Abstract
The purpose of this study was to determine the effect of ABO blood groups on von Willebrand factor–ristocetin cofactor activity (vWF-RCo) and on vWF–antigen (vWF-Ag) in children who have no personal or familial history of bleeding. Material and methods: A survey and testing were performed on 200 children with no personal or familial history of bleeding. In all, 100 of them belonged to blood group O, and the remaining 100 belonged to other blood groups. The blood samples were stored at −80°C for a maximum period of 2 weeks to detect vWF-RCo and vWF-Ag levels. Results: The mean vWF-Ag (±2 standard deviation [SD]) level in children with blood group O was 86% (±20%); and for those with non-O blood group, it was 98.8% (±25%). There was a significant difference between the 2 groups (P < .001). The mean vWF-RCo (±2 SD) level in children with blood group O was 89% (±23%); and for those with non-O blood group, it was 103% (±17%). There was a significant difference between those in the 2 groups (P < .001). The lowest value of vWF-Ag and vWF-RCo levels in children with blood group O was found to be 50%. In conclusion, we showed that the selection of normal ranges based on the ABO group might influence the clinical diagnosis of vWD and that while the approach of using ABO group ranges for a vWF-Ag level lower than 50 IU/dL is scientifically sound, it might not be useful to assist a clinician in identifying people at increased risk of bleeding.
von Willebrand factor (vWF) is a blood glycoprotein essential for normal hemostasis. The mechanisms that control the plasma vWF level are not understood. Researchers have reported that genetic factors account for 66% of the variance in vWF levels and that ABO blood group differences are a major influence. 1 An association between ABO and vWF levels has been acknowledged for many years. On average, the vWF–antigen (vWF-Ag) level in adults is approximately 25% to 30% lower in persons with the blood group O compared to those with other blood groups. 2,3 Low vWF levels are associated with bleeding and lead to the hemorrhagic disorder, von Willebrand disease (vWD).
Mild type 1 vWD (vWF levels ∽25%-50%) causes special problems. One of the main difficulties is differentiating between mild type 1 vWD and normal participants with blood group O. Although it is well known that plasma vWF levels vary according to ABO blood group, the value of using normal reference ranges adjusted for ABO group remains unresolved and is a matter of ongoing debate. 4–6
The purpose of this study was to determine the effect of ABO blood groups on vWF–ristocetin cofactor activity (VWF-RCo) and vWF-Ag in children who have no personal or familial history of bleeding.
Materials and Methods
A survey and testing were done on 200 children with no personal or familial history of bleeding for enrolling in the study. In all, 100 of them belonged to blood group O, and the remaining 100 belonged to other blood groups. (Blood types A, B, or AB were classified as “non-O” for analysis.)
Approval of a family member was required before including a child to the study. Plasma samples were obtained from 100 children with blood group O and 100 children with non-O blood group. Their ages were 2 to 16 years. After filling out a standardized questionnaire, those who indicated they had a positive personal or family history of bleeding were excluded.
Their parents filled out a survey containing the following questions: have you or a blood relative ever needed medical attention for a bleeding problem or been told that you have a bleeding disorder or problem? Have you experienced a bleeding problem in the following circumstances: during or after surgery (particularly circumcision), during dental procedures or extractions, after a trauma, and heavy menses. Have you ever had bruises with lumps? Have you experienced epistaxis (nose bleed)?
Blood samples were drawn from each participant’s antecubital vein using the BD Vacutainer system (Becton-Dickinson; Plymouth, UK). Tests for blood group, complete blood count (CBC), prothrombin time (PT), and activated partial thromboplastin time (aPTT) were done on all samples. If children’s CBC, PT, and aPTT values were not within the age-specific reference range, they were excluded from the study.
The following are the exclusion criteria for vWF assays in children: fasting, unavailibility of an acute or chronic inflammatory, menses, 30 minutes struggling, crying, and exercise (these situations may falsely elevate vWF activity).
For testing the vWF activity 8 mL of whole blood specimens were drawn into 0.109 mol/L (3.2% weight/volume [w/v]) buffered sodium citrate collection tubes and kept at room temperature. Hemolyzed or clotted samples were not used. Plasma was separated from blood cells promptly at room temperature. Then the blood sample was stored at −80°C for a maximum period of 2 weeks to detect vWF-RCo and vWF-Ag levels. Blood grouping was performed using the routine blood banking practice at the Blood Bank, Ege University Hospital. Prothrombin time was measured using Thromborel S and was reported as PT (in seconds) and as an international normalized ratio (INR). An aPTT assay was performed using Pathromtin SL (Dade-Behring, Germany). Both PT and APTT were assayed by automatic coagulation machines (Sysmex, CA-1500 (Dade-Behring, Leusden, The Netherlands)).
The vWF-RCo levels (normal ranges: 60%-120%) were detected using aggregometry (Bio/Data Corporation, Horsham, Pennsylvania). The vWF-Ag levels (normal ranges 60%-150%) were determined through a latex immunoassay using the STA-Compact analyzer (Diagnostica Stago, Asniéres, France).
