Abstract
Background:
Platelet hyperaggregation is known to be associated with arterial and venous thromboembolic events. The prevalence of platelet hyperaggregation in patients with chronic kidney disease (CKD) has not been described to date.
Methods:
Platelet hyperaggregation in patients with renal disease was defined by comparison of platelet aggregation patterns to non-CKD patients without thromboembolic disorders and healthy controls.
Results:
Among the 30 hemodialysis patients and 34 renal transplant recipients, 20 (67%) and 28 (82%) showed significantly decreased median Δ-epinephrine aggregation and increased 0.5 mol/L epinephrine response (65% and 54%) compared to healthy controls and non-CKD patients. In concordance to the laboratory finding of platelet hyperaggregability, renal transplant recipients showed a high rate of thromboembolic events (normal platelet aggregation: 0 events and platelet hyperaggregation: 30 events in 13 of 28 patients).
Conclusions:
Patients with CKD exhibit a hitherto unappreciated high prevalence of platelet hyperaggregability indicating sticky platelet syndrome. Laboratory testing of platelet hyperaggregability may supplement the assessment of thromboembolic complications in patients with CKD.
Introduction
In the laboratory setting, hyperaggregability of platelets is characterized by light transmission aggregometry and dose-independent platelet aggregation solely due to epinephrine and/or adenosine diphosphate (ADP). 1 In the early 1980s Mammen and Bick suggested platelet hyperaggregation as the underlying cause of thrombophilia in patients with unexplained arterial and venous thromboembolism or recurrent spontaneous abortions. The observation of so-called sticky platelet syndromes (SPSs) has been proven by numerous clinical case reports associated with acute myocardial infarction, thromboembolic kidney graft infarction, transient ischemic cerebral attacks, and strokes. 2,3
Family testing of the affected patients suggested an autosomal pattern of heredity. 4,5 Kotuličová et al recently reported the prevalence of selected GP6 gene polymorphisms as independent risk factors for deep vein thrombosis in patients with platelet hyperaggregability. 6,7 In addition, Johnson et al identified associations of 7 loci with platelet aggregation in response to the agonists ADP and epinephrine. 8 In contrast, Yee et al showed in healthy individuals that epinephrine-mediated platelet activation and hyperaggregation are associated with G-protein β-3 polymorphism encoding the β-3 subunit of G proteins involved in platelet activation signaling pathways. 9
Although the cause for platelet hyperaggregation is still unknown, our group has published a mini series of 3 patients with SPS exhibiting thromboembolic complications after kidney transplantation. 10 Based on these findings, we conducted a case–control study in chronic hemodialysis (HD) patients and renal transplant recipients to provide more information about the prevalence of platelet hyperaggregation in patients with chronic kidney disease (CKD). Detailed platelet aggregation analysis was performed and compared to platelet response in healthy controls and non-CKD patients with and without thromboembolic events.
Patients and Methods
Patients and Healthy Control Collective
Platelet aggregation analysis was performed in 64 patients with CKD (chronic HD patients: n = 30, 15 males/15 females, median age 50 years, range 20-75 years; renal transplant recipients: n = 34, 11 males/13 females, median age 56 years, range 26-79 years) who were admitted to the Department of Nephrology at the RWTH Aachen University Hospital. In addition, non-CKD patients (n = 79; median age 34 years, range 23-43 years) were assessed for platelet hyperaggregability at the Department of Gynecological Endocrinology and Reproductive Medicine at the RWTH Aachen University as part of the evaluation for infertility. At the time of laboratory investigation, all patients had normal plasmatic coagulation (data not shown). The healthy control population consisted of 44 volunteers (8 males/36 females, median age 42 years, range 24-63 years).
None of the investigated study populations had disturbed platelet function by neither antiplatelet medication nor physical exertion at the time of blood draws. In patients receiving antiplatelet therapy, the treatment was interrupted for 10 days before platelet function testing. Moreover, patients and healthy controls were questioned about recent ingestion of any nonprescribed medication to exclude drugs that may interfere with platelet aggregation.
Healthy controls were investigated to determine the decisional platelet hyperaggregability cutoffs. Platelet hyperaggregability in patients with CKD was assessed by comparison of platelet function to healthy controls and non-CKD patients with and without thromboembolic events.
