Abstract
We evaluated the use of the turbidimetric HemosIL von Willebrand Factor (VWF) Activity assay (VWF:Act) on the STA-R automated coagulometer (Stago, Asnières, France) for the diagnosis of von Willebrand disease (VWD). For this, we prospectively screened 268 patients. As a second part, we retrospectively assayed 111 patients with well-defined VWD subtype. In the first prospective study, we demonstrate that in most cases of VWD, VWF ristocetin cofactor activity (VWF:RCo) and VWF:Act are highly correlated but that they both cannot be considered a good screening assay when used alone, since they could miss about 25% of VWF abnormalities. However, the association of VWF:Act analysis and the Platelet Function Analyzer-100 (PFA-100) test constitutes an excellent screening strategy. In our second retrospective study concerning VWD subtypes, VWF:RCo and VWF:Act were well correlated but could be very discrepant, especially for some cases of type 2M VWD. We consider that VWF:RCo remains the “reference assay” for VWD subtype classification.
Introduction
von Willebrand disease (VWD), the most commonly inherited bleeding disorder, is caused by a quantitative or qualitative defect of von Willebrand factor (VWF), leading to easy bruising, epistaxis, excessive postoperative or posttraumatic bleeding, gum bleeding, and menorrhagia.
1
Today, making the diagnosis of VWD remains a challenge and requires the correlation of clinical findings with multiple laboratory test results. In theory, VWD could be routinely screened by measuring bleeding time; however, many studies have proven this test insufficiently sensitive for this application. The rapid, simple, and sensitive PFA-100 test (Platelet Function Analyzer-100, Siemens Healthcare Diagnostics, Marburg, Germany) has improved screening and is able to reliably exclude VWD (except type 2N) if results fall within the normal range.
2
To classify VWD in terms of subtype, several biological assays are needed to assess the specific VWF abnormality. The classification of VWD distinguishes quantitative (type 1 or the most rare and severe type 3) and qualitative (type 2) defects.
3
The evaluation of VWF ristocetin cofactor activity (VWF:RCo) together with VWF antigen levels (VWF:Ag) are the most common assays for the diagnosis of VWD. The VWF:RCo/VWF:Ag ratio classically allows the discrimination between type 1 and type 2. For type 2, distinct mutations have been found in the
The VWF:RCo measurement is a very delicate and labor-intensive assay that has poor reproducibility. 5 Recently, the use of a turbidimetric and automated HemosIL VWF Activity assay on ACL TOP or ACL-Futura automated coagulometers (Instrumentation laboratory, Lexington, Massachusetts) has been proposed as an alternative and reliable screening tool in the diagnosis of VWD in patients with a bleeding diathesis6–9 or for the evaluation of VWF activity in concentrates. 10 This reagent contains latex particles coated with a monoclonal anti-VWF antibody directed against VWF’s GPIb binding site. The degree of agglutination is, therefore, directly proportional to VWF.
The aim of this prospective study was to evaluate the use of a turbidimetric HemosIL VWF Activity assay (VWF:Act) on the STA-R automated coagulometer (Stago, Asnières, France) as a potential screening test for the diagnosis of VWD in our routine diagnostics laboratory. VWF:Act results were compared with VWF:RCo levels for all patients and then for each group of patients according to disease subtype.
Materials and Methods
To determine the potential of VWF:Act assay for use in VWD screening, we prospectively studied 268 consecutive patients referred for suspicion or follow-up of VWD in 2 reference medical consultations in Lille (n = 123) or Nantes (n = 145) hemophilia centers. Plasma was collected, respectively, in 0.129 mol/L or in 0.109 mol/L buffered citrate. For all patients, VWF:Act was measured using the HemosIL VWF Activity assay on a STA-R automated coagulometer (Stago) and the VWF:RCo measured by aggregometry using the BC von Willebrand Reagent (Siemens Healthcare Diagnostics). VWF:Ag levels were measured using the Liatest VWF turbidimetric assay (Stago) in the Nantes center or VWF:Ag reagent (Siemens Healthcare Diagnostics) on STA-R (Stago) in the Lille center. VWF methods were performed in duplicate to avoid within-run imprecision. A global evaluation of primary hemostasis was assessed in 248 patients using epinephrine and ADP cartridges on a PFA-100 device (Siemens Healthcare Diagnostics) and was considered abnormal if at least 1 closure time was prolonged. The blood type was systematically determined in cases of low VWF levels. Both VWF:RCo/VWF:Ag and VWF:Act/VWF:Ag ratios were calculated to evaluate their respective interest for use in screening. Quality control was performed according to the laboratory’s operating procedures and as recommended by the manufacturers for all assays. For each patient, a definitive conclusion in terms of a “VWD” or “not VWD” diagnosis was drawn by a hematologist, based on biological test results, clinical data, and blood group.
