Abstract
Left ventricular assist devices (LVADs) are mechanical pumps that enhance cardiac function in patients with heart failure. In all, 7 patients with an LVADs (1.8 international normalized ratio warfarin, 81 mg aspirin) were evaluated monthly for 3 months for platelet and coagulation activation (controls: 5 healthy adults and 5 patients having warfarin). Platelet works revealed greater inhibition of collagen (31.8% vs 7.9%; P = .004), arachidonate- (30.9% vs 8.2%; P = .001), and adenosine diphosphate- (10.9% vs 6.1%; P = .004)-induced platelet aggregation for LVADs. Thrombelastography (recalcified whole blood) showed inhibition of clot initiation time (R; 8.81 vs 6.02 min; P = .001) and stronger clot formation (maximum amplitude; 69.1 vs 64.9 mm; P = .016). Platelet function determined by plateletMapping and flow cytometry was within the normal range. The LVADs had increased ratio of von Willebrand Factor (vWF) antigen and vWF propeptide, indicating increased degradation of vWF (2.04 vs 1.44; P = .144). Coagulation and platelet activation caused by LVAD is suppressed by pharmacotherapy, yielding a profile similar to that of patients on warfarin alone.
Introduction
Implantation of ventricular assist devices (VADs) continues to increase in popularity as a treatment for patients with heart failure. With the risk of heart failure approaching 1 in 100 people at the age of 65 in the United States, 1 there will always be a need for a method to treat end-stage heart failure beyond that of heart transplant. Although the demand for heart transplantation has remained relatively stable in the past 10 years, 2 there remain over 3000 patients on the waiting list. This does not include patients who require surgical intervention yet remain unlisted due to conditions such as advanced age or extensive comorbidities. Nor does it quantify the number of patients who died due to acute onset or exacerbation of heart failure.
The VADs have arisen as the next best alternative to heart transplant. They are used as either a bridge to transplant or a destination therapy depending on the patient. It has been shown that the morbidity and mortality associated with these devices is comparable to that of heart transplant in the short term,3,4 making these devices a viable option for treating select groups of patients.
Despite the use of vitamin K antagonists and platelet antagonists, coagulopathies, particularly uncontrolled bleeding, remain one of the primary complications of VAD use. 5 These events are not associated with an irregular prothrombin time (PT) test. One study found 2 thrombotic and 9 hemorrhagic events in 45 patients using the device. 6 The thrombotic events occurred in patients with an international normalized ratio (INR) of 1.3 and 2.2, while the hemorrhagic events occurred in patients with an INR of less than 2.
Evidence suggests that inhibition due to induced von Willebrand factor (vWF) deficiency is partly responsible for the increased risk of gastric bleeding, hemothorax, and hemopericardium. 7 Alterations in the platelet surface receptors such as glycoprotein Ib (GPIb) and adenosine diphosphate (ADP) have shown inhibition independent of vWF-associated ristocetin cofactor as well confounding the cause of hemorrhage seen in patients with a VAD.8
Since the use of VADs is increasing, there is a need to determine the cause of the coagulopathies associated with their use and also to determine appropriate therapy to avoid such events. Our study is the the first step taken to investigate these. We report here our findings from a homogeneous group of patients with VAD, establishing the typical normal hemostatic response in this population.
Materials and Methods
A single-center, prospective study analyzing hemostatic parameters over time in patients with implanted left VADs (LVADs) was designed to identify laboratory assays useful in detecting abnormal platelet or coagulation function. This study was approved by the Loyola University Health System Institutional Review Board. All patients and control participants gave written informed consent.
Study Groups
Patients
Seven patients (6 male, 1 female; mean age 52, age range 30-68) implanted with HeartMate II LVADs (Thoratec; Pleasanton, California) within the previous 1 to 1.5 years were included in this study. Patients had blood drawn once a month for 3 months during their regular clinic visits. These patients were anticoagulated with 81 mg aspirin daily and warfarin managed to target an INR of 1.8 with a tolerance of 1.5 to 2.0. One patient became ineligible during the study upon the receipt of a heart transplant.
Control groups
Two control groups were included in this study. The normal control group consisted of 5 healthy adults (2 males, 3 females; mean age 42, age range 26-63) with no history of any coagulation disorder and not taking platelet inhibitor drugs or anticoagulants. The warfarin control group consisted of 5 patients (3 males, 2 females, mean age 71, age range 55-85) from the Anticoagulation Clinic with established atrial fibrillation treated with warfarin targeting an INR of 2 to 3. Exclusion criteria for the warfarin controls included known coagulopathy aside from atrial fibrillation, the use of anticoagulant or antiplatelet drugs other than warfarin, cardiac ejection fraction less than 50% by most recent echocardiogram, age greater than 80 years, and pregnancy. All controls had their blood collected once.
Specimen collection
Whole blood was drawn by double-syringe venipuncture during the patients’ regular clinic visits. Whole blood was transferred into 3 Vacutainer tubes (Becton Dickenson, Franklin Lakes, New Jersey) containing sodium citrate, sodium heparin, and EDTA, and reaction tubes for the Plateletworks assay. Certain tests were performed immediately on whole blood after acquiring the samples (thrombelastography, platelet mapping, Plateletworks, and flow cytometry). Citrated whole blood was centrifuged at 3000 rpm for 15 minutes at 22°C and the plasma was stored frozen at −70°C and analyzed at a later time.
