Abstract
The aim of this study was to evaluate the prevalence of aspirin resistance (AR) in patients undergoing hemodialysis (HD) and to assess the effect of HD on the results of the Multiplate test. A total of 54 patients undergoing HD were included in this study. Blood samples were taken just before and after the HD session. To determine AR, we used Multiplate test. Platelet aggregation values of the study population were 363.01 ± 225.69 aggregation unit (AU) × minutes before and 375.33 ± 254.05 AU × minutes after the HD (P = .597). There was strong correlation between the values before and after HD (R = .755, P < .0001). The AR status was changed in 9 (16.6%) patients after HD. Agreement of AR status before and after HD was substantial (κ coefficient = .667, P < .0001). The prevalence of AR in patients undergoing HD seems higher than in most of the studied populations, and this study has shown that the AR statuses of a significant number of patients undergoing HD change after an HD session.
Introduction
Aspirin is the most widely used antiplatelet agent worldwide. It acts by irreversibly inhibiting the platelet cyclooxygenase 1 enzyme, thus preventing the production of thromboxane A2 (TXA2)—a potent vasoconstrictor and platelet activator—from arachidonic acid. 1,2 Aspirin is used in the primary and secondary prevention of thromboembolic vascular events. However, patients taking aspirin might exhibit variable responses to in vitro tests for platelet aggregation and might experience recurrent thromboembolic events under aspirin treatment. This phenomenon has been called aspirin resistance (AR). However, because of the variability in diagnostic tests and absence of controlled randomized trials, there is no uniform definition for AR, and its importance for prognoses of all patient population is not known. The estimates of AR prevalence vary widely (from 2% to 57%), depending on the usage of various laboratory tests, uncertainty about the exact definition, and the broad range of disease states being investigated. 3 –12 The AR is associated with an increased risk of cardiovascular-, cerebrovascular-, and vascular-related events. 8,9
The AR is a multifactorial phenomenon. Possible causes of resistance include genetic polymorphism, factors related to compliance and absorption of aspirin, inadequate dose, upregulation of alternative pathways for thromboxane synthesis, reduced bioavailability, increased platelet turnover, and drug interactions. 13,14
Aspirin is also widely used in patients undergoing hemodialysis (HD) for various indications and in the prevention of access graft thrombosis, cardiovascular, and cerebrovascular events. Approximately 30% of the patients have coronary artery disease at the beginning of the HD treatment, and their risk of 1-year mortality related to cardiac events after myocardial infarction is higher than in the general population. 15 Therefore, aspirin treatment is important for these patients. Some investigators showed increased AR in this patient group. 16 As HD has complex effects on platelet functions, 17 –19 it may affect patients’ responsiveness to aspirin as well as the results of tests used for evaluating aspirin responsiveness.
The aim of our study is to evaluate the prevalence of AR in patients undergoing HD and to evaluate the effect of HD on their AR statuses as determined by a Multiplate test.
Materials and Methods
We studied aspirin responsiveness in 54 patients undergoing HD (19 [35.2%] female, 35 [64.8%] male, mean age of 56.79 ± 12.74) patients. The study was approved by the local ethics committee, and all participants gave written informed consent before participating. The 54 patients in the study were drawn from a population of 150 patients at 2 HD centers. All patients had been on regular aspirin therapy for at least 7 days. Patients who were excluded from the study were those who were taking an antiplatelet therapy other than aspirin (ticlopidine, clopidogrel, dipyridamole, nonsteroidal anti-inflammatory drug, pentoxifylline, and cilostazol); who had been treated with glycoprotein IIb/IIIa inhibitors within the previous 10 days; those with a diagnosis of acute coronary syndrome, active malignancy, or hemorrhagic diathesis; who had thrombolytic treatment within last month; those with liver disease; those with platelet counts <100 000/μL; and who were thought to be noncompliant with the medication.
The patients were dialyzed 3 times a week, for 4 hours every session, with polysulfone low-flux dialyzers. All patients underwent HD by cannulation of arteriovenous fistula and using sterile bicarbonate concentrate, heparin, and reverse osmosis water. Dialysate and blood flow rates were 500 and 350 to 400 mL/min, respectively. Anticoagulation was performed with an intravenous bolus of 2000 units of heparin followed by a 1000 units/h infusion for 3 hours. We used Multiplate tests to determine AR and obtained fasting blood samples to determine blood glucose, hemoglobin, hematocrit, leukocyte, and platelet count.
