Abstract
Background:
We aimed to document enoxaparin use in real world and identify the risk factors for bleeding complications.
Methods:
Postauthorization study in 448 surgical patients receiving enoxaparin prophylaxis. Complete compression ultrasound (CCUS) was performed at day 10 ± 3.
Results:
During treatment, 11 of 448 patients had suspected deep venous thrombosis (DVT) but none confirmed. One patient had symptoms of pulmonary embolism ([PE] 0.22%; 95% confidence interval [CI] −0.21-0.66). There were no asymptomatic cases detected upon CCUS. At the 90-day follow-up, 4 (0.9%) of the 440 patients had DVT symptoms (95% CI 0.02-1.80) and none had PE; 5.4% had major and 11.6% any type of bleeding complications. Major bleeding was more frequent in those with kidney disease (odds ratio [OR] 5.53), those who are bedridden (OR 5.49), those with peridural indwelling catheters (OR 4.01), and those on nonsteroidal anti-inflammatory drugs (OR 3.33).
Conclusions:
Enoxaparin is effective and safe in surgical patients to prevent venous thromboembolism.
Keywords
Introduction
Venous thromboembolism (VTE) is a frequent complication in patients after surgical treatment. 1 Randomized controlled trials investigating the low-molecular-weight heparin (LMWH) enoxaparin in patients with general surgery2-12 may be perceived to have limitations in addressing practical issues that may impact on the benefits and bleeding risk of patients assessed in “real-world” clinical practice. First, patients with renal insufficiency, in whom there is a potential for bioaccumulation of LMWH, were usually excluded from these clinical trials as were patients with severe obesity. Second, the associated bleeding risk was usually not addressed in the context of coadministered platelet inhibition (aspirin or thienopyridines) or nonsteroidal anti-inflammatory drugs (NSAIDs). Third, these randomized trials enrolled selected patients and LWMH was administered in a closely monitored clinical setting. Consequently, the incidence of bleeding complications in these studies may not be representative of the bleeding risk in a real-world clinical setting.
Therefore, we performed a 90-day audit of clinical practice relating to the treatment of patients in general surgery. The objectives of this study were (1) to assess the incidence of symptomatic VTE and asymptomatic deep venous thrombosis (DVT) by complete compression ultrasound (CCUS), (2) to assess the incidence of bleeding complications, and (3) to identify the risk factors for bleeding complications.
Methods
CLexane Prophylaxe nach systematischer EValuation des Thrombo-Embolie-Risikos (CLEVER) was an observational study. Participating physicians were recruited at random in clinics providing surgical care from all regions defined by the Institute of Medical Statistics throughout Germany. The study was conducted according to German Medicines Law (§ 67 (6) Arzneimittelgesetz, AMG) and was duly notified to the federal authorities (Bundesinstitut für Arzneimittel und Medizinprodukte, BfArM). Because of the noninterventional character of the study, no ethics committee approval was required. Patients’ written informed consent was obtained before enrollment.
Patients
Patients of at least 18 years receiving a peri- or postoperative prophylaxis with 20 or 40 mg enoxaparin for a recommended duration of 10 ± 3 days because of an in-hospital surgical intervention were consecutively included into this observational study. Applicable exclusion criteria were based on contraindications outlined in the summary of product characteristics of enoxaparin (Clexane, Sanofi-Aventis, Paris): hypersensitivity against one of the components of Clexane; recent (6 weeks) injury or surgery to the central nervous system, eye, or ear; recent (within 30 days) clinically relevant bleeding; acute or hemorrhagic stroke or other intracranial bleeding less than 6 months ago; acute or anamnestic intracranial disease; clinically relevant coagulation disorder; gastric or gut ulcers; imminent abort; severe liver or pancreatic disease; uncontrolled severe hypertension; endocarditis; acute or anamnestic type-2 heparin-induced thrombocytopenia; suspected vascular retinopathy, vitreous hemorrhage, or other intraocular bleeding.
Enoxaparin Dosing Regimen
A stratification of risk was carried out according to the national guideline for the prevention of VTE and resulted in an assignment of low, medium, and high risk (Table 1). 13 Based on this assignment, the physician decided on the choice of thromboprophylaxis. In patients with a low or moderate risk the recommended dose is 20 mg/d and in patients with high risk the recommended dose is 40 mg/d.
