (Hb), albumin, C-reactive protein (CRP), and leukocyte count.
For the postsurgical bleeding cohort, records captured perioperative bleeding risk, the surgical procedure, any intraoperative complications, and hemostatic drugs or blood products administered perioperatively.
FXIII Measurement
FXIII levels were assessed using one of two established methods depending on the participating center: FXIII activity was determined by a chromogenic method (Berichrom® F XIII; Siemens, Forchheim, Germany), while antigenic FXIII A-subunit levels were measured by a latex-enhanced immunoassay (HemosIL®; Werfen, Barcelona, Spain). Although the latter quantifies the concentration of the catalytic A-subunit rather than direct enzymatic functionality, results from this antigen assay have been shown to correlate well with activity assays in patients without FXIII inhibitors. 21 Specifically, 5 centers have used the Berichrom® F XIII test and 4 centers used the HemosIL® test. In the following analysis, results from both assays are referred to as FXIII activity for readability, while acknowledging that antigen levels serve as a surrogate for activity in this clinical setting. FXIII measurements were performed at the discretion of the treating physician, typically triggered by the onset of bleeding symptoms, diagnosis of wound healing disorders, or as part of routine postoperative monitoring (days 0-5) in high-risk surgical patients.
Statistical Analysis
Data analysis was primarily descriptive. Continuous variables were assessed for normal distribution using the Shapiro-Wilk test and, due to non-normal distribution across most parameters, are reported as medians with interquartile ranges (IQR) and sample sizes. Categorical variables are presented as absolute counts and percentages.
For inferential statistics, subgroup comparisons of continuous variables were conducted using the non-parametric Kruskal-Wallis test, followed by Dunn's post-hoc test for pairwise comparisons where appropriate. A P-value of <.05 was pre-specified as threshold for statistical significance. Given the exploratory nature of some subgroup analyses, no formal adjustments for multiple comparisons were made. Therefore, these findings should be considered hypothesis-generating.
As the study's primary objective was to describe real-world clinical patterns rather than to test a pre-specified hypothesis, a formal sample size calculation was not performed. The sample size was determined by the consecutive enrollment of all eligible patients within a defined time frame to ensure a representative cohort.
Missing values were documented and not imputed. Given the retrospective, observational design, sensitivity or fragility analyses were not performed. To minimize selection bias, participating centers were instructed to enroll consecutive patients who met the eligibility criteria during the defined study period.
All statistical analyses were conducted using SPSS® version 22.0 (IBM Deutschland GmbH, Ehningen, Germany) and GraphPad Prism version 10.3.1 (GraphPad Software, Boston, US).
Results
Demographic Data
The diagnostic cohort of the study included 255 patients. In the treatment cohort, 96 patients were assigned to the postsurgical bleeding cohort, and 78 patients to the WHD cohort (Figure 1). The median age and sex distribution, with a higher proportion of males, were comparable between the cohorts. The median body mass index (BMI), recorded only in the treatment cohort, was slightly higher in patients with WHD compared to those with postsurgical bleeding (Table 1).
Patient Demographics by Study Cohort.