Laboratory Analyses
Thrombelastography
Within 30 minutes of venipuncture, the citrated whole blood was analyzed on the thrombelastograph (TEG; Haemoscope; Niles, Illinois). Coagulation was initiated only with calcium chloride (CaCl2). This assay provides for an innate physiologic coagulation response of the blood without influence from the external activators.
The TEG is a device able to continuously record the dynamic process of blood coagulation. The sample is placed in a reaction cup and a pin connected to a torsion wire is lowered into the sample. The cup gently oscillates. Torsion between the cup and pin, produced as the blood clots, is transmitted to a recorder. As the clot becomes firmer the rotational amplitude is reduced. A time versus torsion tracing is generated from which the following 4 parameters are calculated: R (time to initiate clot formation; related to enzymatic activity of coagulation factors), K (time for the forming clot to reach a standard strength; related to fibrinogen content and kinetics of clot formation), angle (indicative of the kinetics of clot formation; a steep angle equates to a rapidly forming clot), and MA (the maximum strength of the clot; related to platelet function and clot stability).
PlateletMapping assay
Within 30 minutes of venipuncture, the whole blood samples were analyzed on the TEG by the PlateletMapping assay (Haemoscope). This assay utilizes 4 simultaneous TEG runs to generate a complete picture of whole-blood clotting. The test samples and the net result of the 4 TEG tracings were as follows:
Overall coagulation (fibrin, thrombin, and platelets) determined on citrated whole blood and CaCl2. Fibrin only coagulation determined on heparinized whole blood and reptilase activator. Fibrin/activated platelet coagulation determined on heparinized whole blood and reptilase and ADP. Fibrin/activated platelet coagulation determined on heparinized whole blood and reptilase and arachidonic acid (AA).
The R, K, angle, and MA parameters were determined on sample #1. On samples #2, #3, and #4, the ratios were calculated using the MA parameter to determine the percentage inhibition of the platelet ADP receptors (P2Y12) and the platelet AA receptors (TPα, TPβ).
Plateletworks assay
Immediately after drawing into the syringe, whole blood was placed into the vials of the Plateletworks assay (Helena, Beaumont, Texas): EDTA, collagen, ADP, and AA. The EDTA tube was inverted 5 times for mixing; the AA, ADP, and collagen tubes were shaken vigorously 20 times. The AA, ADP, and collagen tubes were gently mixed by inversion every 8 to 10 seconds for 2 minutes after shaking. Then, the Plateletworks testing was performed. The EDTA sample was run first on the ICHOR automated cell counter (Helena) to obtain platelet counts. An additional 5 minutes was allowed to pass before running (in order) the AA, collagen, and ADP samples on the ICHOR. The percentage platelet inhibition was calculated as the difference in platelet count between the AA-, ADP-, or collagen-treated samples and the EDTA sample.
Flow cytometry
Aliquots of citrated whole blood were activated for 3 minutes by 5 μmol/L ADP, 6.25 μmol/L thrombin receptor–activating peptide (TRAP), 5.0 μg/mL collagen, or 0.5 mg/mL AA. Samples were then labeled with antibodies to CD61-PerCP (platelets), CD62P-PE (activated platelets), and PAC-1-FITC (activated fibrinogen receptor) and fixed in cold 1% paraformaldehyde diluted with calcium-free Tyrode buffer. The samples were analyzed on the Epics-XL flow cytometer (Beckman-Coulter; Miami, Florida) for percentage platelet aggregation and median expression levels of CD62 and PAC-1 on the platelets. Sample results were gated based on the size and analysis was done using Windows Multiple Document Interface for Flow Cytometry (WinMDI) version 2.8 software. 9
Plasma coagulation factors
The stored citrated plasma samples were analyzed in batch by the enzyme-linked immunosorbent assay (ELISA) technique for vWF antigen and vWF propeptide (GTI Diagnostics, Waukesha, Wisconsin), ADAMTS-13 (Imubind; American Diagnostica, Stamford, Connecticut), cellular microparticles (Zymutest; Aniara, Mason, Ohio), C-reactive protein (CRP; Zymutest), and soluble vascular cell adhesion molecule 1 (sVCAM-1; Quantikine; R&D Systems; Minneapolis, Minnesota). Manufacturers’ instructions were followed for all the assays.
Statistics
Statistical analysis was performed using SPSS Statistics (IBM, Armonk, New York). Comparisons between the means were performed using Student t test or Welch t test based upon the variance assumption indicated by the Levene test with a 95% confidence interval (CI). An unpaired, repeated-measures t test was used in order to account for the different sampling frequencies between the study groups and the controls as well as the withdrawal of one of the patients due to transplant.
Analysis of variance, performed using the Bonferroni method on all data collected for the group of patients with an LVAD, showed that there was no statistically significant trend over time for any test. Therefore, the repeat-measures t test used to compare study groups is not affected by month-to-month variation in the samples of patients with an LVAD. Thus, the 3-month data of patient with an LVAD were combined for comparison to the control groups.