The AWMF S2 Guideline Resulting into an Assignment of Low, Medium, and High Risk. 13
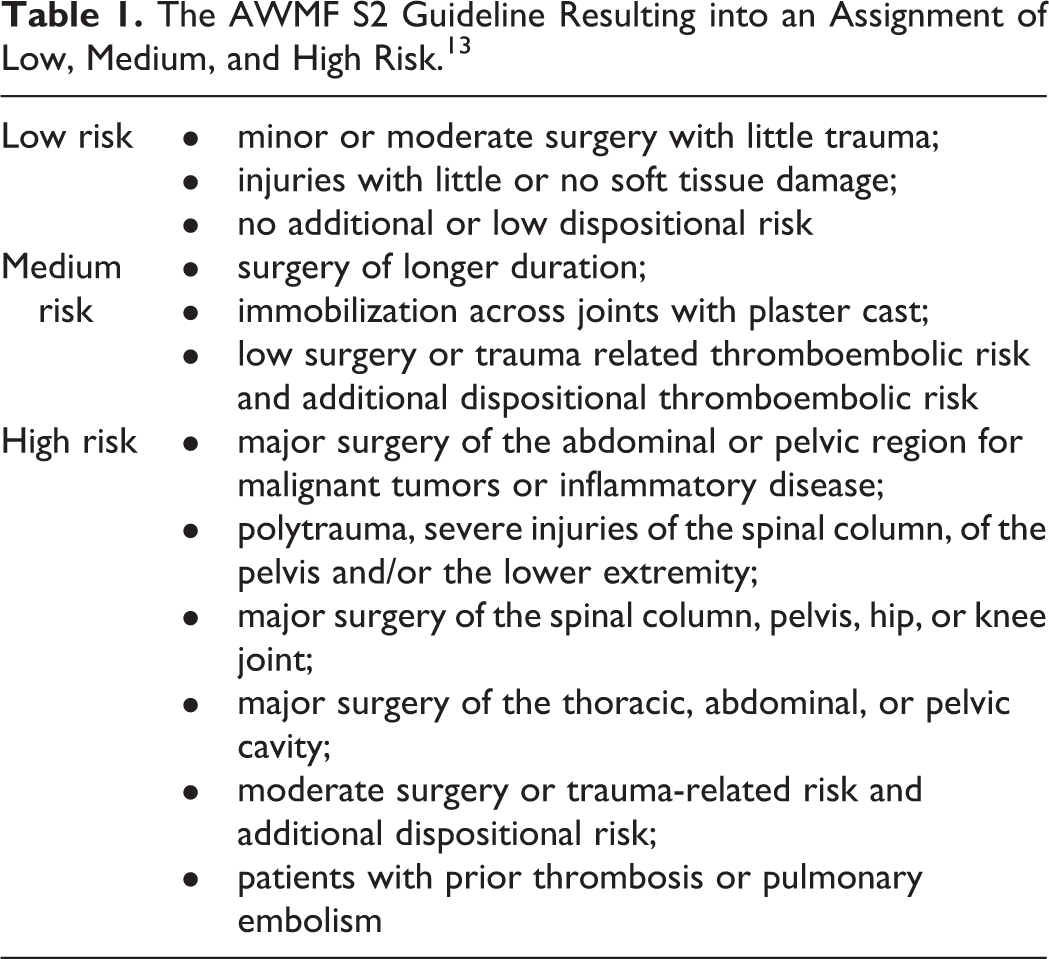
Documentation
At enrollment, patient characteristics, surgical procedures, and data on predisposing risk factors and concomitant medications were obtained. During follow-up a number of variables were obtained including the use of compression methods, interruption of therapy, a change in enoxaparin dose, immobilization, bleeding complications, and any evidence of VTE. In case of unexpected adverse events, these were recorded and announced to the responsible regulatory body.
Documentation of VTE Events
In case of symptomatic DVT or PE, events had to be documented in the case report form. On day 10 ± 3, all patients underwent a compression ultrasound screening for asymptomatic VTE. In case a patient was discharged from the hospital prior to day 10 ± 3 he or she was contacted and asked to return to the hospital for the scheduled investigation within the previously defined time frame. The CCUS investigation was conducted according to a predefined protocol and performed on either leg.
14
Furthermore,
Definition of Bleeding Complications
Major bleeding included serious, life-threatening bleeding or death, a decrease in hemoglobin ≥2 g/dL, blood transfusions of at least 2 units, retroperitoneal, intracranial or intraocular bleeding as well as bleeding complications causing an additional intervention. All other bleeding events were classified as minor.
Statistical Analysis
PASW Statistics version 17 was used for the statistical analysis. For categorical data absolute and relative frequencies and for continuous variables average and standard deviations were calculated. The incidence rates for all patients and for both risk groups (20 and 40 mg enoxaparin) were determined. Odds ratio (OR) with 95% confidence intervals (CIs) were determined from univariable and multivariable logistic regression analysis.
Results
Patient Characteristics
This study was performed between September 2006 and July 2007. A total of 448 patients (mean age 59.0 ± 15.0 years; 48.3% male) were documented. In all, 200 patients (44.6%) were bedridden with an average duration of 3.9 ± 6.1 days. In all, 25.0% of patients had an American Society of Anesthesiologists score of 1, 51.7% had a score of 2, and 23.3% had a score of 3 (Table 2). According to International Classification of Diseases, Tenth Revision, most patients were classified as miscellaneous abdominal surgery (33.7%), gastrointestinal (30.1%), and malignant tumors (22.5%). The mean duration of surgery was 109.7 ± 71.4 minutes. The majority of patients (97.8%) received endotracheal anesthesia; 32.6% of patient had cancer as a predisposing risk factor, 24.6% a medical history of venous disorders or thrombophilic conditions, and 16.5% had cardiopulmonary comorbidity (Table 3).
Patient Characteristics.
