Abstract
We aimed to confirm the results of randomized, controlled trials on enoxaparin prophylaxis in unselected patients undergoing day surgery. The primary end point was the incidence of thromboembolic events during prophylaxis and up to 48 hours thereafter. A total of 11 794 patients, consisting of 52.1% male with mean age of 49.2 ± 15.7 were included. In all, 61.5% had no predisposing risk factors and 67.1% received no concomitant medication with the potential to increase bleeding. Patients were exposed to 20 mg (63.6%) and 40 mg (36.4%) of enoxaparin for a mean of 12.4 ± 9.8 days. Forty-four patients (0.39%) had confirmed symptomatic deep venous thrombosis and 1 patient confirmed pulmonary embolism. Bleeding occurred in 3.47% of patients (3.29% minor bleeding). Differences between 20 and 40 mg enoxaparin were negligible. Adverse drug reactions were experienced by 3.1% of patients. The present study results demonstrate that it is effective and tolerable to use a risk stratified dose of 20 or 40 mg enoxaparin in patients undergoing day surgery.
Introduction
Venous thromboembolism (VTE) is a frequent complication of surgical treatment in patients without appropriate prophylaxis. However, still between 10% and 20% of patients aged between 40 and 60 without additional risk factors develop distal deep venous thrombosis (DVT) and 2% to 4% proximal DVT. These rates are twice as high in patients older than 60 years. 1 This applies not only to hospitalized patients but also to those treated on an outpatient basis, although risk is perceived to be lower because of the extent of the surgical intervention. 2
For the purpose of prophylaxis, unfractionated heparins (UFHs) and low-molecular-weight heparins (LMWHs) are used, which result in a risk reduction of 60% to 70%. 3 While bleeding rates are low with low doses of LMWH (3.8% vs 5.4% with low-dose UFH), bleeding complications are increased with higher doses. 4 Therefore, a stratification of risk is important to balance the benefits and risks of prophylaxis in each individual patient.
A straight forward approach to the assessment of risk is the use of scoring cards such as the one proposed by Haas. 5 The Haas’ algorithm makes use of a number of exposing and predisposing risk factors and assigns either a low, medium, or a high risk to each patient. This risk stratification can be used to guide the dosing of anticoagulants. One such anticoagulant is enoxaparin, which is registered for the prophylaxis of VTE in the outpatient setting. It is used at 20 mg in surgical patients with low or medium risk and at 40 mg in patients with high risk.
When setting up this noninterventional study (NIS), we aimed to assess the efficacy and tolerability of enoxaparin in an outpatient setting, when used in accordance with the labeling, at a proper dosing as outlined above and the applicable guidelines. 1,6 These data are of importance because not all patients in clinical practice have been investigated in randomized controlled trials.
Patients and Methods
The SMART (SysteMatische Datenerfassung im Ambulanten Bereich zur Risikoabschätzung Thromboembolischer Ereignisse bei chirurgischen Patienten—SysteMatic documentation within the Ambulatory setting to assess the Risk of Thromboembolic events in surgical patients) was an NIS in the outpatient setting. Participating physicians were recruited at random from a list of general or orthopedic surgeons providing outpatient surgical care from all regions throughout Germany, defined by the Institute of Medical Statistics (IMS). The study was conducted according to German Medicines Law (§67 (6) Arzneimittelgesetz [AMG]) and was duly notified to the federal authorities (Bundesinstitut für Arzneimittel und Medizinprodukte [BfArM]) and the Association of Statutory Health Insurance Physicians (Kassenärztliche Bundesvereinigung [KBV]). Because of the noninterventional character of the study, no ethics committee approval was required. However, patients’ informed consent was obtained before enrollment.
Patients
Patients ≥18 years receiving perioperative or postoperative prophylaxis with 20 or 40 mg enoxaparin because of a surgical intervention or orthopedic surgery on an outpatient basis were included into this observational study. Applicable exclusion criteria were based on contraindications outlined in the summary of product characteristics (Clexane, Sanofi Aventis, Berlin, Germany). The duration of medication was left to the discretion of the surgeon.
Documentation
At enrollment, patient characteristics and data on predisposing risk factors and concomitant medication were obtained. A stratification of risk was carried out according to Haas’ algorithm (Figure 1 ) that allowed assigning patients a low, medium, or high risk. 5 During pharmacological prophylaxis, a number of variables were obtained including the use of compression methods, interruption of therapy, a change in enoxaparin dose, immobilization, bleeding complications, and any evidence of VTE. In case of unexpected adverse events, these were duly recorded and announced to the responsible regulatory body in due course.
