Abstract
Dysfunctional T-lymphocyte immunity plays an important role in the pathophysiology of immune thrombocytopenic purpura (ITP). Cytotoxic T-lymphocyte antigen 4 (CTLA-4)—a surface marker expressed on T regulatory cells and activated T lymphocytes—is a negative modulator of T-cell responses. Polymorphisms of the CTLA-4 may alter the level of antigen expression and hence may influence immune regulation. The study aimed to evaluate the possible contribution of CTLA-4 exon 1 49 A>G polymorphism to the pathogenesis of ITP and its relation to age of disease onset, clinical course, and response to therapy. Genotyping of CTLA-4 exon 1 49 A>G was performed in 100 pediatric patients with ITP and 259 healthy individuals by polymerase chain reaction–restricted fragment length polymorphism. No significant differences existed in genotype or allele distributions between patients and controls for the studied polymorphism. Comparable genotypes and allele frequencies were obtained between the 2 groups after their stratification by age of disease onset, clinical course, or response to therapy. In conclusion, CTLA-4 exon 1 49 A>G polymorphism is not associated with susceptibility to ITP in the Egyptian population; neither it affects the clinical picture of the disease.
Introduction
Immune thrombocytopenic purpura (ITP) is an acquired organ-specific autoimmune disease characterized by the production of autoantibodies that mediate platelet destruction. 1 Although autoreactive B lymphocytes are considered the primary immunologic defect, dysfunctional T-lymphocyte immunity and regulatory T cells play an important role in the pathophysiology of ITP. 2
Cytotoxic T-lymphocyte antigen 4 (CTLA-4; CD 152) molecule is an important negative regulator of T-cell immune response. It is normally expressed as a surface marker on T regulatory cells and on activated T lymphocytes. 1 Due to its higher affinity for B7, CD80, and CD86, it can reduce the CD28/B7 interactions and compete with CD28 for ligands on antigen-presenting cells, thus inhibiting further T-cell proliferation. 3 Furthermore, CTLA-4 has independent inhibiting properties through its ability to downregulate T-cell responses in the absence of CD28. 4 Several studies reported the involvement of CTLA-4 in the pathogenesis of autoimmune diseases such as systemic lupus erythematosus, autoimmune thyroid disease, type I diabetes, and rheumatoid arthritis. 5 –9
Many single-nucleotide polymorphisms (SNPs) have been identified in the CTLA-4 region; among those the −318, +49, and CT60 polymorphisms have been implicated with autoimmune diseases. 10 –12 Few studies have addressed this issue both in children and in adults, with no evidence of association between any of the studied polymorphisms and the risk of ITP. 13,14 Nonetheless, risk susceptibility studies cannot be extrapolated from one ethnic group to the other due to the well-known variability of the relative frequency of the different alleles among different populations. Furthermore, this study included only adult patients and did not address the impact of the CTLA-4 +49 gene polymorphism on age of disease onset, its clinical course, and response to different lines of therapy, which could lend an in-depth insight into CTLA-4 involvement in the disease. Thus, the aim of this work was to study the CTLA-4 +49 in an Egyptian cohort, compare the relative frequency of the different alleles in pediatric patients with ITP to that in the normal population, verify whether any of the alleles could contribute to risk susceptibility to ITP, and to evaluate the possible impact of CTLA-4 +49 polymorphism on the age of disease onset, clinical course of the disease as well as response to different lines of therapy.
Methods
Participants
The study was carried out in accordance with the Helsinki Declaration. The hospital-based ethics committee approved the study. With informed consent from participants and/or their parents, the study was conducted on 100 pediatric patients with ITP including 42 males and 58 females with a mean age of 7.3 ± 4.4, a range of 18 months to 17 years and a median of 6 years randomly selected from the Haematology Clinic of New Children’s Cairo University Hospital. Patients with ITP were clinically evaluated and diagnosed according to standard methods and treated according to the international consensus report on the investigation and management of primary immune thrombocytopenia. 15 Clinically the patients were classified according to different parameters of the disease adopted from the “Standardization of terminology, definitions and outcome criteria in immune thrombocytopenic purpura of adults and children: report from an international working group.” 16 The severity at time of diagnosis was assessed according to the presence or absence of bleeding; 57% of cases were bleeders and 43% were nonbleeders. At the time of sampling, 15% of the cases were bleeders and 85% were nonbleeders. A total of 259 healthy adults including 137 males and 122 females were used as control group with a mean age of 44.8 ± 15.3 years and a median of 52 years. The control participants were healthy unrelated individuals with no history of ITP or autoimmune disorders. The choice of healthy adults as a control group was based on ethical restrictions to use healthy children as control. In addition, as the present study involved the detection of a genetic polymorphism that does not change with age, the higher age group served as a better control eliminating the possibility of future development of the disease during childhood.