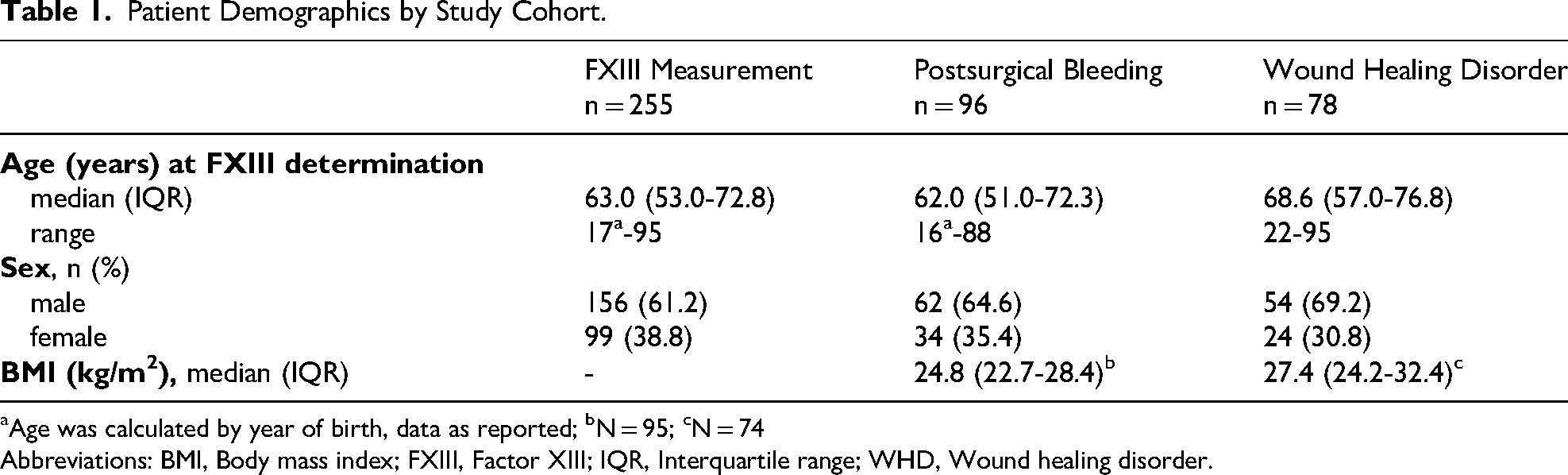
Age was calculated by year of birth, data as reported; bN = 95; cN = 74
Abbreviations: BMI, Body mass index; FXIII, Factor XIII; IQR, Interquartile range; WHD, Wound healing disorder.
Diagnostic Phase: FXIII Measurement
Reasons for FXIII Measurement
The most common reason for FXIII measurement was bleeding (56.9%), followed by WHD and infections. Perioperative status, including postoperative control with or without bleeding, preoperative control and tumor surgery, was cited as the reason in 10.2% of cases, while trauma accounted for 5.9% of cases (Figure 2A).

Diagnostic phase: (A) reasons for FXIII measurement and (B) potential causes for FXIII deficiency. Multiple answers were possible. Other reasons for FXIII measurement: extracorporeal life support/extracorporeal membrane oxygenation (1.6%), cardiogenic shock (1.2%), and anemia (1.2%). Other potential causes for FXIII deficiency: infections (3.7%), organ inflammation (1.4%), tumors (1.4%), pancreatitis or peritonitis (1.4%), multiorgan failure (0.9%), subdural hematoma (0.5%), and thrombocytopathy (0.5%). Percentages refer to n = 255. Abbreviations: FXIII, Factor XIII, Nmiss, Patients with missing data.
Prevalence and Potential Causes of Clinically Relevant FXIII Deficiency
Clinically relevant FXIII deficiency was identified in 214 of the 255 patients (84%) in the diagnostic cohort. Of these deficient patients, 196 (92%) received FXIII supplementation, corresponding to 77% of the total diagnostic cohort. The most frequently documented potential cause of FXIII deficiency was external bleeding in half of the patients, followed by increased consumption and impaired FXIII synthesis. In one in five cases, the cause of FXIII deficiency remained unknown (Figure 2B).
FXIII Activity Measurement
A graphic displays stratified patients by lowest FXIII activity, comparing those with and without clinically relevant FXIII deficiency (Figure 3A). Most patients with a median FXIII activity below 60% (n = 192) were diagnosed with clinically relevant FXIII deficiency, while only 11 patients without clinically relevant deficiency (5.7%) had FXIII activity below 60%. In contrast, most patients with FXIII activity above 70% were not considered to be clinically relevant FXIII-deficient (23 patients without vs 1 patient with clinically relevant deficiency). For patients with FXIII activity between 60%-70%, the majority were considered to have clinically relevant FXIII deficiency (21 patients with vs 7 patients without clinically relevant deficiency) (Figure 3A). The single patient considered to have a relevant deficiency despite activity levels >70% presented with severe, unexplained hemorrhage, leading the clinician to prioritize the severe clinical bleeding phenotype over the isolated laboratory value. The overall median of lowest FXIII activity was 46.8% (IQR: 36-57). Patients with clinically relevant FXIII deficiency had a significantly lower median FXIII activity compared to those without clinically relevant deficiency (P < .0001) (Figure 3B).
