Abstract
Thromboembolic events can be seen in patients with mitral valve prolapse (MVP). It is unclear whether platelet activation may contribute to these events in patients with MVP. Thus, we aimed to evaluate mean platelet volume (MPV) in patients with MVP and its association with the severity of MVP. This study included 312 patients with MVP and 240 control participants. Mean platelet volume was significantly higher in patients with MVP than in controls (8.9 ± 0.7 vs 7.9 ± 0.6 fL, P = .001). In linear regression analysis, MPV was independently associated with the degree of mitral regurgitation (β = .23, 95% confidence interval (CI): 0.14-0.32, P = .001), maximal leaflet displacement (β = .24, 95%CI: 0.17-0.31, P = .001), and mean thickness of the anterior (β = .47, 95%CI: 0.27-0.61, P = .001) and posterior leaflets (β = .22, 95%CI: 0.03-0.41, P = .02). Our findings show that MPV can be elevated in patients with MVP and may be independently associated with severity of mitral regurgitation, leaflet displacement, and thickness of the leaflets.
Introduction
Mitral valve prolapse (MVP) is a valvular disease characterized by the displacement of an abnormally thickened mitral valve leaflet into the left atrium during systole, with or without valvular insufficiency. 1 Primary MVP is characterized by idiopathic myxomatous change in the mitral valve leaflets or the chordal structures or both. 1,2 Its incidence varies from 2% to 6% and it is more prevalent in females. 3,4
Usually, MVP appears to be a benign disorder but can lead to several complications including mitral regurgitation, infectious endocarditis, atrial/ventricular arrhythmias, and thromboembolic events such as stroke, transient ischemic attack, and amaurosis fugax. 2–6
The thromboembolic events, such as cerebrovascular events, and platelet abnormalities have been reported in patients with MVP, particularly in young patients. 7,8 This association may be related to increased systemic platelet activity. 9 In these patients, platelet aggregation and platelet-fibrin complexes may initiated by the following mechanisms: shortened platelet survival, loss of endothelial continuity, and tearing of the endocardium overlying the myxomatous valve. 10 In addition, paroxysmal atrial fibrillation may contribute to the embolic events. 11
Mean platelet volume (MPV) is a simple and easy marker reflecting platelet reactivity or activation. 12,13 Platelet activation is important in the pathophysiology of cardiovascular diseases, 13,14 and larger platelets have higher thrombotic potential. 15 To our knowledge, there is no study investigating MPV and platelet count in patients with MVP. Therefore, we aimed to examine MPV in patients with MVP and its association with severity of MVP.
Methods
Study Population
This study group consisted of 312 consecutive patients with MVP (179 females, mean age = 37 ± 10 years) and 240 controls (139 females, mean age = 37 ± 8 years) matched for age, gender, and body mass index. Patients with MVP were selected from 16 804 patients undergoing echocardiographic examination and having a diagnosis of MVP in our hospital between 2006 and 2011. The diagnosis of MVP was made by physical examination (mid-systolic click with a systolic murmur) mainly 2-dimensional echocardiography. 16 We included the classical form of patients with MVP in the study. Patients with MVP having secondary MVP due to ischemic heart disease or rheumatic heart disease, nonclassic MVP, left ventricular systolic dysfunction, acute coronary syndrome, atrial fibrillation, statin usage, a history of renal or liver disease, malignancy, hematological disorders, acute or chronic infection, and current stroke were not included in the study. The study was approved by the local ethics committee and all patients gave their informed consent.
