Abstract
Background:
Monitoring bypassing agent therapy and observing concordance with clinical hemostasis is crucial in vital hemorrhages and major surgeries in patients with hemophilia having inhibitor.
Objective:
We aimed to investigate the value of the thrombin generation assay (TGA) and thromboelastography (TEG) for monitoring hemostasis in patients with hemophilia having inhibitor, during supplementation therapy with bypassing agents.
Patients and Methods:
The study group consisted of 7 patients with hemophilia having factor VIII inhibitor. All patients were male. The median age of the participants was 10 years. Age range was 6 to 32 years. The median inhibitor level was 10 Bethesda units (BU), with a range of 5 to 32 BU. A total of 17 bleeding episodes were evaluated. Both TEG and TGA tests were assessed in addition to clinical responses. Assessments were made prior to bypass agent therapy such as recombinant factor VIIa (rFVIIa) or activated prothrombin complex concentrate (aPCC) for bleeding episodes, during the first hour and 24 hours after either intervention in patients.
Results:
No relation between clinical response and TGA or TEG parameters was found in patients. There was no difference between clinical responses after rFVIIa and aPCC treatments. However, after aPCC treatment, endogenous thrombin potential and peak thrombin levels and also TEG R, K, and alpha angle degrees were significantly higher.
Conclusions:
In conclusion, we found that the clinical effectiveness of bypass therapy in hemophilia cannot be assessed by TGA and TEG.
Introduction
Development of inhibitors against factor VIII (FVIII) or factor IX (FIX) is one of the major complications to replacement therapy in hemophilia. Inhibitory antibodies to FVIII occur approximately in 15% of patients with hemophilia A following FVIII infusion. 1 The treatment of bleeding for patients with hemophilia having inhibitors relies on the use of the bypassing agents, recombinant factor VIIa (rFVIIa, Novoseven; Novo Nordisk, Bagsvaerd, Denmark), and factor VIII inhibitor bypass activity (FEIBA; Baxter, Glendale, California). These agents are not as effective as native FVIII or FIX in controlling bleeding and importantly, there is no accepted method to monitor the clinical effectiveness. 2
Recently, there have been several attempts to measure overall hemostasis, instead of assessing isolated factor activities, to understand the complex processes by which hemostatic components interact in various bleeding and thrombotic disorders. 3 –5 General clotting tests such as prothrombin time (PT) and activated partial thromboplastin time (APTT) 6 do not reflect overall thrombin generation and are insensitive to hypercoagulation and possibly also hypocoagulation. 4 Thromboelastography (TEG) analyzes whole blood and thus measures the effect of both cellular and plasmatic components on clot formation. The properties of clot formation were assessed by TEG. 3 Whereas PT and APTT do not assess the whole coagulation system. 4 Several authors have suggested that factor dose adjustment and hemostasis follow-up with TEG in major hemorrhages in patients with hemophilia having inhibitor. 7,8
The thrombin generation assay (TGA) measures the actual thrombin concentrations before and after the clot is formed and is very sensitive to variations in 1 or more coagulation factors. 3 In major hemorrhages and operations in patients with hemophilia in particular, it may be possible to calculate the required factor dose and duration of factor replacement therapy by information obtained from the TGA. 3,9,10 Thus, high cost, thrombotic complications, and unnecessary factor use may be prevented in some patients. Also, by determining the rational patient dose, bleeding due to inadequate medication is decreased.
Monitoring bypassing agent therapy and observing concordance with clinical hemostasis is crucial in all major hemorrhages and surgeries in patients with hemophilia having inhibitor. 4 In this study, we aimed to investigate the value of TGA and TEG in monitoring hemostasis during bypassing agent therapy in patients with hemophilia having inhibitor.
Patients and Methods
Ethics committee approval was obtained prior to the start of the study from the Clinical Research Ethics Committee of the Ege University School of Medicine. Patients and parents were well informed of the study requirements.
The study group consisted of 7 patients with hemophilia having FVIII inhibitor. All patients were male. The median age of the participants was 10 years, with a range of 6 to 32 years. The median inhibitor level was 10 Bethesda units (BU), with a range of 5 to 32 BU. Platelet counts of the patients were within normal range.
A total of 17 bleeding episodes were evaluated. Acute bleeding episodes were classified as acute hemarthrosis (N = 10), soft tissue hemorrhages (N = 1), oral cavity hemorrhages (N = 2), or gastrointestinal hemorrhages (N = 4). The median of bleeding episode was 3 for each patient, with a range from 1 to 4 bleeding episodes. Six of the bleeding episodes were treated with rFVIIa and 11 were treated with activated prothrombin complex concentrate (aPCC).
Both TEG and TGA tests were conducted before and after bypass agent therapy during the bleeding episode of patients with hemophilia having inhibitor. Correlations among these tests were evaluated and compared with clinical responses. The results of the basal TGA parameters of the patients were compared with other results obtained from 37 age- and sex-matched healthy volunteers.
