Abstract
We aimed to assess the effective factors on high mean platelet volume (MPV) in patients with stable coronary artery disease (CAD). A total of 411 patients (247 males and 164 females; mean age: 61.7 ± 9.9 years) with angiographically proven CAD were included. The patients were divided into 2 groups according to the median MPV value (MPVlow group <9.5 fL and MPVhigh group ≥9.5 fL). The SYNTAX score, high sensitive C-reactive protein (hsCRP) levels, and frequencies of diabetes and hypertension were higher in MPVhigh group compared to MPVlow group. Aortic distensibility (AD) and platelet count of patients in MPVhigh group were lower than patients in MPVlow group (P < .05, for all). Multivariate linear regression analysis showed that MPV was independently related with diabetes (β = 0.135, P = .007), hsCRP (β = 0.259, P < .001), platelet count (β = −0.144, P < .001), and AD (β = −0.425, P < .001). High MPV value is independently related to AD, as well as diabetes, hsCRP, and platelet count in patients with stable CAD.
Introduction
Platelets play an important role in thrombus formation and the pathogenesis of acute coronary syndromes (ACS). 1 The mean platelet volume (MPV) is a measurement of the average size of the platelets in the blood. 2 Elevated MPV value has been recognized as an independent risk factor for adverse clinical outcomes among survivors of myocardial infarction (MI). 3 On the other hand, patients with increased MPV values had a higher risk of developing MI when compared to those with lower MPV values in stable coronary artery disease (CAD). 4 Increased MPV is associated with higher mortality following MI. 5
Arterial stiffness is a marker for increased cardiovascular disease risk, including MI, heart failure, and total mortality as well as stroke. 6 –11 Aortic distensibility (AD) is a measurement of vascular elasticity, which reflects the stiffness of aorta. 12 It has been shown that AD is inversely proportional to disease severity and presence of cardiovascular risk factors. 13,14 Moreover, high MPV values were observed in healthy participants with increased aortic stiffness. 15 However, relationship between MPV and AD has not been investigated in patients with CAD.
The MPV value increases in patients with CAD, 16,17 but little is known regarding the association of MPV levels with extent and complexity of CAD, AD, and inflammation in patients with stable CAD. In this study, we aimed to assess the relationship between increased MPV values and the extent and complexity of CAD, AD, and risk factors such as diabetes, hypertension, and high sensitive C-reactive protein (hsCRP) in patients with stable CAD.
Methods
Study Population
In all, 411 consecutive patients (247 males and 164 females; mean age: 61.7 ± 9.9 years) with angiographically proven CAD admitted to our cardiology clinic for angiography between January 2012 and July 2012 were included in the study. Coronary angiography was performed for the investigation of ischemic heart disease based on clinical indications (typical chest discomfort and/or abnormal stress test results such as positive treadmill test, dobutamine stress echo, and myocardial perfusion scintigraphy). The patients with coronary lesions with a diameter stenosis ≥50%, in vessels ≥1.5 mm were included in the study. All patients were clinically stable. Exclusion criteria were the presence of neoplastic disease, heart failure, recent major surgical procedure, or liver or kidney disease. Patients with previous MI and angina episodes 48 hours before hospitalization, who had undergone coronary angioplasty or bypass surgery and those with valvular, myocardial, or pericardial disease, were also excluded. The study was conducted according to the recommendations set forth by the Declaration of Helsinki on Biomedical Research Involving Human Subjects. The institutional ethics committee approved the study protocol and each participant provided written informed consent.
After taking detailed medical history and complete physical examination, each participant was questioned for major cardiovascular risk factors such as age, sex, diabetes mellitus, smoking status, and hypertension. In addition, body mass index was calculated and systolic blood pressure (SBP) and diastolic blood pressure (DBP) were recorded.
Blood Samples
Fasting venous blood samples were obtained from all patients to determine their plasma levels of fasting blood glucose, total cholesterol, high-density lipoprotein cholesterol, low-density lipoprotein cholesterol, triglyceride, creatinine, hsCRP, and hemoglobin. We collected the blood samples into tubes containing dipotassium EDTA for the measurements of the platelet count and MPV. The samples were analyzed within 20 minutes after collection using an automated hematology analyzer Sysmex XT 1800i (Roche Diagnostic, Shanghai, China). The patients were divided into 2 groups according to the median MPV value (MPVlow group <9.5 fL and MPVhigh group ≥9.5 fL).