Echocardiography
The M-mode, 2-dimensional, and Doppler echocardiographic examinations were made using GE VingMed System 5 (Norway) to assess the echocardiographic measurements. Echocardiographic diagnosis of MVP was made according to the criteria by Freed et al. 16 Briefly, the patients were classified as having classic prolapse if displacement exceeded 2mm and maximal thickness was at least 5 mm and had nonclassic prolapse if displacement exceeded 2 mm but the maximal thickness was less than 5 mm in the parasternal long-axis view. 16 Left atrial and ventricular dimensions and left ventricular ejection fraction were measured by M-mode echocardiography in the parasternal long-axis view according to the recommendations of the American Echocardiography Society. 17 Valvular regurgitations were evaluated by Doppler color flow mapping. The degree of mitral regurgitation was assessed as the ratio of the maximal regurgitant jet area to the area of the left atrium in the parasternal and apical long-axis and apical 4-chamber views. 18 The degree of mitral regurgitation was considered to be mild, moderate, or severe on the basis of ratios of 0 to 20, >20 to 40, and >40%, respectively. 18
Biochemical Measurements
Blood samples were drawn from the antecubital vein between 08.00 and 10.00
Statistical Analysis
Data were analyzed with the SPSS software version 15.0 for Windows. Continuous variables were presented as mean ± standard deviation and categorical variables as percentages. Student t test or Mann-Whitney U test was used to compare continuous variables where appropriate. Categorical