Abstract
The aim was to evaluate the right ventricular function in patients with inherited thrombophilia and deep vein thrombosis (DVT) without pulmonary embolism. A total of 38 patients with DVT without symptomatic pulmonary embolism and 30 patients with varicose veins were enrolled. Clinical data, echocardiography, and 2 thrombophilic mutations were analyzed. Factor V Leiden (FVL) polymorphism was significantly frequent in the study group (
Keywords
Introduction
Venous thromboembolism (VTE) is a major cause of mortality and morbidity due to its severe acute and chronic complications. It has an annual incidence of 48 to 160 cases per 100 000 person-years (approximately 1 in 1000/annum) in general population. 1 –3 Venous thromboembolism is widely recognized to describe one clinical entity including deep vein thrombosis (DVT) and acute pulmonary embolism (PE). 4 Pulmonary embolism has been reported to occur in 40% to 80% of patients with DVT of lower extremities, and it is one of the most frequent causes of preventable deaths in hospitals. 5 –7
It has been reported that 34 – 55% of patients with proximal DVT without symptoms of PE have high-probability ventilation-perfusion lung scans, which is called silent PE. 4,8 –13 Similar rates have been observed with computed tomography scans and pulmonary angiography. 10,12 Only half of patients (53%) with high-probability lung scans have complaints related to PE. 11 These findings support the occurrence of asymptomatic PE episodes, especially, in patients with proximal DVT. 9,10,14 In addition to silent PE episodes, recurrent VTE may occur. The risk of recurrence after the first episode of VTE is 0.6% to 5% within 90 days, about 13% within 1 year, 20% to 30% within 5 years, and over 30% within 10 years. 15 –17 Recurrent PE episodes are one of the major causes of pulmonary hypertension and related permanent right ventricular dysfunction (RVD). 18
RVD occurs following a PE episode and persists in 20% to 25% of the patients at hospital discharge. 18 –20 The degree of RVD is closely related to the size and the localization of embolus, 21 and there is a strong correlation between the size of perfusion defects and right ventricular (RV) systolic function. 22 RVD evaluated by echocardiography is an important predictor of clinical outcomes in patients with PE. 18,23,24 Pulmonary embolism–related mortality was found to be 2-fold increased in the presence of RVD determined by echocardiography. 19 Also, RVD is a risk factor in clinically stable patients. 23
Inherited thrombophilia including factor V Leiden (FVL) and prothrombin polymorphisms are found to be associated with VTE. Polymorphism of FVL is the most frequent genetic variation held responsible for the occurrence of VTE. 25 –28 Heterozygous FVL mutation increases the VTE risk by 4- to 7-fold, whereas homozygous mutation increases this risk by 9- to 80-fold. 26,27 Combined FVL and prothrombin polymorphisms have a supraadditive risk and are related to a 20-fold increase in the VTE risk. 29
Known occurrences of asymptomatic PE episodes, recurrences, and inherited thrombophilias in patients with DVT without symptomatic PE persuaded us to examine whether these patients had RVD. Although the presence of permanent RVD as a result of symptomatic PE episodes has been well documented in the literature, there is no information regarding whether patients with DVT without clinically proven PE episodes have impaired RV function. The aims of this study were (i) to evaluate RV function and dimensions in patients who have proven DVT without symptoms of PE, and (ii) to compare RV function and dimensions in inherited thrombophilic polymorphisms detected in these patients.
Materials and Methods
The study was approved by Canakkale Onsekiz Mart University Local Ethics Committee (project number EK/2011-25) and conformed to the principles outlined in the Declaration of Helsinki. All patients participating in the study gave informed consent.
Among the patients who were diagnosed previously as having lower extremity DVT and followed up by Cardiovascular Surgery Department of Canakkale Onsekiz Mart University Medicine Faculty, 38 consecutive patients who were admitted to the control visit between May 2011 and October 2011, and have never had any symptoms of PE were enrolled into the study group. All patients were receiving a long-term oral anticoagulation therapy at the time of enrollment and did not have any exclusion criteria. In all patients, DVT was diagnosed by color Doppler ultrasonography (Logiq 9, GE Healthcare, Milwaukee, Wisconsin). Sonographic criteria for diagnosis were identification of hyperechoic thrombus in the veins, lack of compressibility of the vein, increased cross-sectional diameter of the vein, and lack of color Doppler signal. Lack of PE history was confirmed by clinical records. The patients who had any complaints related to PE occurence in their records were excluded. Also, 30 age- and gender-matched patients with varicose veins (the Clinico-Etiological-Anatomical and Pathological classification [CEAP] class C1 and C2) who did not have any venous pathology except junctional reflux detected by color Doppler ultrasonography and who were admitted during the same time period were enrolled into the control group. Exclusion criteria were (i) age over 75 years old, (ii) left ventricular ejection fraction (LVEF) below 50%, (iii) segmental wall motion abnormalities on echocardiogram, (iv) chronic obstructive lung disease, (v) asthma, (vi) congestive heart failure, (vii) malignancy, (viii) chronic renal failure, (ix) haematological disorder, (x) acute myocardial infarction, (xi) valvular heart disease, and (xii) pregnancy and puerperium.