The study protocol was approved by the local ethics committee and conducted in accordance with the ethical standards laid down in the Declaration of Helsinki.
Platelet Aggregometry
To minimize preanalytical activation of platelets and clotting proteins, citrated (sodium citrate 0.11 mol/L) whole blood samples were drawn by venopuncture through wide-bore needles without a tourniquet. Platelet aggregation was performed within 4 hours after blood sample collection. All participating patients and healthy controls had initial platelet counts within the reference range (data not shown).
Platelet function was evaluated by testing aggregation responses to ADP and epinephrine by optical light transmission aggregometry (PAP 8 Aggregometer; mölab, Langenfeld, Germany) according to the method established by Born and Cross 11 and modified by Mammen. 3 The aggregometry testing was performed using native platelet-rich plasma (PRP; without platelet count adjustment), because there is no accepted lower limit of PRP platelet counts for aggregation testing. 12 –15
Microscopic assessment of platelet morphology in citrated whole blood smear was carried out to exclude platelet plugs and spontaneous aggregation prior to aggregation analysis.
Identification of Patients With Platelet Hyperaggregation
There are no validated reference intervals in the literature for platelet aggregation by multiple concentrations of agonists. Therefore, we evaluated dose-dependent platelet aggregation patterns of ADP and epinephrine by stimulating native PRP samples in 44 healthy controls (median platelet count 193/nL). Furthermore, we investigated platelet aggregation in a gynecological non-CKD patient group having infertility. This collective, platelet hyperaggregability, referred to as SPS, is the second most common thrombophilia that causes recurrent spontaneous abortion or fetal loss syndrome. 2,3 Patients were clinically classified into thromboembolic (22 of 79; 28%) and nonthromboembolic categories (57 of 79; 72%) determined by medical history. Platelet aggregation was investigated in all patients after stimulation with ADP and epinephrine at 4 different concentrations according to the recommended concentrations for testing, ADP (µmol/L): 10, 2, 1, and 0.5; epinephrine (µmol/L): 50, 10, 1, and 0.5. 15
The decisional platelet hyperaggregability cutoff for each agonist in patients with CKD was established on the basis of dose-dependent platelet function illustrated in Table 1. Platelet aggregation patterns in patients with CKD compared to non-CKD patients without thromboembolic complications and healthy controls were determined as hyperaggregation under the following conditions: (1) increased platelet aggregation at low-dose epinephrine (0.5 µmol/L) greater than maximum aggregation value in non-CKD patients without thromboembolic events (>39%); (2) significantly increased median aggregability when compared to median low-dose epinephrine aggregation in healthy controls (>27%); (3) decreased Δ-aggregation patterns (difference of platelet aggregation between the highest versus the lowest concentration of each agonist) in patients compared to healthy controls, median Δ-platelet aggregation response significantly lower than 64% for ADP and 56% for epinephrine.