Assessment of AR With Multiple Electrical Impedance Aggregometry
Whole blood aggregation was performed using the Multiplate analyzer (Dynabyte Medical, Munich, Germany), an impedance aggregometer, based on the principle that activated platelets expose receptors on their surface, which allow them to attach to artificial surfaces. Whole blood samples were drawn into the test tubes containing hirudin (25 Mcg/mL) as anticoagulant. Two blood samples were collected from each patient just before starting HD after 1 hour of aspirin ingestion and at the end of the HD session. All analyses were performed within 2 hours of sampling. Arachidonic acid was used as aggregation agonist. The aggregation measured with this device is quantified as area under the curve, aggregation degree, and aggregation velocity. Platelet aggregation results were presented as aggregation unit (AU) × minutes, and values over 300 AU × minutes were accepted as AR. 20
Statistics
Results are presented as mean values and standard deviations for continuous variables or as ratios for categorical variables. Categorical variables were compared using a chi-square test, while continuous variables were compared by a Mann-Whitney U test. Wilcoxon signed-rank test was used to compare before and after dialysis tests results. κ-Values were calculated to determine the AR status agreement before and after HD. Pearson and Spearman correlation tests were used for univariate analysis. A probability (P) value <.05 was interpreted as statistically significant. All statistical tests were performed using the statistics program SPSS 15 for Windows.
Results
The general characteristics and laboratory parameters of the 54 patients included in our study are shown in Table 1.
Demographic Characteristics and Laboratory Values of the Study Group.
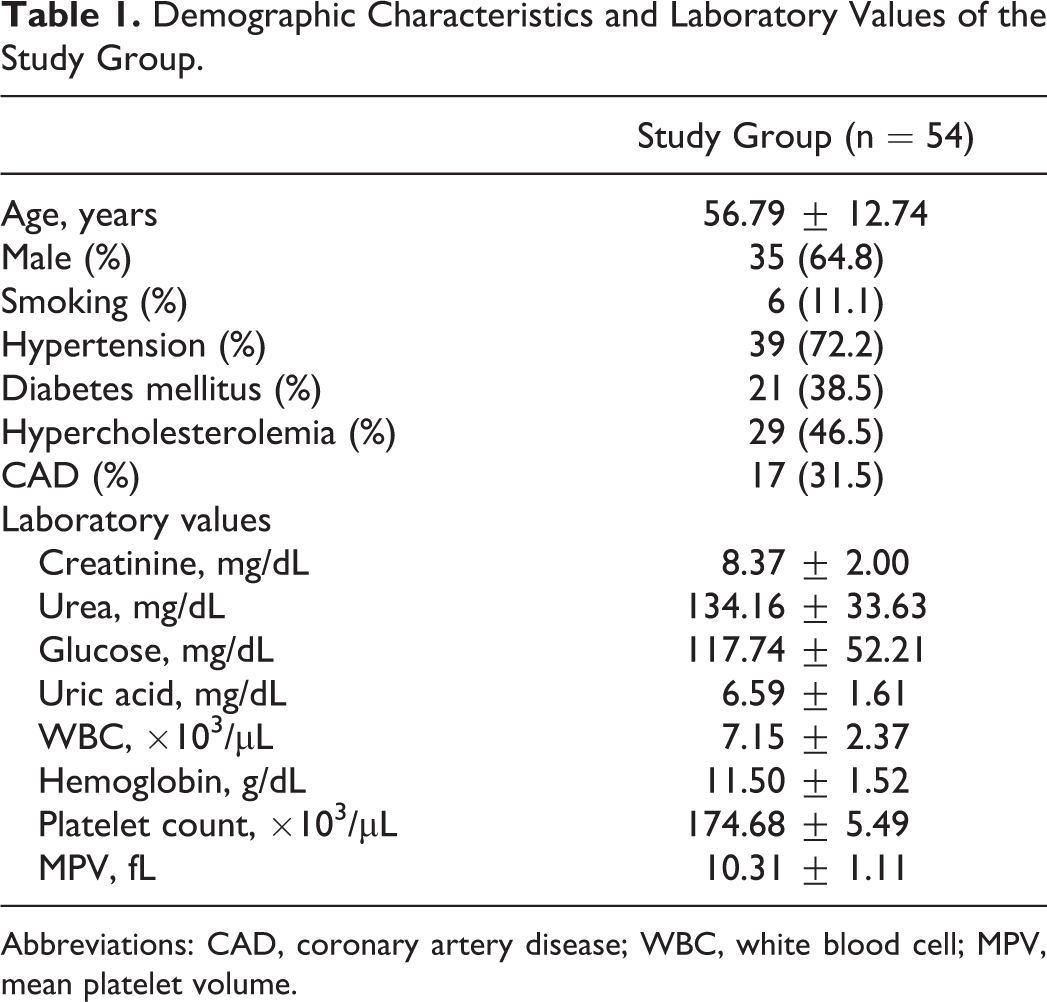
Abbreviations: CAD, coronary artery disease; WBC, white blood cell; MPV, mean platelet volume.