DNA Extraction
Peripheral blood specimens were collected from all participants in vacuum tubes containing EDTA. Genomic DNA was extracted using the Fermentas Life sciences Genomic DNA Purification kit (Cat No. # K0512; Fermentas,Thermo Fisher Scientific Inc., Dublin 1 Ireland), according to the manufacturer’s instructions.
The CTLA-4 Genotyping
The CTLA-4 exon 1 49 A>G polymorphism was analyzed using the polymerase chain reaction–restriction fragment length polymorphism (PCR-RFLP) method: the PCR reaction was performed in a 25-µL mix including 50 to 150 ng of genomic DNA, 10 pmol/L of each primer (forward primer 5′-CCACGGCTTCCTTTCTCGTA-3′ and reverse primer 5′-AGTCTCACTCACCTTTGCAG-3′), 2.5 µL of 10× standard Taq reaction buffer, 0.5 µL of 10 mmol/L deoxynucleotides (Cat No. NO447S; New England BioLabs, Beverly, MA, USA) and 0.125 µL of Taq DNA polymerase (Cat No. MO273S; New England BioLabs, Beverly, MA, USA). Cycling conditions included denaturation at 94°C for 5 minutes, followed by 40 cycles of 30 seconds at 94°C, 30 seconds at 60°C, and 30 seconds at 72°C, and then a final extension for 10 minutes at 72°C. Cleavage was performed with the restriction enzyme BbvI (FastDigest; Fermentas Life sciences), according to the manufacturer’s protocols. Digested fragments were separated on 3% agarose gels and RFLP bands were visualized by ethidium bromide staining under ultraviolet light (Figure 1).
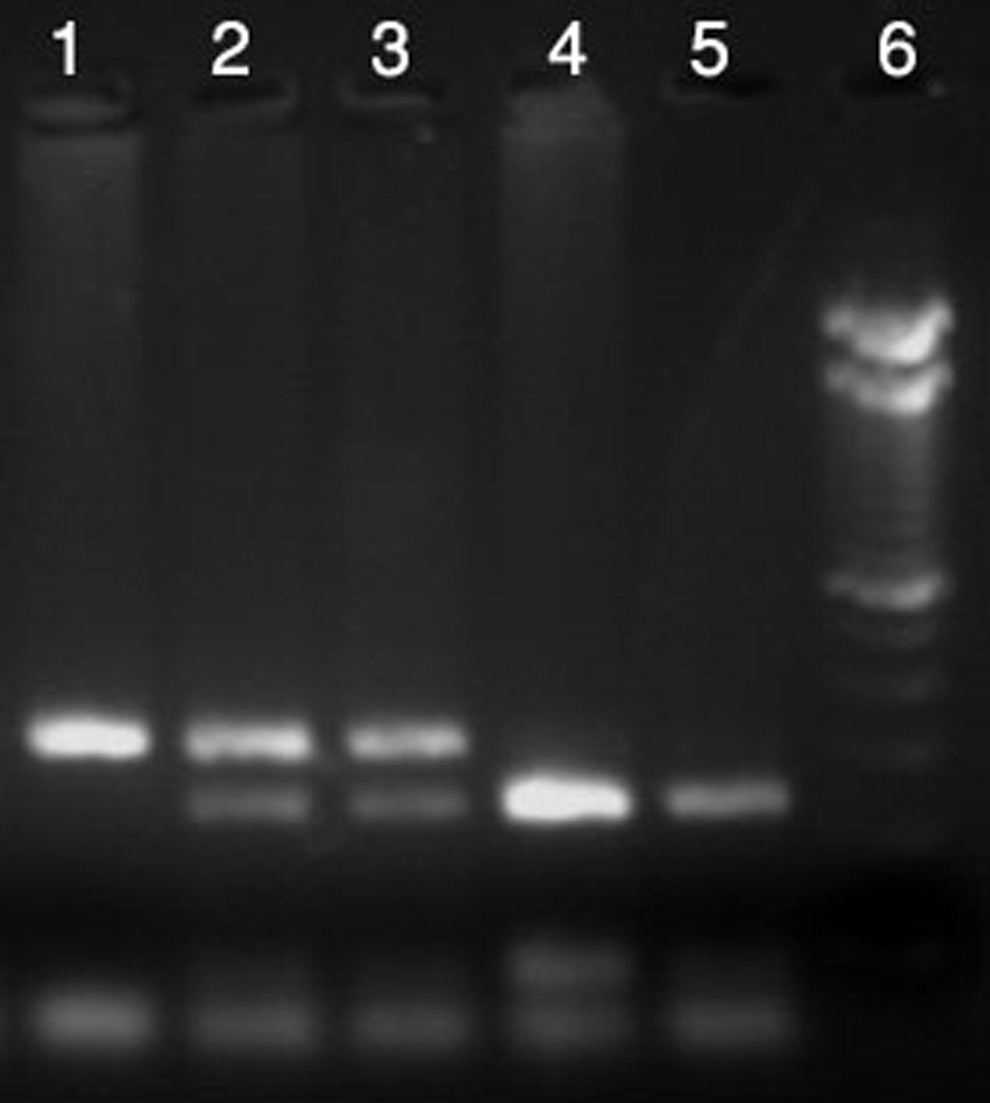
CTLA-4 +49 A>G genotypes after BbvI digestion. Lane 1: wild type: AA, 327 bp. Lanes 2 and 3: heterozygous: AG, 327, 244, and 84 bp. Lanes 4 and 5: homozygous: GG, 244 and 84 bp. Lane 6: 100-bp Ladder. CTLA-4 indicates cytotoxic T lymphocyte antigen 4; bp, base pair.
Statistical Analysis
SPSS version 18.0 was used for data analysis. Mean ± standard deviation described quantitative data. Qualitative data were expressed as percentages. Percentages were compared using chi-square and Fisher exact tests. Quantitative data were compared by both parametric and nonparametric tests for 2 and more than 2 groups (t test and analysis of variance). P value is significant at .05 level.
Results
