Abstract
We aimed to compare cerebral artery velocity and relevant clinical factors in patients with β-thalassemia intermedia (β-TI), β-thalassemia major (β-TM), and healthy individuals. For this study, 60 patients with β-TM and 64 with β-TI were randomly selected. Sixty healthy adults comprised the control group. Time average maximum mean velocity (TAMMV) was measured in large cerebral arteries with transcranial Doppler (TCD) ultrasonography. In all arteries, TAMMV was significantly higher in the β-TI group than that of β-TM or controls (P < .001). Patients with β-TM had significantly higher TAMMV than controls in most arteries evaluated (P < .001). Overall, the lack of regular blood transfusions, splenectomy, and lower age (to a lesser extent) was found to be independent influencing factors contributing to high cerebral artery velocities. Further detailed longitudinal studies are needed to confirm these results and to determine the risk of silent infarction and stroke in patients with thalassemia and abnormal TCD, with particular focus on patients with β-TI.
Keywords
Introduction
Patients with β-thalassemia are prone to hypercoagulable state associated with various mechanisms. 1,2 As a result thromboembolic events are frequent in these patients, especially in those with β-thalassemia intermedia (β-TI). Consequently, cerebral involvement presenting as silent infarction or stroke might occur more frequently in these patients than in the general population. 3 The prevalence of stroke has been reported between 0.25% and 1.4% in patients with β-thalassemia major (β-TM) and between 0.36% and 1.8% in patients with β-TI. 4 –9 However, it seems that asymptomatic brain abnormalities or silent infarctions occur more frequently (28%-60%) in β-TI. 3,10,11 Many studies of children and adults with sickle cell disease (SCD) indicate a higher risk of developing stroke in patients with silent infarcts. 12 –15 Because of the importance of stroke-related complications and mortality, preventive interventions in high-risk patients are critical in patients with thalassemia. Based on the results of the Stoke Prevention Trial in Sickle Cell Anemia (STOP) study, patients with SCD who have a time average maximum mean velocity (TAMMV) ≥200 cm/s are at higher risk of developing stroke. 16 The National Heart Lung and Blood Institute has recommended chronic blood transfusion in these high-risk patients. 17 Transcranial Doppler (TCD) studies in adults with SCD also showed higher TAMMV compared to healthy controls; however, velocities were lower than those reported in children with SCD. 18,19 Many reports have evaluated cerebral artery velocity and stroke risk, especially in children with SCD, but apparently no such studies have been published regarding the assessment of TAMMV in patients with thalassemia.
Transcranial Doppler ultrasonography has been used since 1982 to measure blood velocity in the basal cerebral arteries. This technique is effective in screening for stenosis, vasospasm, or alterations in intracranial hemodynamics related to occlusive carotid disease. 20 Compared to angiography, it is a safe, noninvasive, inexpensive, and portable method that is well tolerated especially by children. 21 –23 Noninvasive diagnosis by Doppler ultrasonography of fetal anemia has been reported in many situations including maternal red cell alloimmunization, 24 fetomaternal hemorrhage, 25,26 and parvovirus infection. 27 It has been used widely in patients with SCD to determine cerebral artery stenosis due to anemia and to predict stroke risk. 28 –30 However, there appears to be no reports of TCD screening in patients with thalassemia.
In this study, we aimed to compare TAMMV of cerebral arteries in patients with β-TI, β-TM, and healthy individuals with TCD ultrasonography. Also, we sought for factors that influence TAMMV findings in patients with thalassemia.
Patients and Methods
The participants in this cross-sectional study were 60 randomly selected patients (age >15 years) with β-TM and 64 patients with β-TI, who were recruited from patients with thalassemia referred to the thalassemia clinic of Fars province in southern Iran, during 2009 to 2010. The diagnosis was based on cellulose acetate hemoglobin electrophoresis and complete blood count. All patients with β-TM were transfusion dependent and received regular blood transfusion every 2 to 4 weeks from the age of less than 2 years. All patients with β-TI were transfusion independent or required only long-interval transfusion (1-2 times per year). The exclusion criteria were history of diabetes mellitus, hypertension, hypothyroidism, thrombosis, cardiovascular disease, cerebrovascular accident, and any neurological abnormality on physical examination. Sixty healthy adults (age >15 years) comprised the control group. They were selected from healthy individuals who were referring to outpatient clinic for checkup. Informed consent was obtained in writing from each patient. The research protocol was approved by the Ethics Committee of Shiraz University of Medical Sciences.