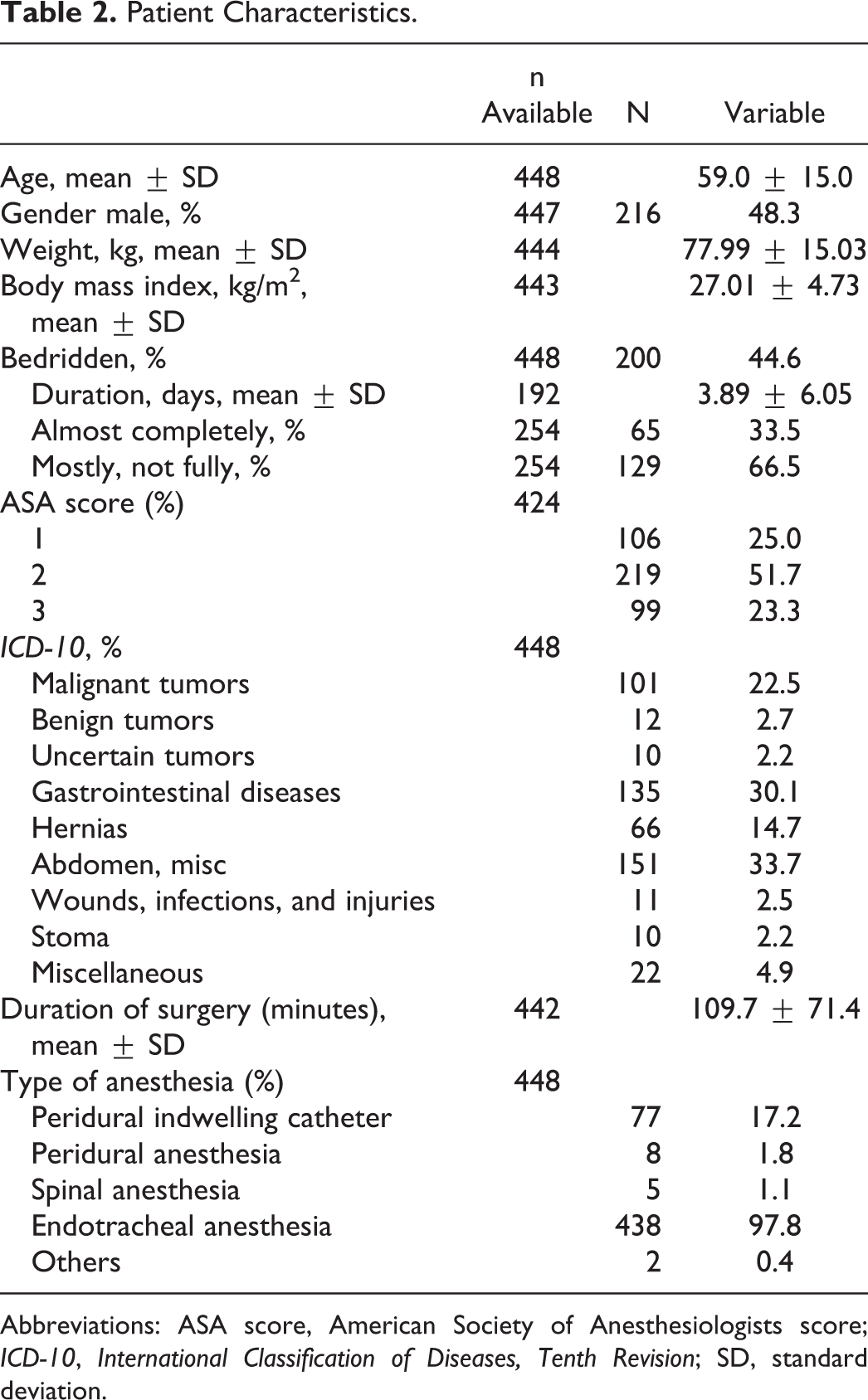
Abbreviations: ASA score, American Society of Anesthesiologists score; ICD-10, International Classification of Diseases, Tenth Revision; SD, standard deviation.
Predisposing Risk Factors.
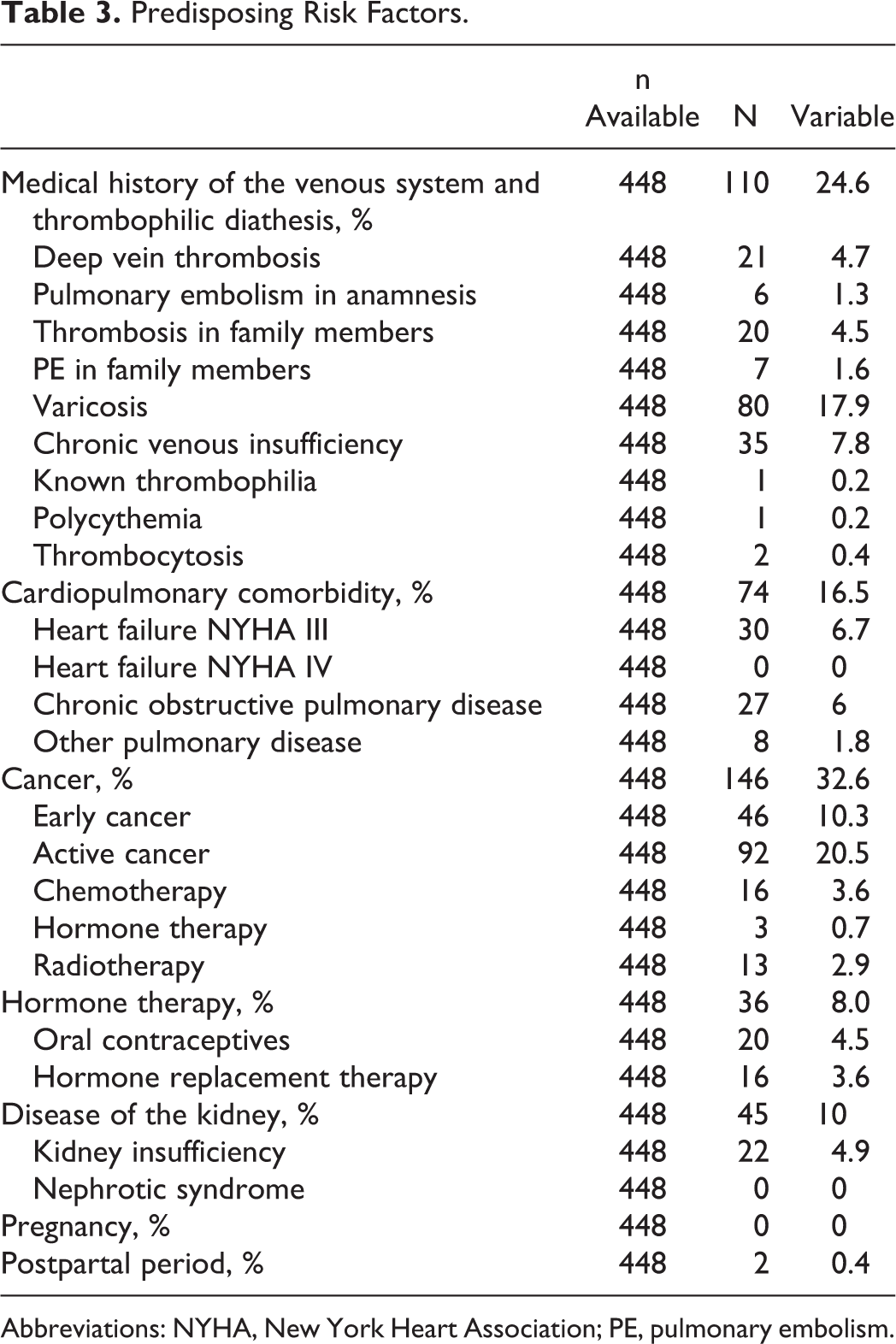
Abbreviations: NYHA, New York Heart Association; PE, pulmonary embolism.
Pharmacotherapy
In all, 43.6% of patients received 20 mg enoxaparin and 54.6% received 40 mg enoxaparin (1.8% other). The