Factor Dosages Used in Bleeding Episodes
Recombinant activated factor VII ([rFVIIa]; NovoSeven/NovoNordisk, Denmark) and aPCC (FEIBA/Baxter) therapies were administered at 80 to 115 mcg/kg per dose and 50 to 100 IU/kg per dose, respectively, for patients with hemophilia having inhibitor according to universal guidelines.
Blood Sampling Procedure and Platelet-Poor Plasma
Peripheral venous blood was collected into Vacutainer tubes containing 0.129 mol/L trisodium citrate. Antecubital venipuncture was completed using 21-gauge needles. Following a double centrifugation at 2500g for 15 minutes, platelet-poor plasma (PPP) was collected from the upper half of the plasma supernatant, quick-frozen, and stored at −80°C. The PPP was always prepared within 30 minutes of venipuncture. The TEG was performed on the complete citrated blood sample within an hour on the day of collection. For the TGA test, aliquoted PPP was stored for 3 months at −80°C, and all samples were assessed together. Both TEG and TGA parameters were evaluated at basal status and after 1 hour of bypass agent therapy. These tests were repeated when clinical hemostasis was obtained.
We utilized the TEG 5000 Thromboelastograph Hemostasis Analyzer (Hemoscope, Skokie, Illinois, USA) with TEG Analytical Software Version 4.1. All the assays were activated by kaolin per the manufacturer’s instructions. The TEG parameters include the following: R is a period of time from initiation of the test to the initial fibrin formation (minute). K is a measure of time from beginning of clot formation until the amplitude of thromboelastogram reaches 20 mm and represents the dynamics of clot formation (minute). Alpha angle is an angle between the line in the middle of the TEG tracing and the line tangential to the developing “body” of the TEG tracing. The alpha angle represents the acceleration of fibrin buildup and cross-linking. Maximum amplitude (MA) reflects the strength of a clot which is dependent on the number and function of platelets and its interaction with fibrin (millimeter).
The calibrated automated thrombogram (Thrombinoscope BV, Maastricht, The Netherlands) method was used for the TGA. 9 The assays were performed according to the manufacturers' instructions for thrombin generation with a mixture of 1 pmol/L tissue factor (TF) and 4 µmol/L phospholipids for PPP. Fluorescence was measured in an Ascent Reader (Thermolabsystems OY, Helsinki, Finland), and thrombin generation curves were calculated with Thrombinoscope software package. Thrombin generation parameters that can be derived from the thrombin generation curve include the following: lag time (time to 16.7% of peak concentration, minutes), endogenous thrombin potential (ETP; area under the curve; nmol/L per min), peak height (nmol/L), time to peak (minutes), and time to tail (minutes).
Evaluation of Clinical Responses After Treatment
Clinical response outcomes were classified either as effective responses or as no response. An effective response was defined as cessation of bleeding following bypass agent therapy in oral cavity patients. In hemarthrosis, a response was considered effective if recovery of arthralgia either improved joint range of motion or not. In soft tissue hematoma, a response was defined as effective if there was a decrease in hematoma size. In gastrointestinal system hemorrhages, a response was considered effective if bleeding stopped or decreased. Clinical response outcomes were classified as effective responses versus no responses. These data were compared with those from other tests.
Evaluation of Data and Statistical Analyses
Data were analyzed by SPSS 15.0. The chi-square, Kruskal-Wallis, and Mann-Whitney U tests were used for analysis. P < .05 considered statistically significant.
Results
No difference was found between either 1st hour or 24th hour of clinical responses (who had no response at first hour) after rFVIIa and aPCC treatment (P = .61 and .56, respectively) in patients with hemophilia having inhibitor. Each of the bleeding episodes of the same patient was treated with rFVIIa or aPCC. Results showed us that the same bypassing agent on the same patient may give different clinical results time to time. There was no association between first hour clinical response and either TGA or TEG parameters after inhibitor bypassing agent treatment (Tables 1 and 2 ). No significant association was found between 24th hour clinically effective responses and TEG or TGA parameters after bypassing agent treatment in patients with hemophilia having inhibitor. Bypass therapy agents had significant effects on all TGA and TEG parameters except time to peak thrombin (Tables 3 and 4 ).