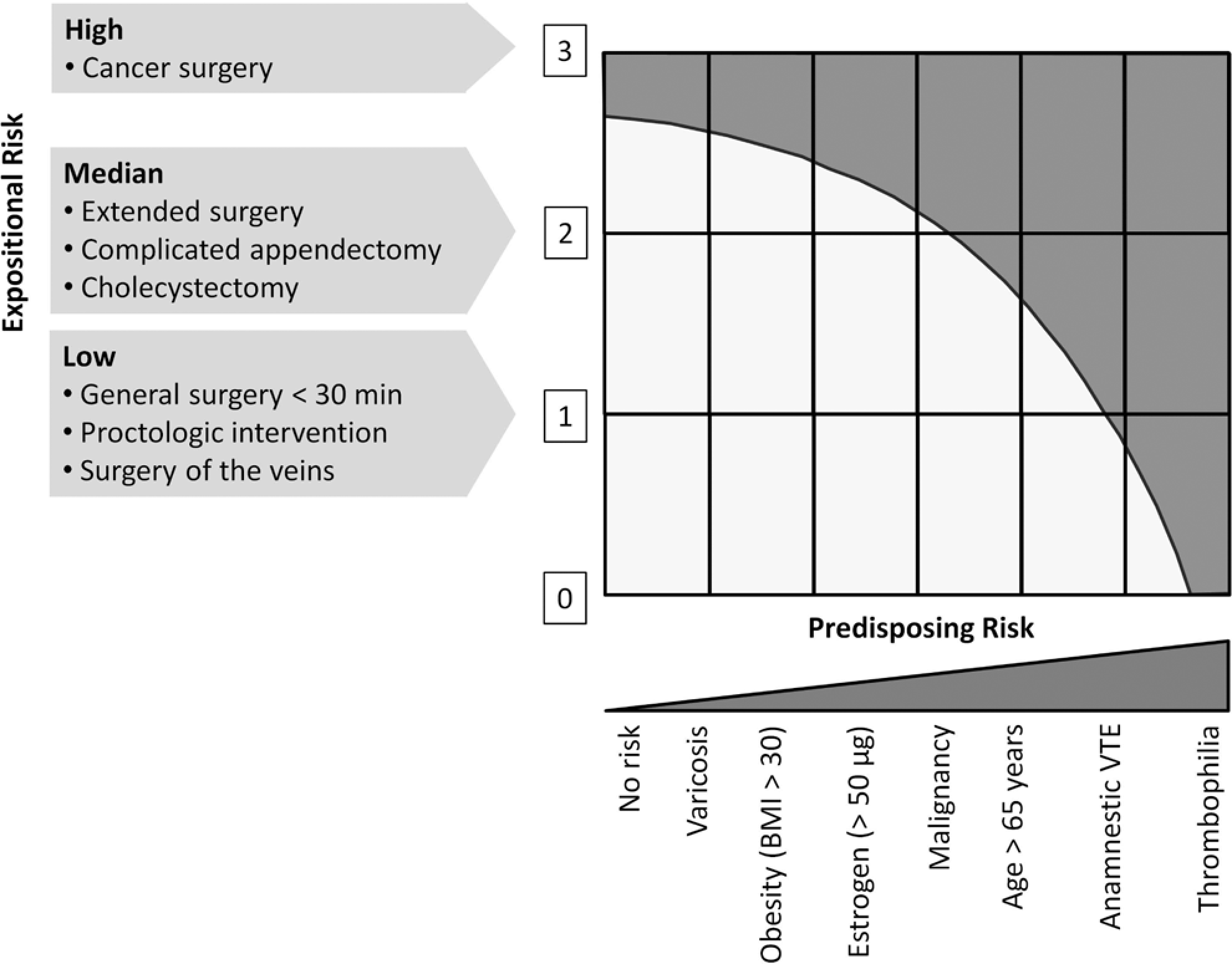
Risk assessment based on the modified Haas’ scheme. 5
End Points
The primary end point of this observation was defined as the incidence of symptomatic thromboembolic events (deep venous thromboembolism [DVT] and pulmonary embolism [PE]) confirmed by objective diagnostic tests such as a sonography, phlebography, computed tomography, or magnetic resonance (MR) tomography within the treatment period and up to 48 hours after last dose. The secondary end point was defined as the incidence of major bleeding (serious, life-threatening events or death, a decrease in hemoglobin ≥2 g/dL, blood transfusions of at least 2 units, retroperitoneal, intracranial, or intraocular bleeding, and bleeding causing an additional intervention) or minor bleeding complications (bleeding events that did not meet the above mentioned criteria for major bleeding).
Statistical Considerations
SPSS for Windows (release 15.0.0) was used for the statistical analysis. For categorical data, absolute and relative frequencies and for continuous variables average and standard deviations (SDs) were calculated. The incidence rates for all patients and for both the dose groups (20 and 40 mg enoxaparin), as defined by risk category, were determined. The expected incidence for the primary end point was 0.8% to 1.5%.
Results
Between March 2007 and December 2008, a total of 11 794 patients were documented in SMART. Of these, 11 791 patients received at least 1 dose of enoxaparin. One patient received enoxaparin but surgery was postponed. Therefore, 11 790 patients were available for the analysis of efficacy.
Patient Characteristics
Baseline characteristics of the patients are summarized in Table 1 . They had a mean age of 49.2 ± 15.7, a mean body weight of 79.2 ± 14.5 kg, and 52.1% were male. Predisposing risk factors were present in 38.5% of patients—with venous disease (19.9%) and hormone therapy (9.1%) being most frequent. One third (32.9%) of the patients received any drugs that may increase the risk of bleeding complications, the majority of which were nonsteroidal anti-inflammatory drugs (29.9%).