Healthy Controls and Patient Characteristics.a
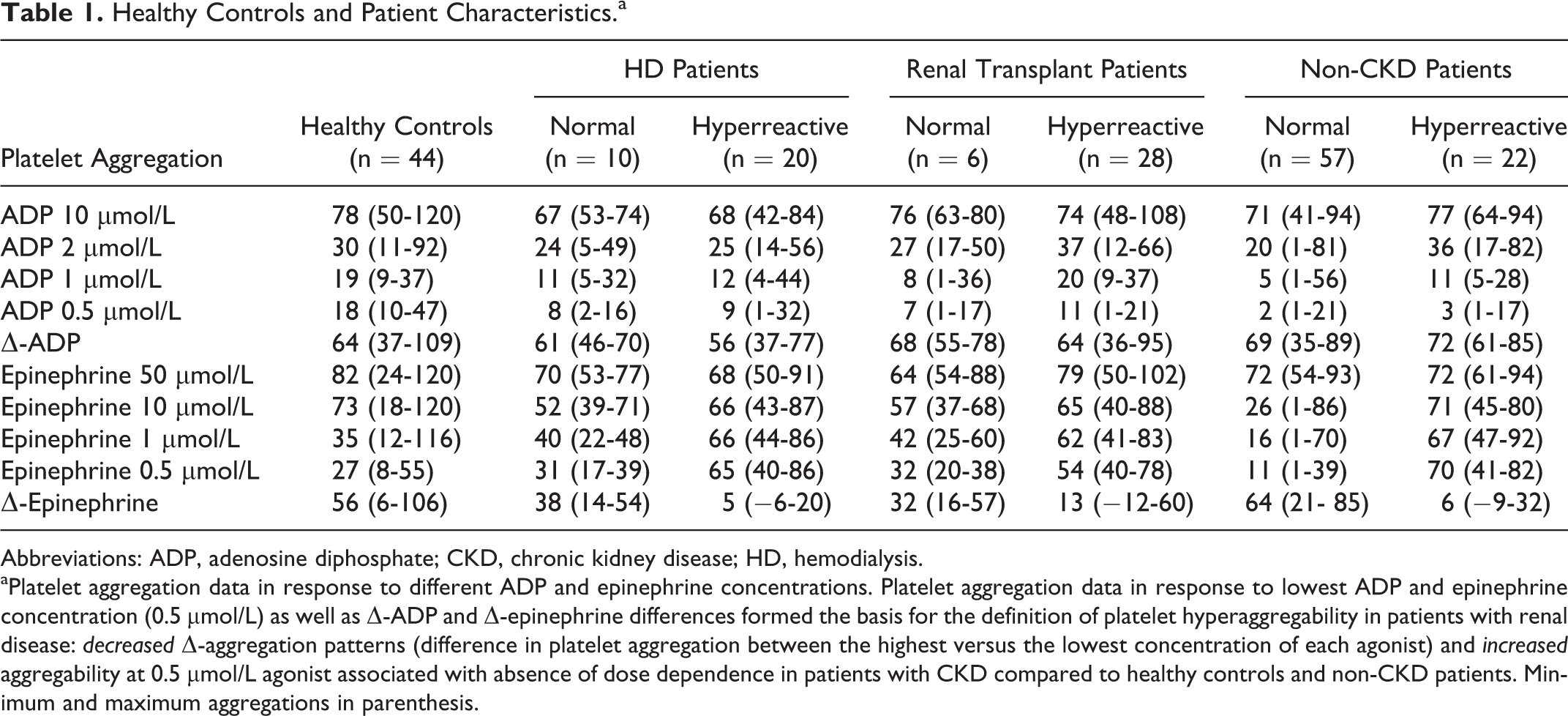
Abbreviations: ADP, adenosine diphosphate; CKD, chronic kidney disease; HD, hemodialysis.
aPlatelet aggregation data in response to different ADP and epinephrine concentrations. Platelet aggregation data in response to lowest ADP and epinephrine concentration (0.5 µmol/L) as well as Δ-ADP and Δ-epinephrine differences formed the basis for the definition of platelet hyperaggregability in patients with renal disease: decreased Δ-aggregation patterns (difference in platelet aggregation between the highest versus the lowest concentration of each agonist) and increased aggregability at 0.5 µmol/L agonist associated with absence of dose dependence in patients with CKD compared to healthy controls and non-CKD patients. Minimum and maximum aggregations in parenthesis.
The ADP-induced aggregation was not considered as significant for potential platelet hyperaggregability due to the presence of dose-dependent platelet aggregation patterns in all the study collectives.
Statistical Analysis
Owing to the skewed distribution of the aggregation parameters, values are given as minimum, maximum, and median. Comparisons between the subgroups are illustrated using Box and -Whiskers plots. Differences between the 2 groups and multiple comparisons among more than 2 groups were assessed by the nonparametric Mann-Whitney U test. The statistical significance level is determined by P < .05. Because multiple comparisons were performed simultaneously, the significance level is adapted by the Bonferroni method (P < .0021). All statistical analyses were performed using MedCalc version 11.4.2 (Mariakerke, Belgium) and SigmaStat version 3.1 (Systat Software Inc, San Jose, California).
Results
Dose-Dependent Platelet Aggregation in Healthy Controls and Non-CKD Patients With and Without Thromboembolic Events
In healthy controls, normal platelet aggregation in native PRP (median platelet count: 193/nL) after stimulation by ADP (10 µmol/L) was determined as 78% (median aggregation; 95% confidence interval: 71%-90%) and 82% (median aggregation; 95% confidence interval: 75%-89%) by 50 µmol/L epinephrine stimulation, respectively (Figure 1A-C). The median platelet aggregation responses to lowest ADP and epinephrine concentrations were significantly and dose dependently decreased (0.5 µmol/L ADP 18%; 95% confidence interval [CI]: 15%-19%; P < .0001 [vs 10 µmol/L] and 0.5 µmol/L epinephrine 27%; 95% confidence interCIval: 23%-35%; P < .0001 [vs 50 µmol/L]). The median Δ-platelet aggregation response defined by the difference between highest and lowest concentrations of ADP and epinephrine was 64% and 56%, respectively (Table 1).