variables were compared with Fisher exact test or chi-square test. The correlation of MPV with other variables was evaluated with Pearson correlation analysis. Variables with P value less than .05 in the correlation analysis were included into multivariable linear regression analysis to identify independent variables for MPV. Beta value and its 95% confidence interval (CI) were calculated for the independent variables. Statistical significance was defined as P < .05.
Results
Clinical and laboratory characteristics are presented in Table 1. There were no significant differences in these characteristics between MVP and control groups. Echocardiographic findings of the study population are shown in Table 2. Prolapsing leaflet was anterior, posterior, and both leaflets in 68%, 14%, and 18% of patients, respectively. Maximal leaflet displacement was 4.8 ± 1.4 mm. The mean thicknesses of the anterior and posterior leaflets were 5.4 ± 0.5 mm and 5.3 ± 0.4 mm, respectively (Table 2).
Clinical and Laboratory Characteristics in Patients With MVP and Controls.
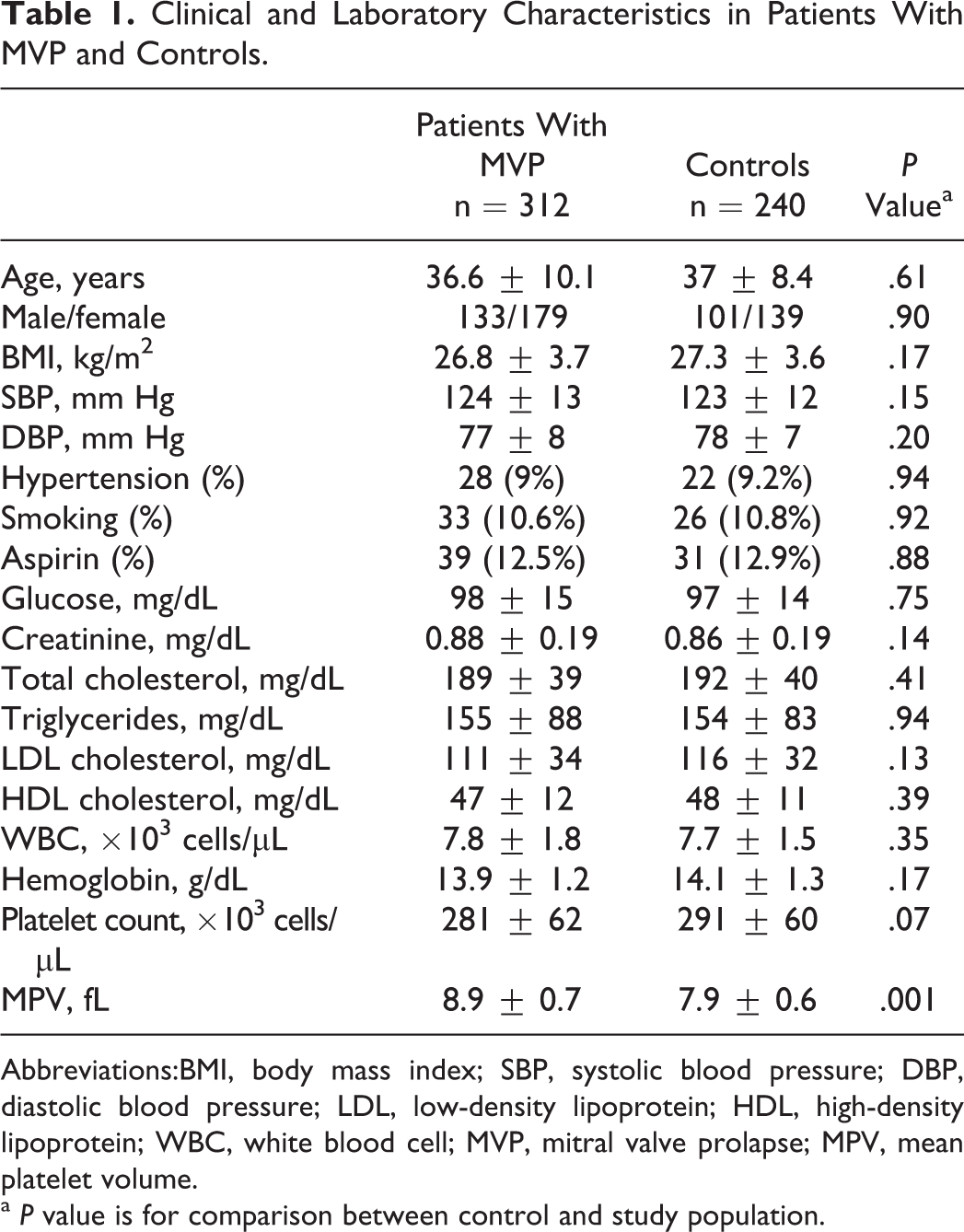
Abbreviations:BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; LDL, low-density lipoprotein; HDL, high-density lipoprotein; WBC, white blood cell; MVP, mitral valve prolapse; MPV, mean platelet volume.
a P value is for comparison between control and study population.
Echocardiographic Findings in Patients With Mitral Valve Prolapse and in Controls.
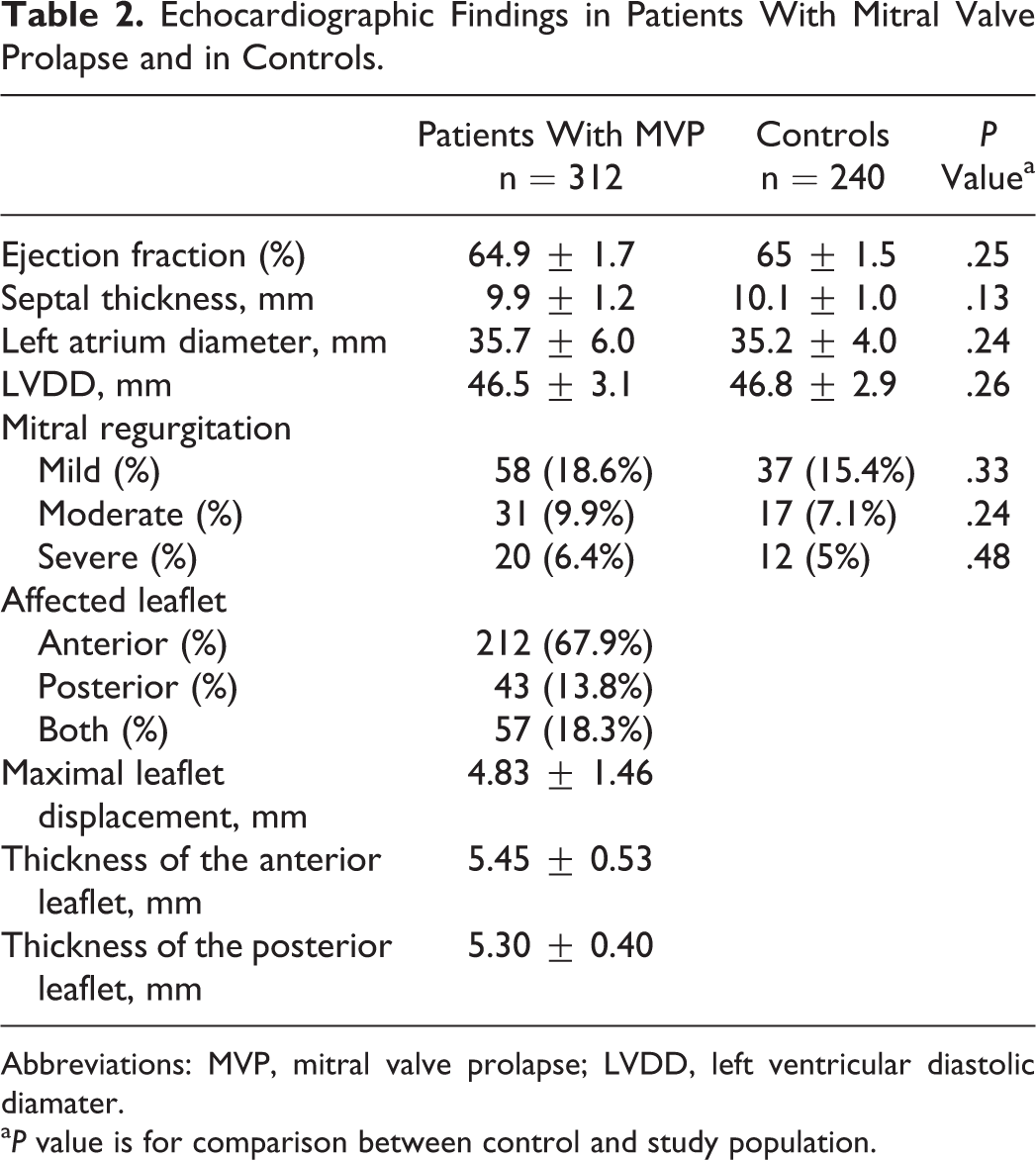
Abbreviations: MVP, mitral valve prolapse; LVDD, left ventricular diastolic diamater.