Patient Characteristics
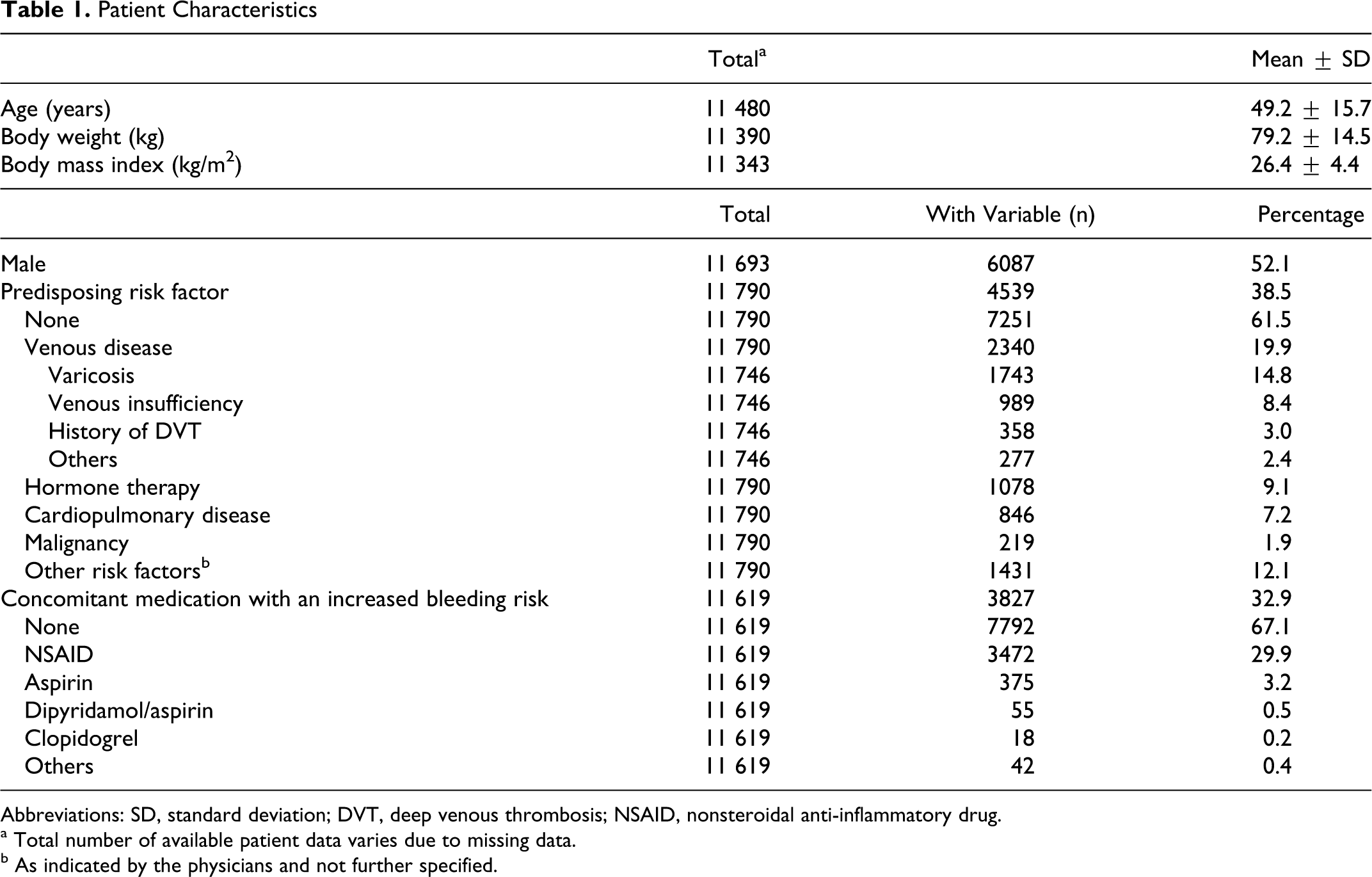
Abbreviations: SD, standard deviation; DVT, deep venous thrombosis; NSAID, nonsteroidal anti-inflammatory drug.
a Total number of available patient data varies due to missing data.
b As indicated by the physicians and not further specified.
Surgical Intervention
Three quarters (75.4%) of patients had orthopedic surgery (the single most frequent intervention was a meniscal resection), 11.9% had abdominal surgery, and 10.1% had vascular surgery (Table 2 ) lasting a mean of 0.72 ± 0.4 hours and was usually performed under general anesthesia (86.2%). A minor proportion (20.8%) was immobilized.
