Abstract
Introduction:
Disturbances in cytokine networks are believed to be associated with increased risk of adverse pregnancy complications.
Methods:
Plasma samples collected from pregnant women with preterm deliveries, high-risk pregnancy complications including postpartum hemorrhage, hypertensive disorders, and multiple gestations, and normal pregnancies were analyzed at different periods throughout gestation and postpartum. Interleukin (IL) 1β , IL-6, IL-8, IL-10, tumor necrosis factor α, and antiprotein Z antibody levels were measured by enzyme-linked immunosorbent assay.
Results:
The IL-6 levels in preterm delivery patients were elevated during pregnancy with statistically significant differences observed at 21 to 32 weeks (P < .01) and 33+ weeks (P < .001). The IL-10 levels were increased in normal pregnancy at all time points compared to the other patient groups (P < .05). The TNF-α levels were elevated in the high-risk pregnancy group versus normal controls (P < .001 at <21 weeks and P < .05 at 21-32 weeks).
Conclusion:
Analysis of the maternal plasma for elevated IL-6 and reduced IL-10 levels may be of value in the early prediction of pregnancy complications.
Introduction
Preterm labor (PTL) resulting in preterm birth, postpartum hemorrhage (PPH), and hypertensive disorders such as pre-eclampsia (PE) are serious complications associated with adverse outcomes in pregnancy. PTL, preterm premature rupture of membranes, and cervical incompetence are major causes of preterm delivery (before 37 weeks of gestation). Preterm delivery is a leading cause of perinatal morbidity and mortality, 1 –3 and it affects 12% to 13% of pregnancies in the United States and 5% to 9% of pregnancies in other developed countries. 2 A major cause of maternal morbidity and mortality is PPH. 4 –6 PPH is often clinically diagnosed as excessive bleeding after delivery that is associated with hemodynamic instability. Blood loss greater than 500 mL after delivery occurs in up to 18% of births, and an estimated 3% of vaginal deliveries are complicated by severe PPH. 4 Another complication is PE, a pregnancy-specific medical condition characterized by hypertension and proteinuria. PE is one of the primary causes of maternal and fetal morbidity and mortality, 5,7 –9 and PE and other hypertensive disorders affect up to 8% of all pregnancies. 10 PE is also a common cause of PTL, 8 and 15% of preterm deliveries are early deliveries indicated for PE. 10 In addition, multiple gestation pregnancies are inherently high risk compared to singleton pregnancies, and they are associated with increased rates of PTL, fetal growth restriction, PE, and placental and cord problems. 11
Although the precise etiologies of many pregnancy complications remain poorly understood, important risk factors and influential pathways associated with immunomodulation and inflammation have been identified. Processes thought to be involved in the pathogenesis of PTL include intrauterine infection and inflammation, uteroplacental ischemia or hemorrhage, and fetal or maternal stress. 2 In addition, there is growing evidence that PE involves an exaggerated maternal inflammatory response to pregnancy 12 and that placental ischemia is an instrumental factor in the preeclamptic state; it is believed that placental ischemia/hypoxia triggers increased production of inflammatory factors, such as interleukin 6 (IL-6) and tumor necrosis factor α (TNF-α), which contribute to endothelial dysfunction in PE. 7 Multiple biochemical markers have suggested value in the prediction and diagnosis of PE, but no single marker alone has proven strength to reliably and safely predict the condition. 8 Likewise, although many ante- and intrapartum risk factors and causes of PPH, including uterine overdistension, retained placenta, and uterine atony, have been identified, single risk factors have low predictive probability for PPH. 6 Thus, the continued search for clinically useful biomarkers that are predictive of adverse conditions is pivotal for identifying asymptomatic, at-risk women, developing strategies for prevention and treatment, and elucidating the pathophysiology of pregnancy complications.
