Abstract
Immune thrombocytopenia (ITP) is an autoimmune disease in which platelets are destroyed by special antiplatelet autoantibodies produced by B cells. Dendritic cells (DCs) are professional antigen-presenting cells involved in humoral immunity and cellular immunity and among them DCs that induce autoimmune tolerance are called tolerogenic DCs (tDCs). As a promising immunotherapeutic strategy for ITP, tDCs have received increasing attention. In this review, we describe the significant role of DCs in regulating autoimmune balances, introduce the manipulation strategies to generate tDCs, summarize recent progress on the experimental application of tDCs for ITP therapy, and finally discuss the perspectives of tolerogenic vaccination for ITP treatment in the clinic.
Background
Immune thrombocytopenia (ITP) is an autoimmune disease characterized by increased destruction of the platelets. It is generally accepted that ITP is mainly caused by the production of antibodies against the antigens on the membranes of platelets. 1 However, recent evidence suggests that the pathogenesis of ITP is complicated. 2 The immune disorders in the patients with ITP not only refer to autoantigen-mediated decrease in the platelets but also involve other processes such as the abnormality of T and B lymphocytes, cytotoxic T lymphocyte (CTL)-mediated destruction of the platelets, and the dysfunction of megalokaryocytes. Notably, these processes are closely related to the function of dendritic cells (DCs) and imply new opportunities for ITP therapy. 3
Immune thrombocytopenia affects not only the adults but also the children in the developed countries where it is commonly encountered in clinical practice. 4 Immune thrombocytopenia is currently treated with immunosuppressive drugs and biological agents such as rituximab 5 and CD44 antibody. 6 However, these therapeutic agents may induce a generalized immune suppression that increases the risk of infectious diseases and cancers.7,8 Therefore, new therapeutic approaches should aim at the suppression of inflammation and reestablishment of tolerance toward platelet antigens without compromising the patient’s immune system. 7 Recent progress on the investigations of immune modulation has shown that a particular subset of DCs could modulate immune responses. 9 These DCs play a key role in maintaining both central and peripheral autoimmune tolerance, and the constitutive ablation of DCs destroys self-tolerance, resulting in spontaneous fatal autoimmunity.10–16 The DCs that induce autoimmune tolerance are called tolerogenic DCs (tDCs). As a promising immunotherapeutic strategy for autoimmune disorders, tDCs have received considerable attention.15,17–19 In this review, we first describe the significant role of tDCs in autoimmune regulation and then introduce the manipulation strategies to generate tDCs and summarize recent progress in the experimental application of tDCs for ITP therapy. Finally, we discuss the perspectives of tolerogenic vaccination for ITP treatment in the clinic.
The Role of DCs in Autoimmune Balance
Subsets of DCs
Human peripheral blood DCs can be classified into 2 major subsets according to the source they are derived from: myeloid DCs (mDCs) and plasmacytoid DCs (pDCs).20–24 In addition to these 2 subsets, the DCs derived from monocytes are called monocyte-derived DCs (mo-DCs) which play important role in innate and adaptive immunity.25–27 Bone marrow–derived murine DCs can be divided into 3 subpopulations based on their lineages during the development: classic DCs (cDCs), pDCs, and tumor necrosis factor and inducible oxide synthase–producing DCs (TipDCs).27,28 In the bone marrow, the DC precursors give rise to pDCs that can travel through the blood to peripheral destinations to differentiate into cDCs. 27 Under inflammatory conditions, the monocytes can differentiate into TipDCs. Thus, the classification of DCs is similar in different species including human. It has been reported that in patients with chronic ITP both mDCs and pDCs numbers were comparable to that of healthy controls, but the percentage of CD4(+) Foxp3(+) regulatory T (Treg) cells was significantly reduced compared with healthy controls. 29
Activation of Antigen-Specific Immune Responses
The important role of DCs in immunity was first recognized by Steinman et al. 30 The DCs can induce the differentiation of naive T cells into T-helper (Th) cells to regulate the functions of other T cells or B cells, into cytotoxic T cells to eliminate the infected cells, or further into memory T cells to defend the body from future infection by the same source by inducing the expression of adhesion molecules and cytokine receptors as well as the production of cytokines, which can activate autoimmune responses (Figure 1 ).31–33 Moreover, the DCs can secrete cytokines such as interleukin 12 (IL-12) 34 and interferon-α (IFN-α), 35 which can influence the differentiation of T-cells into antigen-specific Th1, Th2, Th17 36 or regulator T cells. 18 Although the B cells and macrophages also present the antigens, the DCs are the most efficient and professional antigen-presenting cells. Moreover, the decrease in circulating DCs is associated with increased platelets in the circulation when patients with ITP were treated with prednisone, 1 which suggests that DCs activate antigen-specific immune responses in ITP.