mean duration of prophylaxis was 8.9 ± 7.5 days (Table 4). While interruptions of therapy were infrequent (2.0%), there was a considerable degree of early termination (<7 days; 42.0%). Specific comedication (anticoagulants, platelet inhibitors) was infrequent with aspirin (9.2%) and NSAID (10.7%) being the most often used.
Pharmacotherapy.
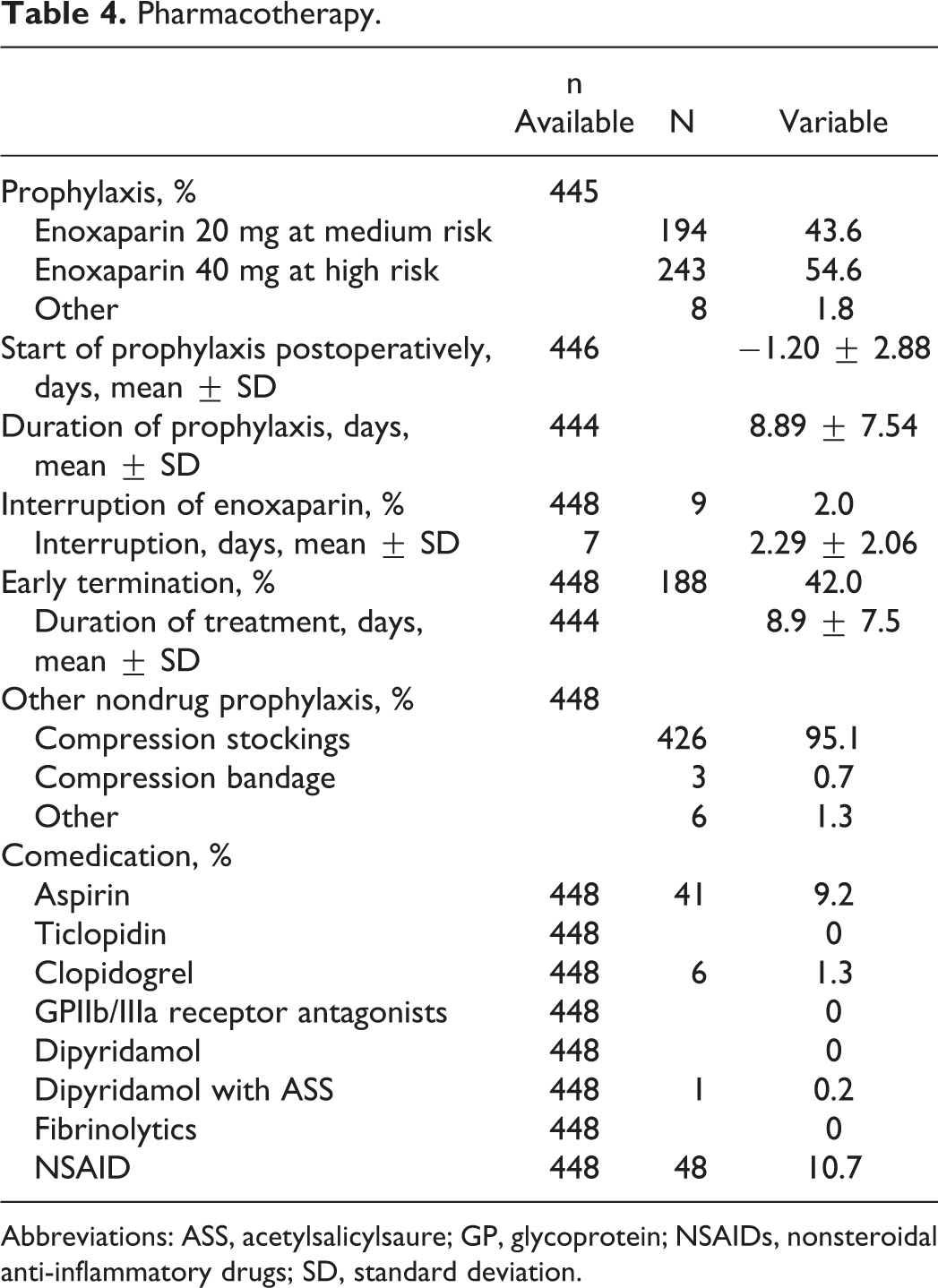
Abbreviations: ASS, acetylsalicylsaure; GP, glycoprotein; NSAIDs, nonsteroidal anti-inflammatory drugs; SD, standard deviation.