Given the substantial involvement of inflammation and the immune system in many pathologic pregnancy states, immune mediators such as cytokines have been logical candidates for predictive biomarkers. 3,13,14 Cytokines are pleiotropic glycoproteins with immunoregulatory functions that are produced by the cells of the immune system (T lymphocytes and maternal and fetal macrophages) and by the uterine epithelium, uterine smooth muscle, cervix, and placenta. 15 Both proinflammatory cytokines, such as TNF-α, interleukin (IL) 1β, IL-6, and IL-8, and anti-inflammatory cytokines, such as IL-10, have key functions in pregnancy. In early pregnancy, the decidua is believed to foster both a proinflammatory environment to support the formation of the placenta and an anti-inflammatory environment that is immunotolerant of the fetal allograft. 15 Continued modulation of the immune system during gestation, involving a reduction in proinflammatory cytokines and an increase in counter-regulatory cytokines such as IL-10, appears to be important for maintaining a healthy pregnancy. 13
Many studies have examined the role of placental and systemic cytokines in pregnancy and parturition and in relation to PTL and PE. Elevated serum IL-614,16,17 and IL-81,17 levels have been observed in PTL and preterm delivery patients compared to normal pregnancies, and IL-10 levels tend to be raised in normal pregnancies 13,14 and reduced in preterm delivery pregnancies. 14 When compared to normal pregnancies, most studies report increased maternal serum and plasma levels of IL-6, IL-8, and TNF-α in pregnancies complicated by PE. 9,18,19 In addition, both increased 20 and decreased 19,21 IL-10 levels have been shown in patients with PE. Another analyte of interest is protein Z (PZ). PZ serves an important role in limiting the coagulation cascade as a cofactor for PZ-dependent protease inhibitor (an inhibitor of activated factor X). 22,23 PZ deficiency 22,23 and the presence of antiPZ antibodies 23,24 also appear to correspond with increased risk of adverse pregnancy outcomes.
Additional studies utilizing a longitudinal approach are needed to investigate the time-dependent relationship of changes in cytokine levels over the course of pregnancy in association with the development of harmful complications. Identifying a relationship between cytokine levels and when they appear elevated or decreased that correlates with adverse pregnancy outcomes may be useful for determining risk and for providing further insight into the pathophysiology of these conditions. Thus, this study aimed to measure, compare, and correlate the concentrations of various cytokines and analytes in plasma samples taken throughout pregnancy to determine whether cytokine levels could be predictive of adverse pregnancy complications such as preterm delivery, PPH, and hypertensive disorders.
Materials and Methods
Plasma samples were collected from a population of pregnant women (Hilleroed Hospital, Copenhagen, Denmark) over the course of pregnancy. All sample collections complied with the Declaration of Helsinki. All patients were at least 18 years old. Women with human immunodeficiency virus, chorioamnionitis, or other infections were excluded from the study. Between 11 and 14 weeks of gestation, all patients had an ultrasound measuring the nuchal translucency thickness and the crown-rump length of the fetus to estimate gestational age. We defined 3 patient groups based on complications encountered during pregnancy: high-risk pregnancy complications including PPH, hypertensive disorders such as PE, multiple gestations, and thrombotic disorders (n = 16), preterm delivery (n = 19), and normal pregnancy (n = 20). Preterm delivery was defined as birth at less than 37 weeks of gestation with a mean gestational age at delivery of 32.1 weeks. PPH was determined by blood loss of 500 mL or more during the first 24 hours after delivery. Criteria for PE diagnosis included hypertension (blood pressure ≥ 140/90 mm Hg) and proteinuria (> 300 mg/24 h) after 20 weeks of gestation. The normal pregnancy control group consisted of uncomplicated gestations and deliveries that resulted in full-term births. Citrated plasma samples were stored at −80°C and shipped on dry ice to the Loyola University Medical Center’s Hemostasis and Thrombosis Laboratories for analysis. The samples were categorized into 3 gestational time periods (<21 weeks, 21-32 weeks, and 33+ weeks) and postpartum according to the time at which they were collected. Several preterm delivery patients did not have 33+ week samples as a result of their early deliveries.