Demographic, laboratory, and medical information from all patients was gathered with a specially designed data collection form. Complete blood count analysis was done with a Lution Beds counter (Sysmex, England). Transcranial Doppler ultrasonography was done for each patient or control participant by an experienced neurologist with a legend TC22 TCD ultrasound machine and 2-MHz transducer (Bristol, United Kingdom). Time average maximum mean velocity was measured in the bilateral anterior cerebral artery (ACA), posterior cerebral artery (PCA), middle cerebral artery (MCA), and internal carotid artery (ICA) with a temporal windows approach, and in the basilar artery and vertebral arteries with a suboccipital approach. In patients with β-TM, TCD examination was done just prior the transfusion.
Data analysis was done with Statistical Package for the Social Sciences v 17 software (SPSS, Chicago, Illinois). Categorical variables were compared between 2 or more groups with the Fisher exact test or chi-square test. Continuous variables were compared between 2 groups with independent sample t tests, and comparisons among 3 groups were done by analysis of variance and analysis of covariance (ANCOVA). The least significant difference test was used as a post hoc test to identify the differences between groups. Multiple linear regression analysis was done to determine which independent factors influenced TAMMV in each artery. Correlations between the 2 continuous variables were assessed by calculating the Pearson correlation coefficient (r). Values of P less than .05 were considered statistically significant.
Results
The characteristics and laboratory data for patients and controls are shown in Table 1. Mean ages of the patients in the β-TM and β-TI groups did not differ significantly; however, both groups were significantly younger than the control group (P = .016). Mean serum platelet count in patients with β-TI was significantly higher than in patients with β-TM (P = .03); however, these groups did not differ significantly with regard to thrombocytosis (platelet counts >500 000/µL).
Characteristics and Laboratory Data of the Patients With β-TM and β-TI and Healthy Participants.
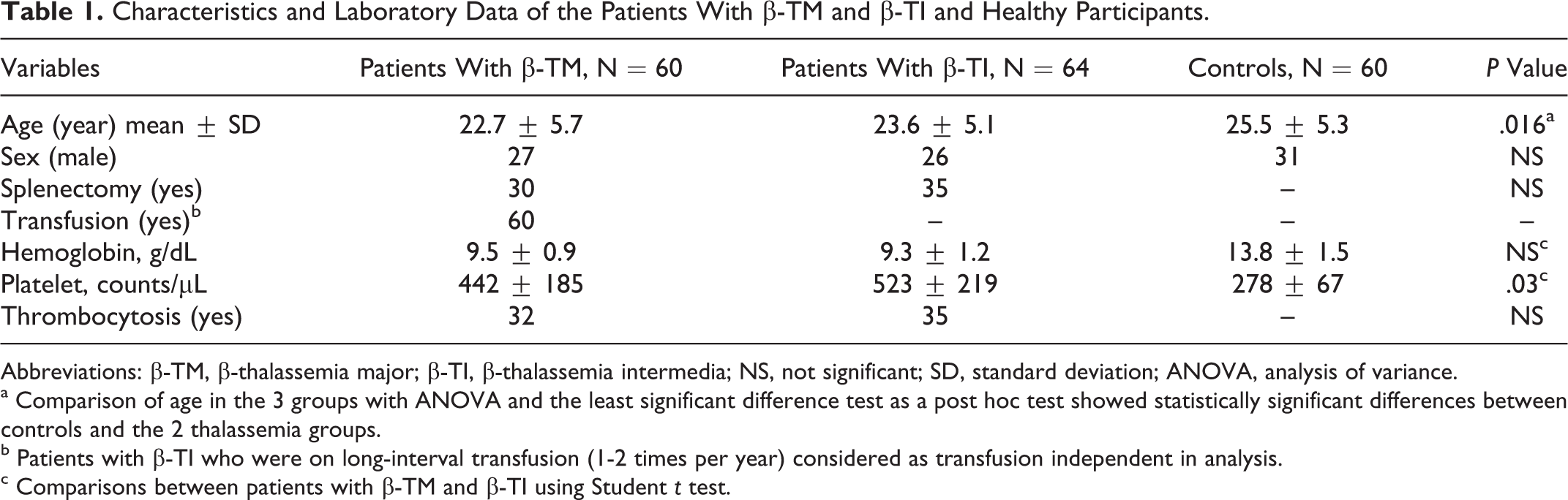
Abbreviations: β-TM, β-thalassemia major; β-TI, β-thalassemia intermedia; NS, not significant; SD, standard deviation; ANOVA, analysis of variance.