Incidence of DVT/PE
During the treatment phase, 11 of 448 patients had suspected symptomatic DVT, but none was confirmed upon
Venous Thromboembolism.
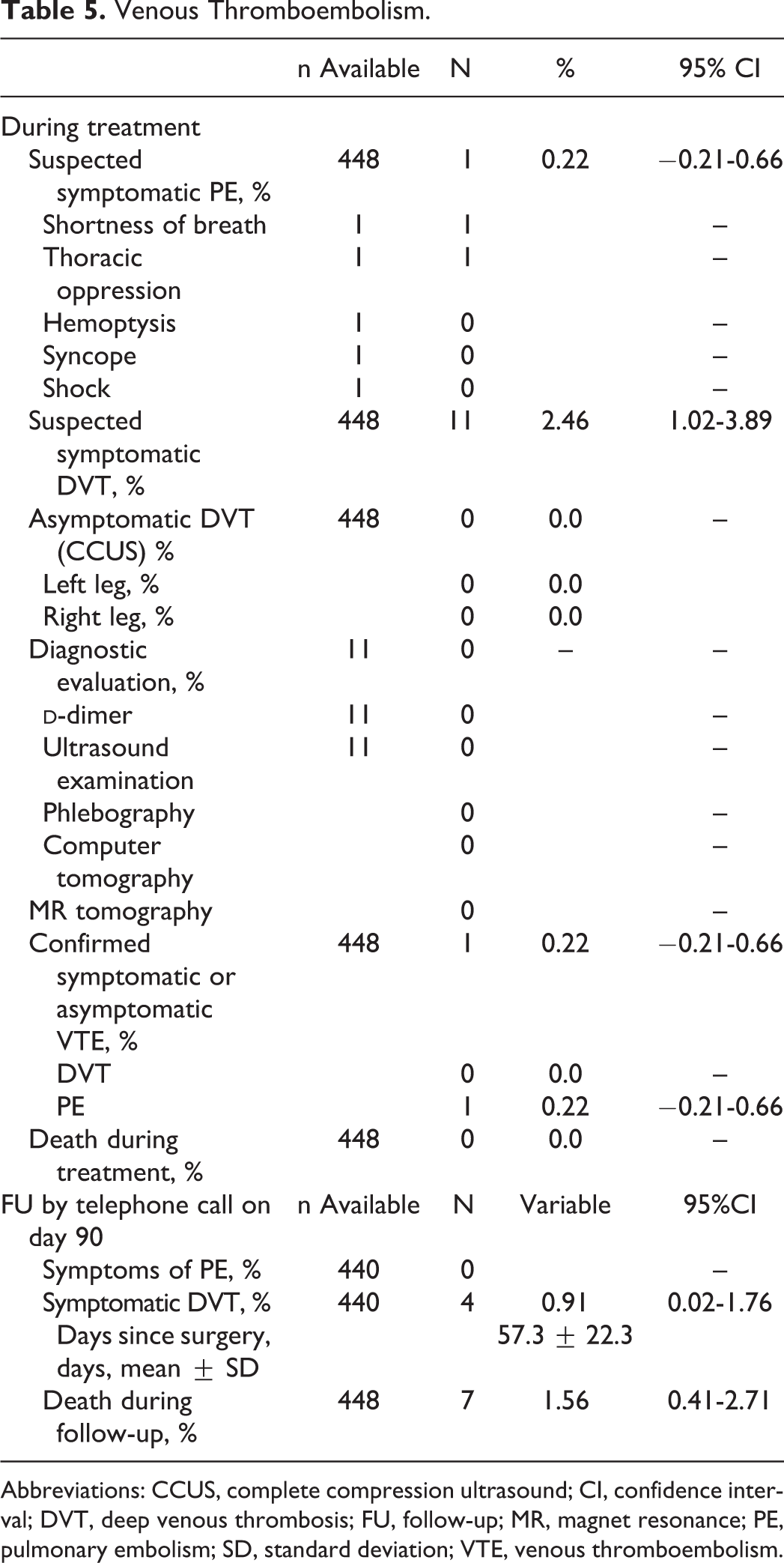
Abbreviations: CCUS, complete compression ultrasound; CI, confidence interval; DVT, deep venous thrombosis; FU, follow-up; MR, magnet resonance; PE, pulmonary embolism; SD, standard deviation; VTE, venous thromboembolism.