Surgical Intervention
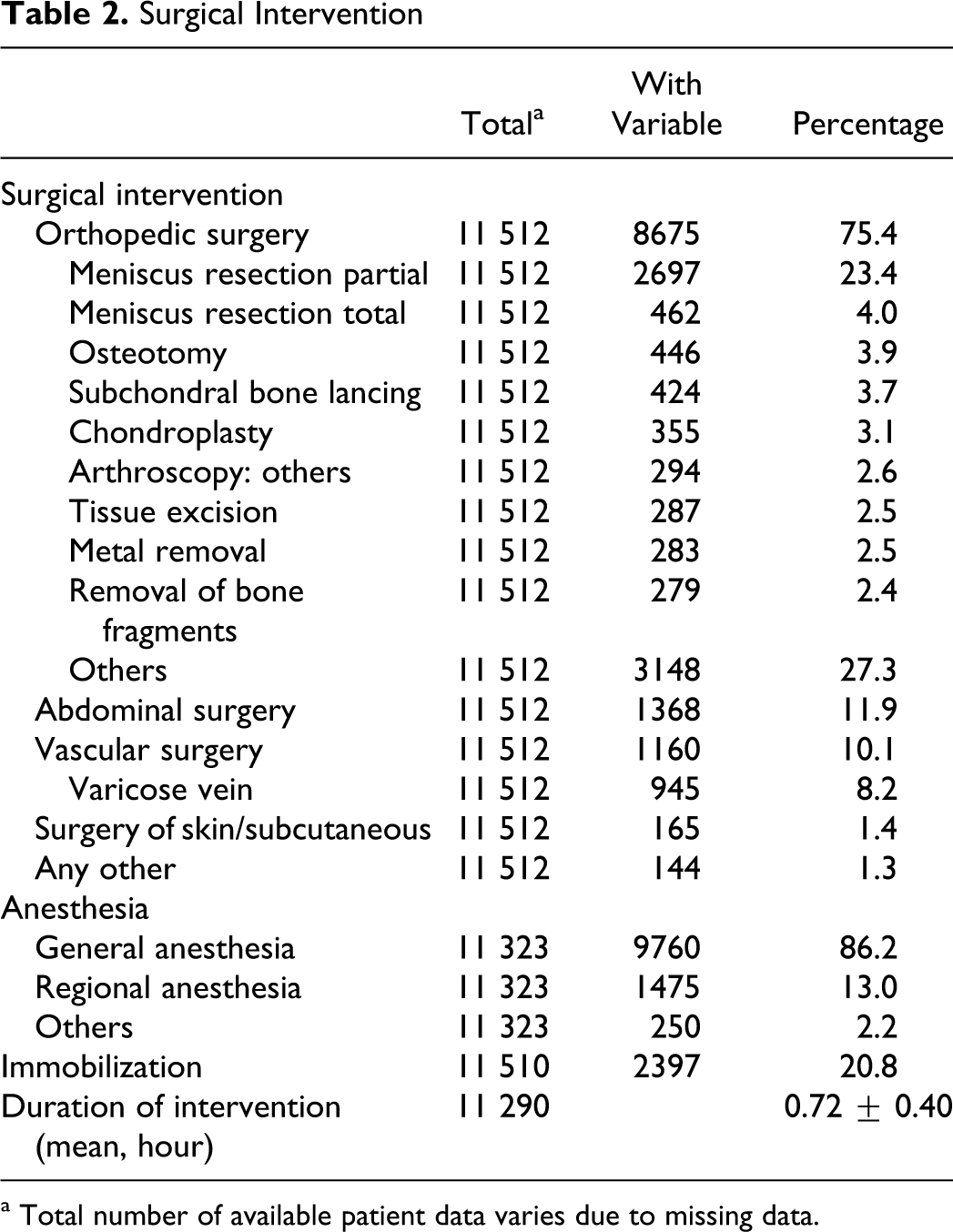
a Total number of available patient data varies due to missing data.
Venous Thromboembolism Prophylaxis
All patients received enoxaparin; of which 63.6% received the lower dose of 20 mg (Table 3 ). The mean exposure to enoxaparin was 12.37 ± 9.84 days, which was longer in patients receiving 40 mg when compared to 20 mg (14.72 ± 12.33 vs 10.96 ± 7.69 days). Enoxaparin was generally started on the day of surgery (75.7%) or before (21.8%). Additional surgical stockings were received by 47.6% of patients; 24.7% received compression bandages. The vast majority of the patients administered the subcutaneous injections themselves (94.5%) after having been instructed by the physician.
Prophylaxis
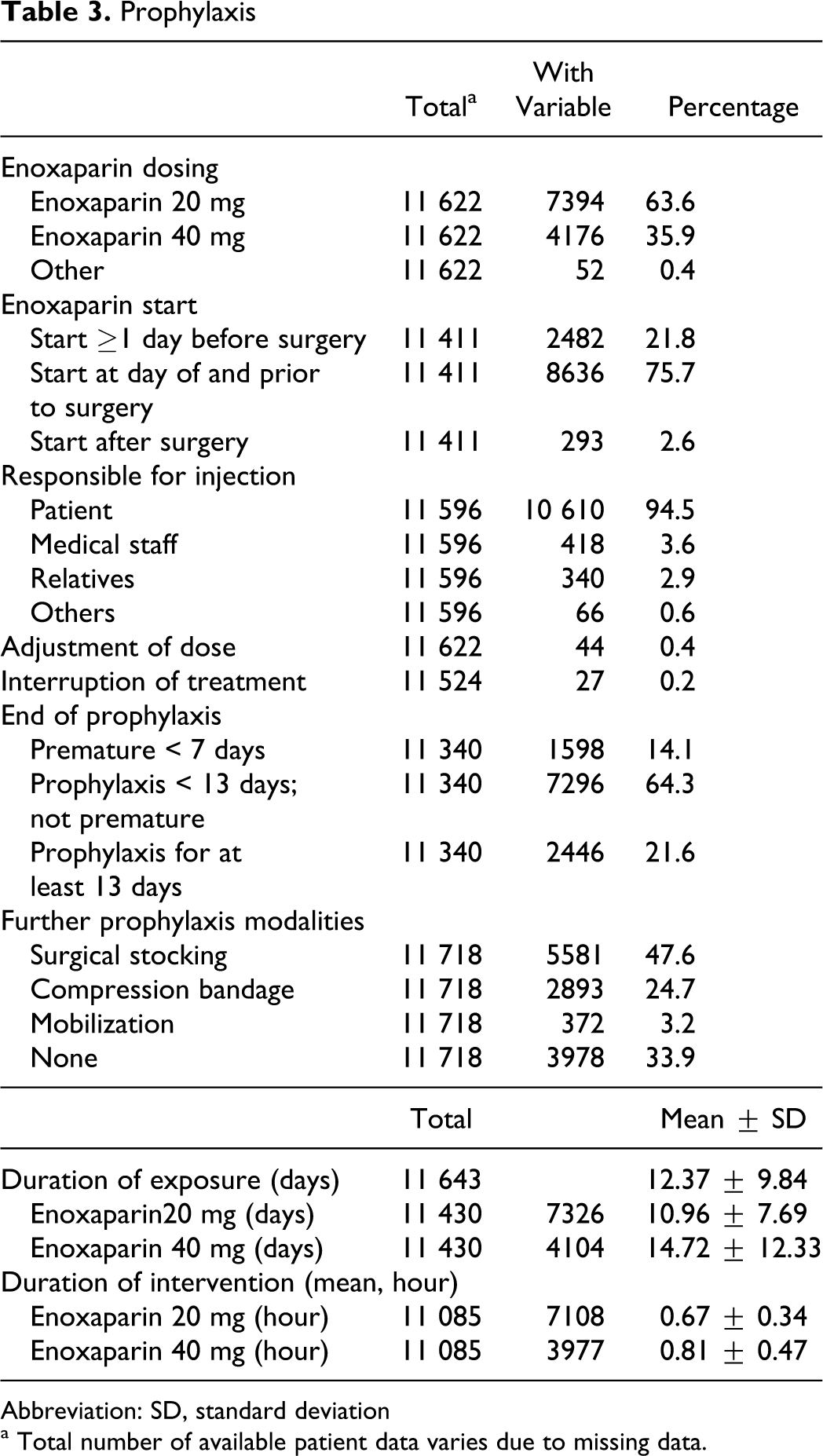
Abbreviation: SD, standard deviation
a Total number of available patient data varies due to missing data.