Citrated plasma samples from normal, healthy volunteers (n = 50) were also included in the study. Volunteers who participated in the study were females aged 21 to 45 years from the United States who did not have chronic medical conditions and who were not taking any medications. The blood samples were drawn from normal volunteers and mixed with 3.2% sodium citrate in a 9:1 ratio. Cytokine levels—IL-1β, IL-6, IL-8, IL-10, and TNF-α—were measured in both maternal and normal control plasma samples using commercially available high-sensitivity enzyme-linked immunosorbent assay (ELISA) kits (R&D Systems Quantikine, Minneapolis, MN). The HYPHEN BioMed ZYMUTEST (Neuville sur Oise, France) antiPZ, immunoglobulin G (IgG) ELISA assay was used to determine the maternal levels of autoantibodies to PZ. According to the assay manufacturers, the sensitivity (expressed as the minimum detectable dose) and the upper limit of detection, respectively, for the various assays are: IL-1β (<1 pg/mL, 4200 pg/mL), IL-6 (<0.7 pg/mL, 17 500 pg/mL), IL-8 (3.5 pg/mL, 100 000 pg/mL), IL-10 (<3.9 pg/mL, 3600 pg/mL), TNF-α (1.6 pg/mL, 7200 pg/mL), and anti PZ, IgG (<1 ng/mL, 125 AU/mL). For all of the assays, the intra-assay precision coefficient of variation (CV%) ranged from 1.6% to 8.5%, and the interassay precision CV% ranged from 3.3% to 9.7%. Optical density values were measured using a microplate reader (SpectraMax Plus384, Molecular Devices, Sunnyvale, California). Analyte concentrations were then calculated using the optical density readings and equations obtained from the standard calibration curve for each assay.
Statistical analysis was performed using GraphPad Prism software (version 3.03, La Jolla,California). One-way analysis of variance and the Bonferroni multiple comparison test were used to determine statistically significant differences between groups and time points for each individual cytokine and to calculate 95% confidence intervals. A P value of <.05 was considered to be statistically significant.
Results
IL-6 levels were elevated in the preterm delivery group compared to the normal pregnancy, high-risk pregnancy, and normal control groups at all time points, as shown in Figure 1. In addition, the mean preterm delivery IL-6 concentration also increased over the course of pregnancy and reached its highest level at 33+ weeks. Statistically significant differences were found between the preterm delivery group and the other groups at 21 to 32 weeks (P < .01) and 33+ weeks (P < .001). Furthermore, for the preterm delivery group, there were no statistically significant differences between the IL-6 concentrations at the individual time points. There was also no correlation between IL-6 levels in preterm delivery and high-risk patients (R 2 = .07 for IL-6 levels from 21 to 32 weeks)
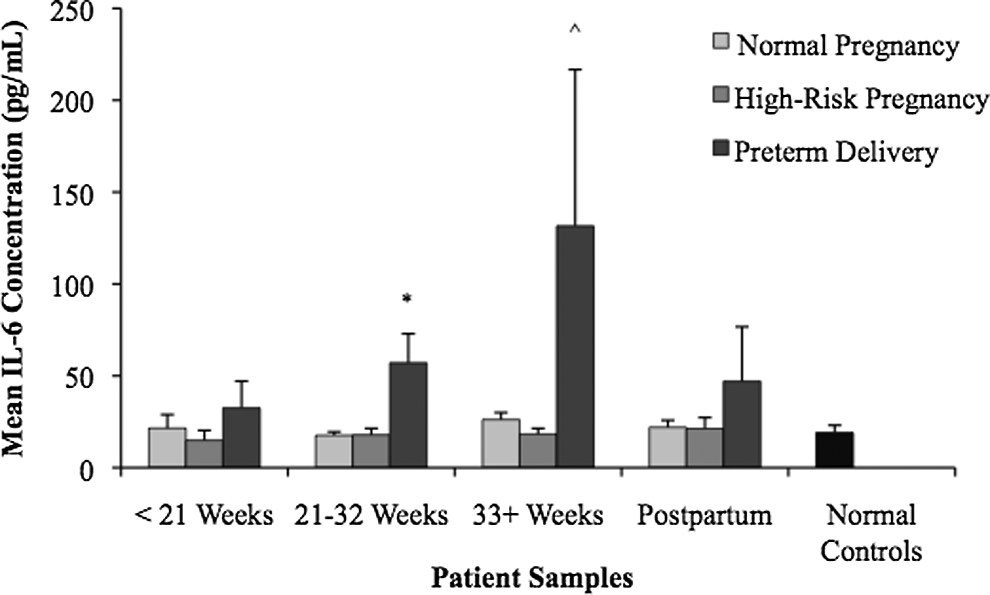
Interleukin 6 (IL-6) levels in normal and complicated pregnancies. Plasma samples were drawn and analyzed for IL-6 levels. The mean IL-6 concentration was elevated in the preterm delivery group. Statistical differences were observed between the preterm delivery group and the normal pregnancy, high-risk pregnancy, and normal control groups at 21 to 32 weeks (P < .01) and 33+ weeks (P < .001).