Results
The TEG and PlateletMapping
The TEG and PlateletMapping results are provided in Table 1. Patients with an LVAD exhibited a significantly prolonged mean R time that was 46% longer than that of normal adults but not significantly different from that of the warfarin control group. Patients with LVAD showed a significantly higher mean MA than that of the normal individuals or patients on warfarin. Patients with an LVAD showed inhibition trends of ADP- and AA- receptor-mediated- coagulation when compared to normal individuals and patients on warfarin that were not statistically significant. The results of the TEG indicate that the patients with an LVAD in this study, in general, have a mild inhibition of the coagulation system and platelet function that is not different from normal. There was no change over the 3-month study period in any parameter.
Coagulation and Platelet Function in Patients With a Left Ventricular Assist Device and Controls as Determined by Thrombelastography.a
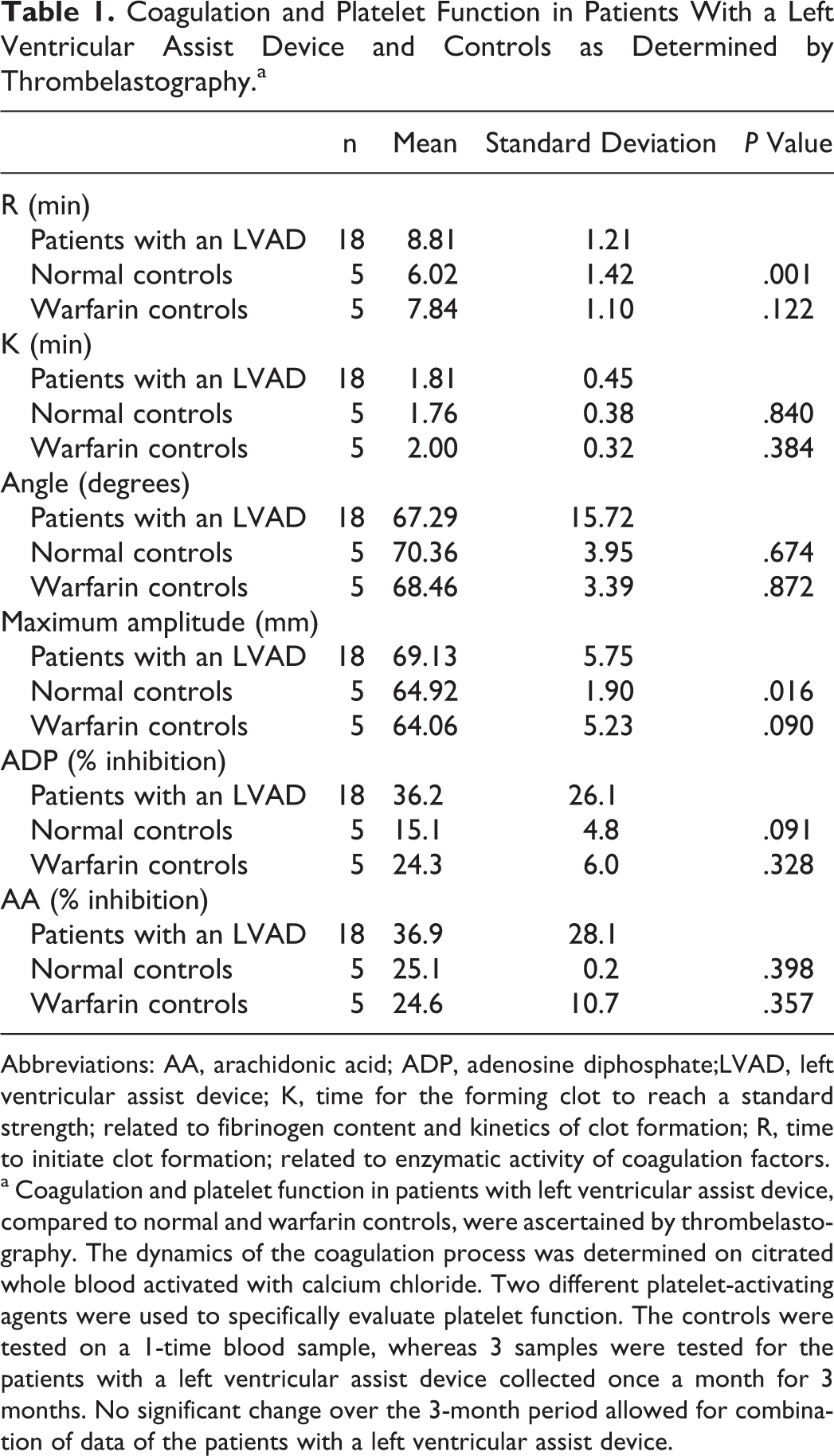
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate;LVAD, left ventricular assist device; K, time for the forming clot to reach a standard strength; related to fibrinogen content and kinetics of clot formation; R, time to initiate clot formation; related to enzymatic activity of coagulation factors.
a Coagulation and platelet function in patients with left ventricular assist device, compared to normal and warfarin controls, were ascertained by thrombelastography. The dynamics of the coagulation process was determined on citrated whole blood activated with calcium chloride. Two different platelet-activating agents were used to specifically evaluate platelet function. The controls were tested on a 1-time blood sample, whereas 3 samples were tested for the patients with a left ventricular assist device collected once a month for 3 months. No significant change over the 3-month period allowed for combination of data of the patients with a left ventricular assist device.