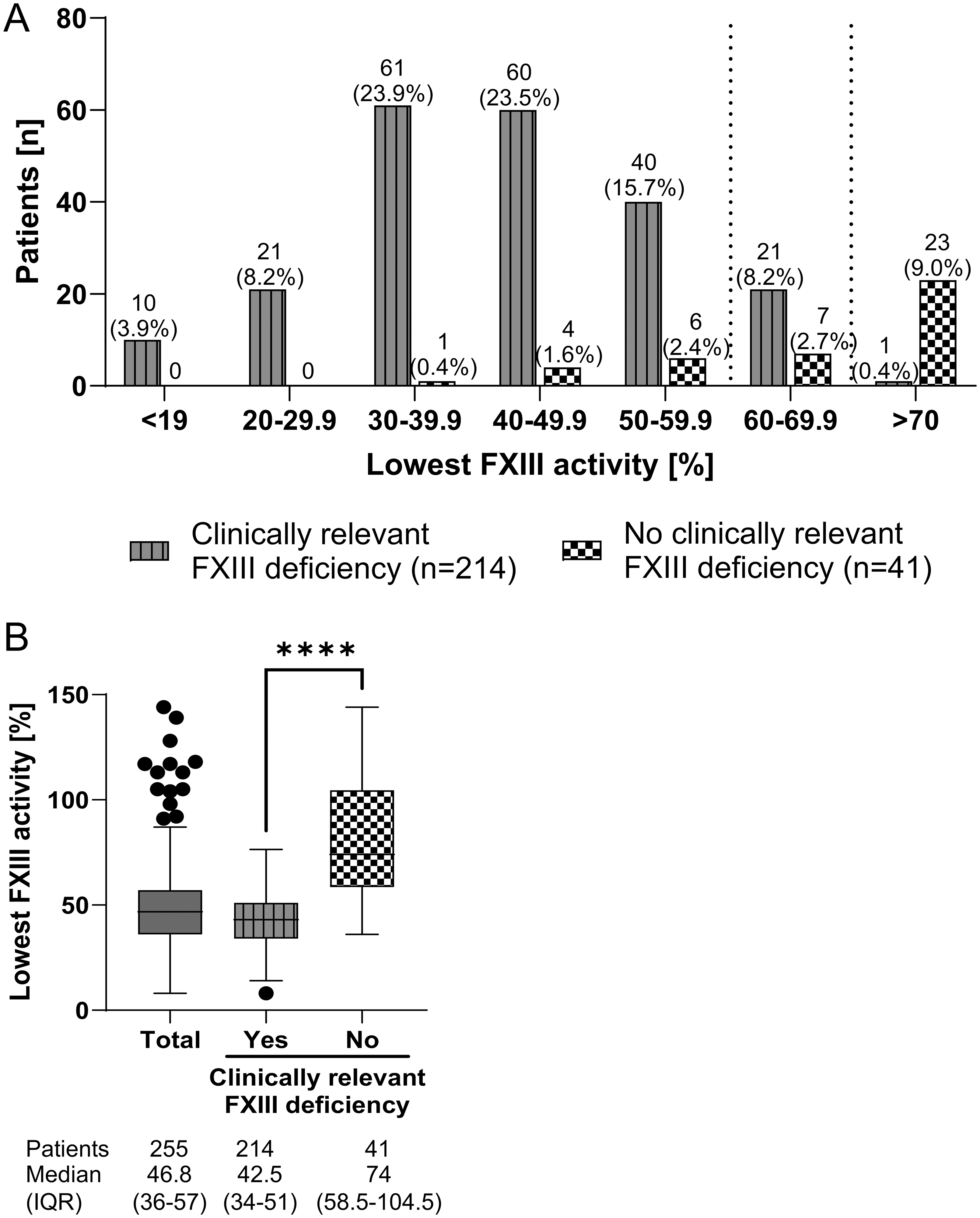
Diagnostic phase: (A) number of patients with and without clinically relevant FXIII deficiency stratified by lowest FXIII activity. Percentages refer to n = 255. (B) Lowest FXIII activity in total patient cohort and in patients with and without clinically relevant FXIII deficiency. Shown is the median with IQR. ****P < .0001. Abbreviations: FXIII, Factor XIII; IQR, Interquartile range.