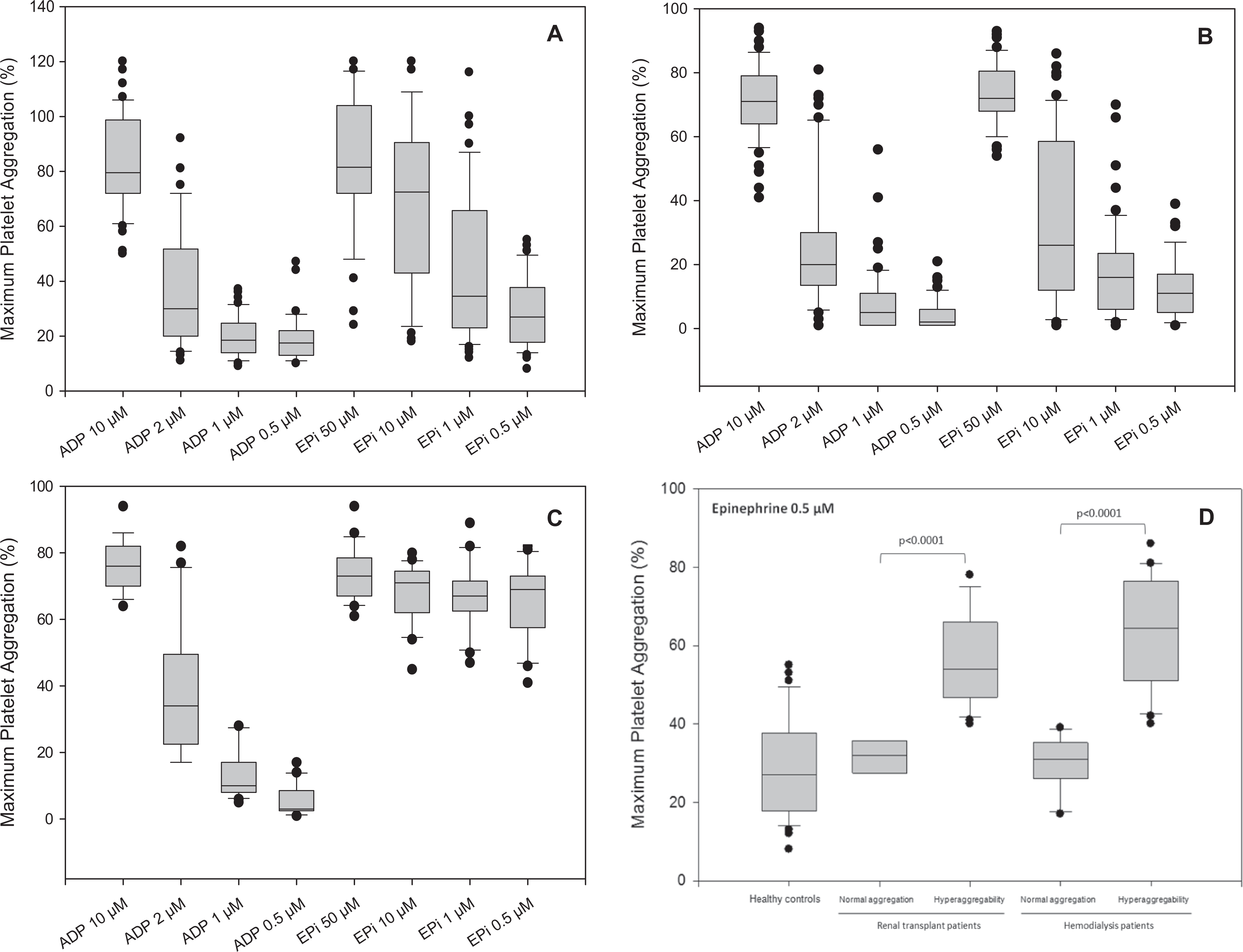
Statistical summary of platelet aggregation by Box-and-Whiskers plots. Dose-dependent platelet aggregation in (A) to (C): (A) healthy controls, (B) non-CKD patients without thromboembolic events, and (C) non-CKD patients with thromboembolic events. Interestingly, significant dose-independent platelet hyperaggregation is found in non-CKD patients with thromboembolic events compared with clearly dose-dependent normal aggregation patterns in healthy controls and non-CKD patients without thromboembolic events. D, Low-dose epinephrine-induced platelet aggregation in healthy controls, hemodialysis patients, and renal transplant patients. Similar to non-CKD patients, platelet aggregation due to low-dose epinephrine is significantly elevated in patients with CKD and related to observed high number of thromboembolic events in renal transplant patients. P values of nonparametric Mann-Whitney U tests are given in (D). Comparisons between collectives are illustrated using Box-and-Whiskers-plots, where the vertical lines in the box indicate the median per group, the box represents 50% of the values, and horizontal lines show minimum and maximum values of the calculated nonoutlier values; dots indicate outlier values. CKD indicates chronic kidney disease.