Plateletworks
Plateletworks results are provided in Table 2. Patients with an LVAD showed significant inhibition of collagen-, ADP-, and AA-mediated platelet aggregation relative to the normal adults. Collagen- and AA-induced platelet aggregation was nearly 400% less responsive in patients with an LVAD compared to normal adults. Trends were similar when comparing the LVAD and warfarin groups; however, no comparison was statistically significant. The results of the Plateletworks indicate that the patients with an LVAD in this study, in general, have a mild inhibition of platelet function. There was no change over the 3-month study period in any parameter.
Platelet Function in Patients With a Left Ventricular Assist Device and Controls as Determined by PlateletWorks.a
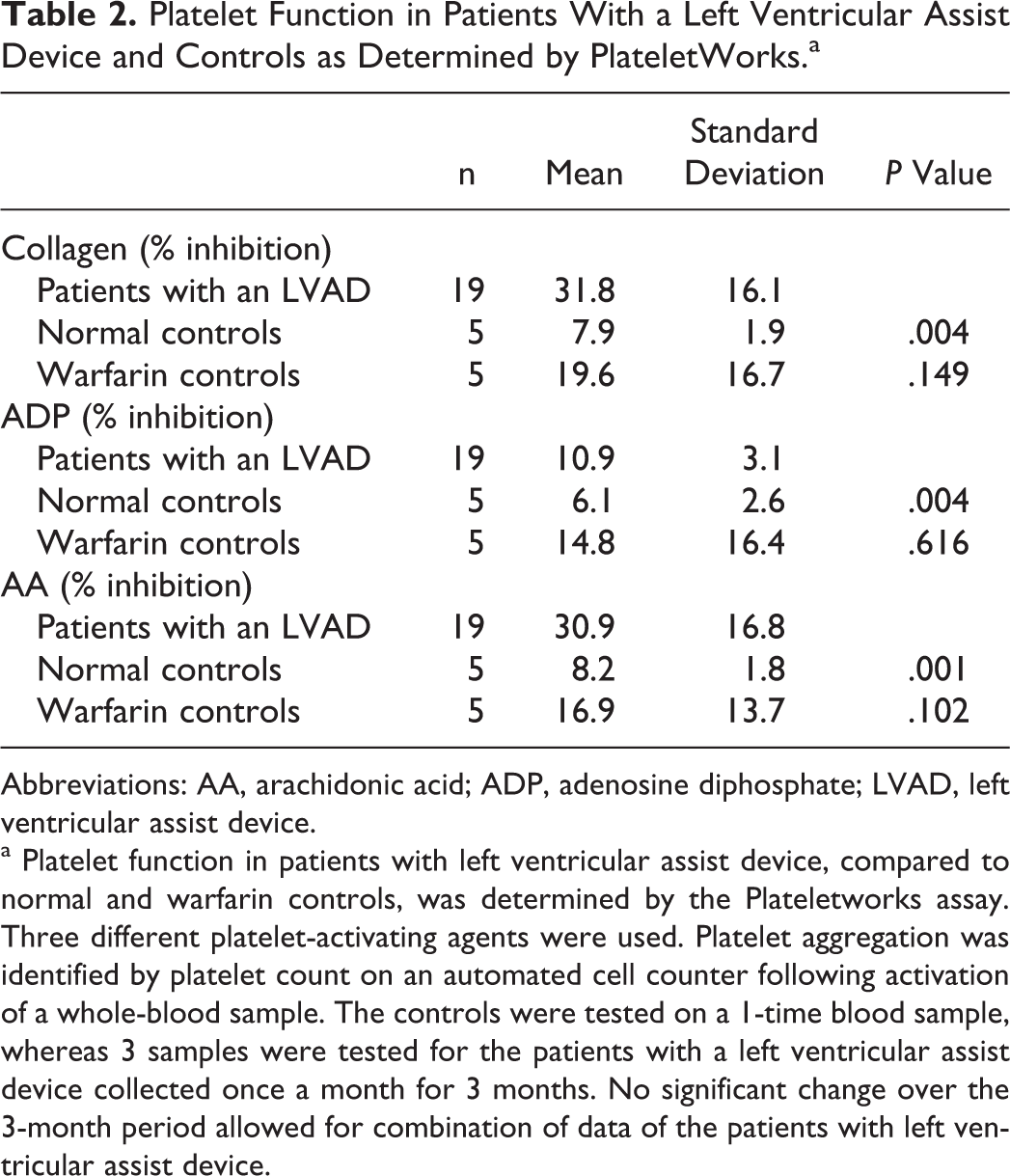
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate; LVAD, left ventricular assist device.
a Platelet function in patients with left ventricular assist device, compared to normal and warfarin controls, was determined by the Plateletworks assay. Three different platelet-activating agents were used. Platelet aggregation was identified by platelet count on an automated cell counter following activation of a whole-blood sample. The controls were tested on a 1-time blood sample, whereas 3 samples were tested for the patients with a left ventricular assist device collected once a month for 3 months. No significant change over the 3-month period allowed for combination of data of the patients with left ventricular assist device.