The IL-10 levels in pregnancy are illustrated in Figure 2. The mean concentration of IL-10 in normal pregnancy was found to be elevated at each time point and statistically significant compared to other groups, with P values ranging from P < .05 to <.001. Statistically significant differences between IL-10 levels in normal pregnancies and preterm delivery pregnancies were observed at <21 weeks (P < .01), 21 to 32 weeks (P < .05), and postpartum (P < .05). Differences in IL-10 levels between normal pregnancy and high-risk pregnancy were statistically significant at 21 to 32 weeks (P < .05) and 33+ weeks (P < .05). Elevated IL-10 levels in normal pregnancy were statistically significant compared with the IL-10 levels in normal controls at all gestational time periods analyzed (P < .001). The IL-10 levels in high-risk pregnancy were relatively steady across pregnancy, whereas in the preterm delivery group, we observed an increase in the IL-10 levels over the course of pregnancy. There was no correlation between the increasing IL-6 and IL-10 levels (observed in Figures 1 and 2) in the preterm delivery group (R 2 = .06). In addition, Figure 2 illustrates that the IL-10 levels were elevated in all 3 pregnancy patient groups compared to the normal controls.
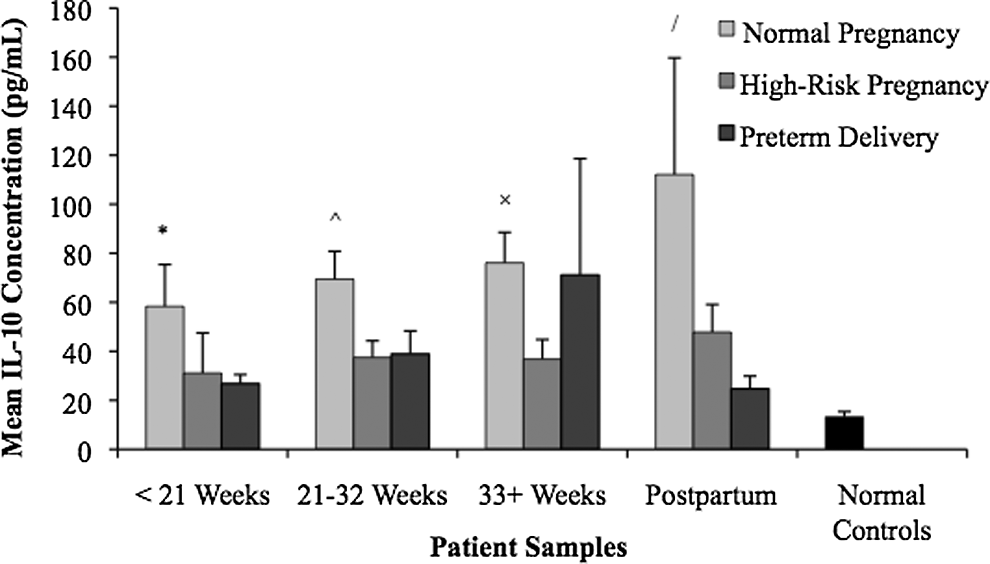
Interleukin 10 (IL-10) levels in normal and complicated pregnancies. Plasma samples were drawn and analyzed for IL-10 levels. The mean IL-10 concentration was elevated in the normal pregnancy group. Statistical differences were observed between normal pregnancy and preterm delivery patients at <21 weeks (P < .01), 21 to 32 weeks (P < .05), and postpartum (P < .05), between normal pregnancy and high-risk pregnancy at 21 to 32 weeks (P < .05) and 33+ weeks (P < .05), and between normal pregnancy and normal controls at all time points (P < .001).
Figure 3 shows that the mean concentration of TNF-α was elevated at <21 weeks in the high-risk pregnancy group. Statistically significant differences were seen in early high-risk pregnancy patients compared to preterm delivery patients at <21 weeks (P < .01) and compared to normal controls at <21 weeks (P < .001) and 21 to 32 weeks (P < .05). TNF-α was also elevated in normal pregnancy and statistically significant from normal control level at 21 to 32 weeks (P < .05). Otherwise, the TNF-α level remained relatively constant throughout mid- to late pregnancy in the 3 patient groups. Furthermore, within the high-risk pregnancy group, the increased mean TNF-α concentration at <21 weeks was statistically significant (P < .01) compared to the other 3 time intervals (21 to 32 weeks, 33+ weeks, and postpartum). Although statistically significant differences were not found at every time point, Figure 3 illustrates that the TNF-α levels were elevated in pregnant patients compared to normal controls.