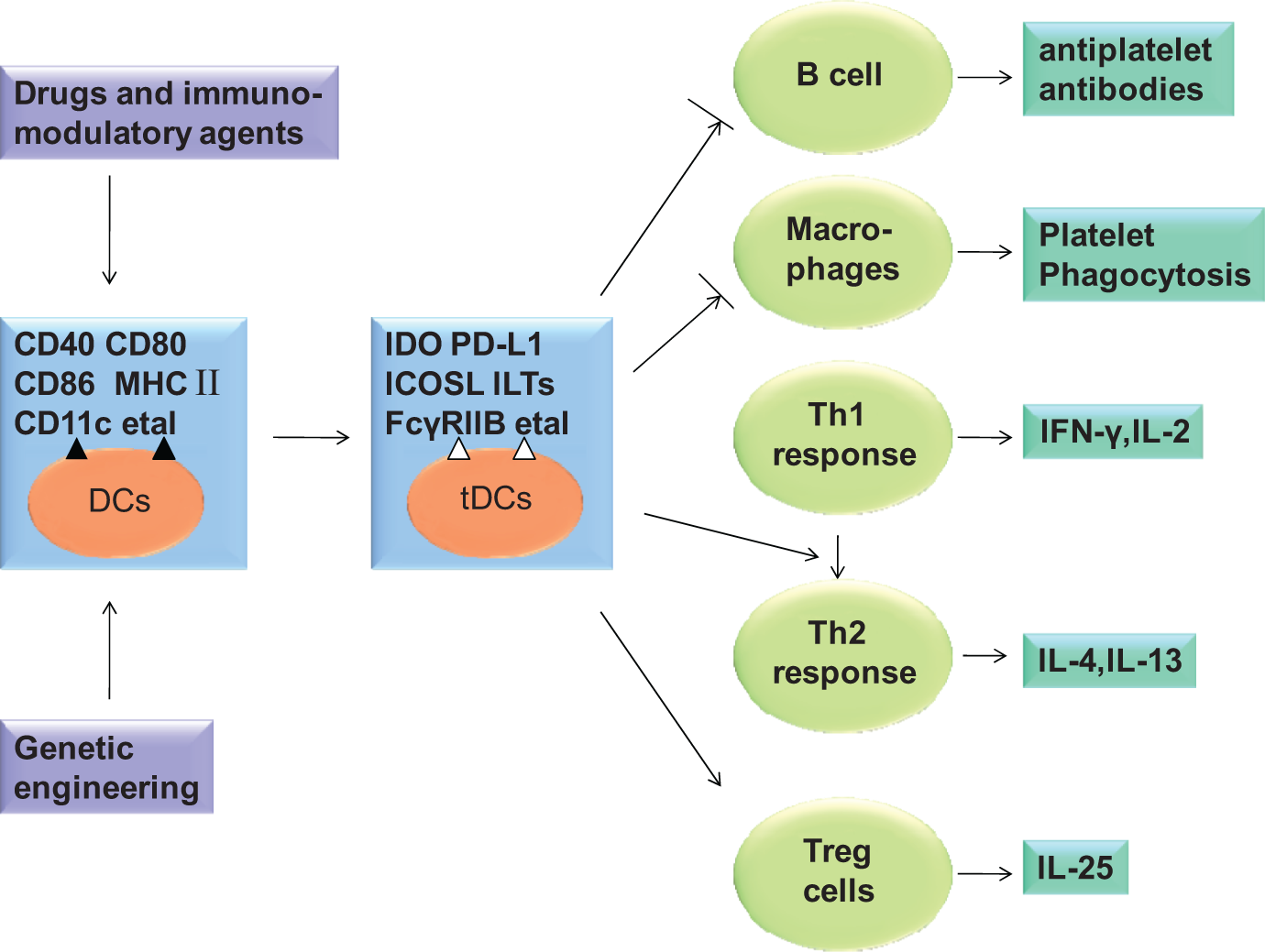
Scheme showing the manipulation strategies to generate tDCs and potential mechanisms by which tDCs promote autoimmune tolerance. Drugs and immunomodulatory agents which target the inhibition of costimulatory molecules such as CD40, CD80, and CD86 and upregulation of inhibitory molecules including IDO, PD-L1, ILTs, and ICOSL can induce tolerogenic phenotype of DCs. tDCs with low expression of MHC II molecule and costimulatory molecules fail to process antigen to B cells with decreased antiplatelet autoantibodies. tDCs with FcγRIIB promote the expression of FcγRIIB on phagocytes, thereby ameliorating the inflammatory response and platelet clearance in ITP. tDCs induce special differentiation of naïve T cells, which cause tolerance of autoimmunity and prevent the development of ITP.