Adverse Events and Bleeding Complications
In all, 52 patients had any adverse event during the treatment period (11.6%; 95% CI 8.64-14.57) of which 10 were considered to be serious (Table 6). Of all, 28 (6.3%) had adverse drug reactions (2 serious), of which general disorders and administration site conditions were most frequent (n = 21). In all, 24 patients had major bleeding at a mean of 1.5 ± 3.7 days after surgery and 52 had any bleeding complications.
Number of Patients With Adverse Events (AEs) Without Casual Relationship or Adverse Drug Reaction (ADR) in the Observation Period and Adverse Drug Reactions Coded According to MedDRA.
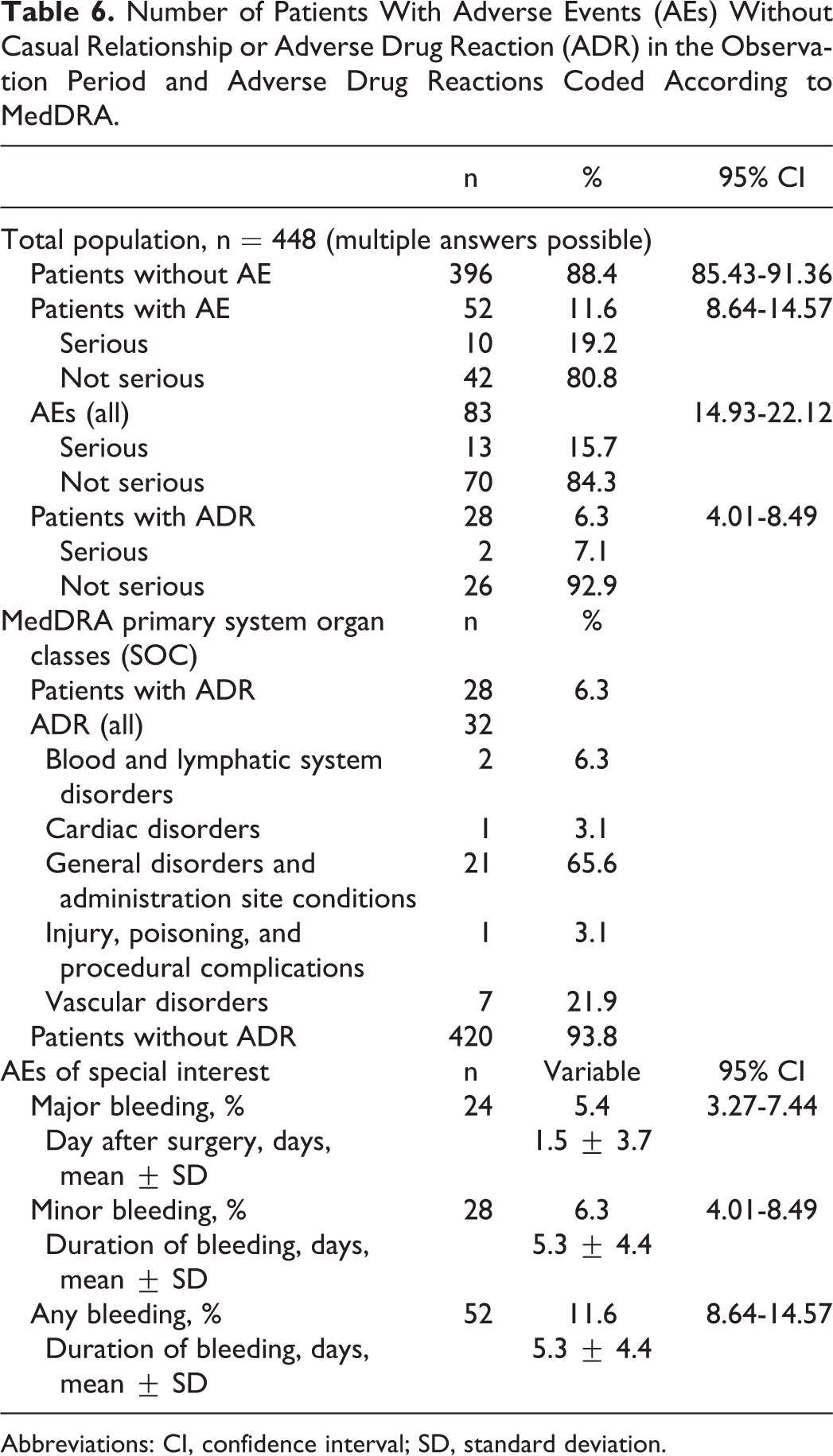
Abbreviations: CI, confidence interval; SD, standard deviation.