Laboratory, Clinical, and Therapy Response Characteristics of Cohort of Patients With ITP
The mean platelet count at disease onset was 16.7 ± 15.6 with a range of 1 to 70 × 109/L. According to the phase of the disease, patients were classified into 37 newly diagnosed (disease duration from 0 to 3 months), 13 persistent (disease duration from 3 to 12 months), and 50 chronic cases (disease duration more than 12 months). Quality of response assessment showed 27% of the cases with a complete response, 24% showed a partial response, and 49% showed no response to the applied line of management at the time of study. The clinical response assessment—which is considered with cessation of bleeding—showed that 82% of cases responded clinically; the percentage of bleeders dropped from 57% to 18%. Another classification was carried out according to the response to the different lines of management. Patients who showed a response or a complete response were considered as responders: 32 of the patients with ITP were put under initial observation, 3 (9.4%) responded spontaneously and 29 (90.6%) did not respond; 63 patients received a trial of steroid therapy, 23 (36.5%) responded and 40 (63.5%) did not respond to steroids; 23 patients received immuran, 8 (34.8%) responded and 15 (65.2%) did not respond; 4 patients with ITP received intravenous immunoglobulin, 1 responded and 3 did not respond; 4 patients had undergone splenectomy to which 2 responded and 2 did not respond; and 3 patients with ITP received a therapeutic trial of cyclosporine, there were no responders.
Genotypes and Allele Frequency Distribution of CTLA-4 +49 in Patients With ITP and Control Groups
We evaluated CTLA-4 +49 genotype and allele distributions in patients with ITP and controls. No significant difference was encountered between both groups in either genotype or allele frequency distribution of CTLA-4 +49 (P > .05). Based on the disease course, the patients were subdivided into 3 phases: newly diagnosed, persistent, and chronic ITP. No statistical differences were detected in genotype and allelic frequencies between the 3 groups and the controls (Table 1).