To assess the prevalence of platelet hyperaggregability in a best-characterized high-risk non-CKD population, we analyzed the reports of platelet aggregation testing in the central laboratory at the RWTH Aachen University between March 2000 and April 2007. During this time, platelet function tests were ordered in 79 patients from the department of gynecological endocrinology and reproductive medicine to assess platelet aggregability as part of the evaluation for infertility. Of these patients, 22 patients had a positive medical history of recurrent spontaneous abortions or fetal loss syndrome, whereas 57 patients were negative for thrombophilia. All 79 patients were negative for plasmatic hypercoagulopathy. Patients with positive medical history had increased and dose-independent 0.5 µmol/L epinephrine-induced platelet aggregation compared to normal platelet aggregation (70%, range 41%-82% vs 11%, range 1%-39%; P < .0001; Fig. 1B and C). Patients with normal dose-dependent platelet function demonstrated a median Δ-epinephrine (difference of the highest vs lowest epinephrine stimulation) of 64% (range 21%-85%; median Δ-ADP 69%; range 35%-89%). The median Δ-epinephrine response in patients with dose-independent platelet hyperaggregation was significantly lower (6%, range −9%-39%; P < .0001; median Δ-ADP 71%, range 61%-85%; P = .0996).
Platelet aggregation response in these patients was used together with the results in healthy controls to determine platelet hyperaggregation in patients with CKD (see Identification of Patients With Platelet Hyperaggregation section and Tables 1 and 2).
Significance Level of Platelet Aggregation of Low Agonist Concentration (0.5 µmol/L) in Healthy Controls and Non-CKD Patients Compared to Patients With Renal Disease.a
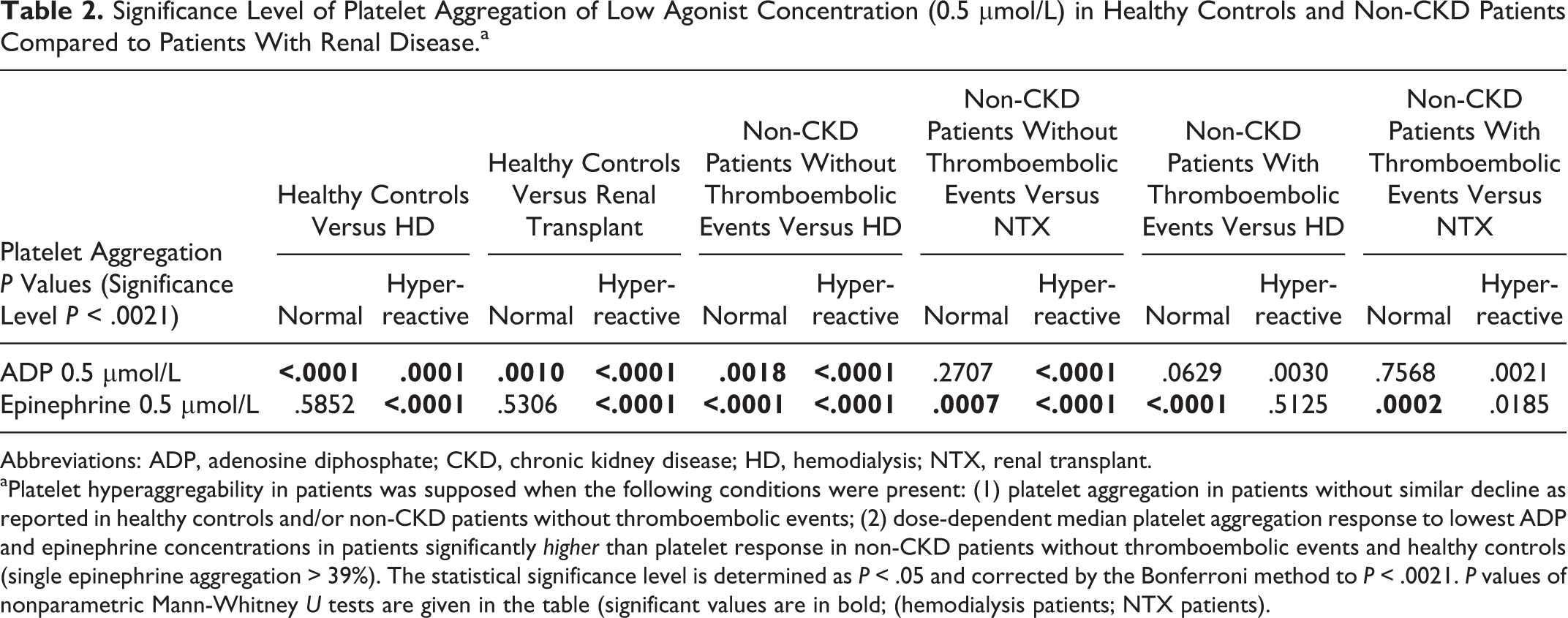
Abbreviations: ADP, adenosine diphosphate; CKD, chronic kidney disease; HD, hemodialysis; NTX, renal transplant.
aPlatelet hyperaggregability in patients was supposed when the following conditions were present: (1) platelet aggregation in patients without similar decline as reported in healthy controls and/or non-CKD patients without thromboembolic events; (2) dose-dependent median platelet aggregation response to lowest ADP and epinephrine concentrations in patients significantly higher than platelet response in non-CKD patients without thromboembolic events and healthy controls (single epinephrine aggregation > 39%). The statistical significance level is determined as P < .05 and corrected by the Bonferroni method to P < .0021. P values of nonparametric Mann-Whitney U tests are given in the table (significant values are in bold; (hemodialysis patients; NTX patients).