To evaluate the interest in using VWF:Act assay in VWD classification, 111 patients previously diagnosed as carrying a VWF deficiency based on VWF:RCo levels were retrospectively studied and results compared. Plasma samples were frozen at −80°C before performing both methods. In all cases of type 2 VWD, molecular analysis allowed a precise diagnosis: type 2A (n = 17); type 2B (n = 9); type 2M (n = 22), VWD 3 (n = 3); type 2A/2M (n = 1): VWD 2M/2B (n = 4); VWD 2N/3 (n = 4); VWD 2 “unclassified” (n = 15) according to the Sheffield VWF database (http://www.vwf.group.shef.ac.uk). Type 1 VWD (n = 36) was defined as proposed by Sadler et al 3 with VWF:RCo < 30 IU/dL and a normal VWF:RCo/VWF:Ag ratio.
Mann-Whitney and Spearman tests were used to perform statistical analysis. Concordance between the 2 methods was analyzed by graphical approach according to Bland and Altman’s method.
Results
Patients
In the first prospective study, among the 268 patients referred for VWD screening, 155 (58%) were considered normal, whereas 113 (42%) were diagnosed as having a VWF deficiency by an expert, according to biological test results and blood group. Mean values for FVIII:C, VWF:Ag, VWF:RCo, and VWF:Act are given in Table 1
. Values could be found to be >100 IU/dL, even in patients with VWD, due to pregnancy or inflammation, something that expert physicians should take into account for diagnosis. As expected, however, there is an overall significant decrease (
Values (Mean ± SD; Range) of FVIII:C, VWF:Ag, VWF:RCo, VWF:Act, VWF:RCo/VWF:Ag Ratio, VWF:Act/VWF:Ag Ratio, and Blood Group According to Diagnosis in 268 Patients Screened for VWD
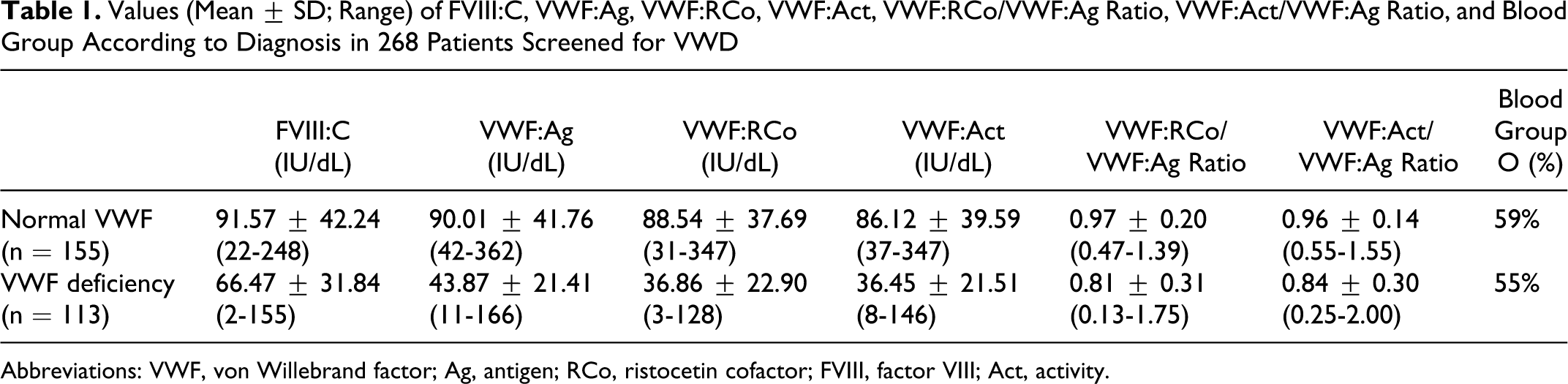
Abbreviations: VWF, von Willebrand factor; Ag, antigen; RCo, ristocetin cofactor; FVIII, factor VIII; Act, activity.