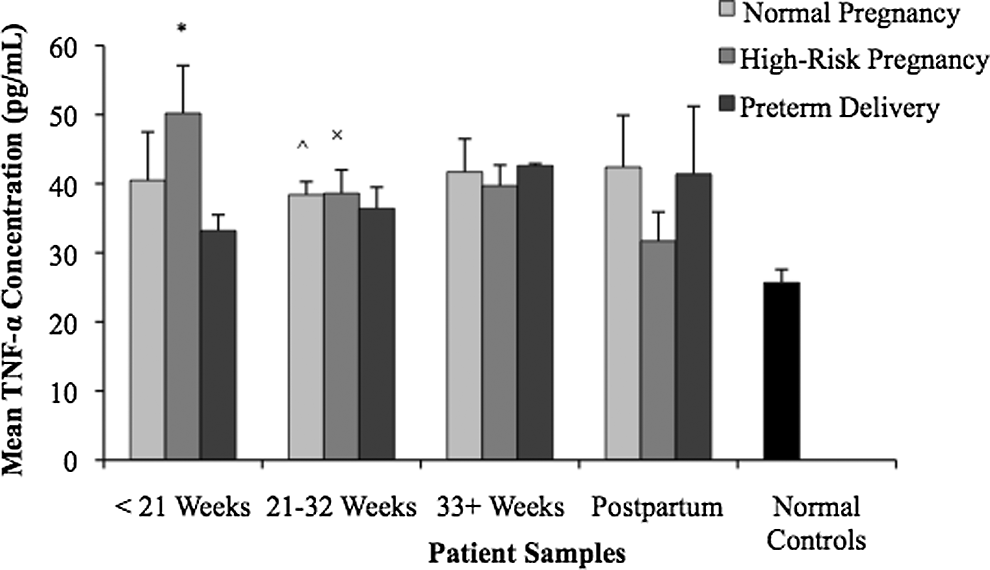
Tumor necrosis factor α (TNF-α) levels in normal and complicated pregnancies. Plasma samples were drawn and analyzed for TNF-α levels. The mean concentration of TNF-α was elevated in the high-risk pregnancy group at <21 weeks. At <21 weeks, statistical differences were observed between high-risk pregnancy and preterm delivery patients (P < .01) and between high-risk pregnancy and normal controls (P < .001). At 21 to 32 weeks, normal pregnancy and high-risk pregnancy TNF-α levels were elevated compared to normal controls (P < .05). The high-risk pregnancy TNF-α concentration at <21 weeks was also statistically different from the high-risk pregnancy concentrations measured in the other 3 time intervals (P < .01).
Few differences in the levels of IL-1β, IL-8, and autoantibodies (IgG) to PZ were observed between groups or gestational time periods. The IL-1β levels, illustrated in Figure 4, did not vary significantly between the groups. In Figure 5, the IL-8 mean levels remained consistently in the 100 to 200 pg/mL range in each patient group across pregnancy. This was a much higher mean cytokine concentration than observed for the other analytes. Furthermore, concentrations of autoantibodies to PZ, as illustrated in Figure 6, were found to be relatively low in both the normal controls and the pregnancy patient groups. No statistically significant differences were found between the groups or time points for IL-1β, IL-8, and antiPZ antibodies.