Statistics Analysis
The patients included in the study were categorized into either the O or non-O blood groups for measuring vWF-Ag and vWF-RCo. Reference ranges were calculated as ±2 standard deviation (SD) to generate 95% confidence intervals for vWF-Ag and vWF-RCo levels for O and non-O blood group, using a 2-tailed distribution with 2.5% of the population expected to be in the lower tail. The significance level was set at .01. Statistical software (SAS, Version 9.1.3; SAS Institute, Inc., Cary, North Carolina) was used for data processing and analysis. P values <.05 were considered to be statistically significant.
Results
Results of the tests of 200 healthy children and adolescents are shown in Table 1 .
Reference Intervals for vWF-Ag, vWF-RCo, and Relationship to ABO Blood Group for Children Without Bleeding Symptoms
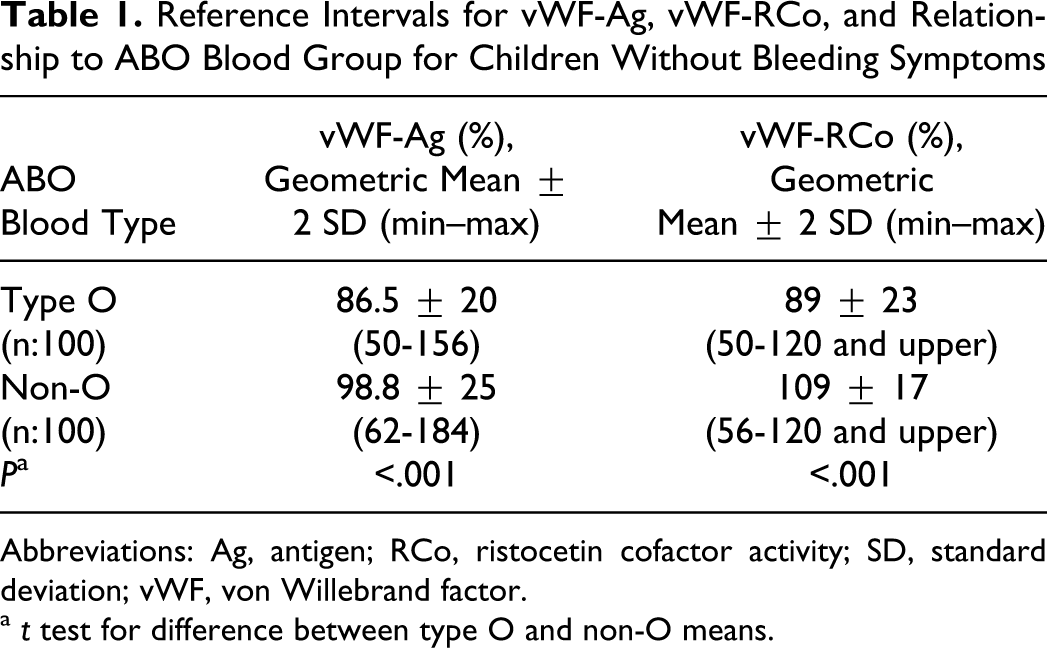
Abbreviations: Ag, antigen; RCo, ristocetin cofactor activity; SD, standard deviation; vWF, von Willebrand factor.
a t test for difference between type O and non-O means.
There were no age-related or gender differences between O blood group (median age, 8.9 years; age range, 2-17 years; and gender F/M: 49/51) and non-O blood group (median age, 7.4 years; age range, 2-15 years; and gender F/M: 52/48).
The mean vWF-Ag (±2 SD) level in children with O blood group was 86% (±20%) and for those in non-O blood group, it was 98.8% (±25%). There was a significant difference between the 2 groups (P < .001).
The median values of vWF-Ag showed that there were approximately 15% higher levels in non-O blood group compared to O blood group.
The mean vWF-RCo (±2 SD) level in children with O blood group was 89% (±23%), and with non-O blood group, it was 103% (±17%). There was a significant difference between those in the 2 groups (P < .001).
The median values of vWF-RCo showed there were approximately 15% higher levels in non-O blood group compared to O blood group. The lowest value for vWF-Ag and vWF-RCo level in children with blood group O were 50%. The lowest 5 values for vWF-Ag in children with O blood group were 50%, 50%, 50%, 51%, and 50%, respectively. The lowest 5 values for vWF-RCo in children with O blood group were 50%, 50%, 50%, 51%, and 52%, respectively. The lowest 5 values for vWF-Ag in children with non-O blood group were 62%, 62%, 62%, 64%, and 64%, respectively. The lowest 5 values for vWF-RCo in children with non-O blood group were 56%,62%, 64%, 67%, and 68%, respectively.