Flow Cytometry
Flow cytometry results are provided in Table 3. There were no statistically significant findings comparing markers for platelet activation (P-selectin upregulation) and activation of the GPIIb/IIIa receptor (fibrinogen binding) between patients with an LVAD and either of the two control groups. This held true when platelets were in a resting state (saline; no agonist) and when they were stimulated with ADP, TRAP, or collagen. When platelets were stimulated with AA, there was a mildly decreased receptor expression in patients with an LVAD compared to normal adult and warfarin controls. This is consistent with the platelet inhibition induced by aspirin treatment in the patients with an LVAD.
Platelet Function in Patients with a Left Ventricular Assist Device and Controls as Determined by Flow Cytometry.a
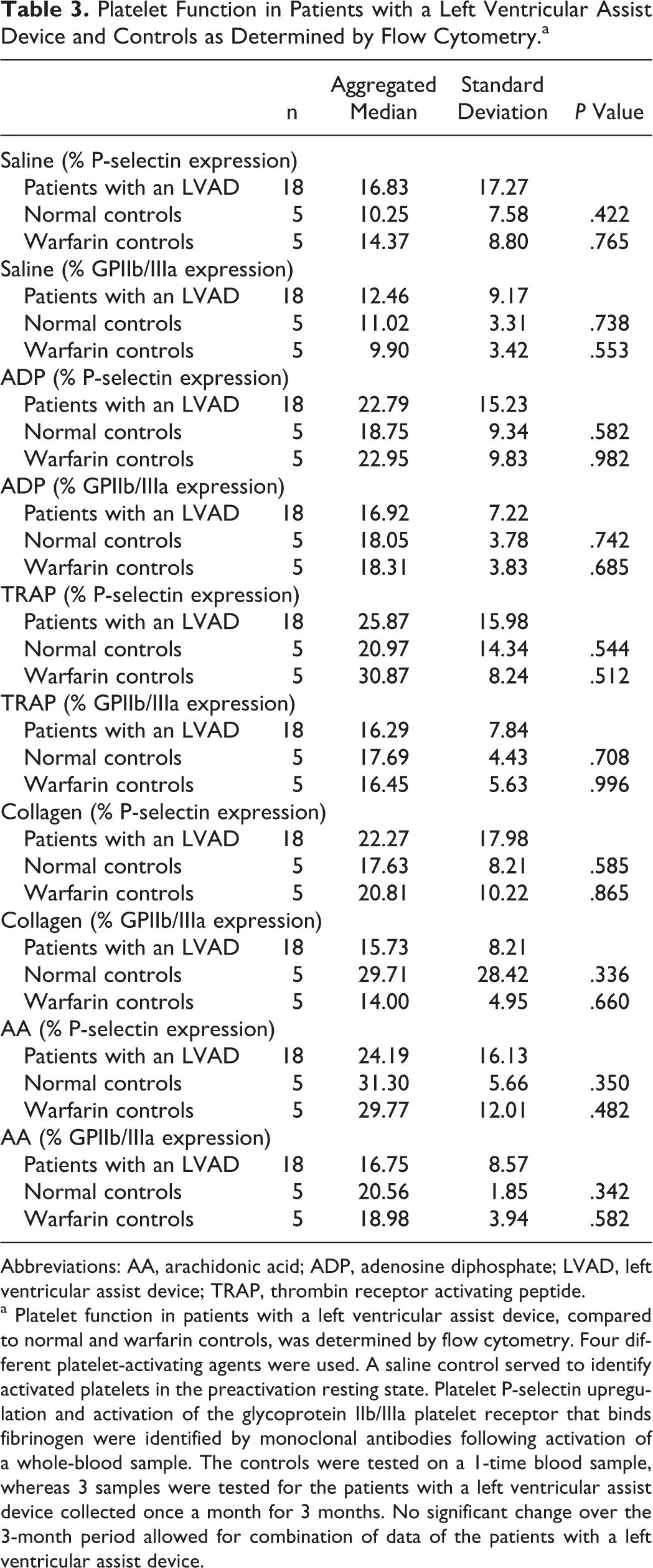
Abbreviations: AA, arachidonic acid; ADP, adenosine diphosphate; LVAD, left ventricular assist device; TRAP, thrombin receptor activating peptide.
a Platelet function in patients with a left ventricular assist device, compared to normal and warfarin controls, was determined by flow cytometry. Four different platelet-activating agents were used. A saline control served to identify activated platelets in the preactivation resting state. Platelet P-selectin upregulation and activation of the glycoprotein IIb/IIIa platelet receptor that binds fibrinogen were identified by monoclonal antibodies following activation of a whole-blood sample. The controls were tested on a 1-time blood sample, whereas 3 samples were tested for the patients with a left ventricular assist device collected once a month for 3 months. No significant change over the 3-month period allowed for combination of data of the patients with a left ventricular assist device.