Echocardiography and AD
Standard 2-dimensional examinations were performed using commercially available equipment (Vivid-7, GE Vingmed Sound, Horten, Norway) with a 2.5- to 35 MHz transducer before angiography. Simultaneous electrocardiogram (ECG) recordings were also obtained. All echocardiograms were performed and analyzed by one observer. Ejection fraction (EF) was determined using the Simpson method, according to the suggestions of the American Society of Echocardiography. 18
Ascending aorta (Ao) diameters were measured from the same view on the M mode tracing at a level 3 cm above the aortic valve. The aortic systolic (AoS) diameter was measured at the maximum anterior motion of the aorta and the aortic diastolic (AoD)diameter was measured at the peak of the QRS complex on the simultaneously recorded ECG. Pulse pressure (PP) was obtained simultaneously by cuff sphygmomanometry of the left brachial artery as SBP minus DBP. The AD was calculated by the formula AD = 2 × (AoSD − AoDD)/(AoDD × PP) (cm2 dyn−1 × 10−6). 12,19
SYNTAX Score and Angiographic Analysis
Coronary lesions leading to ≥50% diameter stenosis in vessels ≥1.5 mm was scored separately and added together to provide the cumulative SYNTAX score (SS) which was prospectively calculated using the SS algorithm on the baseline diagnostic angiogram. 20 Two experienced interventional cardiologists analyzed the SS; the opinion of a third analyst was obtained and the final judgment was made by consensus in cases of disagreement. The final score was calculated from the individual lesion scores by analysts who were blinded to procedural data and clinical outcome.
Statistical Analysis
All analyses were conducted using SPSS 17.0 (SPSS for Windows 17.0, Chicago, Illinois). Continuous variables were expressed as mean ± standard deviation and categorical variables were expressed as percentages. Comparison of categorical variables between the groups was performed using the chi-square (χ2) test. Analysis of normality was performed with the Kolmogorov-Smirnov test. Comparisons of continuous variables between the 2 groups were performed using the independent samples t test. The correlation between MPV and clinical and laboratory parameters was assessed by the Pearson correlation test. Multiple linear regression analysis was performed to identify the independent relations of MPV by including the parameters, which were correlated with MPV on bivariate analysis. A 2-tailed P <.05 was considered as statistically significant.
Results
During the study period, 411 patients with stable CAD were included into the study. The patient groups were defined as MPVlow <9.5 fL (n = 205) and MPVhigh ≥9.5 fL (n = 206).
Comparisons of baseline, clinical, laboratory, echocardiographic, and angiographic findings were summarized in Table 1. Age, SBP, and PP values were higher in MPVhigh group compared to MPVlow group (P < .05, for all). Diabetes and hypertension frequencies of MPVhigh group were higher than MPVlow group (P < .05, for all). Total cholesterol, creatinine, and hsCRP levels were higher and platelet count was lower in MPVhigh group compared to MPVlow group (P < .05, for all). The AD was lower and SS was higher in MPVhigh group compared to MPVlow group (P < .05, for all). Angiotensin-converting enzyme inhibitor use and oral antidiabetic use were different between the groups (P < .05, for all). Bivariate and multivariate relationships of the MPV in patient groups were reported in Table 2.
Comparison of Baseline, Clinical, Laboratory, Echocardiographic, and Angiographic Findings.