VWF: RCo and VWF: Act Assay Comparison
In the first prospective study, we found no statistically significant difference between VWF:RCo and VWF:Act, with both values being very well correlated (
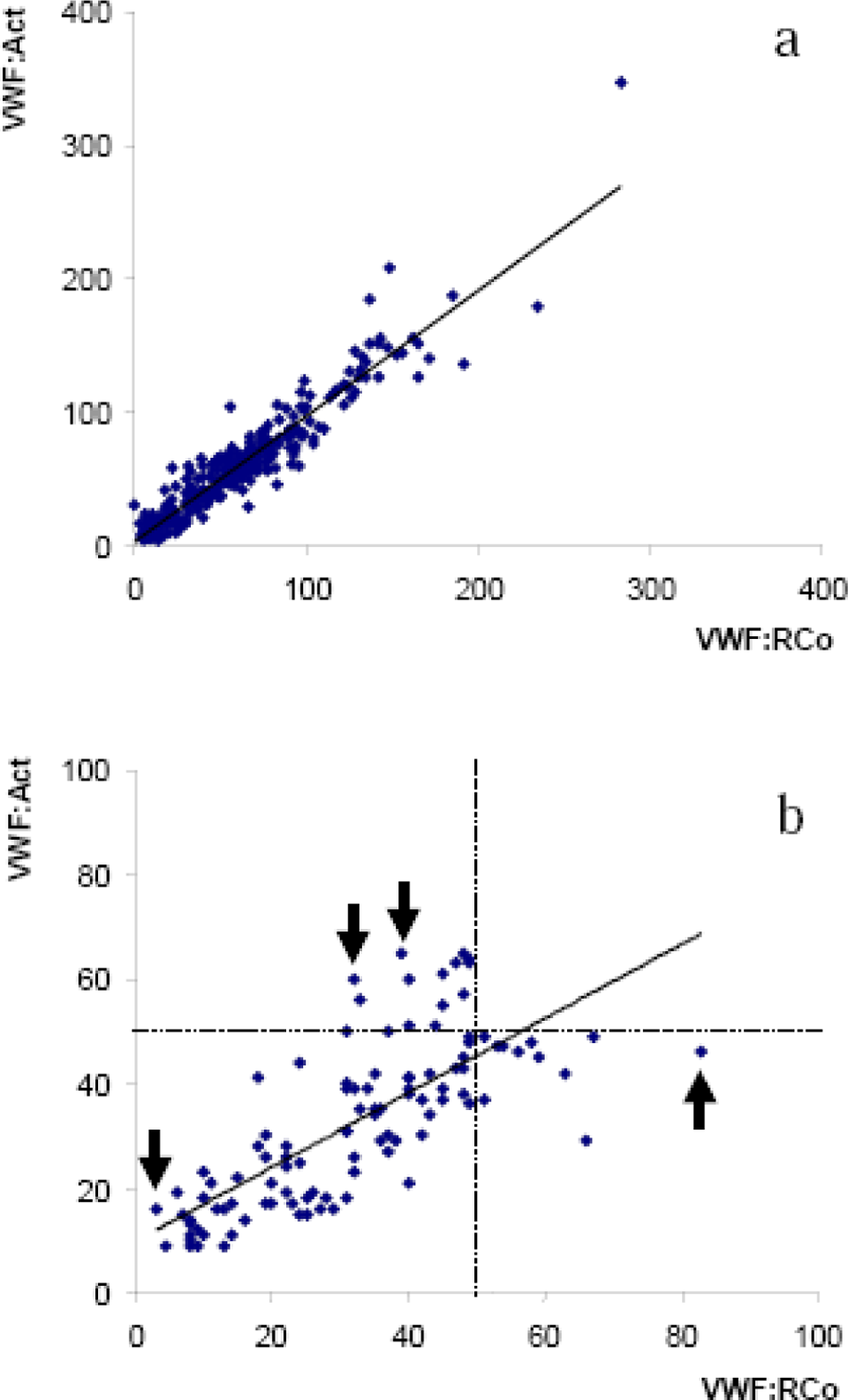
A, Correlation between VWF:RCo and VWF:Act in 268 patients tested for VWD screening. B, Correlation between VWF:RCo and VWF:Act in the 92 patients with, at least, 1 VWF value <50 IU/dL. Arrows indicate patients with particularly highly discrepant results (VWF:RCo vs VWF:Act: 3 IU/dL vs 16 IU/dL; 33 IU/dL vs 56 IU/d; 39 IU/dL vs 65 IU/dL, or 83 IU/dl vs 46 IU/dL). VWF indicates von Willebrand factor; RCo, ristocetin cofactor; Act, activity; VWD, von Willebrand disease.