Demographic (age, gender, height, weight, body surface area [BSA]) and clinical data (type, localization and duration of DVT, risk factors, treatments, recurrences, accompanying disorders) of the patients were recorded by the same cardiovascular surgeon. The surgeon was blinded to the results of echocardiographic and genetic evaluations until the study was over. Provoked DVT is defined as VTE that provoked by an antecedent or precipitating clinical risk factor including surgery, trauma, malignancy, immobility, pregnancy and the puerperium, hormone replacement therapy, or known thrombophilic abnormality (deficiency of antithrombin, protein C, or protein S, presence of the lupus anticoagulant, or presence of hyperhomocysteinemia), and unprovoked DVT is idiopathic VTE. 15,16 Proximal DVT denotes thrombosis of the popliteal vein or higher veins. 30
Echocardiography
Transthoracic echocardiography (GE Vivid 7 Dimension, GE Healthcare, Horten, Norway) was performed in all patients by the same cardiologist. The cardiologist was blinded to the group allocation and genetic test results of patients. Two-dimensional, M-mode and Doppler (spectral and colour flow) images in the left lateral decubitus position were obtained during end-expiratory hold using a 3.5-MHz transducer with electrocardiogram synchronization. Evaluation of these images were carried out in accordance with the recommendations of the American Society of Echocardiography. 31
Following parameters were obtained by echocardiography: (i) Chamber dimensions of right atrium (RA; major dimension, minor dimension, end-systolic area) (ii) right ventricle (RV; basal diameter, wall thickness, RV outflow tract [RVOT] parasternal short-axis [PSAX] distal diameter, RVOT parasternal long-axis [PLAX] proximal diameter, PSAX proximal diameter), (iii) chamber dimensions and volumes of left ventricle (LV; diastolic long-axis dimension, systolic long-axis dimension, diastolic diameter, systolic diameter, end-diastolic volume, end-systolic volume, interventricular septum thickness, posterior wall thickness), (iv) systolic functions of RV (tricuspid annular plane systolic excursion [TAPSE], fractional area change [FAC]) (v) LV systolic functions (LVEF, stroke volume), (vi) systolic pulmonary artery pressure (SPAP), and (vii) right/left ventricular end-diastolic diameter (RV/LV) ratio. The LVEF was calculated with the modified Simpson method in the apical 4-chamber view. The TAPSE that is a measure to evaluate global systolic functions of the RV calculated in M-mode images in the apical 4-chamber view. 32 The average of the 3 consecutive measurements was calculated for each variable.
Genetic Testing
Genetic analysis was performed in all patients by the same geneticist. Similarly, the geneticist was blinded to the group allocation and echocardiogram results of patients. Venous blood samples were taken from all participants. The samples were analyzed for FVL and prothrombin G20210A polymorphisms using the real-time polymerase chain reaction technique (LightCycler 2.0, Roche Diagnostics, Mannheim, Germany). DNA was isolated from K3 EDTA-peripheral blood samples with spin column method (Roche Diagnostics). Briefly, LightCycler FastStart DNA Master HybProbes, master mix (PCR-grade water, MgCl2, Primer-Probe Mix, 15 µL final volume) and DNA template (5 µL) were used for real-time amplification. Amplification conditions for 45 cycles were (i) initial denaturation at 95°C for 0 to 10 seconds, (ii) annealing at 55°C for 5 to 10 seconds, and (iii) extension at 72°C for 5 to 15 seconds; and melting conditions were denaturation at (i) 95°C for 20 to 60 seconds, (ii) 40°C for 20 to 120 seconds, (iii) 70°C to 85°C for 0.1 to 0.2 seconds; and a cooling step at 40°C for 30 seconds. Single quantification was performed for each annealing period, and continuous quantification for melting. The software program (Roche) was used for melting curve analysis and detection of genotype profiles (wild-type, heterozygous or homozygous mutant) of target genes.