Comparing Clinical Response With TGA Parameters After Bypassing Agent Treatment at First Hour in Patients With Hemophilia Having Inhibitor.a
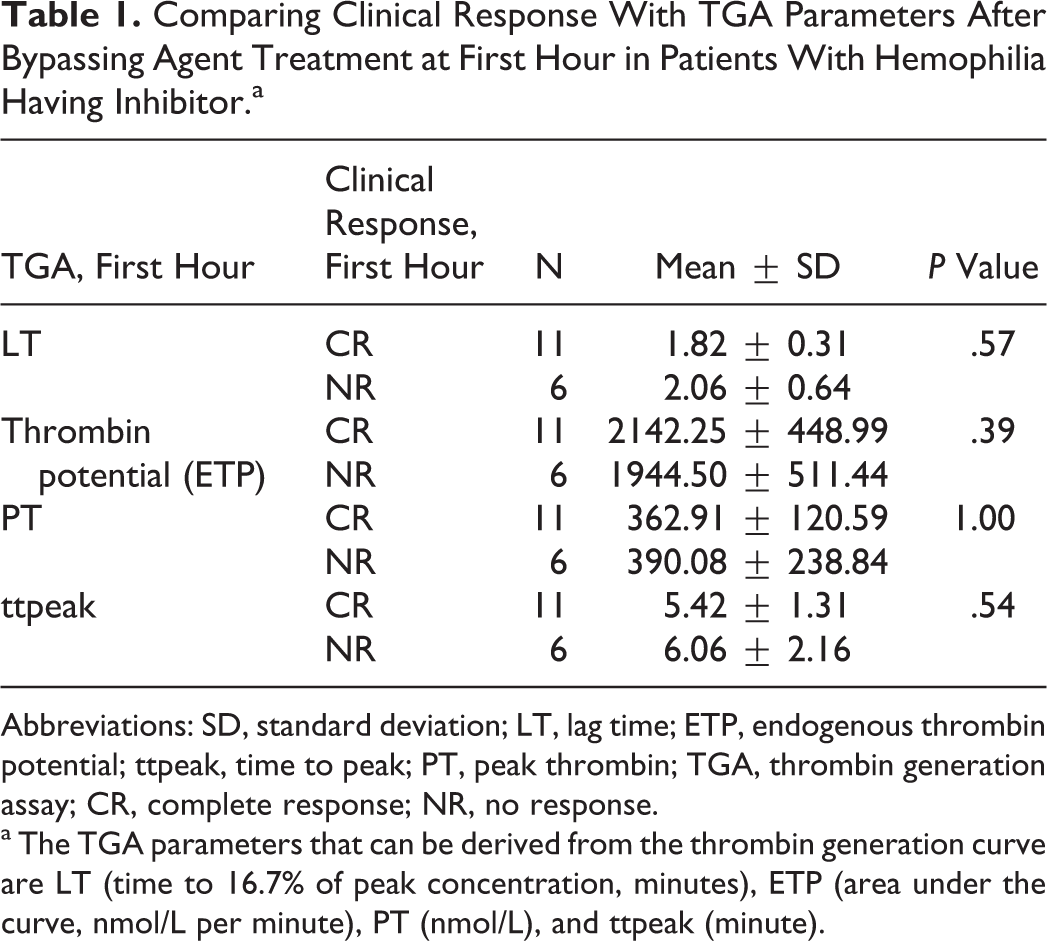
Abbreviations: SD, standard deviation; LT, lag time; ETP, endogenous thrombin potential; ttpeak, time to peak; PT, peak thrombin; TGA, thrombin generation assay; CR, complete response; NR, no response.
a The TGA parameters that can be derived from the thrombin generation curve are LT (time to 16.7% of peak concentration, minutes), ETP (area under the curve, nmol/L per minute), PT (nmol/L), and ttpeak (minute).
Comparing Clinical Response With TEG Parameters After Bypassing Agent Treatment at First Hour in Patients With Hemophilia Having Inhibitor.a

Abbreviations: MA, maximum amplitude; SD, standard deviation; TEG, thromboelastography; CR, complete response; NR, no response.
a TEG parameters—R is a period of time from initiation of the test to the initial fibrin formation (minute). K is a measure of time from beginning of clot formation until the amplitude of thromboelastogram reaches 20 mm and represents the dynamics of clot formation (minute). Alpha angle is an angle between the line in the middle of the TEG tracing and the line tangential to the developing “body” of the TEG tracing. The alpha angle represents the acceleration (kinetics) of fibrin buildup and cross-linking. The MA reflects the strength of a clot which is dependent on number and function of platelets and its interaction with fibrin (millimeter).