Treatment Phase: Causes and Risk Factors for an Acquired FXIII Deficiency
WHD: Wound Cause
Surgery was the leading cause of the wound in the WHD cohort. This was followed by wound infections in 29.5% of cases, of which 82.6% were postsurgical patients (Figure 4A).

Treatment phase: (A) wound cause in the WHD cohort (n = 78). Multiple answers were possible. Percentages refer to n = 78. (B) Type of surgery in the postsurgical bleeding cohort (n = 96). Percentages refer to n = 96. Abbreviations: Nmiss, Patients with missing data; WHD, Wound healing disorder.
Postsurgical Bleeding: Reasons for Bleeding
Most patients in the postsurgical bleeding cohort underwent general surgery, cardiothoracic surgery and orthopedic/trauma surgery (Figure 4B). A total of 17.7% of patients required dialysis. The majority of these patients (11 out of 17 patients) had undergone cardiothoracic surgery and 9.4% received extracorporeal membrane oxygenation (ECMO)/extracorporeal life support (ECLS). Moreover, 17.7% of patients required the support of a heart-lung machine (all cardiothoracic surgery).
Medical History of Indications Associated with Increased Bleeding Risk and Comorbidities
Most patients had no history of major bleeding prior to the WHD (52.6%). Accordingly, 51.3% of patients with WHD were treated with FXIII alone and no other procoagulant was administered prior to FXIII supplementation.
In the postsurgical bleeding cohort, half of the patients had anamnestic findings suggestive of increased bleeding risk. The majority of these patients were receiving anticoagulant therapy, including antiplatelet therapies (Table 2). Additionally, 62.5% of patients experienced intraoperative complications, of which the majority had severe bleedings, which required transfusions (Table 2).
Treatment Phase: Postsurgical Bleeding Cohort: Medical History of Indications Associated with Increased Bleeding Risk and Intraoperative Complications in Patients with Postsurgical Bleeding.

Multiple answers were possible. bThe percentages are based on patients with a medical history that indicates an increased risk of bleeding (n = 50). cOthers include: increased bleeding tendency (rebleeding during previous surgical interventions/generally increased bleeding tendency in the patient/family (5.2%)), liver diseases (alcoholic liver cirrhosis/liver failure (2.1%)), upper gastrointestinal hemorrhage (1.0%) and ECMO (1.0%). dThe percentages are based on patients with intraoperative complications (n = 60). Abbreviation: ECMO, Extracorporeal membrane oxygenation.
Comorbidities
In both cohorts, infection was the most common comorbidity (33.3%), followed by diabetes, renal and hepatobiliary disorders. While the prevalence of diabetes and hepatobiliary disorders was comparable between the postsurgical bleeding cohort and the WHD cohort, more cases of renal and urinary disorders were reported in the postsurgical bleeding cohort (29.2%) than in the WHD cohort (21.8%) (Figure 5).

Treatment phase: comorbidities of patients with postsurgical bleeding (light gray) and wound healing disorders (WHD) (black). Multiple answers were possible. Percentages are based on the respective patient cohort (postsurgical bleeding: n = 96; WHD: n = 78). Abbreviation: WHD, Wound healing disorder.
Coagulation-Related Laboratory Parameters
As expected, FXIII activity was low in patients before FXIII supplementation. The median FXIII activity was 44.0% (IQR: 35-51) in the postsurgical bleeding versus 48.0% (IQR: 36-56) in the WHD cohorts. Most other coagulation-related parameters were within reference ranges. Both cohorts had comparable levels of INR and PTT, while fibrinogen level (P = .119) and platelet counts (P = .057) were higher in the WHD than in the postsurgical bleeding cohort. In the WHD cohort, median fibrinogen levels were moderately elevated, while in the postsurgical bleeding cohort, median platelet counts were slightly below the reference range (Table 3).
Treatment Phase: Laboratory Parameters Before Supplementation by Cohort.