Statistics
Clinical, echocardiographic, and genetic data stored in the separate databases were combined into a single database at the end of the study. Statistical analyses were performed using IBM SPSS Statistics version 19 (IBM Corporation, New York). Numerical data were summarized as the mean and standard deviation (mean ± SD), and categorical data were summarized as frequencies and percentages (n, %). The chi-square goodness-of-fit statistics was performed to test the significant deviations from Hardy-Weinberg Equilibrium. Group comparisons of numerical data were made by Mann-Whitney
Results
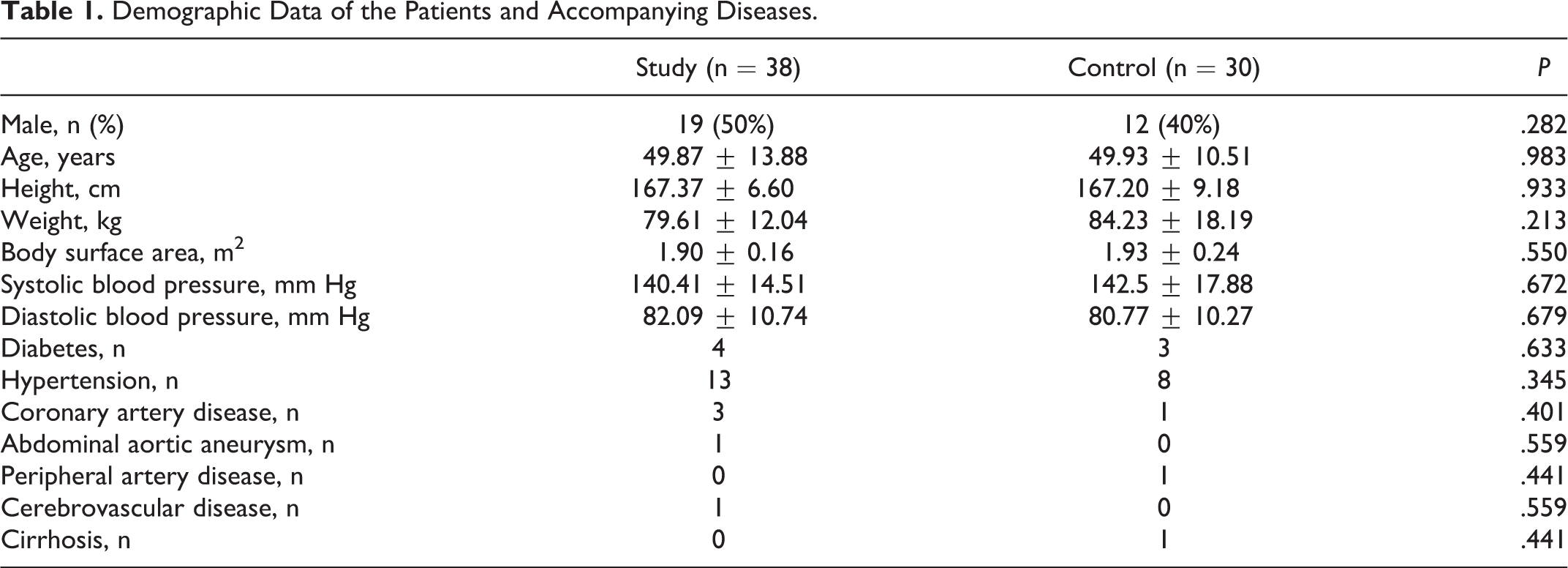
Clinical and demographic characteristics of patients were presented in Tables 1 and 2. The data of control and study groups were similar. The mean age was 49.9 ± 12.42 years (range 25-75 years), and the mean duration of DVT occurence was 68.33 ± 89.72 months (range 0.6-372 months).
Demographic Data of the Patients and Accompanying Diseases.
Clinical Data of the Patients With Deep Vein Thrombosis.