Incidence of Symptomatic Thromboembolic and Bleeding Events
Of 11 266 patients, 58 (0.51%) had clinical suspicion of DVT at day 8.72 ± 8.65 (mean ± SD) with pain upon pressure, spontaneous pain, and swelling being the most frequent symptoms (Table 4
). Ultrasonography,
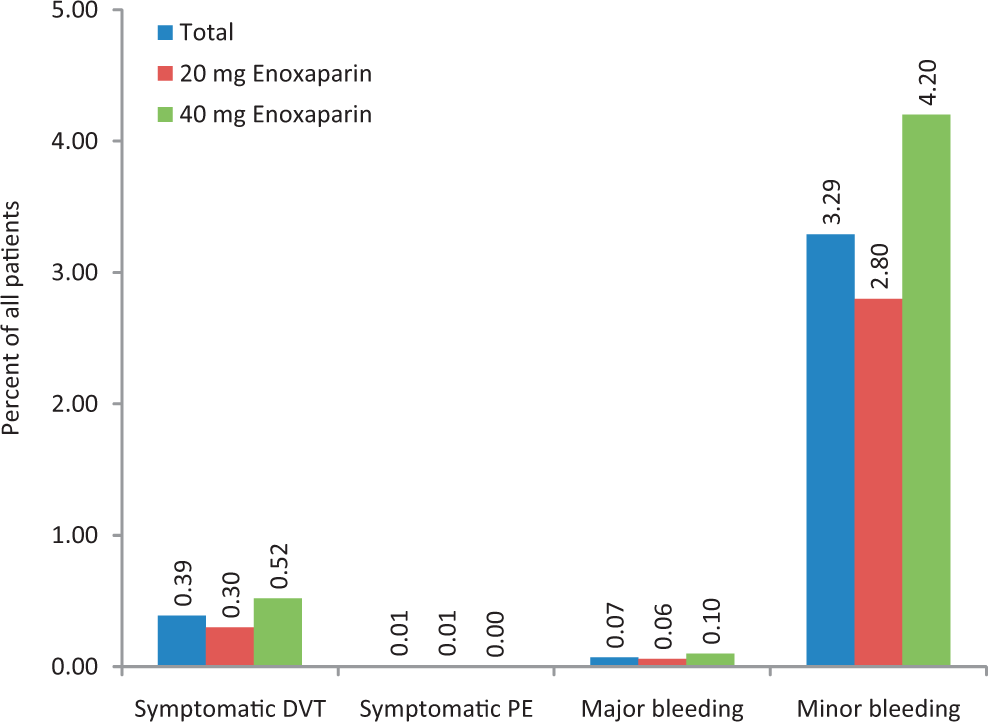
Incidence of major adverse events in SMART. DVT indicates deep venous thrombosis; PE, pulmonary embolism; SMART, SysteMatic documentation within the Ambulatory setting to assess the Risk of Thromboembolic events in surgical patients.