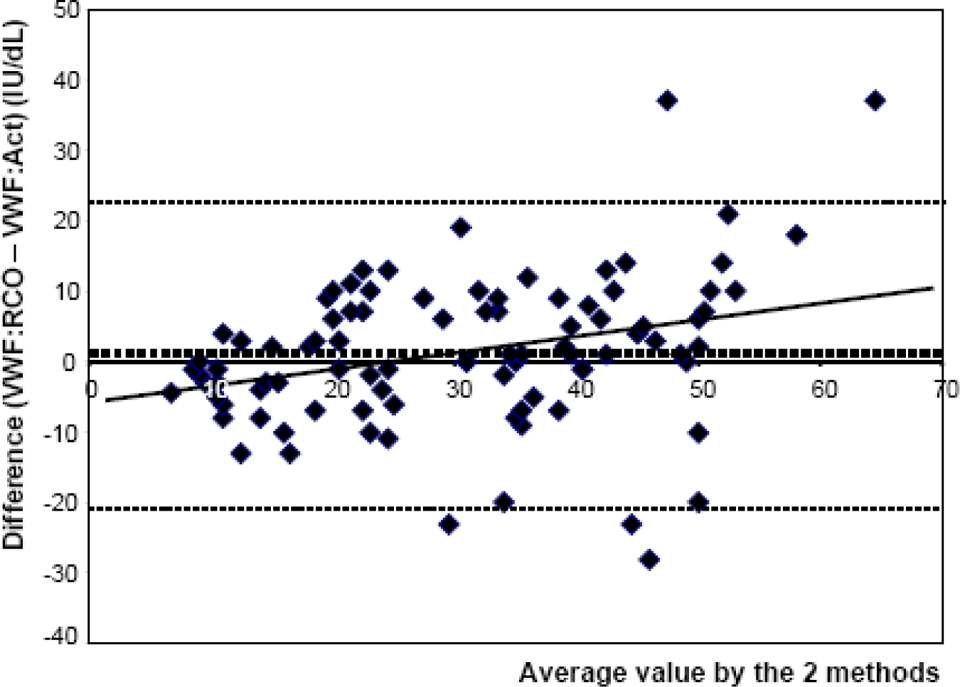
Bland-Altman plot comparing VWF:RCo and VWF:Act on patients with, at least, 1 VWF value <50 IU/dL. Dotted Lines indicate mean ± 2 standard deviations. VWF indicates von Willebrand factor; RCo, ristocetin cofactor; Act, activity.
When used alone, VWF:RCo and VWF:Act analyses have the same level of performance in VWD screening, with a sensitivity about 75%, that is they can miss about 25% of VWD. The association of these methods with the PFA-100 test, however, considerably improves their performance (Table 2).
