Abstract
Available options for the treatment of advanced heart failure have expanded to include the use of mechanical circulatory assist devices to improve quality of life in those both eligible and ineligible for heart transplant. Although there have been significant advancements in device technologies, anticoagulation protocols, and multidisciplinary team management, bleeding and thrombosis are the most common adverse effects. Management strategies for pump thrombosis and their outcomes vary considerably among mechanical circulatory support centers and include intensification of antithrombotic therapy (medical) and device exchange (surgical). We describe a successful case of medical therapy for pump thrombosis with bivalirudin monotherapy.
Introduction
Available options for the treatment of advanced heart failure (HF) have expanded to include the use of mechanical circulatory assist devices to improve quality of life in those both eligible and ineligible for heart transplant. Despite significant advancements in device technologies, anticoagulation protocols, and multidisciplinary team management, bleeding and thrombosis are the most common adverse effects. Confirmed versus suspected left ventricular assist device (LVAD) thrombosis is most often defined as (1) direct observation of obstructive thrombus in the pump or conduit or (2) severe hemolysis, as defined by either a lactate dehydrogenase (LDH) level >4 times the upper limit of normal or a plasma-free hemoglobin >40 mg/dL, and symptoms of decompensated HF. 1 Pump thrombosis can occur due to a variety of factors, including those effecting the patient, device, and/or management strategy. Patient-related factors include history of hypercoagulable state, infection, preexisting left ventricle (LV) thrombus, and malignancy. Device-related factors include axial versus centrifugal flow design, magnetically levitated versus regular bearings, and different heat generation as well as pump material and variable hemocompatibility. Management-related factors include varying levels of anticoagulation and antiplatelet therapy, lower pump speeds, and mechanical issues from a stenosed or kinked cannula(e). 2 The LVAD thrombosis is associated with high morbidity and mortality, and uncertainty remains surrounding safe and effective management. Even with chronic administration of antiplatelet and anticoagulant therapies, suspected/confirmed pump thrombosis with the HeartMate II LVAD and HeartWare devices have been reported. Specifically, the HeartMate II LVAD pump thrombosis rates increased from 2.2% (95% CI = 1.5-3.4) in 2011 to 8.4% (95% CI = 5.0-13.9) in 2013. 3 Similarly, findings were also observed by HeartWare investigators after concern for the high-incidence (0.07 events per patient year) pump thrombosis, which prompted a more detailed analysis. 4,5 Even with recognition of increase in percentage of pump thrombosis and changes in anticoagulation strategies, rates have not decreased.
Management strategies for pump thrombosis vary considerably and include initiation of antiplatelet agents; increased intensity of antiplatelet and anticoagulants; or initiating intravenous antithrombotics, thrombolytic agents, and/or ultimately device exchange. At our institution, if the LDH ranges between 250 and 500 U/L, we perform hemolysis surveillance labs weekly, check for hemoglobinuria defined by urine analysis with large quantitative blood and few to no urine red blood cells. If the LDH is >500 U/L, the hemolysis labs and urine analysis are checked twice weekly. If pump thrombosis is suspected, the patient is admitted, warfarin therapy is held, and bivalirudin and potentially intensifying antiplatelet therapies are started. 6 We target a highly activated partial thromboplastin time (aPTT) of 70 to 90 seconds in the initial admission for suspected or confirmed pump thrombosis, and for subsequent admissions we increase it to 80 to 100 seconds, all dependent upon bleeding history and clinical examination. We describe a successful case of pump thrombosis abortion with bivalirudin monotherapy.
Patient Case
A 56-year-old male with a history of atrial fibrillation, dyslipidemia, diabetes, end-stage familial dilated cardiomyopathy status post (s/p) HeartWare LVAD in October 2014, complicated by pump thrombosis in June 2015, requiring pump exchange (status 1B), was admitted in November 2015, with increase in ventricular assist device (VAD) flows (6-7 L/min, baseline 4.5-5 L/min), LDH 621 U/L (upper limit of normal 135-225 U/L), plasma hemoglobin 11.4 mg/dL (upper limit of normal 0.0-9.7 mg/dL), and darkening of his urine, with a therapeutic international normalized ratio (INR; goal 2.5-3.5 time in therapeutic range, 96% for the 90 days leading up to admission for suspected thrombosis). He was anticoagulated with warfarin dosed by a pharmacist-run anticoagulation clinic and aspirin 325 mg daily. Bivalirudin monotherapy targeting the aPTT range of 80 to 100 seconds (2.5-3.0× control) was started the day of admission, and dipyridamole 75 mg 3 times daily was added to his antiplatelet regimen. During his admission, his LDH continued to rise, peaking 3 days later at 987 U/L, with power spikes and changes in flow. Bivalirudin at this goal was continued, resulting in slow reductions in his LDH. In 16 days of bivalirudin monotherapy, his LDH decreased to baseline, and he was subsequently bridged to warfarin targeting the same INR goal of 2.5 to 3.5 (Figure 1).