Platelet aggregation values of the study population measured by Multiplate test were 363.01 ± 225.69 AU × min before HD and 375.33 ± 254.05 AU × min after the HD (P = .350). There was a strong correlation between the values before and after HD (R = .755, P < .0001; Figure 1).
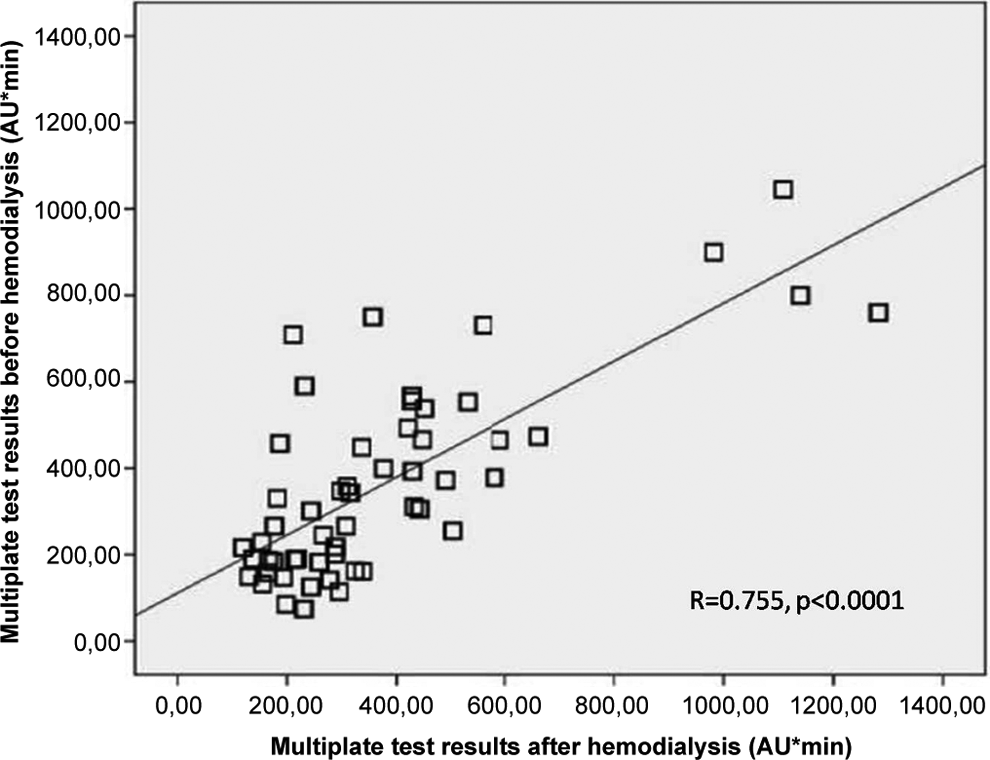
Relation of Multiplate test results before and after hemodialysis.
The AR was detected in 28 (51.9%) patients before HD, whereas in 27 (50%) patients after HD (P < .0001). The AR status was changed in 9 (16.6%) patients after HD. The AR emerged in 4 patients after HD who had no AR before HD, whereas AR disappeared in 5 patients after HD who had AR before HD. Agreement of AR status before and after HD was substantial (κ coefficient = .667, P < .0001).