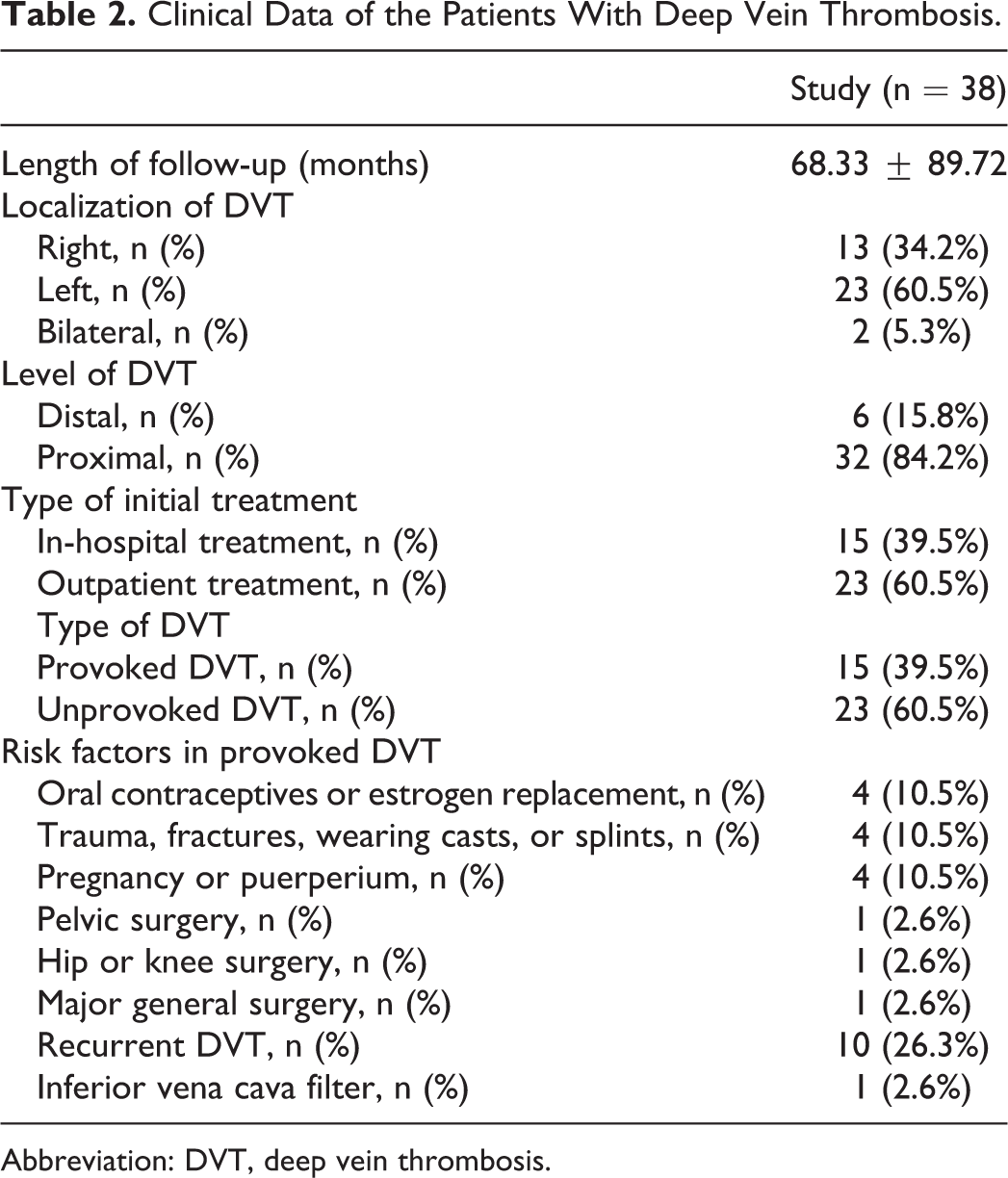
Abbreviation: DVT, deep vein thrombosis.
Results of echocardiographic examinations were presented in Table 3. The results were similar in the groups. There was no association and correlation between age, accompanying diseases, DVT characteristics (duration and level of DVT, presence of recurrence), and echocardiographic measurements.
Results of Echocardiogram in the Groups.
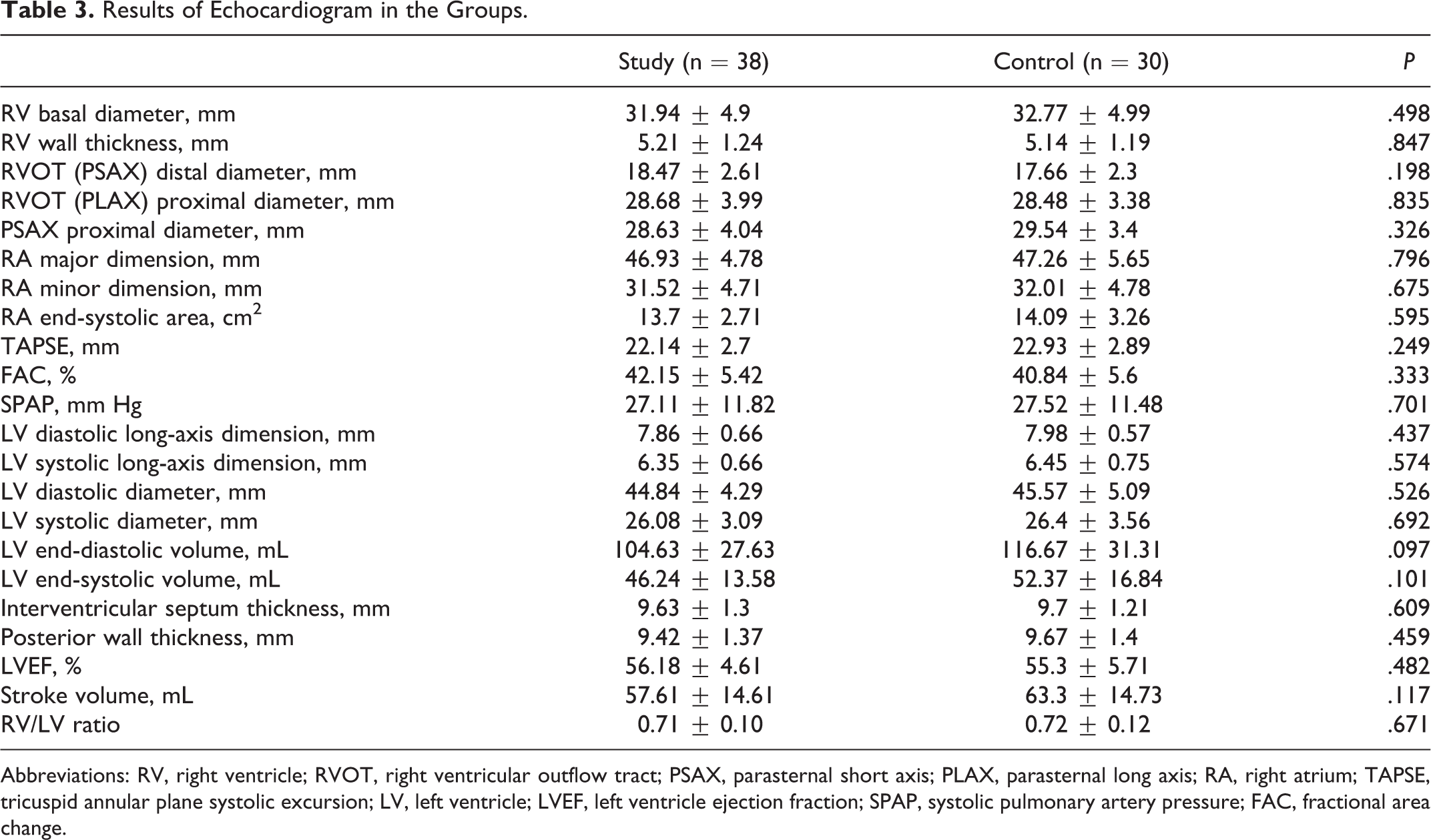
Abbreviations: RV, right ventricle; RVOT, right ventricular outflow tract; PSAX, parasternal short axis; PLAX, parasternal long