Platelet Aggregation in Patients With CKD
Hemodialysis Patients
Platelet function was assessed in 30 HD patients. Ten HD patients were found to have normal platelet aggregation pattern compared to non-CKD and healthy controls. In contrast, median low-dose epinephrine-induced platelet aggregation was significantly higher in 20 HD patients compared to normal aggregation (65%, range 40%-86% vs 31%, range 17%-39%; P < .0001; Table 1 and Figure 1D). In HD patients with normal platelet function, the median Δ-epinephrine was 38% (range 14%-24%; median Δ-ADP 61%, range 46%-70%), while the median Δ-epinephrine response in patients with platelet hyperaggregability was decreased to only 5% (range −6%-20%, P < .0001).
Low-dose epinephrine-induced platelet aggregation in patients with identified hyperaggregability was significantly increased compared to healthy controls and non-CKD patients without thromboembolic events (all P < .0001) and statistically comparable to platelet hyperaggregation in non-CKD patients with thromboembolic events (P = .5125; Table 2).
To exclude activation of platelets during HD as a reason for the high prevalence of platelet hyperaggregation, we analyzed platelet response in 10 patients immediately before and after a HD session. In 8 cases, identical aggregation results were found before and after HD. In 2 patients, hyperreactive platelets changed into normal reactive platelets after HD (data not shown). Taken together, these results rule out mechanical induction of platelet aggregation through the process of HD.
Renal Transplant Patients
Of the 34 patients tested, 6 (18%) were found to have normal platelet aggregation patterns (as described previously) and 28 (82%) showed platelet hyperaggregation (Table 1). Low-dose epinephrine-induced platelet aggregation was significantly higher compared to normal aggregation (54%, range 40%-78% vs 32%, 20%-38%range; P < .0001; Figure 1D). In patients with normal dose-dependent platelet function, the median Δ-epinephrine was 32% (range 16%-57%; median Δ-ADP 68%, range 55%-78%). The median Δ-epinephrine response in renal transplant patients with dose-independent platelet aggregation was significantly decreased (13%, range −12%-60%; P < .0001; median Δ-ADP 64, range 36%-95%; P = .5569).