Differences between patients with an LVAD and normal controls over time for the expression of P-selectin and the GPIIb/IIIa receptor when platelets are stimulated are shown in Figure 1. Throughout the 3-month period, the platelet expression levels remained consistent with no significant variation.
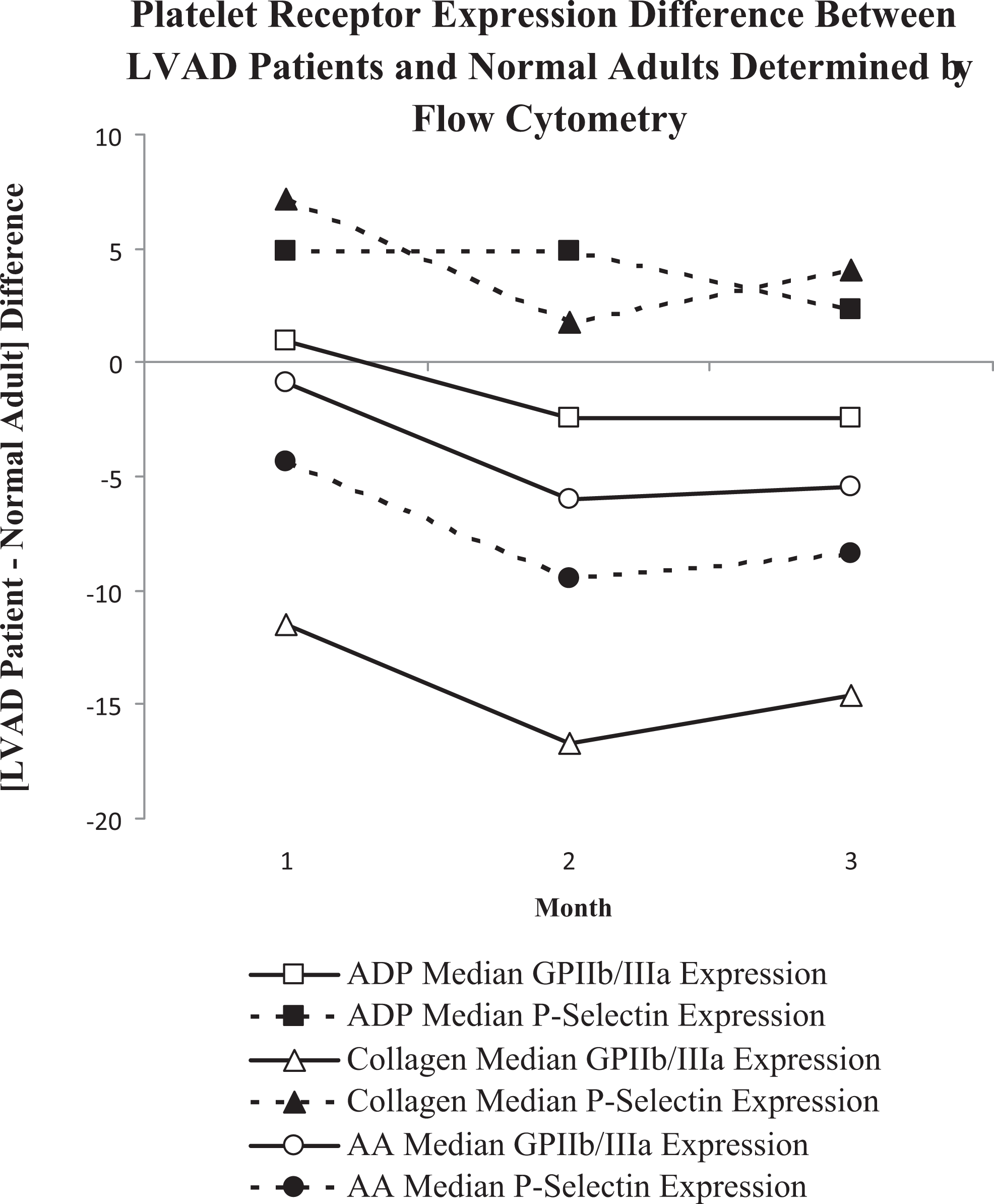
Platelet function in patients with a left ventricular assist device (LVAD) directly compared to normal controls was evaluated over a 3-month period. Using 3 different platelet agonists, platelet activation identified by P-selectin upregulation and activation of the glycoprotein IIb/IIIa receptor were determined by flow cytometry. The obtained results were used to calculate the difference between the groups (LVAD-normal = difference).
Plasma Coagulation Factors
The results of the analyses of coagulation factors in plasma are provided in Table 4. Patients with an LVAD had slightly higher levels of plasma vWF when compared to normal adults. The level of vWF propeptide was higher in patients with an LVAD but not significantly. The ratio of vWF to vWF propeptide was slightly higher in the LVAD group, suggesting increased degradation of vWF. These higher levels could not be accounted for by an alteration in the vWF cleaving protein ADAMTS-13 that was similar in all the groups. Throughout the 3-month period, there was no significant variation in any of these parameters.