The TGA Parameters' Comparison Between Basal and After Bypassing Agent Treatment (First Hour) in Patients With Hemophilia Having Inhibitor.a
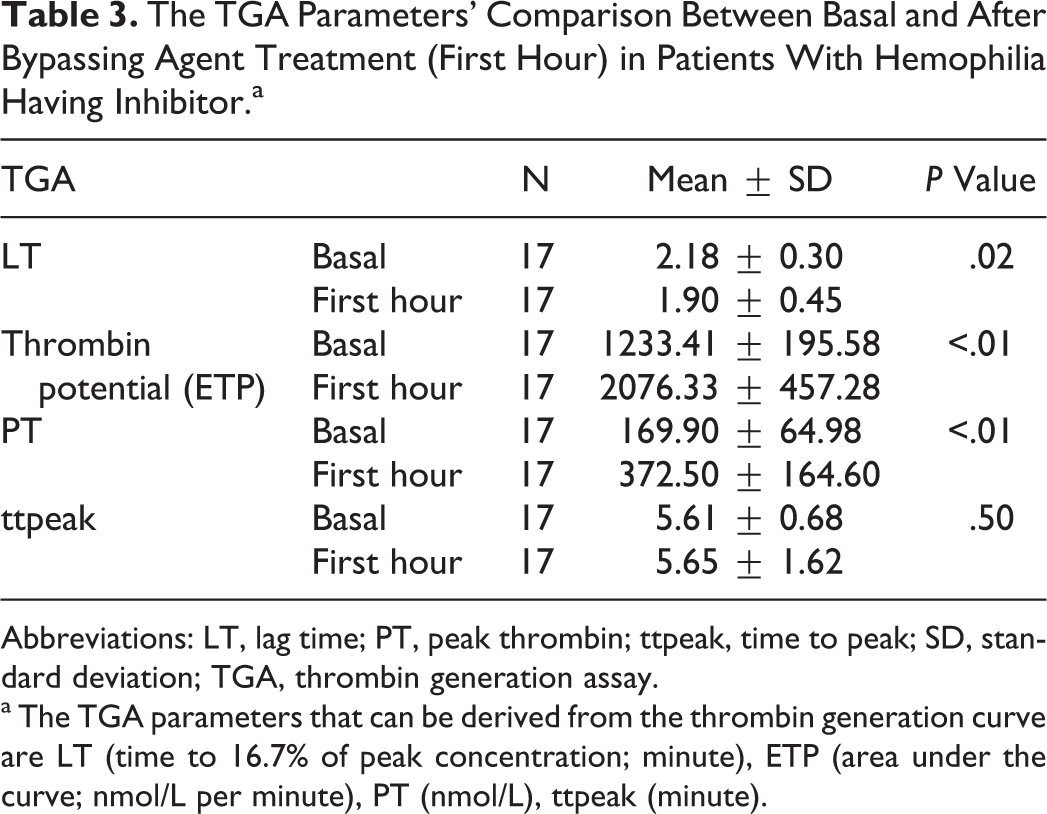
Abbreviations: LT, lag time; PT, peak thrombin; ttpeak, time to peak; SD, standard deviation; TGA, thrombin generation assay.
a The TGA parameters that can be derived from the thrombin generation curve are LT (time to 16.7% of peak concentration; minute), ETP (area under the curve; nmol/L per minute), PT (nmol/L), ttpeak (minute).
The TEG Parameters' Comparison Between Basal and After Bypassing Agent Treatment (First Hour) in Patients With Hemophilia Having Inhibitor.a
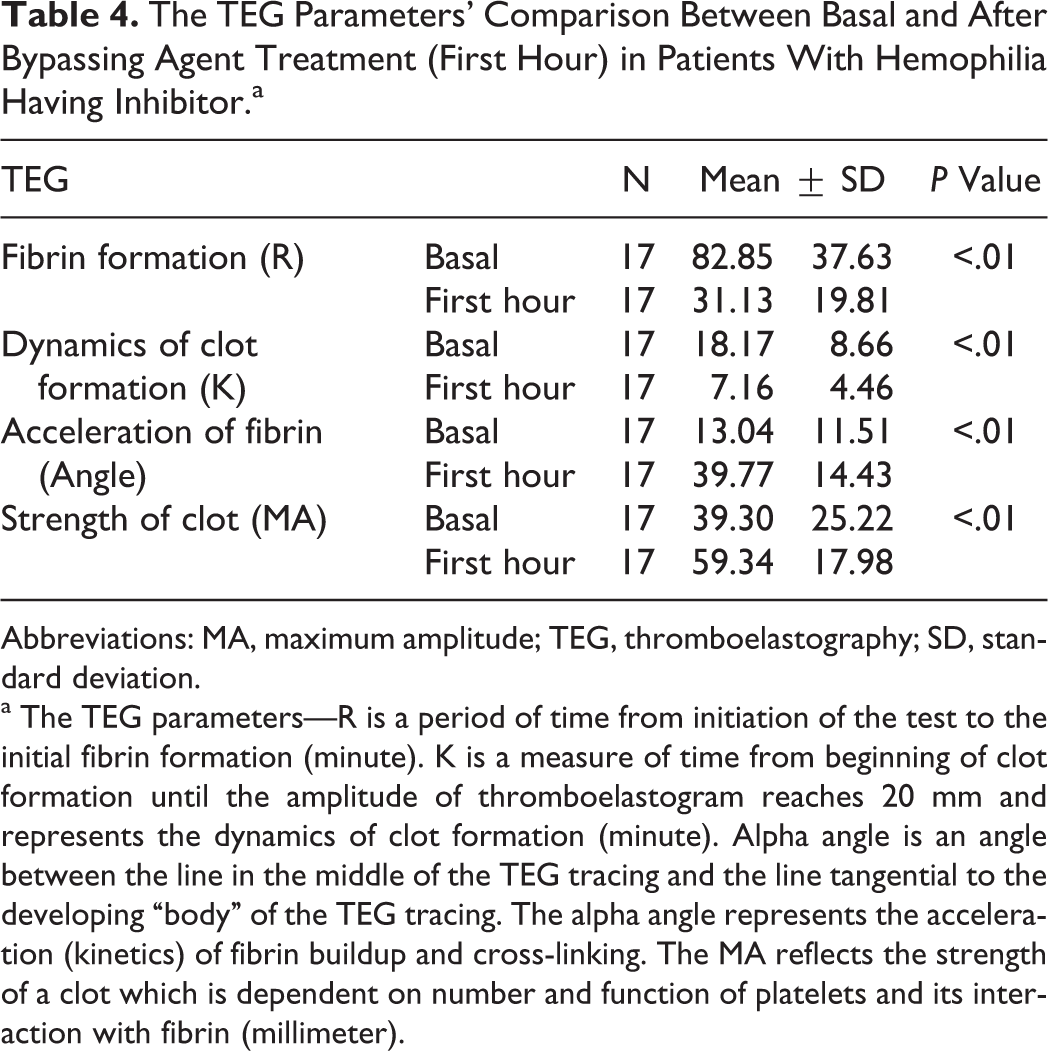
Abbreviations: MA, maximum amplitude; TEG, thromboelastography; SD, standard deviation.
a The TEG parameters—R is a period of time from initiation of the test to the initial fibrin formation (minute). K is a measure of time from beginning of clot formation until the amplitude of thromboelastogram reaches 20 mm and represents the dynamics of clot formation (minute). Alpha angle is an angle between the line in the middle of the TEG tracing and the line tangential to the developing “body” of the TEG tracing. The alpha angle represents the acceleration (kinetics) of fibrin buildup and cross-linking. The MA reflects the strength of a clot which is dependent on number and function of platelets and its interaction with fibrin (millimeter).
The TGA lag time values measured 24th hours post-event were significantly shorter in rFVIIa than aPCC treatment in patients with hemophilia having inhibitor (P = .04). First hour ETP and first-hour and 24th-hour peak thrombin heights were significantly higher in aPCC than in rFVIIa treatment in patients with hemophilia having inhibitor (P = .01, <.01, and .04, respectively; Table 5 ). First-hour TEG parameter (R, K, and alpha angle degree) values were significantly higher in aPCC than rFVIIa treatment (P = .03, .04, and .02, respectively) in patients with hemophilia having inhibitor.
The TGA Parameters' Comparison Between rFVIIa and aPCC After Therapy in Patients With Hemophilia Having Inhibitor.a
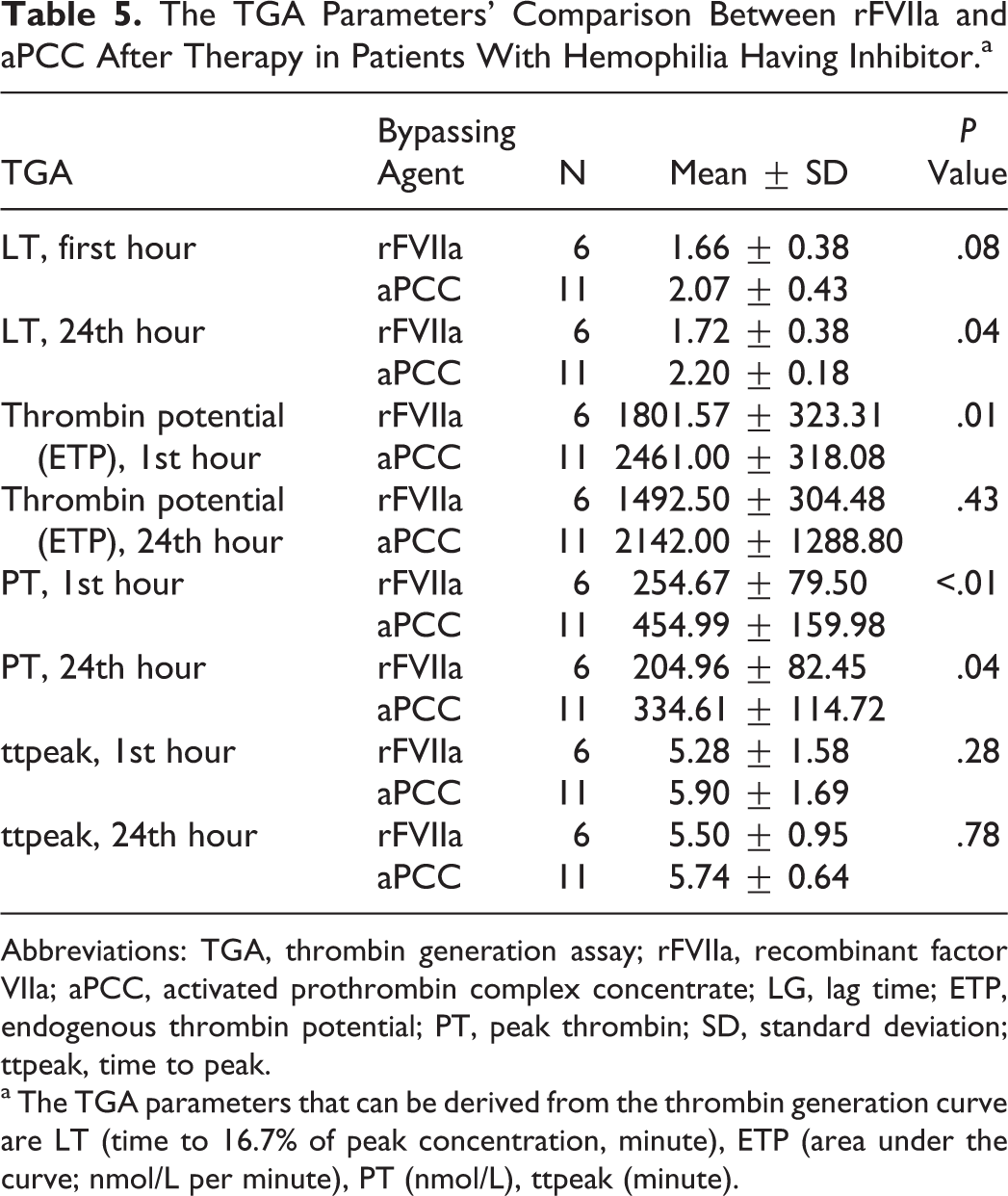
Abbreviations: TGA, thrombin generation assay; rFVIIa, recombinant factor VIIa; aPCC, activated prothrombin complex concentrate; LG, lag time; ETP, endogenous thrombin potential; PT, peak thrombin; SD, standard deviation; ttpeak, time to peak.