Clinical Symptoms Leading to Suspicion of VTE and Diagnostic Workup
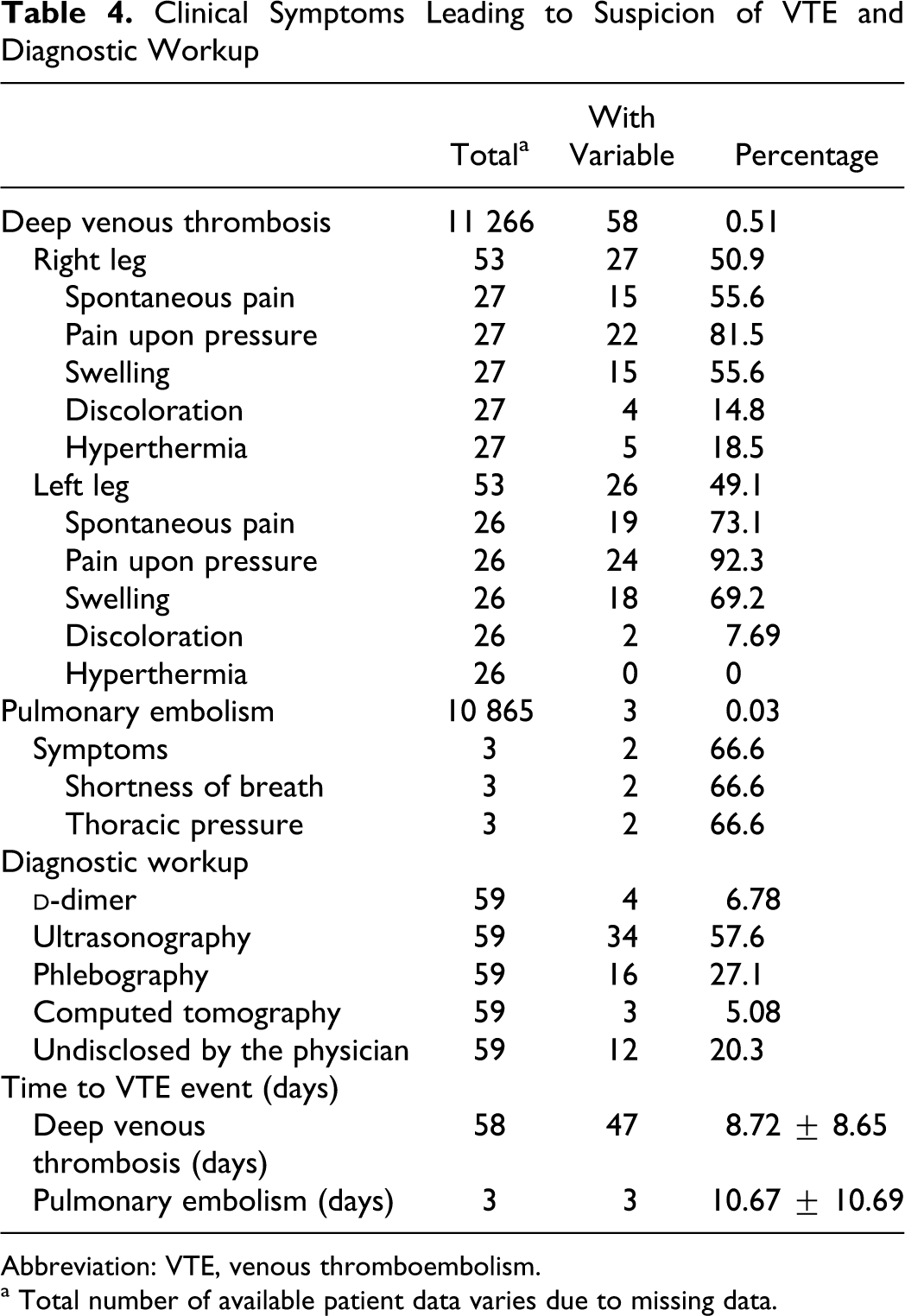
Abbreviation: VTE, venous thromboembolism.
a Total number of available patient data varies due to missing data.
Incidence of Symptomatic Thromboembolic and Bleeding Events
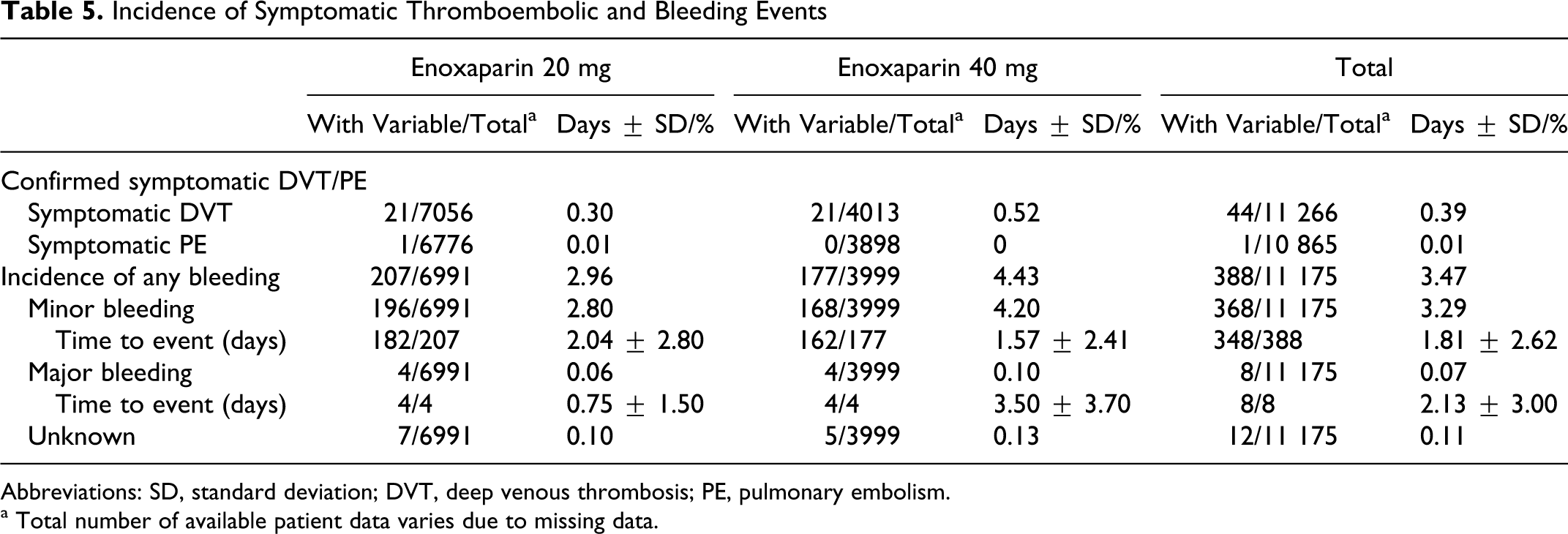
Abbreviations: SD, standard deviation; DVT, deep venous thrombosis; PE, pulmonary embolism.
a Total number of available patient data varies due to missing data.