a P value is for comparison between control and study population.
Compared with the controls, MPV was significantly higher in patients with MVP (8.9 ± 0.7 vs 7.9 ± 0.6 fL, P < .001; Table 1). On the contrary, platelet count was slightly lower in patients with MVP than in controls (281 ± 62 vs 291 ± 60 × 103 cells/µL, P = .07). White blood cell count and hemoglobin level were comparable in 2 groups (Table 1).
In the correlation analysis, MPV was correlated with degree of mitral regurgitation (r = .62, P = .001), maximal leaflet displacement (r = .72, P = .001), mean thickness of the anterior (r = 0.57, P = .001) and posterior leaflets (r = .45, P = .001), smoking (r = .15, P = .01), serum creatinine (r = .11, P = .02), and platelet count (r = −.28, P = .001; Table 3). Of these variables, degree of mitral regurgitation (β = .23, 95% CI: 0.14-0.32, P = .001), maximal leaflet displacement (β = .24, 95% CI: 0.17-0.31, P = .001), and the mean thickness of the anterior (β = .47, 95% CI: 0.27-0.61, P = .001) and posterior leaflets (β = .22, 95% CI: 0.03-0.41, P = .02) were independently associated with MPV in linear regression analysis.
Variables Showing Significant Correlation With Mean Platelet Volume.
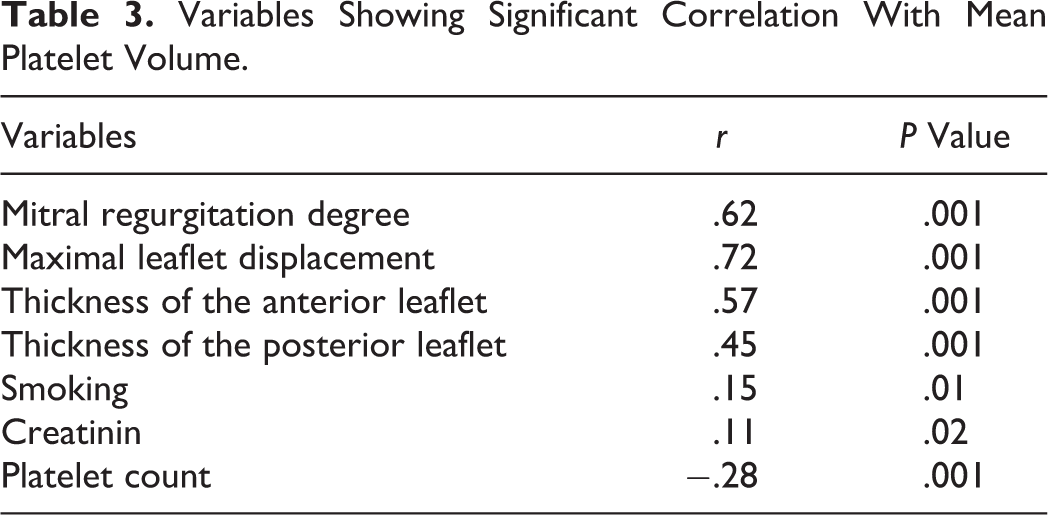
Discussion
In the present study, MPV was higher in patients with MVP than age-, gender-, and risk factor-matched controls. More importantly, it was associated with the degree of mitral regugitation, maximal displacement, and thickness of leaflets. To our knowledge, this is the first study investigating MPV in patients with MVP.