Median time between analysis and supplementation (Range): Postsurgical bleeding: 0 (0-45) days; WHD: 1 (0-62) days.
Abbreviations: CRP, C-reactive protein; FXIII, Factor XIII; INR, International normalized ratio; IQR, Interquartile range; PTT, Partial thromboplastin time (PTT); RR, Reference range.
WHD Cohort: Correlation of Infection and Laboratory Parameters
Most infections in the WHD cohort were caused by cocci (15 of 26 patients, 57.7%), including 9 patients (34.6%) with staphylococcal, 8 patients (30.8%) with enterococcal and 3 patients (11.5%) with streptococcal infections.
Several laboratory parameters correlated with infections, particularly coccal infections. As expected, white blood cell counts and CRP levels were higher in patients with infections compared to those without infections. Median CRP levels were significantly higher in patients with coccal infections compared to those with other infections (P = .021). In addition, fibrinogen levels tended to be higher in patients with coccal infections compared to those with other infections (Table 4). Median FXIII activity was low in all subgroups regardless of the infection status.
Treatment Phase: Laboratory Parameters in the Cohort with Wound Healing Disorders Before Supplementation Stratified According to Infection status.

Significant differences with P < .05 for leukocyte count (cohort without infections vs with infections) and for CRP (cohort with coccal infections vs other infections).
Abbreviations: CRP, C-reactive protein; FXIII, Factor XIII; INR, International normalized ratio; IQR, Interquartile range; PTT, Partial thromboplastin time (PTT); RR, Reference range; WHD, Wound healing disorder.
Among the 78 patients in the WHD cohort, 38.5% received multiple doses of FXIII supplementation. However, while only 17.7% of patients without infections received multiple doses of FXIII, 61.5% of patients with infections required multiple doses of FXIII supplementation. Among those with infections, 80% of patients with coccal infections compared to 36.4% with other infections were given multiple doses of FXIII supplementation.
Discussion
Acquired FXIII deficiency is frequently underestimated and unrecognized, increasing the risk of secondary hemorrhage and delayed wound healing. Data from this registry characterizes the clinical settings, risk factors, typical clinical presentation, and real-world management of acquired FXIII deficiency, aiming to support targeted testing and improve hemostatic management.
In the diagnostic cohort, 84% of tested patients were assessed as having a clinically relevant FXIII deficiency, indicating a high degree of diagnostic experience and confidence among experts. Although external blood loss explained several cases, the underlying cause often remained unclear, underscoring the need for FXIII testing in patients with diffuse or unexplained bleeding. This could help identify previously undetected cases and treat patients more effectively. 22 However, there is currently no clearly defined cut-off level for FXIII activity that would indicate the need for supplementation in acquired FXIII deficiency. A previous publication by Kleber et al has highlighted that an FXIII activity of <50%-70% may be appropriate to diagnose acquired FXIII deficiency. 4 Our findings reveal that in this expert setting an activity threshold of 60%-70% was predominantly used to diagnose a clinically relevant deficiency in high-risk patients. This observation is consistent with current interdisciplinary guidelines, such as the German S3 Guidelines on Polytrauma and Peripartum Hemorrhage, which recommend maintaining FXIII activity >60% in bleeding patients to ensure clot stability. Furthermore, prospective data from high-risk populations, such as neurosurgery, have identified postoperative activity levels <60% as an independent predictor for re-bleeding. It is important to note that these findings are based on the collective clinical judgment of experienced practitioners in a retrospective setting rather than a prospectively validated diagnostic cut-off. Therefore, this threshold should be regarded as a real-world evidence-based reference point for future interventional trials rather than a definitive diagnostic standard.