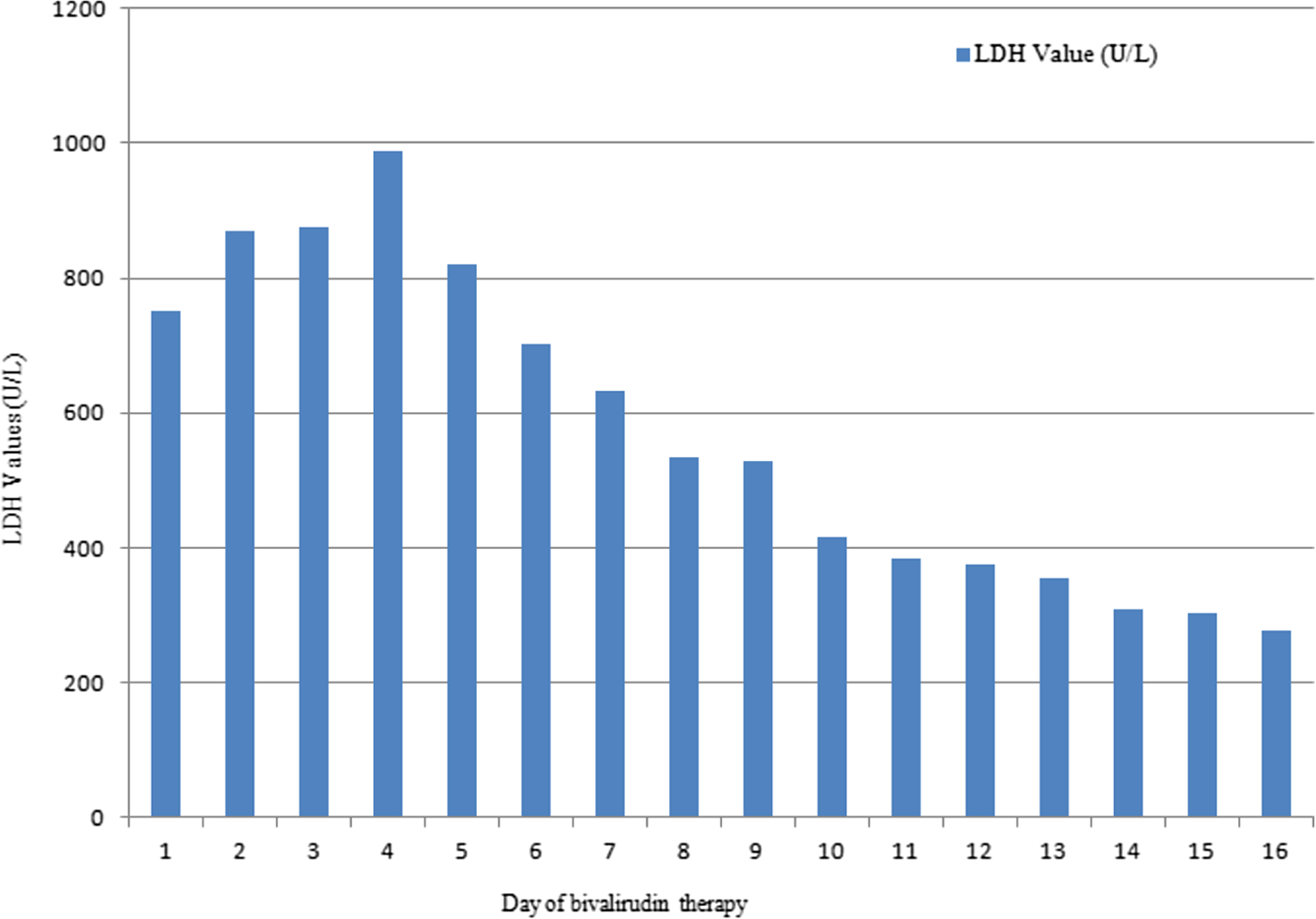
The LDH values while receiving bivalirudin therapy. LDH indicates lactate dehydrogenase.