Patients with major bleeding complications usually received the higher enoxaparin dose (Table 7). Further predictors were complete bedridden (OR 10.24), kidney disease (OR 9.71), and other chronic obstructive pulmonary disease (OR 6.33). These predictors also applied to patients with any bleeding complications, but the association with enoxaparin dose was not significant. Based on multivariable analyses major bleeding was more frequent in those bedridden (OR 5.49), with kidney disease (OR 5.53), peridural indwelling catheters (OR 4.01), and those using NSAID (OR 3.33).
Odds Ratio of patient profiles for Bleeding Events (Any or Major Bleedings in Comparison to No Events) During the Study.
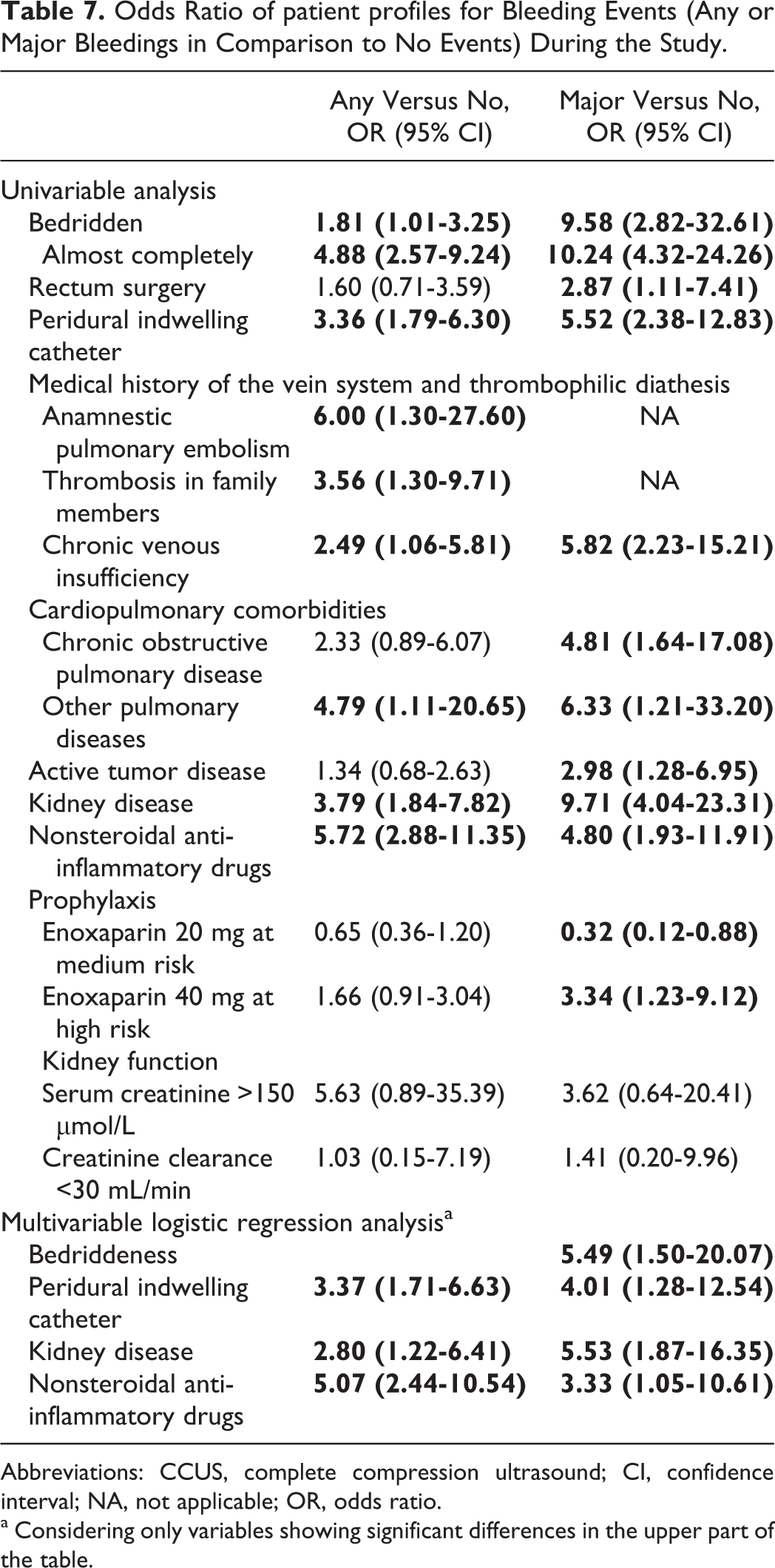
Abbreviations: CCUS, complete compression ultrasound; CI, confidence interval; NA, not applicable; OR, odds ratio.
a Considering only variables showing significant differences in the upper part of the table.
Discussion
Enoxaparin is highly effective in preventing VTE after surgical treatment. The present analysis of 448 patients that included a CCUS protocol to detect asymptomatic DVT identified no case of symptomatic or asymptomatic DVT and only 1 patient with PE during the treatment period. Four cases with symptomatic DVT (no case of PE) were observed during a 90-day follow-up. A total of 24 patients experienced major (5.4%) and 28 nonmajor bleeding complications (6.2%). Major bleeding was more frequent in those bedridden, with kidney disease, peridural indwelling catheters, and those using NSAID.