a Comparison of age in the 3 groups with ANOVA and the least significant difference test as a post hoc test showed statistically significant differences between controls and the 2 thalassemia groups.
b Patients with β-TI who were on long-interval transfusion (1-2 times per year) considered as transfusion independent in analysis.
c Comparisons between patients with β-TM and β-TI using Student t test.
Because age is a potential confounding variable in comparisons of TAMMV between groups, we conducted ANCOVA to account for the effect of age. As shown in Table 2, TAMMV in all arteries was significantly higher in patients with β-TI than in patients with β-TM and controls (P < .001). Patients with β-TM had significantly higher TAMMV than controls in all arteries except the left and right PCA (P < .001).
Comparison of the Mean Velocities of Different Intracranial Arteries in Patients With β-TM and β-TI and Healthy Participants.a
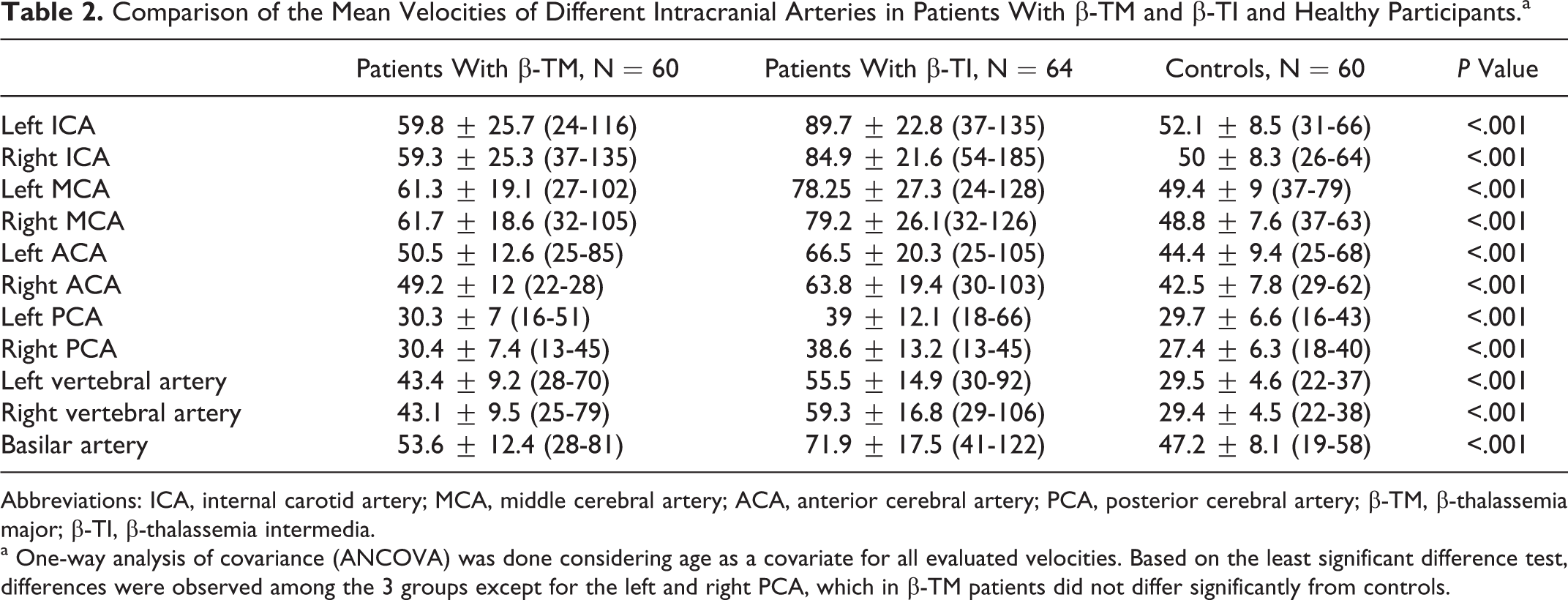
Abbreviations: ICA, internal carotid artery; MCA, middle cerebral artery; ACA, anterior cerebral artery; PCA, posterior cerebral artery; β-TM, β-thalassemia major; β-TI, β-thalassemia intermedia.