axis; RA, right atrium; TAPSE, tricuspid annular plane systolic excursion; LV, left ventricle; LVEF, left ventricle ejection fraction; SPAP, systolic pulmonary artery pressure; FAC, fractional area change.
Results of genetic test were presented in Table 4. Genotype frequencies of each polymorphism conformed to the distributions predicted by the Hardy-Weinberg equilibrium (FVL
Frequencies and ORs of Mutant Alleles (Either Heterozygous or Homozygous) and Frequencies of Genotypes in the Groups.
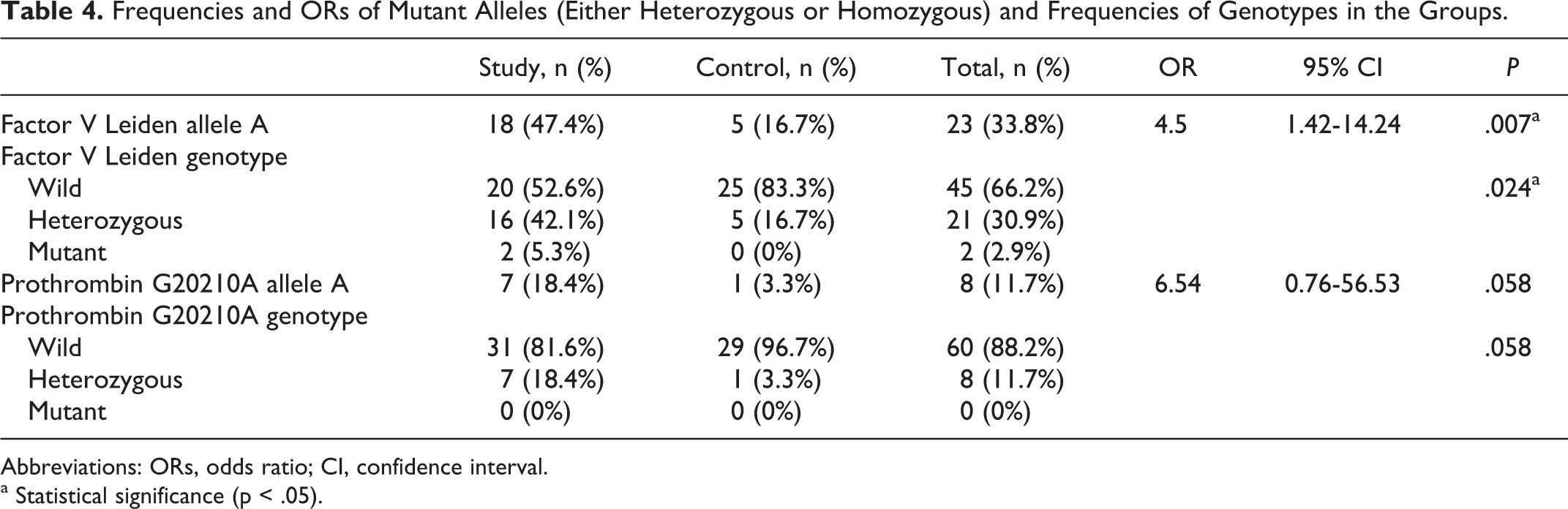
Abbreviations: ORs, odds ratio; CI, confidence interval.