To identify FXIII deficient patients, clinicians should consider FXIII testing in specific high-risk situations that could be caused by FXIII deficiency. The results from the diagnostic phase of the study indicate that FXIII deficiency is often suspected in patients with bleeding and WHD. In particular, surgery was identified as a major risk factor for acquired FXIII deficiency. The most common surgical interventions in the postsurgical bleeding cohort were general or cardiothoracic surgeries, which were also surgeries associated with ECLS. However, the high incidence of cardiothoracic surgery may be influenced by the routine measurement of FXIII prior to these procedures.23,24 Activation of the coagulation system by extracorporeal circulation, such as heart-lung machine support/ECMO, can lead to increased consumption of coagulation factors. In particular, the plastic surface of the ECLS cannula and tubing can promote coagulation through fibrin deposition and ultimately overconsumption of coagulation factors, while ongoing hyperfibrinolysis contributes to the bleeding phenotype.23,24
Furthermore, therapeutic interventions such as plasmapheresis, often utilized in the context of renal transplantation or rejection treatment, can induce significant acquired FXIII deficiency due to the physical removal of plasma proteins, representing another distinct high-risk group that warrants monitoring. 21
Additionally, FXIII has the longest biological half-life of any coagulation factor, at 120-200 h. 25 The high intraoperative blood loss and the slower secretion kinetics may impede compensation for sudden drops in FXIII activity. Specific attention should therefore be paid to patients with ECLS, as they appear to be at particular risk of FXIII deficiency.
In line with current literature, trauma was reported in all three patient cohorts (between 6% in the diagnostic phase; 12% (postsurgical bleeding) and 20% (WHD) in the treatment phase).4,10,19 In particular, trauma patients with massive soft tissue injury or traumatic brain injury were shown to be at significant risk for FXIII deficiency.19,26–28 In the early stages of trauma-induced coagulopathy, hypocoagulability may occur due to significant bleeding preventing effective hemostasis.
In general, there is little data on the impact of infections in general on FXIII deficiency. As part of the innate immune response, coagulation can enable the immobilization and killing of streptococci by cross-linking the bacteria to fibrin via FXIII. 29 Persistent infections could therefore lead to excessive consumption of FXIII, as demonstrated in a previous case study. 30 Data from a mouse model demonstrated that a local activation of FXIII reduces systemic complications and improves survival after a skin infection with Staphylococcus pyogenes. 31 Our cohort confirms an association between infection and acquired FXIII deficiency. It was the most frequent comorbidity (33%) and most WHD patients had postoperative wound infections. Notably, within the WHD group, coccal infections triggered a more pronounced acute-phase response and necessitated repeated FXIII supplementation far more often than other pathogens (80% vs 36%), implicating them as a key risk factor for persistent FXIII deficiency. A significant drop in FXIII in patients with persistent infections can precede changes in standard coagulation tests, likely reflecting an early-stage consumptive coagulopathy where FXIII is a more sensitive marker.
Of note, more than half of the patients in the WHD cohort received FXIII alone and no other procoagulant prior to FXIII supplementation. This indirectly indicates and supports the hypothesis of an isolated acquired FXIII deficiency with clinical relevance.
It should be noted that potential confounders, such as underlying liver disease, anticoagulant therapy, and surgical complexity, were not adjusted for in a multivariate model. Therefore, the observed associations may be influenced by these factors, and the results should be interpreted as exploratory.
Limitations
The primary methodological limitation is the post-hoc, investigator-driven definition of ‘clinically relevant’ deficiency, creating a risk of confirmation bias. Consequently, our findings on activity thresholds should be interpreted as a description of clinical practice, not as validated diagnostic criteria. A major limitation is a selection bias, as the registry only included patients selected for FXIII testing based on clinical suspicion. Consequently, the high prevalence of deficiency (84%) reflects a pre-selected, high-risk population and cannot be generalized to the broader, unselected critical care population. The study design also underrepresents certain high-risk groups like neurosurgery or burn patients.32,33
Additionally, comprehensive coagulation profiles (INR, aPTT, fibrinogen, platelet count) were not systematically collected for the entire diagnostic cohort. However, this data was available and analyzed for the treatment cohorts. This finding reinforces the concept of the diagnostic gap, where standard tests fail to predict FXIII deficiency.
A further limitation arises from the multicenter design, where different assays were used to quantify FXIII. While some centers utilized chromogenic activity assays, others used antigenic immunoassays. Although these methods correlate well in acquired deficiency states resulting from consumption or reduced synthesis, applying both may lead to variability, especially if rare functional defects or inhibitors would be present. 21 Furthermore, heterogeneity existed between centers regarding specific supplementation protocols and local thresholds for intervention, reflecting the diversity of clinical practice in the absence of unified guidelines.