a One-way analysis of covariance (ANCOVA) was done considering age as a covariate for all evaluated velocities. Based on the least significant difference test, differences were observed among the 3 groups except for the left and right PCA, which in β-TM patients did not differ significantly from controls.
We considered β-TM and β-TI patients together and used univariate analysis to compare TAMMV. Independent variables included sex, hemoglobin, age, transfusion, splenectomy, and thrombocytosis in this population. There was no significant relationship between the TAMMV and sex (P > .05). Time average maximum mean velocity was not significantly correlated with hemoglobin level in any of the arteries (P > .05). With regard to age, we detected significant inverse correlations between this variable and TAMMV only in the left ACA (r = −0.189, P = .035), right ACA (r = −0.201, P = .025), and right PCA (r = −0.187, P = .037). Time average maximum mean velocity was significantly lower in all arteries in transfusion-dependent patients compared to patients without transfusion (P < .001). In splenectomized patients, TAMMV was significantly higher in all arteries (P < .05), except the left and right vertebral arteries as well as the basilar artery. This pattern was also observed in patients who had thrombocytosis compared to patients without thrombocytosis. However, after adjustment for splenectomy as a confounding variable, it was not significant in each of the evaluated arteries (P > .05). The highest velocity was observed in the right ICA: 135 cm/s in the β-TM group and 185 cm/s in the β-TI group. Figure 1 shows TAMMV of the right ICA in a healthy participant (A) and a patient with β-TI (B).
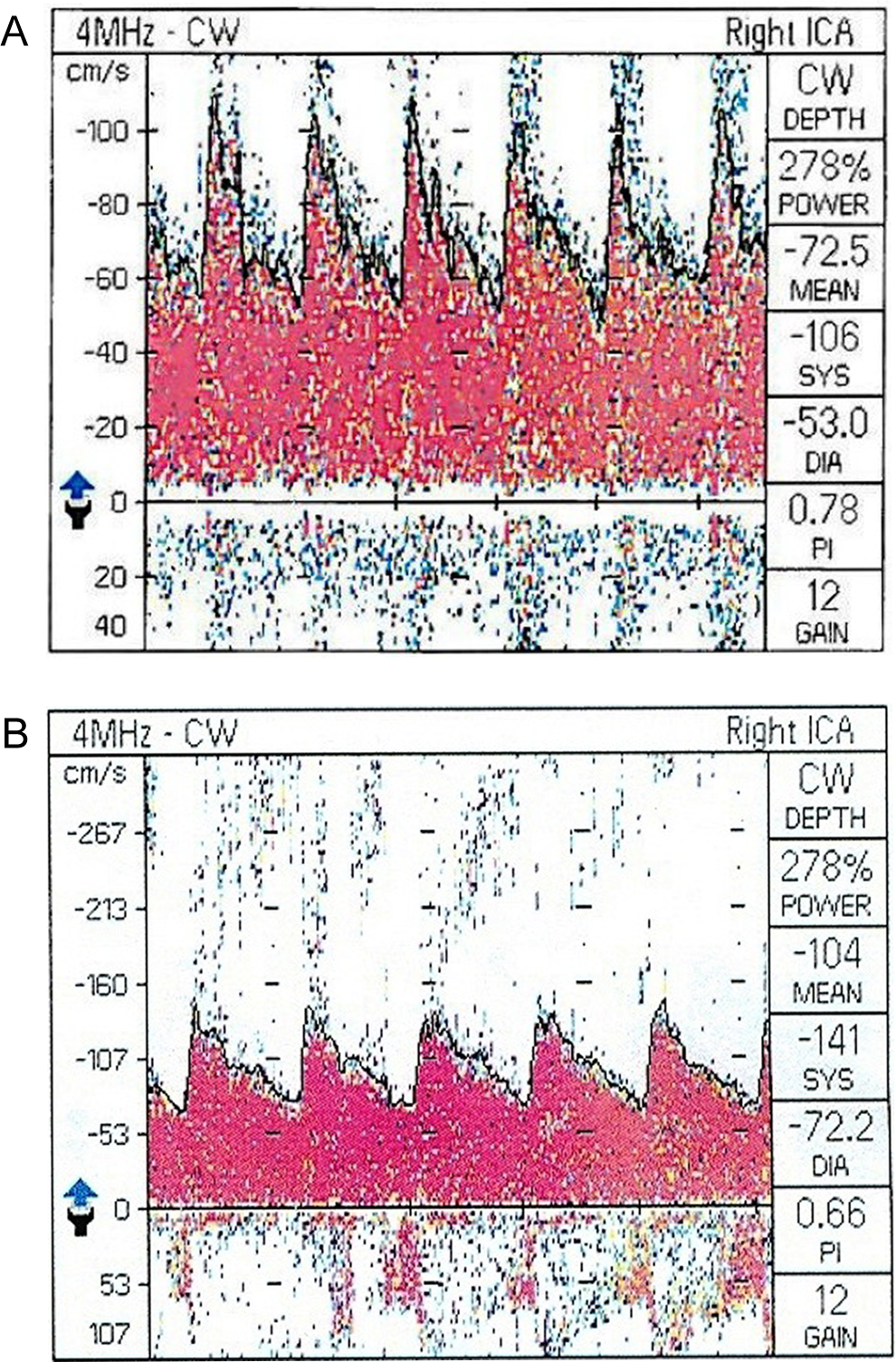
Time average maximum mean velocity of right internal carotid artery in a 25-year-old healthy participant (A) and in a 25-year-old patient with β-thalassemia intermedia (B).
Multiple linear regression analysis was used to further characterize the relationship between TAMMV in each artery and related independent variables that were significant based on the univariate analysis (Table 3). Since left and right vertebral arteries and basilar artery showed significant association only with transfusion in univariate analysis, these arteries were not entered in this model. In all other evaluated arteries, transfusion and splenectomy showed significant negative and positive correlation, respectively, with TAMMV (P < 2.05). Age was inversely correlated with TAMMV only in the right ACA, left ACA, and right PCA (P < .05).