In Figure 1, if 4 points reflecting the patients with extreme Multiplate test results and possibly disrupting the equal distribution are excluded, correlation of before and after HD Multiplate test results is decreased (R = .550, P < .0001) but still moderate. Also, after exclusion of these points, agreement of AR status before and after HD was still substantial (κ coefficient = .639, P < .0001), and the AR status change ratio is increased to 18%.
There was not any statistically significant difference except sex ratios between AR and aspirin responsive groups according to the test results before and after HD (Tables 2 and 3).
Comparisons of Characteristics and Baseline Laboratory Values of Aspirin Resistant and Responser Groups Before Hemodialysis.
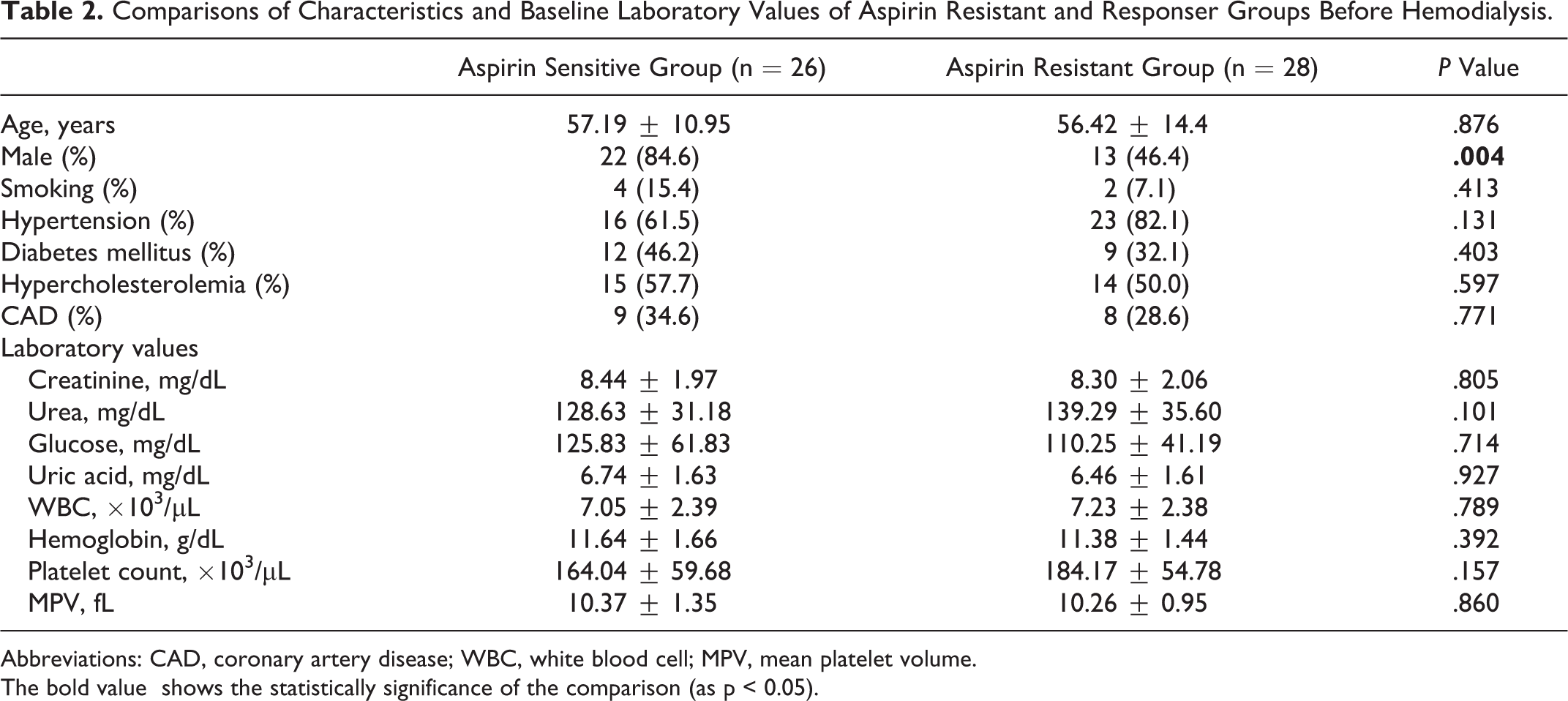
Abbreviations: CAD, coronary artery disease; WBC, white blood cell; MPV, mean platelet volume.