a Statistical significance (p < .05).
The comparison between echocardiographic measures and thrombophilic polymorphisms revealed that there was statistically significant decrease in TAPSE values in patients with FVL and prothrombin G20210A polymorphism (TAPSE in FVL polymorphism 23.06 ± 2.62 vs 21.37 ± 2.84 mm,
Results of Echocardiographic Measurements of the Right Heart in Factor V Leiden and Prothrombin G20210A Mutations.
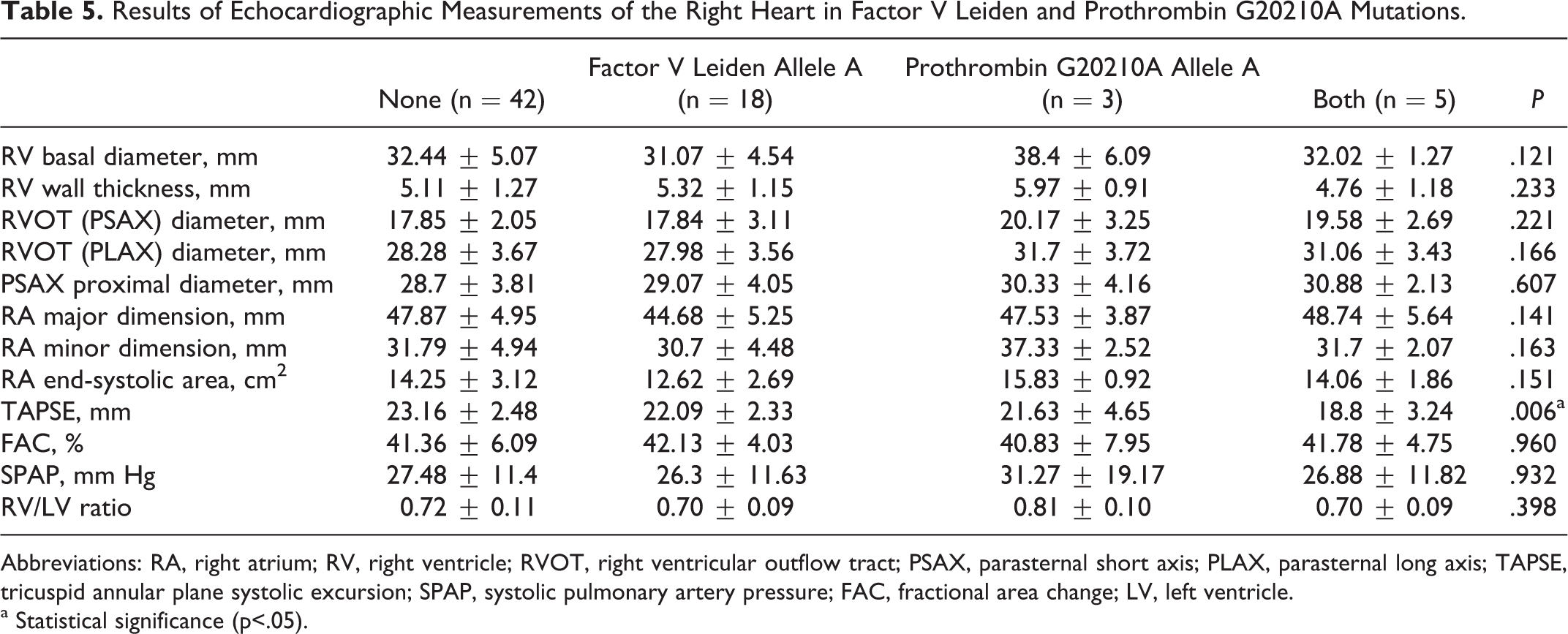
Abbreviations: RA, right atrium; RV, right ventricle; RVOT, right ventricular outflow tract; PSAX, parasternal short axis; PLAX, parasternal long axis; TAPSE, tricuspid annular plane systolic excursion; SPAP, systolic pulmonary artery pressure; FAC, fractional area change; LV, left ventricle.
a Statistical significance (p<.05).