Multiple Linear Regression Analysis of Covariates Associated With the Mean Velocities of Different Intracranial Arteries in Patients With β-TM and β-TI.a
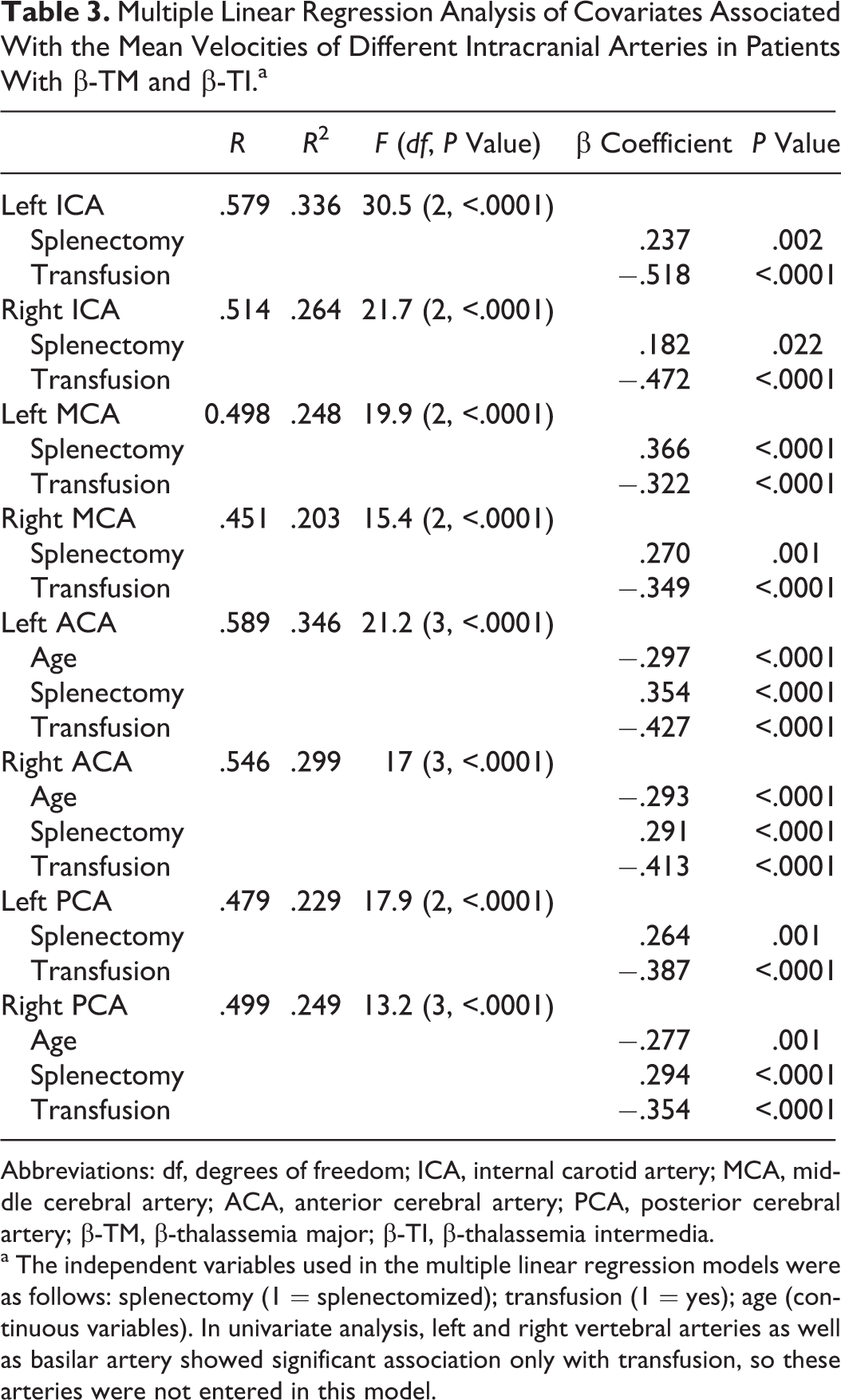
Abbreviations: df, degrees of freedom; ICA, internal carotid artery; MCA, middle cerebral artery; ACA, anterior cerebral artery; PCA, posterior cerebral artery; β-TM, β-thalassemia major; β-TI, β-thalassemia intermedia.
a The independent variables used in the multiple linear regression models were as follows: splenectomy (1 = splenectomized); transfusion (1 = yes); age (continuous variables). In univariate analysis, left and right vertebral arteries as well as basilar artery showed significant association only with transfusion, so these arteries were not entered in this model.
Discussion
We found that TAMMV in all arteries was significantly higher in patients with β-TI than in patients with β-TM and healthy individuals. Patients with β-TM also had significantly higher TAMMV than controls in all arteries except the left and right PCA. Transfusion was found to be an independent protective factor in all arteries. Splenectomy was a risk factor in all evaluated arteries except the left and right vertebral arteries and basilar artery, and older age was a protective factor only in the right ACA, left ACA, and right PCA.