Status of Selected Coagulation and Inflammatory Biomarkers in Patients With a Left Ventricular Assist Device and Controls.a
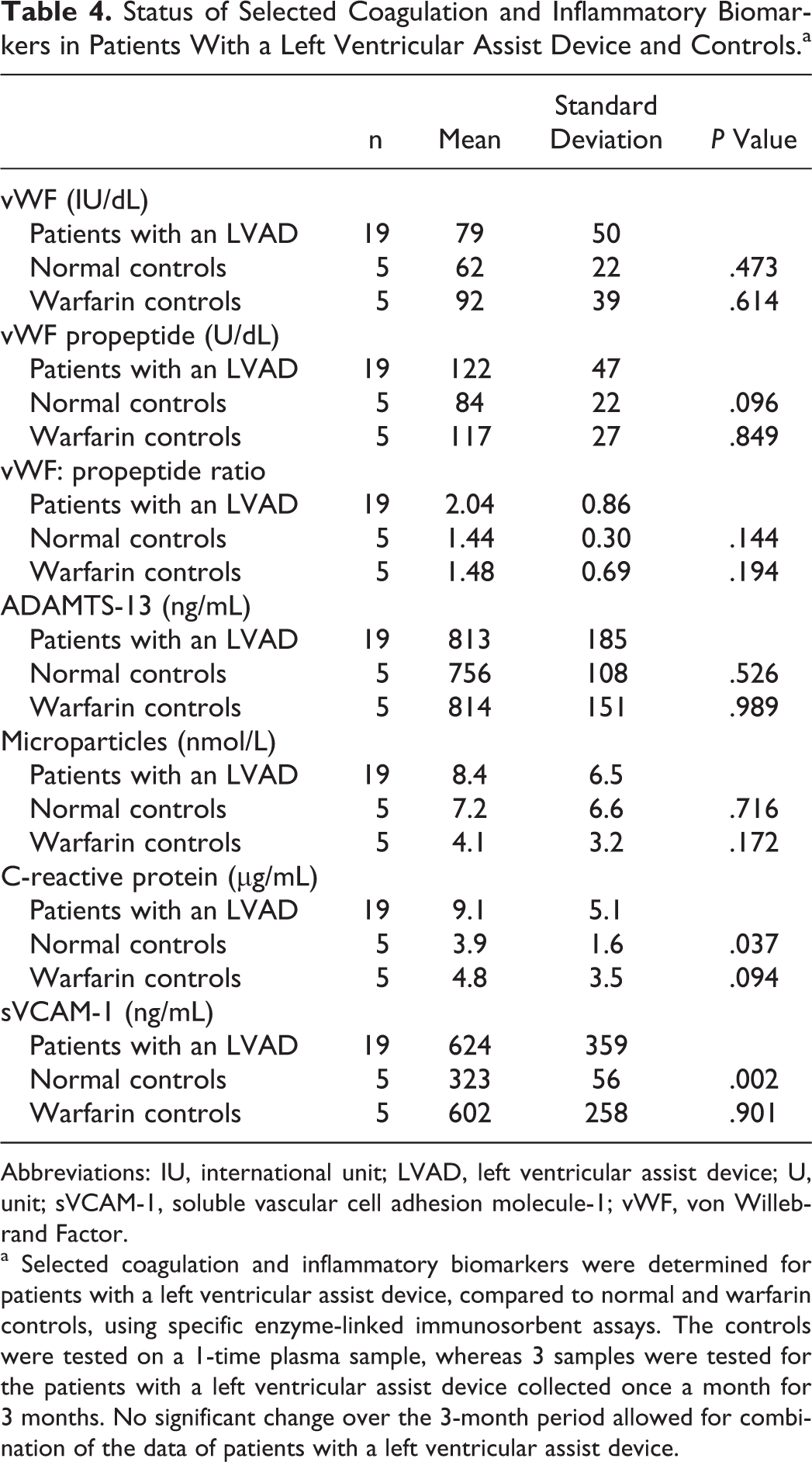
Abbreviations: IU, international unit; LVAD, left ventricular assist device; U, unit; sVCAM-1, soluble vascular cell adhesion molecule-1; vWF, von Willebrand Factor.
a Selected coagulation and inflammatory biomarkers were determined for patients with a left ventricular assist device, compared to normal and warfarin controls, using specific enzyme-linked immunosorbent assays. The controls were tested on a 1-time plasma sample, whereas 3 samples were tested for the patients with a left ventricular assist device collected once a month for 3 months. No significant change over the 3-month period allowed for combination of the data of patients with a left ventricular assist device.
Patients with an LVAD had levels of CRP and sVCAM-1 more than twice that of normal adults, although cellular microparticles were not different between the groups. Together these findings indicate a mild upregulation of an inflammatory response in the patients with an LVAD. Throughout the 3-month period, there was no significant variation in these parameters.
Discussion
The focus of this study was to evaluate coagulation and platelet function in patients with implanted LVADs. A secondary aim of our study was to identify a practical way for the clinical laboratory to assess these responses in patients with an LVAD over the time period of their implant. Due to the complexities of hemostasis and varying sensitivities and specificities of the assays, several different assay systems were used. Multiple platelet aggregating agonists were chosen to gain a full assessment of platelet function including the effect of aspirin therapy. Coagulation was assessed by a global whole-blood system (TEG) that is available in our hospital, as well as by certain factors selected based on prior published information.8,10
Our study showed that patients with an LVAD have a mild inhibition of coagulation and a mild inhibition of platelet function consistent with their treatment of warfarin (INR 1.8) and low-dose aspirin (81 mg daily). By comparing the elevated levels of activation of all hemostatic parameters observed in all artificial heart implant patients, 10 the “baseline” hemostatic system of a patient with an LVAD does not appear to be very different from the normal population. There was also no change in any parameter studied over a 3-month period of LVAD use.