Genetic engineering generate tDCs by suppressing the expression of costimulatory molecules (CD40, CD80, and CD86) and promoting the expression of inhibitory proteins (eg, IDO).
Induction of Antigen-Specific Tolerance
The DCs play an important role in immune tolerance. The nonresponse to the challenge with specific antigen can be generated in the thymus (central) or peripheral tissues. The DCs in the thymus principally generate central immune tolerance.37,38 The central immune tolerance is achieved by the deletion of potentially self-reactive antigen-specific T cells 39 as well as the generation of Treg cells.40,41 Both cDCs and pDCs resident in the human thymus, when activated with CD40 ligand (CD40L) plus IL-3, efficiently induced the differentiation of autologous thymocytes into CD4(+) CD25(+) Foxp3(+) Tregs.42,43 On the other hand, several mechanisms are responsible for the induction of peripheral tolerance by DCs, including the generation and expansion of Treg cells, T-cell deletion, or the induction of anergy.44–46 It is shown that tDCs are essential for the development of CD4+ CD25+ Foxp3+ Treg cells both in vitro and in vivo. 47
Modulation of the Functions of Other Cells
It is well known that DCs can modulate the functions of other immune cells, such as B cells and natural killer (NK) cells. The DCs are equipped with both nondegradative and degradative antigen uptake pathways to facilitate antigen presentation to B cells.48,49 The NK cells are innate immune cells that have the ability to modulate the adaptive immune system through cytokine production or direct cell–cell interaction. The DCs can activate NK cells by NK cell-activating receptors, NKp46. 50 When the pDCs were injected into C57BL/6 mice, they efficiently recruited the NK cells. 51 In addition, DCs are required for myeloid homeostasis. Depletion of murine DCs leads to systemic myeloid expansion in the mice. 52
Strategies for the Generation of tDCs
Given the essential role of DCs in autoimmune balance, especially in tolerance maintenance, it is promising to generate DCs with tolerogenic properties for the therapy of specific autoimmune diseases. The commonly used strategies for the generation of tDCs include inhibiting the activation of DCs, disrupting primarily costimulatory pathways, or inducing overexpression of inhibitory molecules.
Recently, various immunomodulatory agents and pharmacological agents have been tested to confer tolerogenic properties on DCs and regarded as clinically applicable options.53–56 For instance, primary and mo-DCs in hyper-IgE syndrome (HIES) showed defective responses to IL-10 and thus failed to become tolerogenic. When treated with IL-10, patients' DCs showed an impaired upregulation of inhibitory molecules on their surfaces, including programmed death 1 ligand (PD-L1) and immunoglobulin-like transcript (ILT-4). Moreover, IL-10-treated DCs from the patients displayed impaired ability to induce the differentiation of naive CD4+T cells to Treg cells. 57
The activation of nuclear factor (NF)-κB is crucial for DC maturation. 58 Thus, the blockade of NF-κB by certain drugs (eg, andrographolide or rosiglitazone) is a new approach to enhance the tolerogenic capacity of DCs. 59 Other inhibitory molecules include ILT-2, ILT-3, ILT-4, and ILT-6 in the ILT family, 60 indoleamine 2,3-dioxygenase (IDO),61,62 and inducible costimulator ligand (ICOSL). 15 On the contrary, it has been shown that the stimulation of DCs through the engagement of P-selectin glycoprotein ligand 1 (PSGL-1) with P-selectin enhanced their capabilities to generate Treg cells, thus contributing to the tolerogenic functions of DCs. 63 Similarly, the activation of E-cadherin-/β-catenin-signaling pathway promoted DC maturation without the induction of inflammatory cytokines, and generated tDCs that provide protection against autoimmune diseases in experimental autoimmune encephalomyelitis (EAE). 64
Moreover, genetic engineering of DCs with enhanced/stabilized tolerogenic potential via the expression of anti-inflammatory modulators such as IL-10, transforming growth factor (TGF)-β, IL-4, and IDO emerges as a novel strategy.65,66 The primary techniques employed include the knockdown of costimulatory factors such as CD40, CD80, and CD86, and the expression of immunosuppressive molecules in DCs.67–69
The Implication of tDCs in ITP
Emergence of Antiplatelet Autoantibodies
The emergence of antiplatelet autoantibodies begins with a failure of the immunological tolerance to self-antigens. The etiology for this failure of tolerance in ITP remains unclear. However, DCs were shown to take up and process platelet proteins by major histocompatibility complex (MHC) II molecule and promote the production of autoantibodies by B cells. 70 Moreover, it was demonstrated that the activated mo-DCs from patients with ITP could directly increase B-cell effector function. 71
The tDCs induced by several strategies described above express low levels of MHC II molecule and costimulatory molecules and exhibit potent immunoregulatory abilities in autoimmune diseases. 72 Indeed, the tDCs have been utilized for the treatment of autoimmune diseases such as rheumatoid arthritis (RA), psoriasis, multiple sclerosis, type 1 diabetes, and systemic lupus erythematosus.73,74 Thus, tDCs should have the potential for ITP treatment.