There are few published data discussing the factors influencing cerebral blood flow (CBF) velocity, in general. Cerebral blood flow velocity decreases with age possibly due to reducing CBF, increased vascular resistance downstream from insonated vessels, and decreased vascular compliance. Cerebral blood flow velocity has been reported to be approximately 3% to 5% higher in women than that of men in the age range of less than 50 years. It may be as a result of the effect of estrogen on lower vascular resistance. Hematocrit is inversely correlated with CBF velocity. Vessel diameter and fibrinogen have inverse relationships with blood flow velocity. Other possible influencing factor is abnormal cardiac physiology, such as hypertension and itracardiac shunt. However, the mechanism is not clearly understood. Conditions with increasing cerebral blood volume including arteriovenous malformations, post-stenotic segment of artery, and tortus vessels are associated with elevated velocity. Medications may also contribute in the alteration of CBF velocity. Insonation angle used in TCD examination and examiner variability are other influencing factors of CBF velocity. 31,32 In our study, all TCDs in patients and controls were performed by single insonation angle and 1 expert neurologist, so these possible confounding effects were subsided. In patients with β-TM and β-TI in addition to the mentioned factors, hypercoagulable state that is clinically expressed by thromboembolic events could influence CBF velocity. 33
Time average maximum mean velocity did not correlate significantly with hemoglobin level. In contrast, higher velocities were found to be related with lower hematocrit values in other studies of patients with SCD. 19,34 –36 However, in sickle cell anemia, the presence of intracranial occlusive disease could also contribute to elevated velocities. 31
We found a significant correlation between TAMMV and splenectomy. Only few studies evaluated the relationship between splenectomy and abnormal TCD. Kalpatthi et al 37 reported a rate of 34.2% of at least 1 abnormal TCD value after splenectomy in children with SCD. Moreover, splenectomy could be a possible risk factor for hypercoagulable state and subsequently thromboembolic events in patients with thalassemia 38 and for silent infarction especially in patients with β-TI. 10,11 Our results showed that thrombocytosis by itself may not contribute to increased TAMMV in patients with thalassemia. This does not support the hypothesis that platelet counts may influence blood viscosity and subsequently blood flow velocity. 36 We found no similar studies in patients with thalassemia to compare with our results. However, Bernaudin et al 39 evaluated this association in patients with SCD. In their univariate analysis, they found no significant association between cerebral artery velocity and platelet count. In their multivariate model, they found that glucose-6-phosphate dehydrogenase deficiency, the absence of α-thalassemia, and increased baseline reticulocyte count were the only significant independent predictive factors for abnormal TCD.
In the present study, age was found to be an independent predictive factor in multiple regression models. Age correlated inversely with TAMMV but only in the right ACA, left ACA, and right PCA. Similarly, Venketasubramanian et al 21 detected an inverse correlation of age with MCA velocity (r = −.58, P < .01) in 10 patients with SCD. In addition, Neish et al 30 reported a possible inverse correlation between age and velocity, probably due to higher hematocrit levels in older individuals as well as other mentioned factors in relation with older age discussed previously. 31
Based on our results, TAMMV was significantly lower in patients with thalassemia undergoing transfusion compared to those not undergoing transfusion. In the multiple regression model, the only independent predictive factor for abnormal TCD findings in all arteries was transfusion. Our results are in accordance with those of the STOP II trial reported by Adams and Brambilla. 40 They showed that the discontinuation of prophylactic transfusion in children with SCD resulted in a high rate of reversion to abnormal blood flow velocities in TCD studies. The protective effects of transfusion against initial or recurrent stroke 16,39,41 as well as silent infarct have been documented in patients with SCD who have abnormal TCD findings. 12 However, the mechanism through which transfusion prevents silent infarcts in patients with SCD has not been clearly identified.