Distribution of Genotypes and Allele Frequency of CTLA-4 +49 A>G in the Studied Children With ITP and Control Groups and Association With Phase of the Disease.

Abbreviations: tITP, total immune thrombocytopenic purpura; ndITP, newly diagnosed immune thrombocytopenic purpura; cITP, chronic immune thrombocytopenic purpura; pITP, persistent immune thrombocytopenic purpura; CTLA4, cytotoxic T-lymphocyte antigen 4; ITP, immune thrombocytopenic purpura.
Studying the Relation of CTLA-4 +49 Genotypes in Patients With ITP With Other Parameters
No statistically significant association was found between the CTLA-4 +49 genotype and any of the studied parameters, namely, platelet count and age at disease onset, gender, severity at diagnosis of the disease, the quality of response, clinical response, and response to different lines of treatment.
Discussion
To our knowledge, only 1 previous study investigated the role of CTLA-4 +49 genotype polymorphism as a risk factor for ITP. However, it did not address its possible impact on the different laboratory and clinical aspects of the disease.
The CTLA-4 gene is located on chromosome 2q33, closely associated with CD28 and ICOS (Inducible T-cell co-stimulator) genes. It contains 4 exons that code the leading peptide, ligand-binding domain, transmembrane domain, and cytoplasmic tail. 17 The CTLA-4 has 2 isoforms, a full-length and a soluble CTLA-4 which lacks a transmembrane domain. 18 Upon binding to B7 molecules, both inhibit CD28/B7 interaction, resulting in downregulation of the immune response. 19 CTLA-4- −/− mice show a severe lymphoproliferative disorder with massive autoimmune tissue destruction. 20 The CTLA-4 is also deficiently expressed in nonobese diabetic mice, which is an animal model of autoimmune diabetes. 21 Furthermore, an A>G polymorphism at position 49 of the CTLA-4 first exon has been associated with the development of insulin-dependent diabetes mellitus in several ethnic groups. 22 This polymorphism results in an amino acid substitution (Thr-Ala) at codon 17 of the CTLA-4 leader peptide. A high prevalence of the CTLA-4 G allele, which apparently confers susceptibility to autoimmune disease, has also been reported in patients with Graves disease, Hashimoto thyroiditis, rheumatoid arthritis, multiple sclerosis, primary biliary cirrhosis, and in some studies of patients with systemic lupus erythematosus. 23 –27 Furthermore, the number and functions of T cells were reported to be abnormal in ITP, 28 and it was found that CTLA-4-Ig was associated with platelet-specific T-cell anergy. 29,30 Hence, it was speculated that there might be a correlation between CTLA-4 polymorphisms and the clinical implications of ITP. 13
The present study evaluated the possible contribution of CTLA-4 +49 polymorphisms to the pathogenesis of ITP and their possible relation to the laboratory and clinical aspects of the disease. Statistical comparisons for the distribution of genotype and allele frequency for the CTLA-4 +49 between patients with ITP and controls showed no statistically significant difference (P value >.05). These results agree with the study of Pavkovic et al, 14 which indicated that the polymorphism might not play an important role as a genetic risk factor in the pathophysiology of ITP. We then stratified the patients by the age of disease onset, platelet count, gender, phase of the disease, severity at diagnosis, quality of response, clinical response, and response to different lines of therapy. No statistically significant association was found between the genotype and any of the studied parameters. Although previously no association was found between this polymorphism and ITP, 14 we decided to conduct this study for several reasons: the possibility of the variation in the relative frequency of the different alleles among different populations, correlating the polymorphism with the different laboratory and clinical parameters, an issue that was not tackled in the other study and also conducting the study in a cohort of pediatric patients to verify whether there is a variation in the association of the polymorphism and the risk of ITP in different age groups.
To our best knowledge, this is the first report investigating the distribution of CTLA-4 +49 polymorphisms in Egyptian pediatric patients with ITP and their role in different aspects of the disease. The present study did not detect a relation between the selected CTLA-4 SNP and susceptibility to ITP in Egyptian children. However, we cannot discard a possible association of other variants within the human CTLA-4 gene covering region with ITP as a genetic risk factor; further studies on different loci are recommended.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