a The TGA parameters that can be derived from the thrombin generation curve are LT (time to 16.7% of peak concentration, minute), ETP (area under the curve; nmol/L per minute), PT (nmol/L), ttpeak (minute).
It was observed that patients with hemophilia having inhibitor had significantly worse TGA parameters such as peak thrombin height and time to peak thrombin values than the healthy control group (P = <.01 and <.01, respectively). No significant difference was found in ETP and lag time between the healthy control group and patients with hemophilia having inhibitor (P = .87 and .20, respectively; Table 6 and Figure 1 ). None of the patients had side effects related to bypass therapy.
The TGA Parameters' Comparison Between Patients With Hemophilia Having Inhibitor Before Bypassing Agent Therapy and Healthy Control Group.a
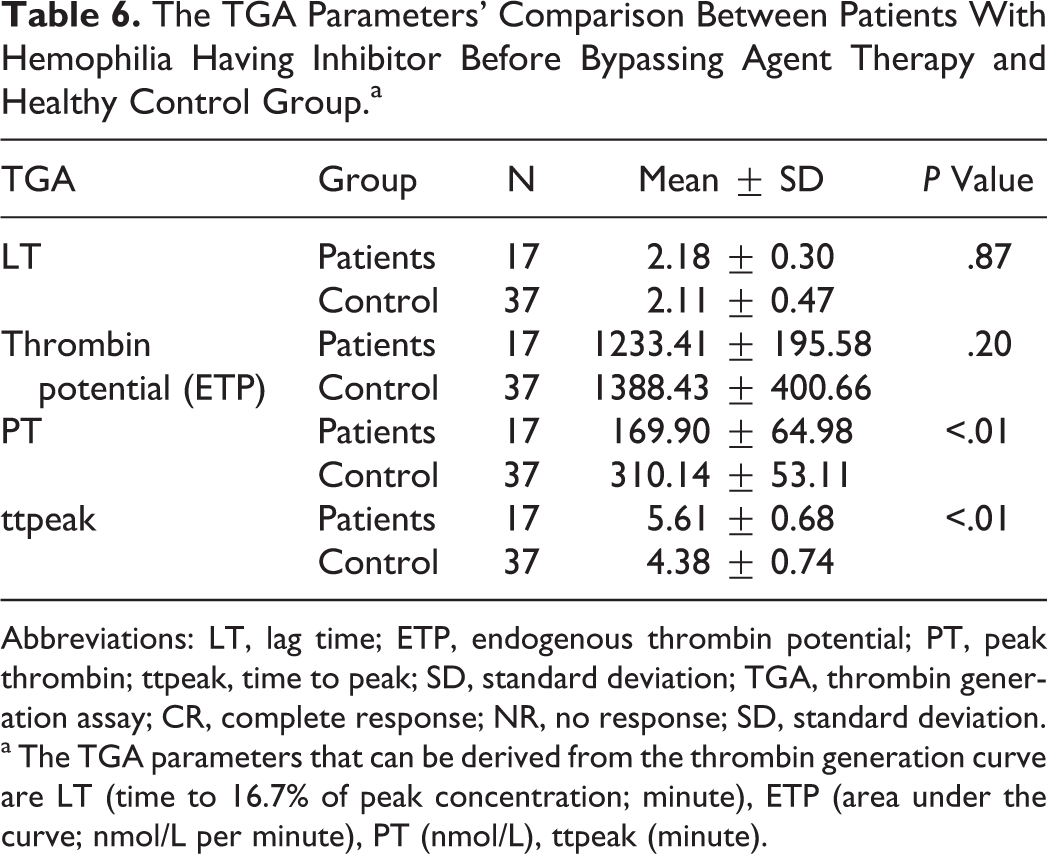
Abbreviations: LT, lag time; ETP, endogenous thrombin potential; PT, peak thrombin; ttpeak, time to peak; SD, standard deviation; TGA, thrombin generation assay; CR, complete response; NR, no response; SD, standard deviation.