Transcranial Doppler studies in children and young adults with SCD documented a direct correlation between high cerebral artery velocity and stroke risk, so the primary prevention of stroke in SCD can be achieved by regular TCD examination. It helps to select patients who need transfusion appropriately. An efficient screening program may be a cost-effective way to prevent stroke-related morbidity and mortality. 29 In our patients with thalassemia, TAMMV was higher than in healthy individuals but lower than in adult patients with SCD. 18 However, according to STOP classification scheme, none of our patients had abnormal TAMMV of the MCA or internal carotid arteries (more than 200 cm/s). Only 1 patient with β-TI had a conditional velocity (between 170 and 199 cm/s) as characterized by Adams et al. 16,29 In patients with thalassemia, there are no detailed studies that aimed to determine the risk of silent infarction or stroke in relation to cerebral artery velocity or the precise effect of transfusion on cerebral artery velocity. More powerful longitudinal studies will be needed to determine the decision-making criteria for the use of TCD examination as a screening tool in these patients.
Based on our literature review, it seems that stroke occurs more frequently in SCD than in patients with β-thalassemia. Cerebrovascular accidents 42 occur in about 12% of SCD patients by age 21 compared to 0.25% to 1.4% in patients with β-TM and 0.36% to 1.8% in patients with β-TI. 4 –9 However, silent infarctions have been reported in 28% to 60% of different populations of patients with β-TI 3,10,11 compared to 13% to 35% of patients with SCD. 14,43 –46 In our study, we found that patients with β-TI had higher TAMMV than patients with β-TM. We also showed that the lack of regular transfusion was the only important risk factor for higher velocity in all arteries evaluated here and that younger age was a risk factor in some arteries. Although patients with β-TM and β-TI did not differ significantly with regard to age or splenectomy in our sample, it seems that the higher rate of cerebral artery velocity in patients with β-TI compared to β-TM may be related to the lack of regular transfusion in this group. However, there may be other unknown risk factors in β-TI contributing to increased cerebral artery velocity. An important finding, which has been clearly documented in patients with SCD, is that high cerebral artery velocity may be a predisposing factor for silent infarction and stroke. In this connection, transfusion is known to be an effective prophylactic method to prevent these events. Silent infarction documented by magnetic resonance imaging (MRI) in patients with SCD should be an indication for performing TCD; conversely, high velocity detected by TCD merits conducting brain MRI because the coexistence of these 2 abnormalities increases the risk of stroke or silent infarction. 44,47
We were unable to compare our study with earlier research because there appears to be no similar comprehensive studies in patients with thalassemia. However, we showed that our patients with thalassemia had higher cerebral artery velocities than the healthy individuals. Because these patients are likely to have a higher subsequent risk of infarction based on the results of SCD studies, we suggest that patients with thalassemia and history of stroke or in whom silent infarction is detected by MRI warrants further attention, close observation, and TCD examination with special attention to patients with β-TI because of their higher cerebral artery velocity, higher rate of silent infarction, and infrequent transfusion.
A limitation of our study is its cross-sectional rather than longitudinal design, which precluded attempts to determine the prophylactic effect of transfusion on high cerebral artery velocities in patients with thalassemia. In order to determine the correlation between cerebral artery velocity and risk of silent infarction and stroke in patients with thalassemia, cohort studies such as those done for patients with SCD will be needed. Moreover, TCD ultrasound can be used to evaluate only large cerebral arteries and is not able to reliably detect abnormalities in cortical arteries or distal parts of the cerebral arteries.
In conclusion, we show that patients with β-TI have a higher TAMMV than patients with β-TM and healthy individuals. For most of the arteries, we examined, TAMMV is higher in patients with β-TM than in healthy individuals. Regular blood transfusion and probably older age (to a lesser extent) may have a protective effect against high cerebral artery velocities in these patients. On the other hand, splenectomy may be a risk factor for high cerebral artery velocities. Further detailed longitudinal studies are needed to confirm the protective role of transfusion against high cerebral velocities. Moreover, to determine the risk of silent infarction and stroke in patients with thalassemia and abnormal TCD findings, highly structured studies are needed, such as those done for patients with SCD, but with a focus on patients with β-TI because of their higher cerebral artery velocities and infrequent transfusions compared to patients with β-TM.
Footnotes
Acknowledgment
We thank Shirin Parand at the Hematology Research Center for help with manuscript preparation, and K. Shashok (Author AID in the Eastern Mediterranean) for improving the English in the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: we thank the Vice Chancellery for Research of Shiraz University of Medical Sciences for support.