Low-dose epinephrine-induced platelet aggregation in patients with hyperaggregability was significantly increased compared to healthy controls and non-CKD patients without thromboembolic events (all P < .0001) and statistically comparable to platelet hyperaggregation in non-CKD patients with thromboembolic events (P = .0185; Table 2).
The degree of renal function impairment did not seem to influence platelet aggregation as transplant patients with platelet hyperaggregation were evenly distributed over the CKD stages 1 to 3. By chance, no patients with CKD stage 4 were assessed. Two patients with thrombotic complications immediately after kidney transplantation who never gained sufficient transplant function also tested positive for platelet hyperaggregability (CKD stage 5; data not shown).
There was no difference in the kind of immunosuppressive medications used or the amount of immunosuppression in transplant patients with or without platelet hyperaggregability (Table 3).
Use of Immunosuppressive Medication in Renal Transplant Patients.
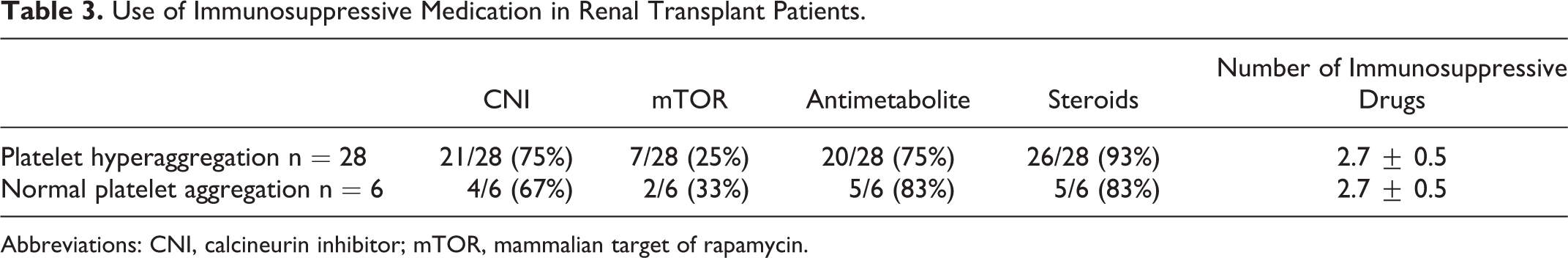
Abbreviations: CNI, calcineurin inhibitor; mTOR, mammalian target of rapamycin.
Thromboembolic Complications in Patients With CKD Having SPS
There was a numerical trend toward more thromboembolic complications in the cohort of renal transplant recipients that tested positive for platelet hyperaggregability. Renal transplant recipients with normal platelet aggregation had no history of thromboembolic events while we could record 30 thromboembolic events in 13 patients diagnosed with platelet hyperaggregation (P = .07; Table 4). On the other hand, 15 of the renal transplant recipients with platelet hyperaggregability had no history of thromboembolic events.
Thromboembolic Events in Renal Transplant Patients Due to Platelet Aggregability.a

aIn 28 patients with platelet hyperaggregability, there were 30 events in 13 patients, whereas none of the 6 patients without platelet hyperaggregability showed thromboembolic events.
Discussion
We found a high prevalence of platelet hyperaggregability in patients with CKD (82%; HD patients, 67% in renal transplant recipients). This prevalence markedly exceeded that of a high-risk disease control, namely, women with recurrent spontaneous abortion (28%). In addition, none of the healthy individuals tested had dose-independent platelet hyperaggregability.
Currently, there is no other study available, which examines platelet aggregation in patients with CKD using ADP and epinephrine-induced light transmission aggregometry. Patients with CKD have a high risk of thromboembolic complications and cardiovascular events. 16 In general, the increase in risk is thought to be multifactorial. In part it might be the result of the same underlying diseases causing vascular disease and CDK (hypertension, diabetes, hyperlipidemia, etc) and in part secondary to CKD-induced disturbances in calcium and phosphate homeostasis and uremic toxins. In addition, in the situation of renal transplantation, there is a high incidence of deep-vein thrombosis, probably due to mechanical alterations by the surgical procedure and the transplant. 17 Overall, our study suggests that increased platelet aggregability might reflect these factors and contribute to the increased risk of thromboembolic events in patients with CKD.