Bleeding occurred in 3.47% of patients, with the majority being minor bleeding events (3.29%) with a latency of 1.81 ± 2.62 days after the start of treatment (0.11% unknown; Table 5). Major bleeding was less frequent (0.07%) and occurred after 2.13 ± 3.00 days of prophylaxis. The risk of bleeding was insignificantly higher in the 40-mg enoxaparin group (4.43% vs 2.96%), mostly on the account of minor bleeding complications.
Adverse Events
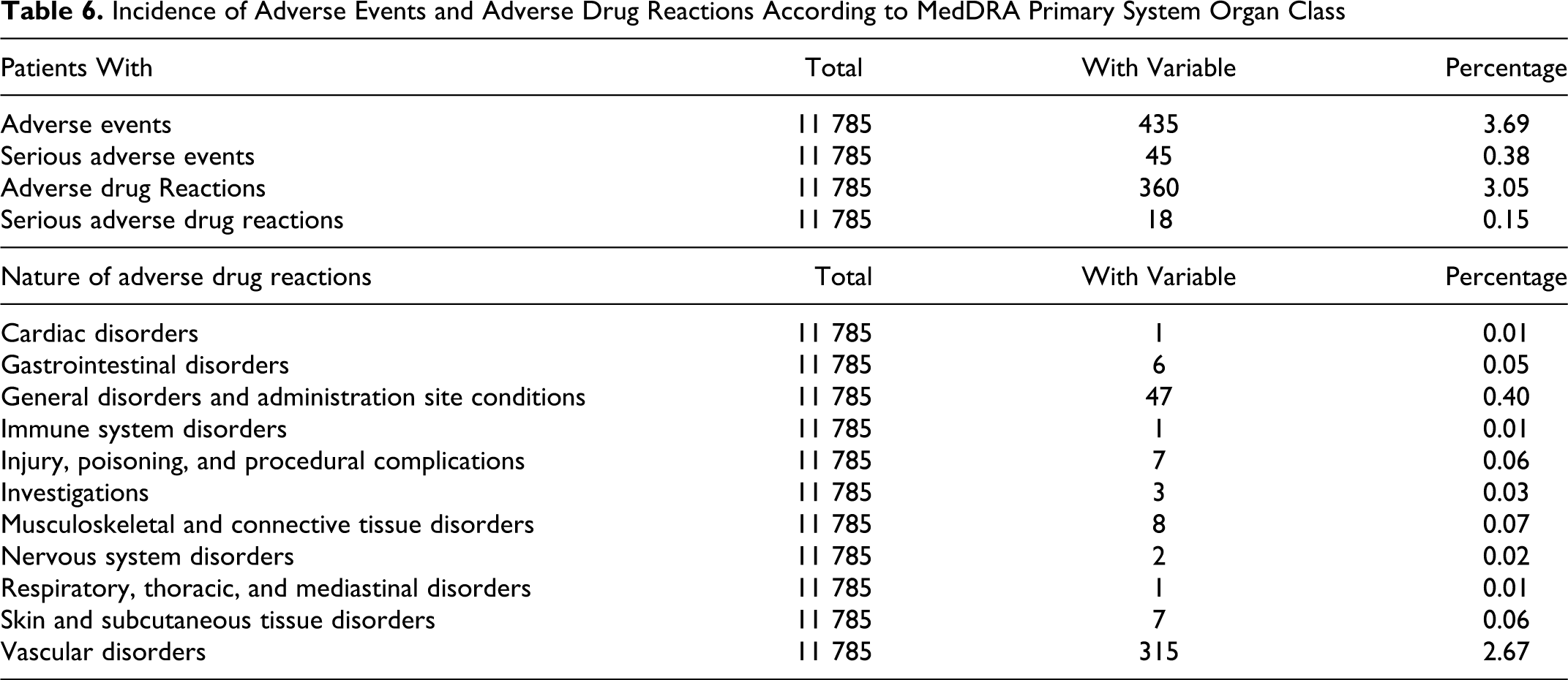
Adverse events were recorded in 3.69% of patients; of which 0.38% were serious. Three percent were related to drug use (0.15% serious). None of the patients died during this NIS (Table 6 ).
Incidence of Adverse Events and Adverse Drug Reactions According to MedDRA Primary System Organ Class
Discussion
The present study results illustrate that the use of a risk-stratified dose of 20 or 40 mg enoxaparin in patients after orthopedic or general and vascular surgery on an outpatient basis is effective and safe for the prevention of VTE. In total, 0.39% of patients had confirmed DVT and 0.01% suffered confirmed PE while having a 0.07% risk of major bleeding.