Specific safety outcomes and adverse events related to FXIII supplementation were not systematically collected or adjudicated in this retrospective registry, making a formal safety analysis infeasible.
Finally, no pre-specified sensitivity analyses were performed to test the robustness of the findings. Such analyses should be integral to future trials aiming to validate the risk factors identified herein.
Implications
In summary, the results of this study confirm the need for FXIII deficiency screening in patients with massive bleeding, substantial soft tissue injury,4,19,26 coccal infections, or unexplained delayed wound healing with diffuse bleeding after major and/or frequent surgery 22 or large wounds. Moreover, FXIII deficiency is associated with specific risk factors such as high intraoperative blood loss, persistent infections (especially with cocci), extracorporeal procedures (especially after cardiothoracic surgery), and impaired liver function. Acquired FXIII deficiency should be considered if these risk factors are prevalent in a patient. In these clinical situations and when FXIII activity is less than 60%-70%, FXIII supplementation should be considered (Figure 6). 4
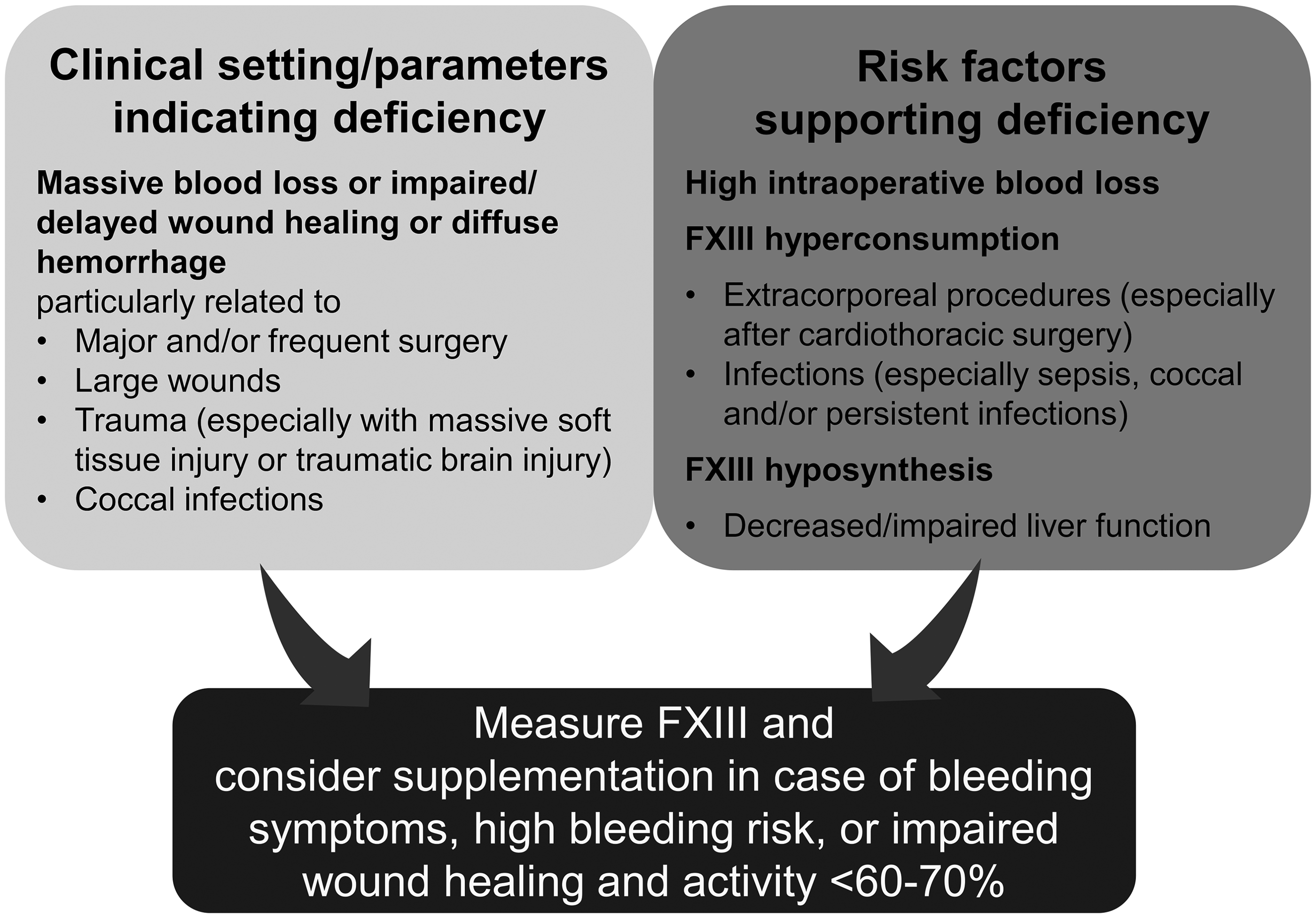
Clinical setting indicating and risk factors supporting an acquired FXIII deficiency. Abbreviation: FXIII, Factor XIII.