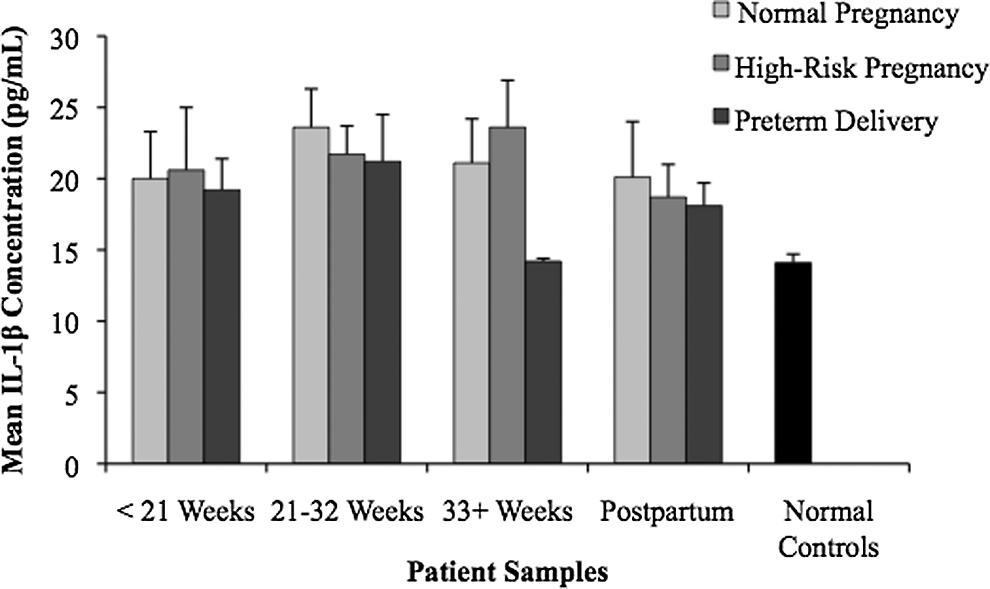
Interleukin 1β (IL-1) levels in normal and complicated pregnancies. Plasma samples were drawn and analyzed for IL-1 levels. No statistically significant differences between groups or time points were observed.
Interleukin 8 (IL-8) levels in normal and complicated pregnancies. Plasma samples were drawn and analyzed for IL-8 levels. No statistically significant differences between groups or time points were observed.
Levels of anti-protein Z (antiPZ) and IgG antibodies in normal and complicated pregnancies. Plasma samples were drawn and analyzed for levels of antiPZ and IgG antibodies. No statistically significant differences between groups or time points were observed.
Discussion
Preterm delivery, PPH, and hypertensive disorders are serious complications of pregnancy and major causes of maternal and perinatal morbidity and mortality. Given the crucial role and interplay of proinflammatory and anti-inflammatory cytokines in processes of normal pregnancy and in high-risk pregnancy complications, cytokines are feasible biomarkers for early screening and detection of adverse pregnancy states. Interestingly, in this preliminary study, certain plasma cytokine profiles helped to differentiate between our 3 pregnancy patient groups. Preterm delivery patients had a higher tendency to have elevated IL-6 levels that increased over the course of pregnancy. In normal pregnancy, the IL-10 levels were elevated at each time point, and an increased TNF-α concentration at <21 weeks was characteristic of the high-risk pregnancy group. These observations are consistent with previous studies of IL-6 in preterm delivery 14,16,17 and IL-10 in normal 13,14 and abnormal 14,19,21 pregnancies. These findings suggest that certain changes in cytokines could be specific to a given condition, and, with early recognition, this could allow for the initiation of risk-specific interventions. Furthermore, we identified a time course of when and how cytokine levels change throughout gestation. Our results indicate that changes in IL-10—increased levels in normal pregnancy and reduced levels in high-risk and preterm delivery pregnancies—can be identified before 21 weeks of gestation. In addition, elevated IL-6 levels were also observed at <21 weeks in preterm delivery patients and were statistically significant at 21 to 32 weeks. These time–cytokine relationships may serve as a good starting point for determining the gestational age at which cytokines have the greatest predictability of adverse outcomes and for distinguishing key periods when implementing interventions will be most beneficial. In addition, plasma cytokine level testing has many clinical advantages as a potential screening method due to the relative ease, low cost, minimal discomfort to the patient, and little maternal or fetal risk involved with venipuncture for sample collection. Nonetheless, one of the limitations of this study is that all patient samples were collected at a designated location in Denmark. Further multicenter studies with larger sample sizes would be helpful in validating the results from this study.