Values of PPV, NPV, Specificity, and Sensitivity According to Screening Test (VWF:RCo, VWF:Act, PFA-100 Alone or in Association With PFA-100 Results) a
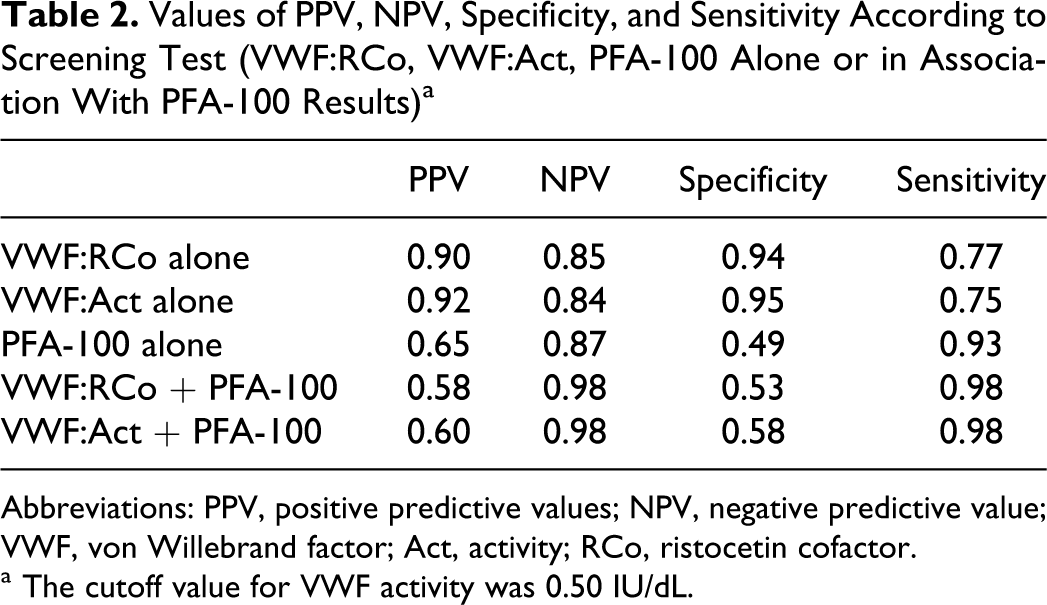
Abbreviations: PPV, positive predictive values; NPV, negative predictive value; VWF, von Willebrand factor; Act, activity; RCo, ristocetin cofactor.
a The cutoff value for VWF activity was 0.50 IU/dL.
VWF: RCo and VWF: Act Assays in VWD Classification
Concerning the retrospective study of 111 patients with identified VWD subtypes, VWF:RCo and VWF:Act are well correlated (
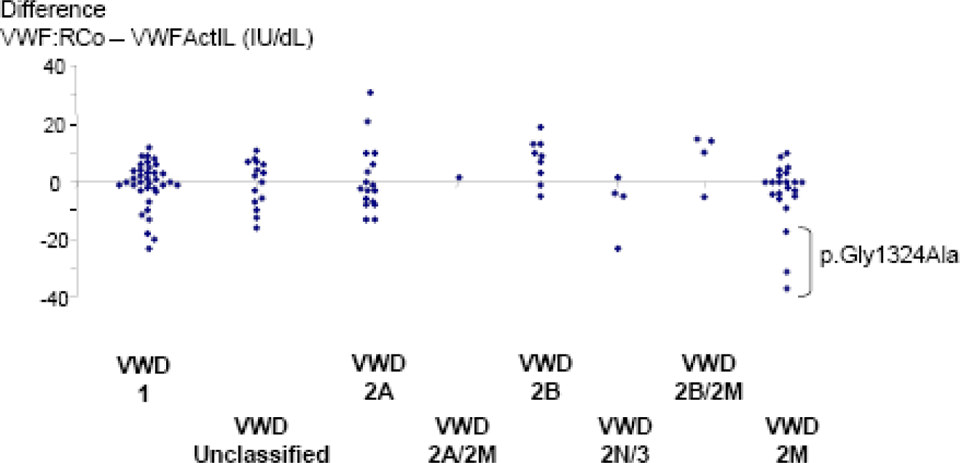
Differences between VWF:RCo and VWF:Act according VWD subtypes. Results from patients with p.Gly1324Ala mutation are specified in type 2M VWD subtype. VWF indicates von Willebrand factor; RCo, ristocetin cofactor; Act, activity.