Platelet Phagocytosis
The pathogenesis of ITP is known to involve autoantibody-mediated phagocytosis of platelets. There are several Fc receptors that bind IgG specifically to the surface of human macrophages. 75 Receptor for the Fc fragments of immunoglobulin G IIA (FcgRIIA) on the surface of human macrophages binds to anti-glycoprotein (GP) IIb/IIIa IgG antibody that has been coated with platelets, leading to the engulfment of the platelets. However, FcγRIIB promotes inhibitory signaling upon binding to the Fc portion of an immune complex and is implicated in ITP.76,77 Interestingly, intravenous immunoglobulin which induces the production of tDCs ameliorates ITP via activating FcγRIIB on DCs. 78 In addition, tDCs modulate the immunosuppression in an adoptive cell transfer model and promote the expression of FcγRIIB on phagocytes, thereby ameliorating the inflammatory response and platelet clearance in ITP. 79
Abnormality of T Cells in ITP
Chronic ITP is prone to a predominant Th1-type immune response characterized by the production of IL-2 and IFN-γ.80,81 In this condition, there is an increased secretion of Th1-type cytokines (IL-2 and IFN-γ) and the levels of Th2-type cytokines (IL-4 and IL-5) decreased, Furthermore, the Th1/Th2 ratio increases compared to the control. 82 DNA microarray screening revealed that the expression of genes involved in the Th1 immune response increased in patients with ITP. 83 Further studies showed that the DCs play a critical role in inducing Th1 immune responses.84,85 In addition, the prophylactic treatment with tDCs is associated with a reduced specific IgG2a/IgG1 ratio, indicating a switch from a Th1- toward a Th2-driven specific immune response to inhibit autoimmune disorders in experimental arthritis models, which is also prone to Th1-type immune response.86,87 These results suggest that the tDCs have a potential therapeutic effect for ITP, a Th1-type autoimmune disease.
It has been well established that Treg cells contribute to the long-term unresponsiveness of T effector cells, resulting in the suppression of autoimmune response.88–90 In patients with chronic ITP, the number of CD4+ Foxp3+ Treg cells was significantly decreased before the treatment with high-dose dexamethasome. 29 Additional study showed that the percentage of Treg cells was significantly decreased in patients with ITP in active state and the suppressive activity of Treg cells was impaired. 91 Based on these data, we speculate that the failure of immune tolerance mediated by the deficiency of Treg cells may also play a role in the pathogenesis of chronic ITP.
The tDCs can act as the inducer of Treg cells in human84,92 or mouse.93,94 Moreover, pDCs express tolerogenic phenotype and induce Treg cell through the expression of IDO.61,62,68,95 In addition, the tolerogenic potential of semi-mature DCs in experimental autoimmune thyroiditis (EAT) is mediated through the selective activation of CD4(+) CD25(+) Treg cells,96–98 which provides new insights into the regulatory mechanisms underlying the immunoregulation in autoimmune diseases.
Perspectives of tDCs in ITP Treatment
Recent studies have shown that the circulating mature DCs reverted to their immature state after HD-DXM 29 or intravenous immunoglobulin 78 treatment in ITP. Self-antigen recognition by T cells presented on immature or intermediary DCs in the absence of inflammatory stimulus induces anergy and/or Tregs. The tDCs have been proven effective in the therapy of autoimmune diseases such as systemic lupus erythematosus and RA.99–101 Thus, the tDCs induced via drugs or other agents might have therapeutic effects on ITP.
Recent progress on the understanding of the role of tDCs in the suppression of autoimmune diseases has contributed greatly to the discovery of novel therapeutic targets. The application of tDCs-based cellular vaccine for tumor, infection, and some autoimmune diseases has been well reviewed.102–106 Further evaluation of the therapeutic effects of tDCs in ITP should pave the way for the application of tDCs in ITP treatment in the clinic.
Footnotes
Acknowledgments
The authors wish to thank Dr Ning Bin and Zhao Kai for their critical reading of the manuscript. This work was supported by the grants from the National Natural Science Foundation of China (No. 30801258 M.H.) and the Star of Jinan Youth Science and Technology Project (No. 20100114).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