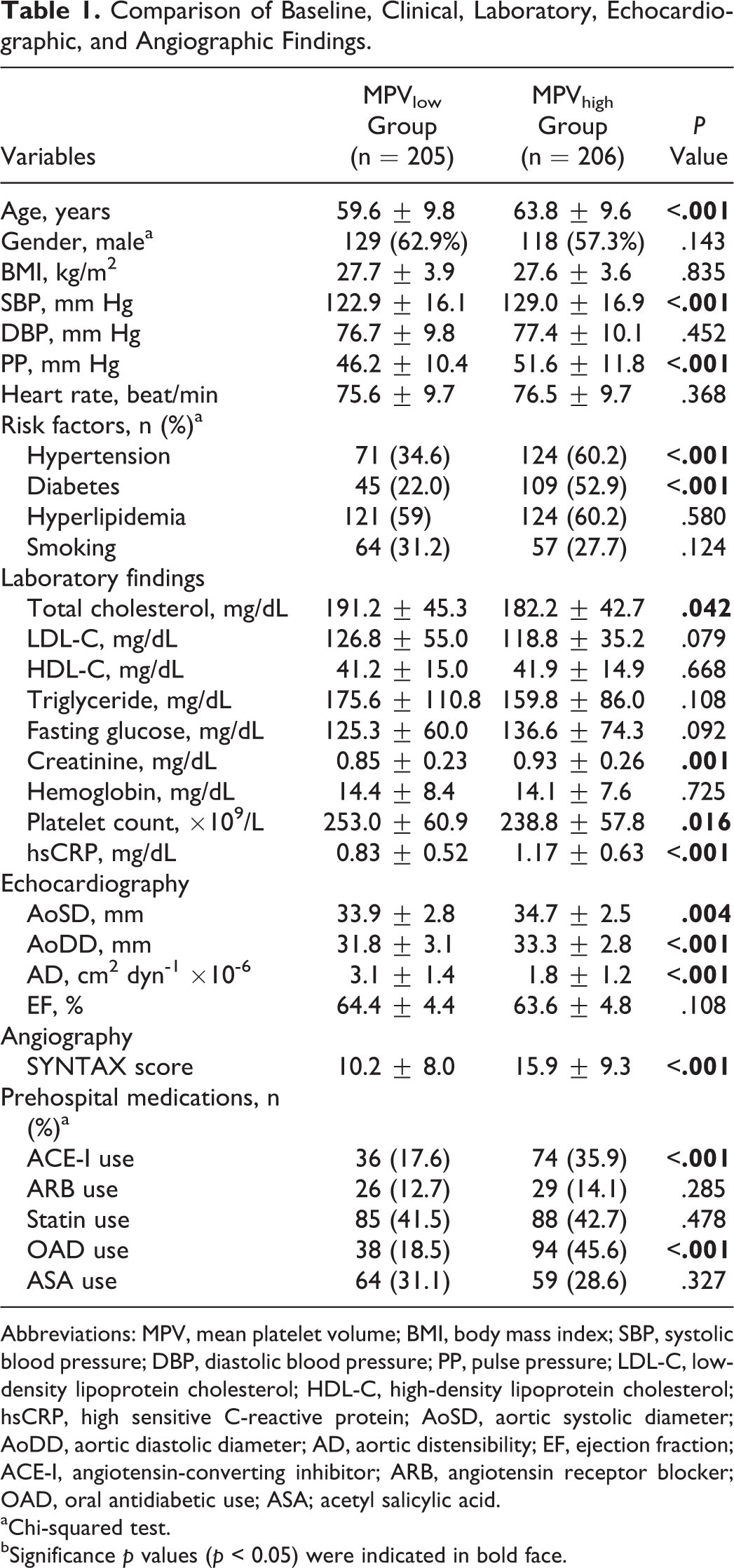
Abbreviations: MPV, mean platelet volume; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; PP, pulse pressure; LDL-C, low-density lipoprotein cholesterol; HDL-C, high-density lipoprotein cholesterol; hsCRP, high sensitive C-reactive protein; AoSD, aortic systolic diameter; AoDD, aortic diastolic diameter; AD, aortic distensibility; EF, ejection fraction; ACE-I, angiotensin-converting inhibitor; ARB, angiotensin receptor blocker; OAD, oral antidiabetic use; ASA; acetyl salicylic acid.
aChi-squared test.
bSignificance p values (p < 0.05) were indicated in bold face.
Bivariate and Multivariate Relationships of Mean Platelet Volume.