Patient Population and Incidence of Thromboembolic Events
Rates of VTE in patients undergoing general surgery without prophylaxis have been determined to be about 14.5%, (asymptomatic DVT) 0.5% for symptomatic PE, 0.9% for symptomatic VTE, and 0.9% for death in patients receiving placebo or no treatment. 15 The introduction of LMWH resulted in a risk reduction of VTE of up to 75% in these patients (relative risk [RR] 0.25-0.54). 15
The enoxaparin trials available for this indication show a large variability in the incidence of DVT ranging from 0% for symptomatic and 0.4% for asymptomatic DVT to as high as 1.3% (symptomatic) and 14.4% (asymptomatic).2-12,16 The broad range of event rates might be explained by a variety of disease conditions leading to surgery such as cancer in 100% of the Enoxaparin and Cancer (ENOXACAN) study patients which were having the highest risk and the longest exposure. 6 Pulmonary embolism risk is low with the highest rate reported by Gazzaniga at 0.5%. 4
The population in our analysis might be classified as having an intermediate risk because of 34% being almost completely bedridden, 25% having a medical history indicating increased risk, and 33% any type of cancer. Despite this, the risk of symptomatic DVT was low (0% during treatment and about 1% thereafter) and is compatible with the low-risk trials such as the ones by Samama9-11,16 (general surgery), Kaaja 7 (gynecology), and Gazzaniga 4 (general and vascular surgery). Risk was substantially lower compared to trials published by Nurmohamed 3 (general surgery), McLeod 12 (colorectal surgery), and the ENOXACAN 6 study (abdominopelvic surgery). We had only 1 patient experiencing PE during the treatment period (0.2%).
Predictors of Bleeding
In clinical trials, the benefits of LMWH have been shown to be accomplished at an acceptable risk of (major) bleeding.15,17 The risk of major hemorrhage is about doubled versus placebo (RR 2.03; 95% CI 1.37-3.01), accounting for an estimated incidence rate of 2.8%. 15 In our observation, the risk documented was 5.4% which is almost twice as high as compared to the aforementioned meta-analysis. 15 Positive predictors for major bleeding complications were bedriddeness (OR 5.49; 95% CI 1.50-20.07), peridural indwelling catheters (OR 4.01; 95% CI 1.28-12.54), kidney disease (OR 5.53; 95% CI 1.87-16.35), and the concomitant use of NSAIDs (OR 3.33; 95% CI 1.05-10.61). A 40-mg dose was predicting major bleeding complications only in univariable but not in multivariable analyses.
For enoxaparin, a dose reduction of up to 50% has been suggested for patients with renal insufficiency needing prophylaxis.1,18 This is of clinical importance because enoxaparin is widely used in patients with acute venous thrombosis and those at risk of developing VTE. Of those, approximately 50% have impaired renal function and a substantial fraction has severe renal insufficiency.19-21 Our results strengthen the recommendation that renal function, usually assessed via serum creatinine, has to be checked in patients requiring anticoagulation undergoing general surgery and doses need to be adjusted appropriately. Furthermore, NSAID use appears to be frequent in patients undergoing general surgery (about 10% of patients in our real-world setting). This is usually an avoidable concomitant medication that can easily be reduced for the limited period requiring anticoagulation.
Limitations
The CLEVER was a postauthorization survey in a real-world general surgery setting focussing on documenting actual treatment patterns. This implies a high external validity with direct relevance of the results for physicians treating these patients. 22 Compared to randomized controlled trials, a number of critical patients are included in noninterventional studies (NIS) that may have renal insufficiency, a high body weight, a number of coadministered platelet inhibitors or NSAIDs. On the other hand, these NIS are usually limited such as a lack of a control group and by the fact than unknown biases, for example, through patient selection, may have interfered with the results, making comparisons to other data more difficult. Further, the validity of CCUS has been questioned recently based on the finding of the VENography versus UltraSound (VENUS) validation study. 23 The VENUS results were surprisingly disappointing with an overall sensitivity of 31% (proximal < distal DVT) and a specificity of 93% in the patient-based comparison. However, the results seem to be characteristic for the orthopedic setting. By contrast, this strategy has been shown to be reliable in internal medicine, with trials like the Prospective Evaluation of Dalteparin Efficacy for Prevention of VTE in Immobilized Patients Trial (PREVENT), 24 the Extended Prophylaxis for Venous ThromboEmbolism (VTE) in Acutely Ill Medical Patients With Prolonged Immobilization (EXCLAIM) study,25,26 and the Apixaban Dosing to Optimize Protection from Thrombosis (ADOPT) study, 27 even in the setting of multicenter studies that tend to be prone to interoperator variability. 28
Conclusions
Enoxaparin is highly effective in patients to prevent VTE after surgical treatment. Clinical predictors may be used to identify patients at high risk for treatment-related complications. Particular caution appears warranted in patients with kidney disease and those receiving potentially avoidable concomitant medications such as NSAIDs.
Footnotes
Acknowledgment
The support of the participating physicians, their staff, and patients is acknowledged.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Sebastian Schellong, Albrecht Encke, Peter Bramlage, and Sylvia Haas report to have received compensation for consulting and lecture honoraria from Sanofi-Aventis Deutschland GmbH. W. Dieter Paar is an employee of Sanofi Aventis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