Conclusions
This study provided valuable insights into the clinical presentation of FXIII deficiency and its risk factors. Due to the registry-based study design, the results should be interpreted as exploratory. We observed that clinicians in this real-world setting primarily used FXIII activity levels below 60%-70% as the diagnostic threshold to diagnose clinically relevant FXIII deficiency. FXIII testing should be considered in patients with bleeding complications and impaired wound healing of unknown etiology, as well as in the perioperative period, following major trauma or persistent infections. High intraoperative blood loss, ECLS, especially after cardiothoracic surgery, and impaired liver function are specific risk factors that can support the development of FXIII deficiency.
Furthermore, many patients with persistent FXIII deficiency had high infection rates, particularly with cocci, suggesting infections could be associated with FXIII deficiency. Our data suggest that FXIII can be a critical early marker for an evolving consumptive coagulopathy, often masked by normal coagulation tests. The presented results are intended to raise awareness of the importance of routine clinical FXIII testing in specific risk constellations.
Supplemental Material
sj-docx-1-cat-10.1177_10760296261440412 - Supplemental material for Acquired Factor XIII Deficiency in Critical Care: Registry-Based Analysis of Features, Symptoms and Risk Factors
Supplemental material, sj-docx-1-cat-10.1177_10760296261440412 for Acquired Factor XIII Deficiency in Critical Care: Registry-Based Analysis of Features, Symptoms and Risk Factors by Michael Hetz, Michaela Wilhelmi and Lutz Mirow, Richard K. Ellerkmann, Alice Bernard, Ingvild Birschmann, Thorsten Perl, Nils Kunze-Szikszay, Hartmut Heinze, Felix C.F. Schmitt, Christian Kleber in Clinical and Applied Thrombosis/Hemostasis
Footnotes
List of Abbreviations
Acknowledgements
The authors thank all patients and their families and caregivers for their contribution to the study, together with all investigators and study teams. The authors thank all participating centers and study teams for their contributions. All centers were compensated per patient.
ORCID iDs
Ethics Approval
Ethical approval was obtained from the Ethics Committee of the Medical Faculty of the University of Heidelberg before the study commenced (S-970/2021, Ethics Commission Heidelberg, dated February 4, 2022). Trial registration: DRKS00030706.
Consent to Participate
The requirement for informed consent to participate was waived by the Ethics Committee of the Medical Faculty of the University of Heidelberg (S-970/2021) due to the retrospective, non-interventional design of the registry.
Consent for Publication
Not applicable.
Authors’ Contributions
All authors read and approved the final manuscript and have agreed both to be personally accountable for the author's own contributions and to ensure that questions related to the accuracy or integrity of any part of the work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by CSL Behring, Hattersheim, Germany. The sponsor was not involved in the data collection and data analysis. Medical writing assistance was provided by Dr Alexandra Ehrens (med:unit GmbH, Germany) and was funded by CSL Behring. The authors had full editorial control over the manuscript content and approved the final version for submission.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Authors received financial compensations from CSL Behring for the contribution of patient data to the study registry and for participating in advisory boards related to the study.
Data Availability
The datasets used and analyzed during the current study are available from the corresponding author on reasonable request.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