Discussion
The main results of the study were that (i) the presence of DVT without PE was not related to impaired echocardiographic measures of RV function and dimensions, and (ii) the presence of FVL and prothrombin G20210A polymorphisms were related to moderate impairment in RV systolic function independent of the presence of DVT.
In the literature, echocardiography parameters widely used to assess RV dysfunction are RV dilatation, hypokinesis, RV/LV ratio, and tricuspid regurgitant jet velocity. 19,33 But especially FAC, TAPSE, RV index of myocardial performance, and tricuspid lateral annular systolic velocity are recommended in the assessment of RV systolic function by the guideline of the American Society of Echocardiography. 32 Although TAPSE is a measure of RV longitudinal function, it is well correlated with techniques estimating RV global systolic function. 32 In this study, we preferred TAPSE and FAC to assess RV systolic function. To detect the presence of any left-sided factor affecting RV function and dimensios, measures indicating LV function, LV dimensions, and the function of heart valves were assessed echocardiographically.
The RV dysfunction following a symptomatic PE episode has been reported in the previous studies. 18 –20 The hemodynamic changes associated with RV overload, pulmonary vasoconstriction, platelet activation, and persistent myocardial injury are the mechanisms of RVD occurrence following PE. 18,21 Also, it was shown that silent PE may lead to pulmonary hypertension. 13 It would be expected that these frequent asymptomatic PE episodes and recurrences occurred in patients with DVT may cause RV dysfunction, but the difference in RV function and dimensions between the control and study groups was statistically insignificant. The results indicated that DVT not complicated by PE did not cause an impairment in RV function. Although we were not able to clearly demonstrate the presence of embolization in these patients, this result was mainly attributed to the size of embolus originated from DVT. Episodes of PE caused by only a small amount of thrombus which is not enough to cause clinical symptoms were assumed not to cause an impairment in RV function. It was known that there is a relationship between RVD and the size of perfusion defects on lung scans. 21 Small-sized embolus occluding only a limited segment of pulmonary vascular bed is not sufficient to produce RV overload and associated myocardial injury. To establish a direct and definitive relationship between the size of embolus and RV function, additional diagnostic methods such as lung scan should be performed in these patients.
Mean age of the patients enrolled into the study was lower than that reported in the literature (54-63 years). 3,15,16 This low mean age was associated with the exclusion criteria. Also, the mean duration of DVT in our patients was 5.7 years. Younger age and the short duration of DVT may be related to preserved RV function. Although, as mentioned earlier, there is a relationship between the degree of RVD and the size of perfusion defect, each PE episode regardless of the size of embolus plugging a portion of pulmonary circulation may cause some degree of myocardial damage and a subclinic reduction in RV function. Recurrent PE causes repeated cycles of myocardial damage and healing, but a certain amount of residual myocardial damage may remain in each cycle. The cumulative effect of this residual damage may cause clinically evident RVD in the course of time. It can be assumed that the short period of having DVT is not enough to cause such accumulation of myocardial damage. But we did not find any relationship between the duration of DVT and RV function, and there was no evidence to suggest that the longer duration of DVT is related to worse RV function. Additionally, although this theory associates recurrences to RVD, we did not observe a relationship between them.
Thrombophilic mutations, especially FVL polymorphism, are regarded as an evolutionary advantage reducing the risk of bleeding. 34 However, again, especially FVL polymorphism is held responsible for an increased VTE risk. The prevalence of FVL polymorphism has been reported as 1.2% to 7.8% in the general population and 11.4% to 22.7% in the patients with VTE. 25,27,28,35 In the studies from Turkey, a higher prevalence of FVL polymorphism has been reported as 3.1% to 15% in patients without DVT and 21% to 34.2% in patients with DVT. 26,36 –38 But in this study, it was 16.7% in patients without DVT and 47.4% in patients with DVT. Considerable variations in the prevalence of FVL in the general population have been reported in different ethnic groups. For example, the prevalence of FVL among Turkish Cypriots was reported as 12.2% versus 8.0% in Greek Cypriots who lives together with Turkish Cypriots on the same island for centuries. 39 Although it was reported that the FVL prevalence is 7.9% in the highly cosmopolitan cities of Turkey, which represents a mean prevalence among various ethnic groups, the prevalence among healthy individuals in the northeastern region of Turkey (Black Sea region) was 15% in the report of Agaoglu et al. 38 It was 16.7% in our study. The city where the study was conducted is one of the most western cities in the country and has a relatively homogeneous population due to low immigration rates from the east and middle part of the country. This ethnical characteristic of the region may be responsible for the higher prevalence of FVL than the country mean. Furthermore, the participants enrolled into the control group did actually not represent the general population because they were a refined group due to the exclusion criteria and had lower extremity varicose veins. Darvall et al reported a higher prevalence of thrombophilia in patients with venous diseases including varicose veins, venous insufficiency, and ulceration although the results for FVL in these patients were inconclusive. 40 Similarly, MacKenzie et al reported a higher thrombophilia prevalence in chronic venous ulceration. 41 The higher prevalence of FVL in the control group may also be influenced by the presence of varicose veins.