a The TGA parameters that can be derived from the thrombin generation curve are LT (time to 16.7% of peak concentration; minute), ETP (area under the curve; nmol/L per minute), PT (nmol/L), ttpeak (minute).
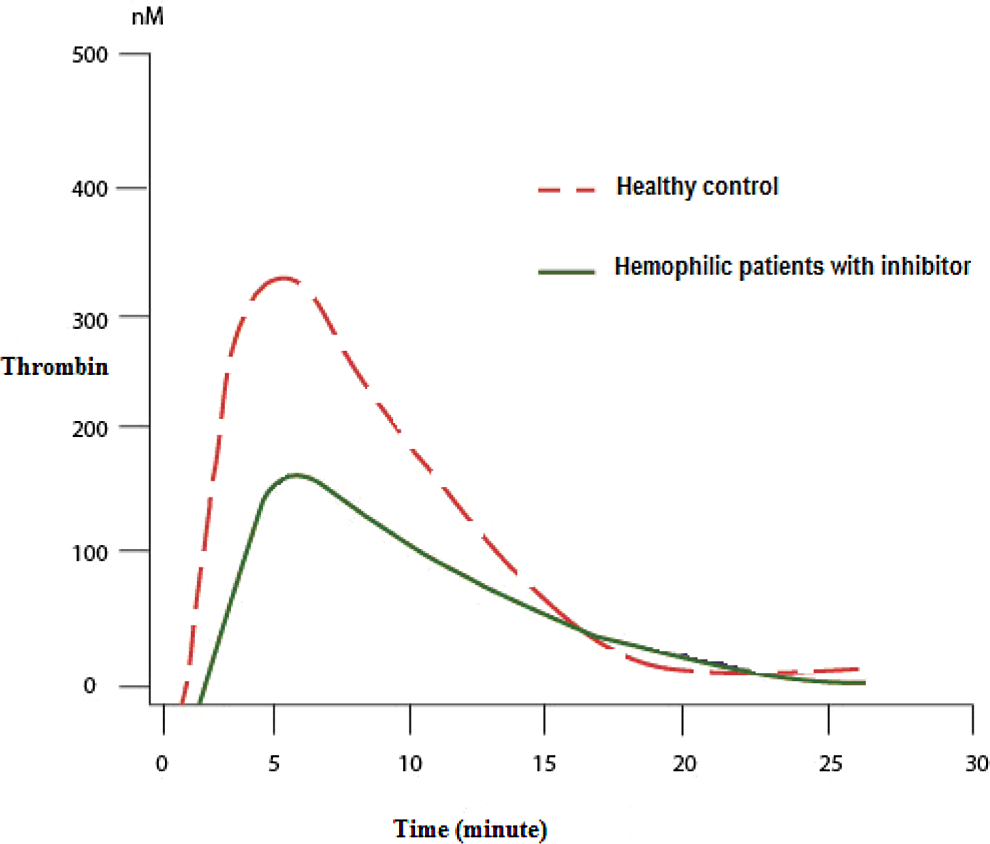
Thrombin generation assay curve of healthy controls and patients with hemophilia having inhibitor.