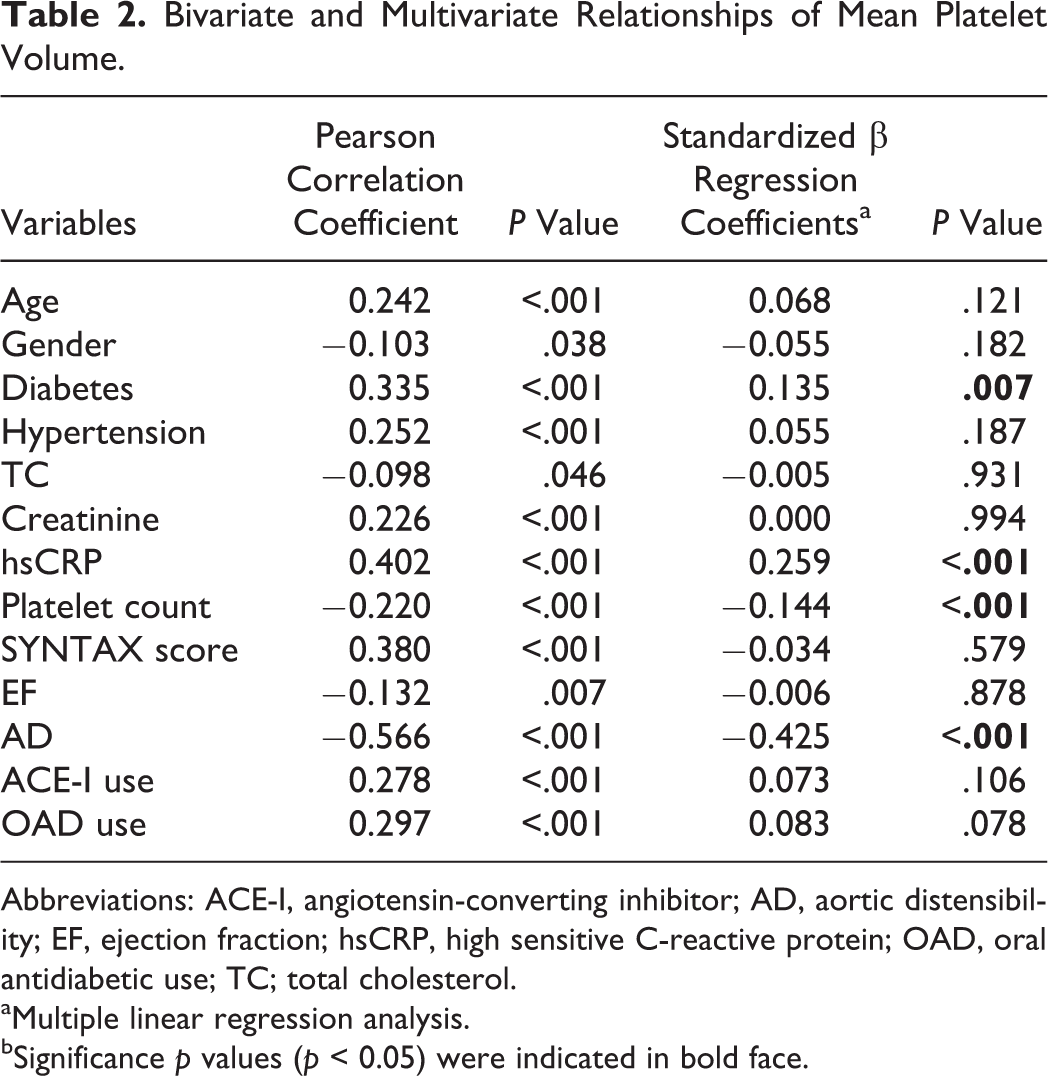
Abbreviations: ACE-I, angiotensin-converting inhibitor; AD, aortic distensibility; EF, ejection fraction; hsCRP, high sensitive C-reactive protein; OAD, oral antidiabetic use; TC; total cholesterol.
aMultiple linear regression analysis.
bSignificance p values (p < 0.05) were indicated in bold face.
Bivariate and Multivariate Relationships of MPV
Mean platelet volume was associated with age (r = .242, P < .001), gender (r = −.103, P = .038), diabetes (r = .335, P < .001), hypertension (r = .252, P < .001), total cholesterol (r = −.098, P = .046), creatinine (r = .226, P < .001), hsCRP (r = .402, P < .001), platelet count (r = −.220, P < .001), SS (r = .380, P < .001), EF (r = −.132, P = .007), angiotensin-converting enzyme inhibitor use (r = .278, P < .001), oral antidiabetic use (r = .297, P < .001), and AD (r = −.566, P < .001) on bivariate analysis.