Despite the associations between cytokine changes and pathologic pregnancies that have been observed by this study and others, there is some uncertainty regarding the clinical usefulness of cytokines as biomarkers for early detection of pregnancy complications. 2,16,25,26 For example, Bahar et al 25 did not observe a significant difference in maternal serum IL-6, IL-8, and TNF-α in women in PTL versus normal control pregnancies (healthy preterm women not in labor and women in term labor), and the authors acknowledged that there is doubt regarding the usefulness of testing serum cytokine levels to identify early intrauterine or fetal infection in PTL. Several studies, including that by Bahar et al, 25 have assessed cytokine levels in symptomatic patients following the onset of labor. 14,17 Similarly, cervicovaginal fetal fibronectin, currently one of the most valuable biomarkers for determining risk of preterm birth, is clinically used for its high negative predictive value when measured at the onset of PTL. 3 Unfortunately, once labor begins, little time remains for prevention. The earlier a biomarker associated with adverse complications can be detected during pregnancy, the earlier a given pregnancy can begin to be monitored to prepare for, manage, and/or treat the condition. Thus, the purpose of this study was to identify an earlier biomarker in asymptomatic women, and we used a longitudinal study design to follow the plasma cytokine levels prior to the onset of PTL symptoms.
Another general limitation of screening tests for PTL and preterm birth is the lack of an intervention that effectively prevents or treats PTL and decreases the rate of preterm delivery. 3,26 Current strategies, including enhanced prenatal care, patient education, cerclage, bed rest, and hydration, are not universally effective and have had minimal success at reducing preterm delivery incidence. 26,27 However, the problem may not be simply unsuccessful interventions but rather ineffective initiation of these interventions in the high-risk patients who are most likely to benefit from them. 26 This demonstrates the importance of accurately and reliably identifying the high-risk patients. In doing so, targeted patient education and closer monitoring of truly high-risk pregnancies may achieve better results than previously described. For instance, educating patients about PTL and preterm birth can encourage awareness of contractions and earlier transport to the hospital such that, when a patient presents, her labor is in the early stages where treatment (such as tocolytic agents) may still be effective. In addition, for a patient with risk factors for PPH and low IL-10 levels, early discussions about blood transfusions and avoiding an episiotomy could be helpful for safely managing the patient’s delivery in the future. 4
Our findings may also have implications in the pathophysiology of these conditions. It appears that increased IL-10 expression in normal pregnancy helps suppress proinflammatory cytokine responses and is important for maintaining a healthy pregnancy. 13 Although increased IL-10 seems to offer some degree of protection in normal pregnancy, the reduced IL-10 levels seen in preterm delivery and high-risk patients could be permissive of an excessively inflammatory, destructive local environment. Indeed, we observed both decreased IL-10 and increased IL-6 concentrations in our preterm delivery group (compared to normal pregnancy), and these results are in accord with the concept of a proinflammatory cytokine bias in PTL/preterm delivery 14,28 that may contribute to the pathogenesis of PTL. For instance, elevated IL-6 is believed to induce production of prostaglandins and matrix metalloproteinases which then promote uterine contractions, cervical ripening, and weakening/rupture of chorioamniotic membranes. 1,14,28 Finally, cytokines may be an effective target for prevention or treatment strategies. For example, dydrogesterone, an orally administered progestogen similar to endogenous progesterone, has been shown to inhibit proinflammatory cytokine production and to increase anti-inflammatory cytokine levels, making it a possible therapeutic intervention for preterm delivery patients. 29 If further research were to establish a causal relationship between elevated IL-6 or reduced IL-10 and adverse complications, correcting the cytokine imbalance could be of value in the management of the condition.
Conclusions
Our results suggest that analysis of maternal plasma for IL-6 and IL-10 may be beneficial for the early prediction of preterm delivery and other high-risk conditions in asymptomatic patients. Elevated IL-6 levels can be observed in preterm delivery pregnancy patients as early as 21 to 32 weeks; thus, examining plasma IL-6 levels over the course of pregnancy may be useful for identifying pregnancies at risk of preterm delivery. Detecting reduced maternal plasma IL-10 levels compared to normal pregnancy may identify patients at risk of preterm delivery and other high-risk conditions including PPH and hypertensive disorders. Through early risk determination, screening for characteristic cytokine profiles has the potential to guide effective implementation of patient education and prevention strategies, and it facilitates the search for new interventions and further elucidation of the immune mechanisms underlying adverse pregnancy complications.
Footnotes
Acknowledgments
The authors wish to acknowledge Merete Bak Bertelsen from Clinical Trial Unit, Copenhagen University Hospital Glostrup for her technical assistance in the handling and processing of the samples for this study.
Authors’ Note
The Abstract was accepted for poster presentation (“Pathophysiology of Thrombosis” session) at the American Society of Hematology Annual Meeting, December 11, 2011, at San Diego, CA.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