Benefits and Risk of Anticoagulant Use
In the present NIS, there were 63.6% of patients at low risk of thromboembolic events as indicated by the frequent use of 20 mg enoxaparin after risk assessment by the Haas’ score card. Patients were usually young (meniscal resection was the most frequent intervention), 61.5% had no predisposing risk factors and 67.1% received no concomitant medication with the potential to increase the risk of bleeding.
It is difficult to put these results into perspective because of a missing control group. Epidemiological data point to a low risk, however. 2,8 In a Danish survey covering 16 048 patients undergoing various types of day surgery, the incidence of VTE was 0.04% (45 per 100 000) and of hematomas/hemorrhage was 0.4%, within a follow-up of 60 days. 8 The VTE incidence was higher than the 60-day incidence in the general population without a previous operation, which is estimated to be 16 to 33 per 100 000, 9 suggesting that surgery and hospitalization does have an impact on the development of VTE. In particular, for knee arthroscopy, highly variable rates from 0.24% (only clinical symptoms) to 17.9% (clinical and nonclinical symptoms) for DVT have been reported. 9,10 In a German NIS, including patients undergoing arthroscopy, the rate of VTE was 0.5% and of PE was 0.11% in those receiving enoxaparin prophylaxis. 11 These rates are in good agreement with our incidence rate of about 0.4%. Differences compared to the higher of the previously reported rates, can probably be explained by the missing identification of asymptomatic VTE in the present study but also from differences in the surgical procedures, duration of surgery, application of a tourniquet, and mobilization procedures.
A randomized study by Michot et al 12 enrolled 130 patients undergoing outpatient knee arthroscopy. The patients were assigned either to LMWH postoperatively for 4 weeks or to a control group without thromboprophylaxis. All patients were screened by bilateral compression ultrasonography before surgery as well as 12 and 31 days thereafter. Thromboprophylaxis resulted in a significantly lower incidence of VTE; DVT was detected in 1 of 66 patients receiving LMWH and in 10 of 64 control patients. All DVTs were confined to the calf of the operated leg, and there was only 1 patient (in the treatment group) with both DVT and PE. No severe side effects of LMWH were observed; there was no case of major bleeding or heparin-induced thrombocytopenia.
We only documented symptomatic VTE in the present NIS, leaving room to speculate that there might have been asymptomatic events that were not verified. This assumption is not unfounded because 13 of 24 patients were asymptomatic in the aforementioned study by Demers et al, 10 about 50% in a documentation of 102 consecutive patients undergoing outpatient knee arthroscopy by Delis et al, 13 and 17 of 19 patients in a 335 patient cohort study by Hoppener et al. 14 This limitation cannot be alleviated in the present data set but may point at the possibility that the efficacy of enoxaparin may also extend into the prevention of asymptomatic VTE at a risk of major bleeding that has been determined to be 0.07% in the present study. The short duration of follow-up can be perceived as a further limitation.
The results are in principal agreement with our findings, pointing to a beneficial effect of thromboprophylaxis in this patient population. It may become even more important as day surgery expands and includes more extensive procedures in older patients and with more serious coexisting diseases. Furthermore, adjusting prophylaxis to the individual patient’s risk has been shown to be effective and safe in our study, with 0.4% of patients experiencing any VTE (DVT and PE), while 0.07% had major bleeding complications.
Timing and Duration of Prophylaxis
The majority of patients were started on enoxaparin on the day of surgery prior to the intervention (75.7%) and prophylaxis continued for a mean of 12 ± 10 days (40 mg > 20 mg). This is in line with the recommendations for the use of enoxaparin. However, it has been reported that when prophylaxis is initiated within 2 hours after the surgical intervention, it increases the risk of major bleeding and initiation at more than 6 hours after surgery is less effective. 15 In our study, 2.6% of patients received thromboprophylaxis only after surgery; because of the low number of events however no reasonable data are available on the relative efficacy and bleeding complications.
The usual duration of thromboprophylaxis is 10 ± 3 days. Shorter durations have been used by some in clinical practice, but without sound evidence to support any benefits of this strategy. 16,17 On the other hand, a prolongation of prophylaxis, which has been suggested for patients with major orthopedic surgery, should be performed only in those patients who are at increased risk of VTE. 1,18,19
Limitations
The fact that SMART had no control group and unknown biases, for example, through patient selection, may have interfered with the results, making comparisons to other data more difficult. A further potential limitation is that we focused on symptomatic VTE only, although asymptomatic VTE are more common and are also relevant for the evaluation of anticoagulants’ efficacy as suggested by Mismetti et al. 3 On the other hand, SMART has been an NIS in an outpatient setting after general or orthopedic surgery, focusing on documenting actual treatment patterns. This implies a high external validity with direct relevance of the results for physicians treating these patients. 20 Compared to randomized controlled trials, a number of “critical patients” included into the NIS may have renal insufficiency, a high body weight, a number of coadministered platelet inhibitors, or nonsteroidal anti-inflammatory drugs. Further patients were observed in an outpatient setting as opposed to the close monitoring in typical clinical trials.
Conclusions
The present study results demonstrate that it is effective and tolerable to use a risk-stratified dose of 20 or 40 mg enoxaparin in patients undergoing day surgery.
Footnotes
Acknowledgments
The support of the participating physicians, their staff, and patients is acknowledged. The study was conducted by Sanofi Aventis Deutschland GmbH.
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. SH and PB report to have received compensation for consulting and lecture honoraria from Sanofi Aventis. VH has been an employee of Sanofi Aventis until 2010.
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: sponsored by Sanofi Aventis Deutschland GmbH.