Discussion
We evaluated the influence of the ABO blood type on plasma vWF-Ag and vWF-RCo levels in healthy children with no bleeding symptoms. The plasma vWF concentration varies widely in healthy individuals, and the most important genetic modifier of plasma vWF levels, other than mutations within the vWF gene, may be the ABO blood group. 1,7 The majority of the variability found in vWF-Ag remains unexplained. The amount of A and B antigens expressed on vWF explains approximately 18% of the variation in vWF levels. 8 Approximately 70% of those with type 1 vWD were in O blood group. 2,4
In our previous study, 62% of those in the O blood group ratio were type 1 vWD patients. 9 Since the O blood group had a higher number of patients with type 1 vWD than found in the normal population, a recurring question is where to draw the line between the lower end of the ABO-specific vWF reference range of the healthy population and the type 1 vWD diagnosis. 10
The diagnosis of moderate or mild type 1 vWD is problematic when detected vWF levels are within the range 25% to 60% because of an overlap of test results with normal individuals (ie, those presenting with similar assay values but without a clinical or family history of bleeding). 2,11
We confirmed a clear association between ABO blood group status and levels of vWF. In children, we confirmed the findings of Klarmann et al that mean vWF-Ag values were significantly lower in members with O blood group than in members with non-O group, which is similar to the trends in previous studies of adults reported in Table 2 . 8,12,13 A study by Gallinaro et al demonstrated that the mechanism responsible for the effects of a blood group on lower plasma vWF levels is characteristic of individuals with the O blood group, due to a shorter vWF survival rate, mainly attributable to a faster clearance, and that circulating levels of vWF are greatly influenced by its half-life. 13 Plasma average vWF levels of O group were generally lower than those in non-O group, but this was unlikely to be clinically significant.
The Effect of Blood Groups on Mean vWF-Ag Levels in Children and Adults in Studies in the Literature
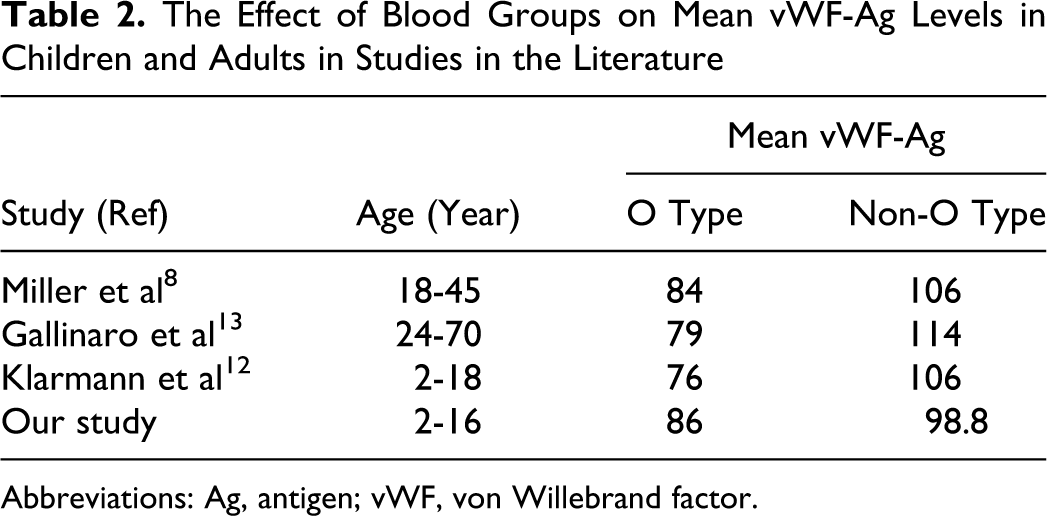
Abbreviations: Ag, antigen; vWF, von Willebrand factor.
In most previous ABO-related studies, the focus has been on the effect on the bottom end of the normal range and the influence on vWD diagnosis. 4,14 A vWF level lower than 50 IU/dL is often considered to be the cutoff when considering a diagnosis of vWD. 15 Our study and previous studies have showed that vWF levels in healthy children without bleeding in O blood group was not lower than 50 IU/dL. 12,16 Davies showed that 2.4% of O blood group donors had a vWF level of 50 IU/dL or lower. Coinheritance of O blood group and C1584 markedly increases the likelihood of a low vWF level that could lead to a false-positive diagnosis of type 1 vWD if a patient by chance had a bleeding history. 17,18
For patients with low vWF levels who are in the O blood group, a DNA analysis is not useful to distinguish whether they exhibit type 1 vWD according to a study of the effect of molecular and clinical markers on diagnosis and management of vWD type 1 (MCMDM-1vWD). It showed that the proportional linkage of vWF-Ag level between 35 and 50 IU/dL was only 0.51. 19 Recent studies of vWD Type 1 revealed that a combination of bleeding symptoms and a vWF-Ag level lower than 50 IU/dL is the key to an accurate diagnosis. 20
In conclusion, we showed that the selection of normal ranges based on the ABO group might influence the clinical diagnosis of vWD and that while the approach of using ABO group ranges for a vWF-Ag level lower than 50 IU/dL is scientifically sound, it might not be useful to assist a clinician in identifying people at increased risk of bleeding.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