Discussion
Here we have first demonstrated that in most cases of VWD, values for VWF:RCo and VWF:Act are highly correlated and that the turbidimetric HemosIL VWF Activity assay can be considered “good” as VWF:Rco assay, since they could both miss about 25% of VWF abnormalities when used alone for screening. PFA-100 system is usually reputed very reliable for VWD screening. 2 In this prospective study, we could calculate the sensitivity for VWD of 93%, but a poor specificity of 49%. However, we suggested that the association of VWF:Act or VWF:RCo assay and PFA-100 test results constitutes an excellent screening strategy with better specificity and sensibility and, overall, an excellent negative predictive value (NPV). We went on to demonstrate that for each of the different subtypes, VWF:RCo and VWF:Act were generally well correlated but could be very discrepant, as was particularly true for some cases of type 2M VWD.
To date, our clinical evaluation of the HemosIL VWF Activity assay is the most rigorous since 2 aspects of VWF activity determination have been investigated: first, its use in screening for VWD in a prospective population and its interest in differentiating among disease subtypes. All diagnoses were established as proposed by Sadler et al 3 by expert physicians in the 2 reference centers of hemostasis, that is these diagnoses were not only based on the presence of VWF deficiency according blood group but also on clinical and familial history, which must be associated to biological results to assert VWD. In our experience, we found the results of the HemosIL VWF Activity assay to be highly reproducible (data not shown).
The VWF:RCo assay explores the interaction of VWF with platelet GPIbα and remains the standard method used for measuring VWF activity.3,11 It is based on the property of the antibiotic ristocetin to agglutinate formalin-fixed normal platelets in the presence of VWF. This method is classically performed using platelet agglutination 12 but can be also automated.13,14 This method does, however, remain difficult to standardize and the between-laboratory variation remains too high and the within-laboratory repeatability not satisfactory. 5 The HemosIL VWF activity assay, use coated latex particles in a turbidimetric method and can be automated, giving much better coefficients of variation than can be obtained with VWF:RCo measurements. 9
Although a better correlation was found between VWF:Act and GP Ib-binding enzyme-linked immunosorbent assay (ELISA) or VWF:CB assays, 6 VWF:Act and VWF:RCo values were usually well correlated in healthy controls and in patients.6,7,9 Using a cutoff value of 50 IU/dL for VWF:Act, De Vleeschauwer et al demonstrated the VWF:Act assay to have a sensitivity of 94.1% and a specificity of 92.8%, thus suggesting its potential usefulness in screening VWD. 9 In our experience with a similar cutoff value of 50 IU/dL however, the sensitivity is not so good, calculated as 75%. As a screening test, the performance of VWF:Act is significantly improved (up to 98%) when combined with a PFA-100 test. Our results show that VWF:RCo levels follow a similar pattern, however confirmed VWF abnormalities have been associated with VWF:RCo > 50 IU/dL (particularly in type 2B VWD). Favaloro et al, thus, proposed that, if only 1 functional VWF test is to be performed, VWF:CB must be the preferred choice since measuring only the VWF:RCo will miss a substantial number of type 2 VWD individuals and could misidentify many others. 13 Today, the best screening test to eliminate VWD appears to be the PFA-100 test, however the specificity is poor.2,13
Results from VWF:Act assay have previously been shown to be well correlated with those of the standard VWF:RCo assay in all type 1 and 2 VWD variants. 7 Our retrospective study of 111 patients with well-identified VWD subtypes confirmed that VWF:RCo and VWF:Act are similar and well correlated. However, for certain patients, the results were very discordant with some cases of type 2M VWD being associated with normal VWF:Act. This could lead to major misdiagnosis. However, the fact that we had previously used VWF:RCo to detect and classify the patients with VWD in the retrospective study could be considered a flaw in the selection of the patients. Furthermore, concerning the family with a p.Gly1324Ala missense mutation for which we underlined very discrepant results, Flood et al recently proposed that a similar phenotype with decreased VWF:RCo could represent an artifact of measurement without any VWF dysfunction. 15 More physiologically relevant assays for VWF function need to be developed
In conclusion, the use of the HemosIL VWF Activity assay offers an automated assay to measure VWF activity with results that are well correlated with VWF:RCo. As previously shown by De Vleeschauwer et al in 2006, methodological considerations are in favor of VWF:Act, since the coefficients of variation appear to be much better. 9 However, a screening test should be very reliable and, in our experience, VWF activity determination by HemosIL VWF Activity assay alone does not appear sufficient for VWD diagnosis and needs to be combined with a PFA-100 test.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