The bold value shows the statistically significance of the comparison (as p < 0.05).
Comparisons of Baseline (Before HD) Characteristics and Laboratory Values of Aspirin Resistant and Responser Groups After HD.
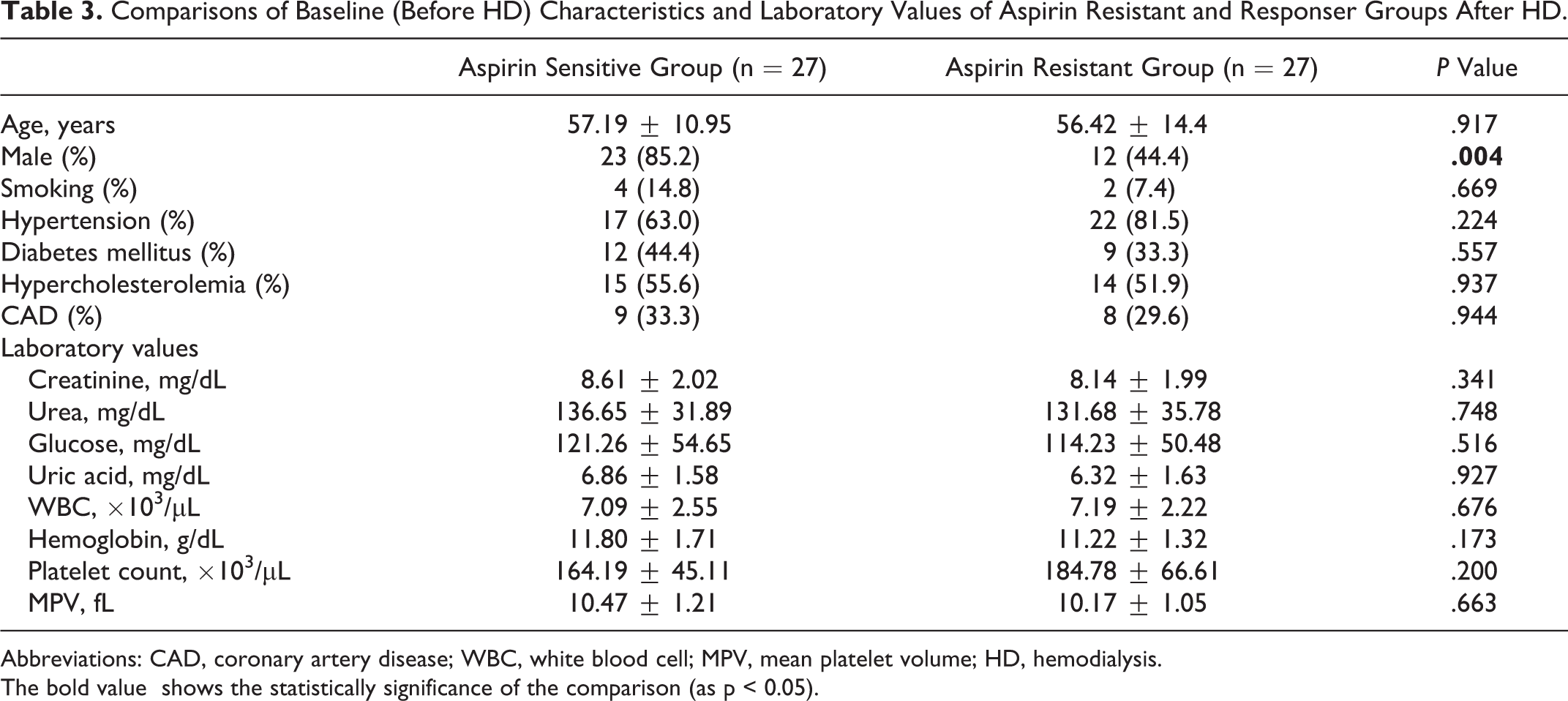
Abbreviations: CAD, coronary artery disease; WBC, white blood cell; MPV, mean platelet volume; HD, hemodialysis.
The bold value shows the statistically significance of the comparison (as p < 0.05).