Multivariate linear regression analysis showed that MPV was independently related to diabetes (β = .135, P = .007), hsCRP (β = .259, P < .001), platelet count (β = −.144, P < .001), and AD (β = −0.425, P < .001). Relationship between MPV and AD was demonstrated in Figure 1.
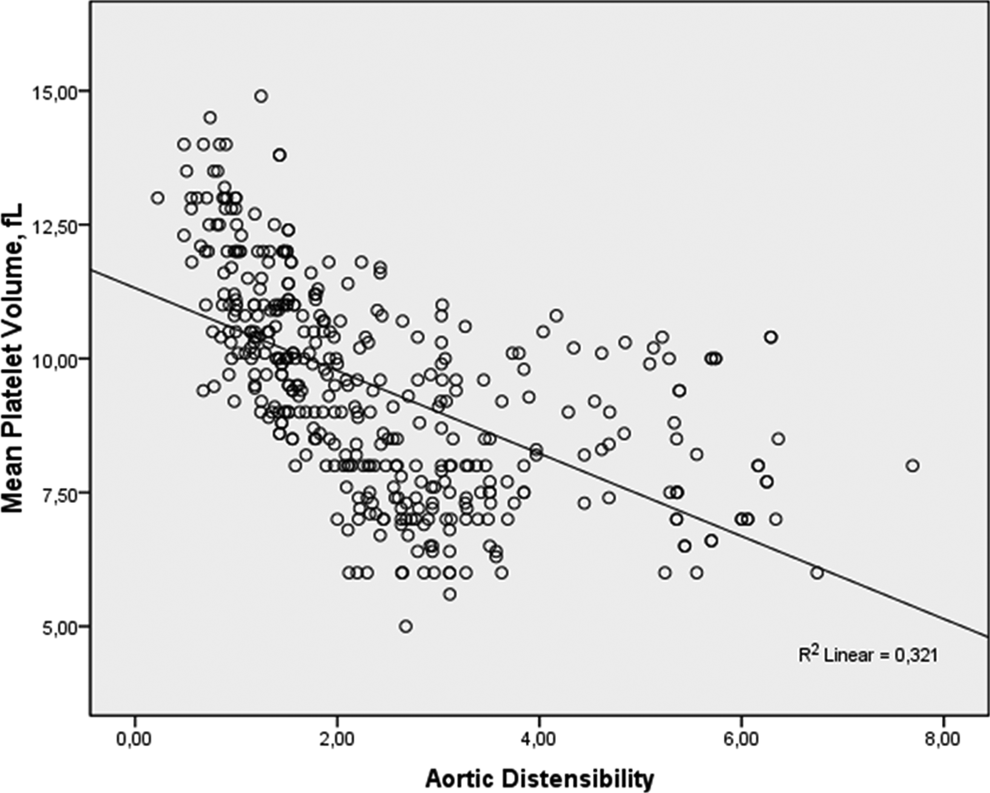
Scatter graph showing the correlation between mean platelet volume and aortic distensibility.
Discussion
The present study showed that MPV was independently associated with AD, as well as diabetes, hsCRP, and platelet count in patients with stable CAD. Although MPV was associated with extent and complexity of CAD assessed with SS on bivariate analysis, similar relationship was not observed in multivariate analysis.