Discussion
The TGA measures the actual thrombin concentrations before and after the clot is formed and is very sensitive to variations in one or more coagulation factors. 3 Both ETP and peak thrombin generation measured by TGA can discriminate between normal individuals and patients with varying degrees of hemophilia severity. 11,12 We found that TGA parameters such as peak thrombin height were significantly lower and time to peak thrombin were significantly longer in patients with hemophilia having inhibitor than in the healthy controls in our study. There was no significant difference in ETP values between patients with hemophilia having inhibitor and healthy controls as has and has not been reported in some studies. 4,11,12 Some researchers also showed a high degree of variability in thrombin generation between patients with similar FVIII/FIX activity. 11,12 Normal lag time was observed in TGA with low tissue factor concentration (1 pmol/L) in patients with hemophilia, 13 –16 which could be explained by the fact that FVIII/FIX activities play a minor role in determining lag time, and measuring lag time by TGA only shows the initial phase of coagulation. 16
No significant association was found between the clinically effective response and TGA parameters, TEG at 1st hour and 24th hours after bypassing agent therapy within bleeding episode in patients with hemophilia having inhibitor.
The clinical effectiveness of monitoring inhibitor bypass therapy in hemophilia cannot be assessed by traditional coagulation tests. In vitro spiking of high-titer inhibitor plasma with increasing concentrations of aPCC (Feiba) resulted in the dose-dependent restoration of the thrombin-generating capacity of the FVIII-inhibitor plasma. 3,4,17,18 When thrombin generation was measured at different times in patients with hemophilia having inhibitor after treatment with single doses of FEIBA, increased thrombin generation was observed within 15 to 30 minutes after injection, which gradually returned to baseline values 8 to 12 hours after treatment. 17,18 The TGA monitoring was suggested in a patient with inhibitors undergoing surgery. 19 Several studies have examined the effect of rFVIIa (NovoSeven) on thrombin generation in different settings. 20 –29 Different techniques and methodologies were used, and only some reports demonstrated an effect of rFVIIa on thrombin generation, whereas all showed a shortening of the lag phase.
In this study, similar to several studies, 3,4,17,18 we established decreased lag time after treatment with a bypassing agent in patients with inhibitor. However, no significant difference was found in lag time between the healthy control group and patients with inhibitor-positive hemophilia, and detection of basal lag times similar to healthy controls supports the variable outcomes in lag time observed in other studies. 13 –16
Numerous studies have demonstrated that 3,4,17,18,20 –26 a bypassing agent can shorten lag time and increase thrombin generation in patients with hemophilia having inhibitor. In this study, rFVIIa and aPCC were applied to patients with hemophilia having inhibitor during bleeding episode treatment. No significant difference was found between both treatments for clinical responses at 1st or 24th hours after either event. The 24-hour lag time values were significantly shorter in the rFVIIa treatment group than in the aPCC treatment group in patients with inhibitor. First hour ETP and the 1st-hour and 24th-hour peak thrombin height values were significantly higher in the aPCC treatment group than in the rFVIIa treatment group (Table 5).
The TEG enables global assessment of hemostatic function. This method measures the viscoelastic properties of blood induced to clot under low shear conditions, corresponding to venous flow. 30,31 The TEG analyzes whole blood and thus measures the effects of both cellular and plasmatic components on clot formation. 31 While other conventional tests stop with the formation of the first fibrin strands, the TEG begins to evaluate clot formation at this point and collects data as clotting continues through to eventual clot lysis or retraction. Therefore, one single test produces information about several steps in the coagulation process. The TEG evaluates the kinetics of formation, stabilization, and subsequent lysis of the clot. Poor correlations have been observed between most conventional coagulation tests and TEG measurements. 32,33
Young et al
34
have demonstrated the utility of TEG for the individualization of bypass therapy in patients with hemophilia having bleeding episodes. In this study, we found no correlation between clinically effective responses and TEG parameters (R, K, angle, and MA) at the 1st and 24th hours after bypassing agent therapy in patients with hemophilia having inhibitor. As in the literature,
34
–37
we found that TEG parameters after bypassing agent treatment were significantly better than the basal TEG parameters in patients with hemophilia having inhibitor. The TEG parameters, such as R, K, and angle degree values, were significantly better in the aPCC treatment group than in the rFVIIa group in patients with hemophilia having inhibitor. The limiting factors in this study included the following: Wide interval of second evaluation after bypassing agent treatment, Insufficient number of patients to compare clinical responses and laboratory efficiencies of rFVIIa and aPCC treatments in patients with hemophilia having inhibitor, Using fresh samples as platelet-rich plasma (PRP) would be fine for better analyses of TGA. However, we should collect PPP samples for cumulative analysis. Moreover, in daily practice, using PPP is a more convenient approach.
Currently, TGA tests are only used in academic studies, as opposed to daily devices such as a coagulometer, and due to the technical details of the TGA, samples are often accumulated for months and processed at once. Evaluation of the treatment processes in patients with inhibitor-positive hemophilia will be improved with further studies. In conclusion, we found the clinical effectiveness of monitoring inhibitor bypass therapy in hemophilia cannot be assessed by TGA and TEG.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