Multiplate test results were correlated only with sex status (R = .292, P = .032) before HD but with sex status (R = .361, P = .007), hemoglobin values (R = .289, P = .036), and platelet counts (R = .347, P = .011) after HD. The AR status was correlated only with sex status either before or after HD (consecutively, R = .400, P = .003; R = .427, P = .001).
Discussion
Aspirin resistance is a multifactorial phenomenon that has been shown to have bad prognostic effects. The AR has been studied in different patient groups using different methods. The AR was studied in patients with chronic renal failure, and its incidence was found to be higher, especially in HD groups. 16 However, AR was assessed only before HD in these studies, despite it being known that HD has effects on platelet functions. 17 –19
In our study, we assessed aspirin responsiveness in 54 patients with chronic renal failure immediately before and after an HD session using the Multiplate method. We found AR in 28 (51.9%) patients before the HD and in 27 (50.0%) patients after the HD. In their study evaluating AR in patients with chronic renal failure, Tanrikulu et al found AR in 46.1% of the patients undergoing HD assessed before undergoing HD. 16 This result is consistent with our study results. Geara et al found AR in 34.8% of the patients undergoing HD. 21 This is lower than our result, but this difference may result from differences in the study population and in the methods that were used to determine AR.
The exact cause of increased AR in patients undergoing HD is not known. There is a complex platelet dysfunction in patients with uremia. Patients undergoing HD and patients with uremia are prone to both hemorrhagic and thrombotic complications. Half of the vascular access is clotted in 2 years. 22 Increased oxidative stress and inflammation were shown in patients with chronic renal failure and patients undergoing HD. 23 HD exacerbates oxidative stress and disturbances in antioxidant enzymes in patients with uremia. 24 The increase in TXA2 production, because of this increased inflammation and oxidative stress, may cause AR development in patients undergoing HD.
Previous study results, as well as our before and after HD study results, suggest that the incidence of AR in patient undergoing HD is higher than in the general population. The effect of increased AR on the prognoses of patients undergoing HD has not yet been studied. However, increased AR may contribute to increased cardiovascular mortality in patients undergoing HD. A determination of a patient’s response to aspirin and the addition of another antiplatelet drug according to the patient’s AR status may be helpful in patients undergoing HD.
In our study, we found that although platelet aggregations of the study participants—as determined by Multiplate tests before and after HD—were not significantly different or strongly correlated, the AR statuses changed significantly, and agreement of the AR status was moderate. Studies that compare AR status before and after HD do not currently exist. Geara et al assessed clopidogrel resistance in patients undergoing HD before and after HD sessions; they found differences in results using a VerifyNow test. 25
The exposure to a dialysis circuit affects both platelet counts and functions. Platelet count decreases during the 15 to 30 minutes of HD and usually resolves by the end of the session. The HD can cause some complex changes in platelet reactivity. These are biphasic effects, because they increase in reactivity on initiation of HD and then decrease by the end of the procedure. 17 The exact cause of these changes is not well known, but they may be due to adenosine diphosphate-mediated activation of platelets, 18 exposure of blood to the roller pump segment of the dialysis tubing, or microbubbles. 19 The clinical consequences of these HD effects on platelets are unknown.
In our study, the percentage of women with AR was higher than the percentage of men with AR. As well, females were related to AR both before and after HD. This is consistent with many other studies showing increased AR in females. 16,7
This is the first study evaluating the effect of HD on a patient’s AR status and testing this status twice, once before and once after the HD session. In our study, Multiplate tests after HD sessions demonstrated changes in the AR statuses of some patients with chronic renal failure. More studies are needed to evaluate the right time and the right test to be used in this patient population. New cutoff values for the AR status of patients undergoing HD may be needed.
Study Limitations
The observational nature of our study did not allow the demonstration of factors contributing to increased AR statuses in patients undergoing HD. Aspirin compliance was based on a face-to-face interview with the patient and with the person responsible for the patient’s primary care, but salicylate levels were not measured. Finally, no locally determined reference ranges for the Multiplate test exist in our country; we therefore used ranges from the existing literature.
Conclusions
The prevalence of AR in patients undergoing HD seems higher than in most of the studied populations, and this study has shown that the AR statuses of a significant number of patients undergoing HD change after an HD session.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