It appeared that the Plateletworks was the most sensitive of the assays used for platelet function in this study. Quantifying the kinetics and strength of clot formation by the TEG provided much useful information of global whole-blood coagulation from a single sample.
The general outcome of this study was consistent with the previous literature for LVADs7,8 and for warfarin therapy. 11 Of interest, however, were the following several observations. Despite being on an anticoagulation regimen targeting a lower INR than the warfarin group, there was no statistical difference between the 2 groups in the TEG analysis. This may be due to the variability in the management of warfarin that is well documented. 12
The patients with an LVAD had increased overall clot strength (increased MA parameter of the TEG) compared to the normal. This observation is unexpected in that warfarin and aspirin therapy would lower the MA as this parameter is largely dependent on the platelet function and fibrinogen level. The increased MA is not necessarily indicative of the ability of a clot to adhere to an endothelial surface, however, as this is accomplished through the interaction of vWF and exposed collagen. 13
The patients with an LVAD exhibited inhibition of platelet activation inducible by ADP, unanticipated as these patients were not on clopidogrel or prasugrel antiplatelet medications. It is unclear why this similar finding was also observed in the warfarin control group. It may prove to be of interest to study whether there is an association of platelet aggregation mediated by vWF, which requires ADP but which is resistant to aspirin, 14 with LVAD use.
The vWF is a multimeric plasma GP that acts as an adhesive intermediate between platelets and exposed collagen on the surface of a laceration or other endothelial disturbance. The vWF-dependent platelet adhesion is most pronounced at high shear rates. The vWF is, therefore, a critical component in producing a stable platelet plug and hemostasis. With regard to the vWF directly, several findings come from this study.
The vWF propeptide is secreted in equal amounts with vWF but is not degraded by the same pathway. The ratio of vWF-to-vWF propeptide, used as an indicator of in vivo vWF degradation, 15 revealed a higher ratio in the patients with an LVAD, indicating increased degradation of vWF. This is likely due to proteolysis of the long-chain vWF multimers by the ADAMTS-13 metalloproteinase, an effect that is more efficient at high shear stresses 16 (such as when blood passes through a VAD). It has been documented that patients with an LVAD have increased short-chain vWF and decreased long-chain vWF multimers.7,8 Smaller vWF multimers are less able to cause vWF-mediated platelet-collagen adhesion.17,18 The potential effect of increased degradation of vWF would be the increased risk of bleeding.
Consistent with this is that the patients with an LVAD in our study had decreased collagen-induced platelet aggregation. Collagen-induced platelet activation requires vWF to cause platelet aggregation via the GP Ib receptor complex. Furthermore, the lack of inhibition of P-selectin expression and activated GPIIb/IIIa on the platelet surface in response to various agonists quantified by flow cytometry suggests that the documented platelet aggregation inhibition in patients with an LVAD8,19 is likely the result of extrinsic factors such as vWF rather than simply decreased agonist-receptor platelet response.
The patients with an LVAD were in an inflammatory state, not observed in the two control groups, indicated by the well-known biomarkers of CRP and sVCAM-1. Cellular microparticles, released by cells and platelets in response to stress, serve as a further inflammatory signal. 20 The interaction between inflammation and coagulation is complex and not well understood; however, in general inflammation can be seen as lending itself to hypercoagulability. This coupled with the activation of hemostatic pathways unaffected by warfarin and aspirin, such as those that involve vWF, opens more possibilities to understand clinical events that occur in patients with LVADs.
The primary limitation of this study was the small number of patients and controls unmatched for age and gender. We were also limited in our ability to evaluate the effect of 81 mg aspirin alone, and we did not include this control group in our study. As none of the patients had a diagnosed bleeding or thrombotic event during the study period, we were unable to determine the full clinical utility of the chosen laboratory assays. Given the small number of patients with an LVAD an individual institution sees in 1 year, a multicenter study is required for increased power of such studies.
In conclusion, the patients with an LVAD in our study were in a complicated hemostatic state characterized by increased proteolysis and generation of vWF, decreased platelet response to collagen, and in general a proinflammatory state. This was shown by ELISA and flow cytometry and is consistent with the studies published previously. Despite these factors, the patients with an LVAD seem to have a less robust response to warfarin than the warfarin-only control group as shown by the TEG R and K times. Although the power of this study was too low to make absolute conclusions, it does provide justification to continue this work to be able to identify sensitive assays and pharmacotherapy that can reliably predict and regulate hemostasis in patients with VADs.
Footnotes
Acknowledgment
The authors are indebted to Linda K. Dusek, RN, BSN, CCTC for her kind and generous assistance with the patients.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Partial funding for this study was provided by Loyola University Chicago. Plateletworks laboratory assay kits were donated by Helena Laboratories.