Discussion
The LVAD thrombosis is associated with high morbidity and mortality due to device malfunction, thromboembolic events, and hemolysis. Patient, device, or anticoagulation management-related factors contribute to pump thrombosis. The LVAD thrombosis occurs in 2% to 13% of adult patients with a continuous-flow LVAD, 4% to 13% with axial-flow devices, and 2% with centrifugal-flow devices. 7 Pump thromboses have been associated with both fibrin- (red) and platelet-rich (white) thrombi that can form anywhere on the device, including the inflow/outflow cannula, outflow graft, LV, or the propeller. Red thrombi form as stagnant blood coagulates under low pressure, whereas white fibrin thrombi form in areas of turbulence. Successful management has included surgical and pharmacological therapies, though consensus guidelines are not available as data are controversial. 7 –10 Data on the management of device thrombosis are sparse, with no randomized clinical trials assessing optimal use of intravenous thrombotic agents such as fibrinolytics (eg, alteplase) and glycoprotein IIb/IIIa (GPIIb/IIIa) inhibitors (eg, eptifibatide). Intraventricular alteplase (tPA) at varying doses and duration of administration have shown inconsistent results, leading many clinicians to feel it is an unreliable option (Table 1). A large case series demonstrated success rates ranging from 23% to 50% at the expense of stroke rates of 10% to 15%, bleeding complications of 65%, and mortality rates of 17% to 52%, leaving the role of intraventricular tPA in therapy inconclusive. 3,11 –15 Similarly, inconsistent results are seen with eptifibatide, where in 1 case series 65% of patients experienced a bleeding event requiring dose reductions and/or subsequent discontinuation of the GPIIb/IIIa inhibitor. 16,17
Data Evaluating Medical Management With the Use of Thrombolytics and GPIIb/IIIa Inhibitors in Ventricular Assist Device Thrombosis.
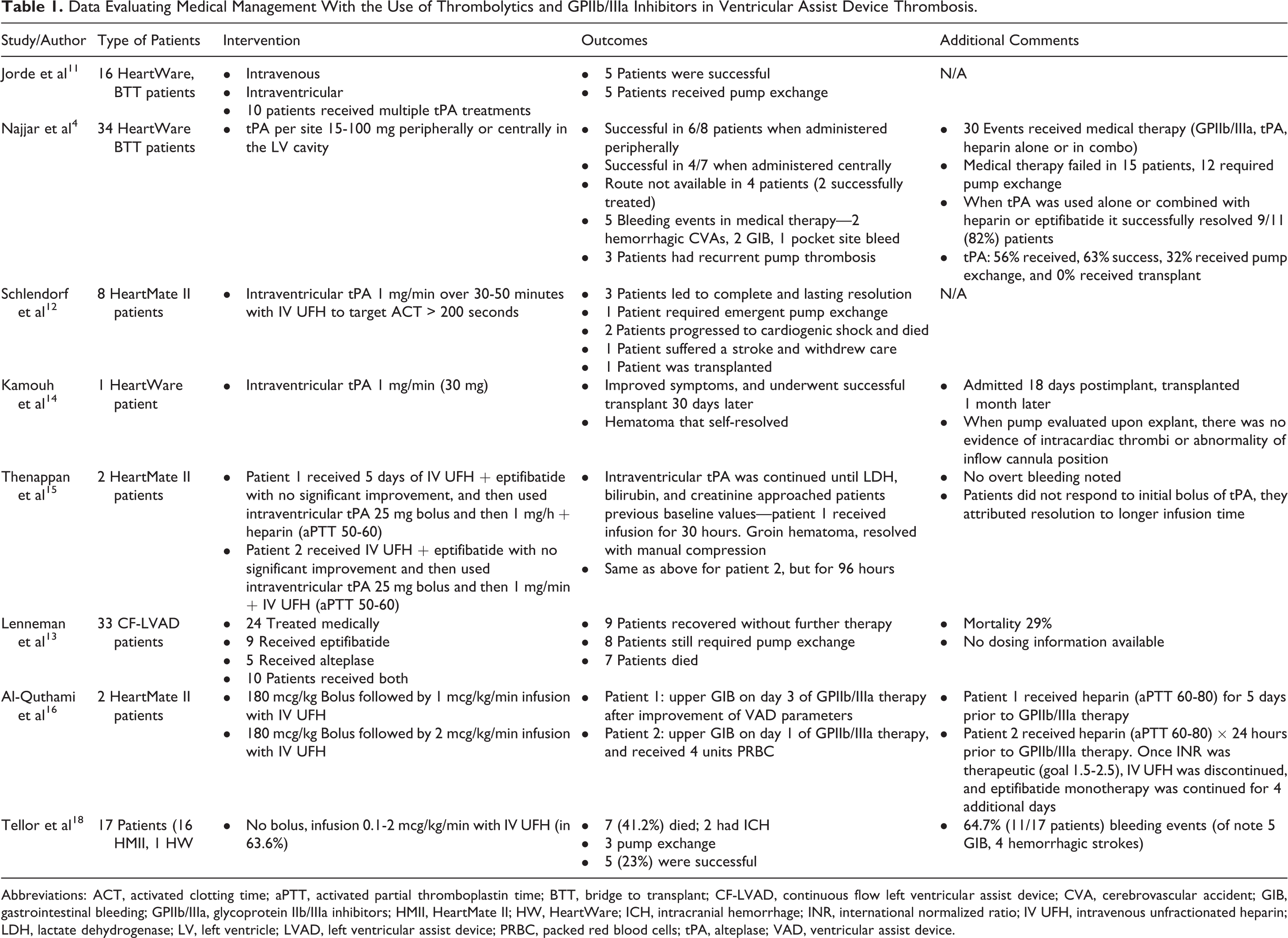
Abbreviations: ACT, activated clotting time; aPTT, activated partial thromboplastin time; BTT, bridge to transplant; CF-LVAD, continuous flow left ventricular assist device; CVA, cerebrovascular accident; GIB, gastrointestinal bleeding; GPIIb/IIIa, glycoprotein IIb/IIIa inhibitors; HMII, HeartMate II; HW, HeartWare; ICH, intracranial hemorrhage; INR, international normalized ratio; IV UFH, intravenous unfractionated heparin; LDH, lactate dehydrogenase; LV, left ventricle; LVAD, left ventricular assist device; PRBC, packed red blood cells; tPA, alteplase; VAD, ventricular assist device.