The MPV is a measurement of the average size of the platelets in the blood and is associated with a variety of prothrombotic and proinflammatory disease states. 2 –5,21 The present study showed that MPV was independently associated with AD, which reflects to the stiffness of aorta and is a measurement of vascular elasticity. The relationship between MPV and arterial stiffness was investigated in a limited number of studies. 15,22 Wang et al 15 reported that high MPV values were positively correlated to arterial stiffness in healthy participants. In that study, the study group, unlike our study, was consisted of healthy individuals. However, the current study included patients with stable CAD. Recently, it has been shown that high MPV values were found in patients with bicuspid aorta and high MPV values were associated with aortic stiffness in this patient group. 22 Multiple mechanisms may be involved for relationship between MPV and AD. Larger platelets are metabolically and enzymatically more active and have greater prothrombotic potential and aggravate inflammation and endothelial dysfunction. 23 –26 Reduced nitric acid synthesis caused by endothelial dysfunction contributes to arterial stiffness. 25,26 Activated platelets release more platelet-derived growth factors that stimulate the proliferation of smooth muscle cells. Increased matrix metalloproteinase activity related to activated platelets further leads to a reduction in arterial elasticity by degradation of elastic fiber. 27 Furthermore, upregulation of matrix metalloproteinase 2 and 9 contributes to stiffening and vasomotor dysfunction in patients with chronic kidney disease. 28 On the other hand, a previous study reported that arterial stiffness was associated with platelet activation by measuring platelet surface expression of P-selectin and platelet–neutrophil complexes. 29 Impaired AD, which reflects increased aortic stiffness, can lead to increased shear stress. It is known that shear stress in turbulent flow induces platelet activation. In addition, AD is associated with aortic atherosclerosis. 30 It is known that there is a close relationship between aortic atherosclerosis and coronary atherosclerosis. 13 In the present study, high MPV values may be associated with increased aortic atherosclerosis in patients with impaired AD.
Previous studies showed that MPV is an inflammatory indicator in different diseases. 21,31,32 Inflammation plays an important role in the rupture of arterial plaques, which is the usual precipitating cause of arterial thrombotic events. 33 Circulating markers or mediators of inflammation such as C-reactive protein 33,34 and interleukin 6, 35 are associated with the risk of atherothrombotic events. The C-reactive protein also contributes to platelet activation and thus increases the risk of coronary heart disease. 33,34 On the other hand, high MPV value in adult nondipper hypertensives was shown to be a part of low-grade inflammation, with a correlation between MPV and hsCRP. 36 The present study reported that high MPV value was independently associated with hsCRP, which reflects low-grade chronic inflammation. High MPV values may be part of the low-grade chronic inflammation in stable CAD.
In the present study, MPV was independently associated with diabetes in patients with stable CAD. Higher MPV value is observed in patients with diabetes mellitus. 16,37 Tavil et al reported that diabetic patients with CAD have significantly higher MPV values compared to the control participants without diabetes and angiographically normal coronary arteries. 16 The mechanism of high MPV in DM is osmotic swelling due to raised blood glucose and due to raised levels of some glucose metabolites. 38 Insulin resistance, diabetes mellitus, and other risk factors associated with endothelial dysfunction might trigger a series of cytokine production which in turn may stimulate the production of large platelets in the bone marrow. 23 In the present study, the above-mentioned mechanisms may be responsible for higher MPV values in diabetic stable CAD.
Our study showed that high MPV value was associated with extent and complexity of CAD assessed with SS on bivariate analysis, but similar relationship was not observed in multivariate analysis. The studies investigating the relationship between MPV and the extent of CAD are controversial. 17,39 De Luca et al reported that MPV is not related to platelet aggregation, the extent of CAD, and carotid intima media thickness (IMT) in stable CAD. 39 Recently, Murat et al showed that high MPV level was independently associated with multivessel CAD and age. 17 However, in that study, the authors did not include patients with stable CAD. The pathophysiology of ACS is different from stable CAD. In our study, MPV increased with extent and complexity of CAD, but this increase is not dependent on the severity of CAD.
Also, in the present study high MPV value was independently associated with the decrease in platelet count. This result is consistent with the previous studies. 23,40 Several studies have reported that increases in platelet volume are often associated with decreases in platelet count. 23,40 It was suggested that small platelets be consumed in order to maintain a constant platelet functional mass. 41
Study Limitation
The MPV does not report a routine part of the complete blood count, because of the anticoagulant-induced changes over time. With the use of EDTA in complete blood count, MPV increases from 7.9% within 30 minutes to 13.4% over 24 hours if measured by the impedance method. 42 Moreover, some investigators have reported inconsistent increases in MPV with EDTA storage up to 50%. 43,44
Conclusion
High MPV value in patients with stable CAD is independently related to AD as well as diabetes, hsCRP, and platelet count. However, high MPV value was not independently associated with the extent and complexity of CAD assessed with SS. High MPV values may be responsible for the atherothrombotic complications related to impaired AD in stable CAD. Also, a high MPV value may be a part of low-grade chronic inflammation in stable CAD.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, and/or publication of this article.