So far, platelet hyperaggregability has been examined in the setting of recurrent miscarriages and unexplained arterial and venous thromboembolic events in non-CKD patients as a reason for vascular morbidity. Bick et al examined 351 women having recurrent miscarriages. 2 Of this population, 20% was found to have SPS. The same authors reported on 153 patients referred for the evaluation of unexplained arterial or venous events and found SPS prevalence of 21% for patients with arterial events and 13% in those with venous events. 18 Kubisz et al investigated 64 patients with a history of arterial or venous thromboembolic events and found SPS in 14% of those patients. 19 In contrast, Ruiz-Argüelles et al found 6 of 10 patients with primary hypercoagulable state to be affected by SPS of which most patients had more than 1 thrombophilic abnormality. 20,21
The prevalence of platelet hyperaggregability in the form of SPS in the general population has never been formally assessed. So far, only patients with clinical events of thromboembolism have been tested for SPS, and no data is available on the rate of thromboembolic events in patients with SPS. In healthy controls without thromboembolic events, we could not find platelet hyperaggregability. In addition, none of the patients with recurrent miscarriages and SPS had any record of clinical thromboembolism other than the gynecological disease. In our study, the relationship between platelet hyperaggregability and thromboembolic events in patients with CKD was not clear. Although there was a high number of thromboembolic events in renal transplant recipients with SPS (30 events in 13 of 28 patients with hyperreactive platelets) compared to patients with normal platelet function (0 events in 6 patients with normal platelet function), it did not reach statistical significance. It is likely that platelet hyperaggregability itself does not cause thromboembolic events, similar to APC resistance or protein C deficiency, but only predisposes to them and that a second hit may be required to develop overt clinical disease. In the literature, it has already been proposed that epinephrine release in vivo may act as an inducer of platelet aggregation. 22 In the case of renal transplant recipients, endothelial injury caused by treatment with immunosuppressive drugs such as cyclosporine A and tacrolimus 23,24 might represent a trigger for the induction of thrombosis. In addition, renal transplant patients have many other risk factors for induction of the coagulation system such as perioperative stress with the release of adrenalin, surgical damage to the vasculature, rejection episodes, and uncontrolled hypertension. These triggers might explain the higher incidence of thromboembolic diseases in the renal transplant population. In the case of HD patients, platelet hyperaggregability-induced thrombophilia might be counteracted by uremic bleeding disorder. However, although there was a numerical increase in thromboembolic events in patients with CKD having platelet hyperaggregability after renal transplantation, the number of patients assessed was too small to allow a meaningful statistical analysis. Larger prospective studies in patients with CKD will have to be performed to assess the risk of thromboembolic complications added by platelet hyperaggregability.
A limitation of this study is that from a methodological standpoint, the definition of platelet hyperaggregability is not standardized to date. 9,14 Furthermore, interpretation of platelet aggregability is heavily affected by preanalytical errors and, so far, it depends on nonstandardized laboratory methods. There are also uncertainties about the limits of acceptable platelet counts. The PRP platelet count shows little relationship to maximum aggregation in samples with normal platelet counts. 12,13 Furthermore, there are no validated reference intervals for platelet aggregation by multiple concentrations of agonists. However, Yee et al reported in 2005 that light transmission aggregometry using 0.4 µmol/L citrated PRP reliably identifies a distinct subgroup of healthy individuals with platelet hyperaggregability. 25
In conclusion, our study highlights that increased platelet aggregability can be found in patients with CKD who are at a high risk of thromboembolic events. This change in platelet function might contribute to the increased rate of vascular events in patients with CKD. Platelet function testing in patients with CKD should be taken into clinical consideration and might thus indicate a potentially increased risk of thromboembolic events. Whether screening for platelet hyperaggregability in patients with CKD and treatment with acetylic salicylic acid, particularly in those patients with CKD and thromboembolic events, will ultimately affect patient outcomes have to be tested in prospective trials.
Footnotes
Acknowledgment
The authors gratefully acknowledge Mrs Claudia Brügmann for revising the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