As a simple marker, MPV has been considered to reflect the platelet reactivity or activation. 12 –14 Larger platelets are metabolically more reactive than smaller ones. Reactive platelets contain more prothrombotic material, with increased thromboxane A2 and B2 per unit volume and glycoprotein IIb-IIIa receptor expression 19 –21 with greater aggregability in response to adenosine diphosphate (ADP) 22 and decreased inhibition of aggregation by prostacyclin in vitro. 23 Similarly, larger platelets create a tendency to the development of thrombosis. 21 Previous studies have reported that elevated MPV can increase the risk of short- and long-term adverse cardiovascular outcome in patients with established cardiovascular diseases. 24 –26 Similarly, the increased platelet activation and aggregation can be related to cardiovascular complications. 14
Previous studies have demonstrated some platelet activation markers in patients with MVP. 27 –29 Fisher et al have reported that platelet factor 4 was elevated in patients with MVP without a history of stroke. 27 Similarly, Cudillo et al have shown that plasma beta-thromboglobulin levels were significantly higher in children with MVP than in normal participants. 28 Zuppiroli et al have shown that platelet function tests were found to be normal with the exception of platelet aggregation rate. 29 In their study, there was a significant rise in von Willebrand factor and fibrinopeptide A levels, suggesting the existence of a particular subset of patients with MVP with a higher risk for thromboembolic events. 29 We evaluated the platelet reactivity by measuring MPV. Our findings are in accordance with those of previous studies. 27 –29
It is well known that platelets play a key role in the development of thrombi. Thromboembolic events have been described in patients with MVP. 30 –33 Mitral valve prolapse is considered to be a predisposing factor for cerebral ischemia, especially in young adults. 7,34 Walsh et al have reported that platelets are frequently activated in asymptomatic patients with MVP and play a part in the pathogenesis of thromboembolism in these patients. 8 They have suggested that measurement of platelet activation may allow identification of a subgroup of patients with MVP at increased risk for emboli. 8 Barnett et al have considered that ischemic events can be related to embolus that resulted from the abnormal mitral valve with or without associated paroxysmal cardiac arrhythmia. 30 Similarly, Kostuk et al have suggested that the surface changes in the leaflets may initiate platelet adherence and aggregation, resulting in the formation of a platelet–fibrin thrombus. 31 In addition, valvular small thrombi have been visualized by echocardiography in patients with MVP-associated cerebral ischemia. 35 On the other hand, MVP-mediated atrial fibrillation may cause left atrial clot as a source of larger emboli. 8,27,36 As a result, platelets may be activated and contribute to the thromboembolic events in patients with MVP.
Platelet survival may be shorter in patients with MVP compared with normal participants. 36 Platelet turnover may be increased in thromboembolic events 37 and subsequently younger and larger platelets can appear in the circulation. 38,39 Therefore, it can be expected that there may be increased platelet turnover and larger platelets in patients with MVP.
Some studies imply that platelet activation may be associated with the degree of mitral regurgitation. 8,9,27 Tse et al 40 have reported that mitral regurgitation due to mitral valve disease was associated with systemic platelet activation. Mitral valve prolapse itself was not associated with increased platelet activation in contrast to our result. However, the percentages of mitral insufficiency were comparable in MVP and control groups, in our study. We also found that MPV was positively correlated with maximal displacement and thickness of the leaflets in patients with MVP in addition to the degree of mitral regurgitation. Thus, we can speculate that platelets may be more reactive in patients with more severe MVP, and that such patients may have more risk for thromboembolic events. More aggressive antiplatelet therapy may be necessary to overcome platelet reactivity in such patients.
Conclusion
Our findings suggest that MPV may be elevated in patients with MVP and independently associated with severity of mitral regurgitation, displacement, and thickness of the leaflets. Patients with MVP having elevated MPV may need more antiplatelet therapy to prevent thromboembolic events.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