On the other hand, the highest prevalence in patients with DVT was attributed to the FVL paradox. The FVL paradox can be defined briefly as the lower prevelance of FVL in patients with PE alone than that in patients with DVT alone. 27,42,43 Such a characteristic has not been observed in other thrombophilic polymorphisms. 42,44 Two clinical circumstances are discussed under this topic: (i) FVL polymorphism increases the risk of a first DVT but not the risk of PE and (ii) FVL polymorphism significantly increases the risk of a first DVT but not the risk of recurrent DVT. 34 Because the patients with PE, who had a lower prevalence of FVL polymorphism, were excluded from the study, as expected, the highest prevalence of FVL polymorphism was found in the remaining patients presented with DVT alone. The prevalence of prothrombin G20210A polymorphism has been reported as 0.6% to 4% in various general population and 3.8% to 16% in patients with VTE. 25,28,36 Its prevalence in our study was 3.3% in patients without DVT and 18.4% in patients with DVT.
Reduced RV systolic function represented by TAPSE was observed in patients with FVL and prothrombin G20210A polymorphisms. This reduction was more evident in patients with combined FVL and prothrombin G20210A polymorphisms. This finding has not been reported previously. Although this impairment in RV systolic function was found to be independent of the presence, type, and localization of DVT, the relationship between thrombophilic polymorphisms and reduced RV systolic function was attributed to a thromboembolic mechanism. The observation that the impairment in RV function occurred only in thrombosis-related polymorphisms, and other polymorphisms not related to DVT occurrence had also preserved RV function supports this hypothesis. It was concluded that although patients with FVL and prothrombin G20210A polymorphisms do not have a symptomatic lower extremity DVT, recurrent and asymptomatic thromboembolism episodes may originate from some other part of the venous network or from small crural veins. Lifelong microembolism episodes in patients with these thrombophilic polymorphisms may be responsible for reduced RV function. It seems contradictory to discuss the likelihood that the recurrent microembolic events impair RV function in patients with thrombophilia while it was not accepted as a predominant mechanism in DVT alone. But it was not concluded in DVT, because symptomatic DVT persists for a relatively short while, at least not for a lifetime, and involves only major veins of the lower extremities, which are likely to produce larger emboli causing excluded symptomatic PE. On the contrary, thromboembolic events occur throughout the lifetime of patients with thrombophilia and involve the entire venous network of the body. As stated earlier, each PE episode might be related to some degree of residual myocardial damage. These patients have enough time for the accumulation of this residual damage reducing RV function in the course of time. Additionally, it was reported that FVL may enhance the local thrombin generation, augmenting the local inflamation, and inhibit fibrinolytic response. These processes may be related to thromboembolic events and impaired RV function. The mechanism involving recurrent microembolism was held responsible for other clinical situations. In a study of Juul and collegues, 45 homozygous FVL polymorphism was found to be associated with severe dyspnea and decreased pulmonary function. The presence of homozygous FVL polymorphism has increased the severe dyspnea risk by 5-fold. This reduced pulmonary function among FVL homozygotes was explained by the occurrence of recurrent, small, and clinically unrecognized PE episodes. The authors did not evaluate cardiac function of the patients, and we did not explore pulmonary function of our patients. But there can be a connection between pulmonary and cardiac dysfunction.
The major limitation of the study was that lung scans which is capable of revealing the presence of asymptomatic PE episodes, and, if there is, the size and the location of perfusion defects were not performed in our patients due to ethical issues. Lung scans could provide more accurate assesment of the relationship between thrombophilic polymorphisms, silent PE, and RV function. Other important limitations of our study were small sample size (especially in prothrombin G20210A and homozygous FVL polymorphisms) and the absence of data about homocysteine levels, antithrombin III deficiency, and protein C and S deficiencies.
In conclusion, (i) RV function and size are preserved in patients with DVT without symptomatic PE and (ii) a moderate and nonpathologic reduction in RV systolic function occurs in patients with FVL and prothrombin G20210A polymorphisms independent of the presence of DVT. It is thought that recurrent asymptomatic venous thrombosis and PE episodes are responsible from this impairment. Echocardiographic evaluation of the RV is advisable to detect and to treat possible RVD in these patients, especially with combined polymorphisms, irrespective of whether they have DVT.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