In contrast, recent analyses have observed that medical therapy is associated with substantial treatment failure and may be associated with a high risk of complications due to intensification of antithrombotic therapies. 17 –20 In those patients who failed medical therapy and received subsequent surgical therapy, the treatment response to initial medical therapy was only 29%. The surgical intervention resulted in less mortality, stroke, and resolution of hemolysis than a medical strategy alone. 21
Our VAD thrombosis management includes typically increasing or intensifying antiplatelet therapies (increase Aspirin 325 mg if at 81 mg daily, add dipyridamole 75 mg 3 times daily, or clopidogrel 75 mg daily), increasing the INR goal when warfarin therapy is resumed, and/or admitting for bivalirudin monotherapy targeting a higher aPTT goal. Our dosing strategy of bivalirudin is considerably different than that observed for percutaneous coronary intervention (PCI) (0.75 mg/kg bolus then 1.75 mg/kg/h infusion) or heparin-induced thrombocytopenia (HIT), which targets an aPTT goal of 60 to 80. Bivalirudin is an intravenous direct thrombin inhibitor that inhibits both circulating and fibrin-bound thrombin, is less immunogenic than heparin, and inhibits platelet adhesion. 22 Other benefits include the decreased risk of HIT in VAD patients requiring cardiopulmonary bypass for device exchange or heart transplant, short half-life, and is primarily enzymatically cleared. Because of its action on both bound and unbound thrombin, it is thought to have a more extensive effect on thrombin compared to heparin. Hemolysis labs and pump flows are monitored daily to evaluate for pump thrombus resolution. Once labs are stabilized and LDH levels return to baseline, warfarin is reinitiated and bivalirudin is continued until a therapeutic INR is reached.
At our institution, bivalirudin is the intravenous anticoagulant of choice versus heparin for VAD patients that are bridge to transplant or presenting with suspected or confirmed pump thrombosis due to the incidence of HIT. The rates of antibody development are significantly higher than the clinical diagnosis of HIT following heparin exposure. The adult medical population rates are estimated to be 10% to 20% and 0.8% to 3%, respectively, compared with 20% to 50% and 1% to 2% in adult patients undergoing cardiac surgery. 23 Following VAD implantation, the incidence of PF4 antibody positivity has been reported as high as 65% with polyspecific enzyme-linked immunosorbent assay testing and as many as 10% of patients develop clinical HIT based on functional platelet testing. 24,25 Recognition, treatment, and even prevention of HIT in the end-stage HF population can limit the risk of potentially catastrophic thrombotic events and prevent significant delays in VAD implantation or heart transplant.
Although resolved VAD thrombosis cannot be confirmed, we consider halted thrombus when LDH, free hemoglobin, urine color, and VAD flows return to baseline. Notably, in this case, LDH returned to normal range after 16 days of bivalirudin therapy, thus making it hard to draw conclusions; therefore, further studies are warranted to assess the benefit to risk balances of the available therapeutic options. Duration of therapy can be extensive as demonstrated in this case, and this approach does not go without expending significant health-care costs, where the cost of surgical treatment of pump thrombosis is approximately 10 times as much as a week on bivalirudin therapy. As VADs represent a lifesaving opportunity for patients with end-stage HF, they do not come without major risks such as VAD thrombosis. Despite years of experience, the optimal treatment/prevention strategy for VAD thrombosis is still not known.
Footnotes
Authors’ Note
Dr Connors is a consultant for St Jude’s Thoratec.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
