Abstract
Background
The benefit of acetylsalicylic acid (ASA) treatment is well established in several subgroups of patients with coronary artery disease (CAD). Many patients, however, suffer recurrent cardiovascular events despite antiplatelet therapy. Persistent high residual platelet reactivity (RPR) despite ASA therapy—a laboratory-defined phenomenon—was shown to correlate with higher risk of recurrence of clinical cardiovascular events.1–4 The extent of platelet function suppression by ASA have been shown to vary between individuals. There are several reasons for an inadequate suppression of platelet aggregation ranging from patient’s noncompliance, inadequate dosage of the drug, poor absorption from gastrointestinal tract, interaction with other drugs, increased cyclooxygenase 2 (COX-2) gene expression, synthesis of thromboxane A2 (TXA2), increased catecholamine release due to exercise and mental stress, increased platelet aggregation triggered by cigarette smoking, erythrocyte-enhanced platelet aggregation, increased platelet sensitivity to collagen, and genetic polymorphisms (platelet glycoprotein IIIa [GpIIIa], COX, and ADP receptor P2Y1 [P2Y1]).5–7 Degree of platelet response to ASA therapy can be characterized as a normal, bell-shaped (Gaussian) distribution and dichotomic labeling as “responder” or “non-responder” is arbitrary and based upon different cutoff points of different platelet function tests. Laboratory documentation of platelet aggregation beyond an arbitrary cutoff point, which remains despite ASA administration, should be correctly termed “high residual platelet reactivity” and should be differentiated from “ASA resistance.” Characterization of “resistant” status is typically based on a single determination of platelet function, with the underlying assumption that this determination represents a stable phenotype. 8
There are, however, still some concerns about the consistency and reliability of the laboratory tests for the antiplatelet treatment assessment. The aim of our study was to assess the long-term reproducibility (LTR) and short-term reproducibility (STR) of light transmission aggregometry (LTA) in patients treated with ASA.
Materials and Methods
Study Population
Consecutive patients admitted to the Department of Cardiology on chronic ASA therapy (100 mg once daily [od] for at least 5 days) were prospectively enrolled into the long-term part of the study. Altogether 207 patients, 56 females, 151 males, age 43 to 85 (mean 67 ± 9) years were included. Residual platelet reactivity was assessed in all the patients using LTA, with second control LTA being carried out at a 10- ± 6-month interval.
In all 15 additional participants were examined in the second part of the study in order to assess the short-term intraindividual variability of the LTA measurements. Correlation between LTA RPR measurement with cationic propyl gallate (CPG) and Aspirin (ASPI) test with arachidonic acid (AA) on Multiplate analyser (Dynabyte) was also assessed in this subgroup.
Exclusion criteria were contraindications to antiplatelet therapy, hemodynamic instability, thrombocytopenia, thrombocytosis, administration of GpIIb-IIIa inhibitors, anemia, polycythemia, end-stage renal disease, hematologic diseases and malignancies, noncompliance, and reluctance to sign informed consent.
Methods
Basic clinical data, resting electrocardiogram (ECG), and biochemistry samples were obtained in all patients. Compliance to ASA intake was confirmed in all participants (basic assessments were made in hospitalized patients and correct drug intake was assured by direct supervision of an attending nurse in order to exclude patients with noncompliance). Light transmission aggregometry was performed in all patients at the time of primary hospitalization. The dose of ASA was unchanged at the second control. We asked all the patients by a special letter to take ASA thoroughly before the control examination and then the ASA intake for the control examination was verified in all patients by self-reported adherence to therapy. Only patients with confirmed ASA intake were included.
For the short-term intraindividual variability of the LTA, blood samples were drawn in exactly the same manner in the morning on 2 consecutive days and 5 measurements were performed from each sample. Laboratory personnel performing the assessment were blinded to the origin of the samples. All patients signed informed consent and the study was approved by Institutional Ethical Committee. The study complies with the Declaration of Helsinki.
Blood Samples for LTA
Whole blood was obtained from peripheral vein of each patient under standard conditions. The patients were fasting for at least 8 hours before blood withdrawal. Two blood samples were collected in 3 mL tubes with 3.8% sodium citrate (buffered, pH 5.5). Platelet-rich plasma (PRP) and platelet-poor plasma (PPP) were prepared by centrifugation for 10 minutes. The centrifugation speed was 114 to 145g and 2191 to 2862g for PRP and PPP, respectively. The platelet count was adjusted to 100 × 109/L in PRP. The entire determination of aggregation response was performed on the optic aggregometer APACT (APACT 4—American Labor Pool, San Diego, California; software: APACT Application, version 1.4). The platelet aggregation response was induced by CPG in a concentration of 300 μmol/L. Aggregation curve was constructed and the slope of platelet aggregation onset was assessed in PRP at 37°C with PPP serving as the reference. Cutoff value of 65%per min and more was considered as a high RPR.
Platelet Aggregation Assessment With AA
Platelet aggregation was assessed also with Multiplate analyser (Dynabyte) based on impedance aggregometry. Blood was then collected into 4.5 mL tubes containing 25 μg/mL hirudin (Dynabyte) as an anticoagulant. For measurement, 300 μL of preheated saline (37°C) and 300 μL hirudin-anticoagulated whole blood were placed into the test cell, and the sample was stirred using a teflon-coated electromagnetic stirrer (800 rpm) over a 3-min incubation period. Platelet aggregation was initiated using AA (ASPI test, 0.5 mmol/L). Increased impedance due to attachment of platelets to the electrodes was continuously measured for each sensor unit over a period of 6 minutes. Data were transformed to arbitrary Arbitrary Units (AU) and plotted as 2 separate aggregation curves versus time. Aggregation measured by Multiple Electrode Aggregometry (MEA) was quantified as the area under the aggregation curve (AU*min). Alternatively, the software of the analyzer allows for the expression of area under the curve (AUC) values in units (U), where 10 AU*min correspond to 1 U.
Statistical Analysis
All data were processed with Microsoft Excel. Categorical variables are presented as frequencies and percentages. Mean values and standard deviations were calculated for continuous variables. Intraclass correlation coefficient (ICC) was calculated to assess the degree of correlation between baseline and control sets of RPR measurements. Reproducibility was also calculated using Bland-Altman plots. 9 In Bland-Altman graphical method the differences between the baseline and control measurements were plotted against the averages of these 2 measurements. Horizontal lines were drawn at the limits of agreement, which were defined as the mean difference ± 1.96 times the standard deviation of the differences. A P value ≤.05 was considered significant for all calculations.
Results
Long-Term Reproducibility
Baseline demographic characteristics of the studied cohort are shown in Table 1 .
Baseline Demographic Characteristics of the Studied Cohort in the Long-Term Part
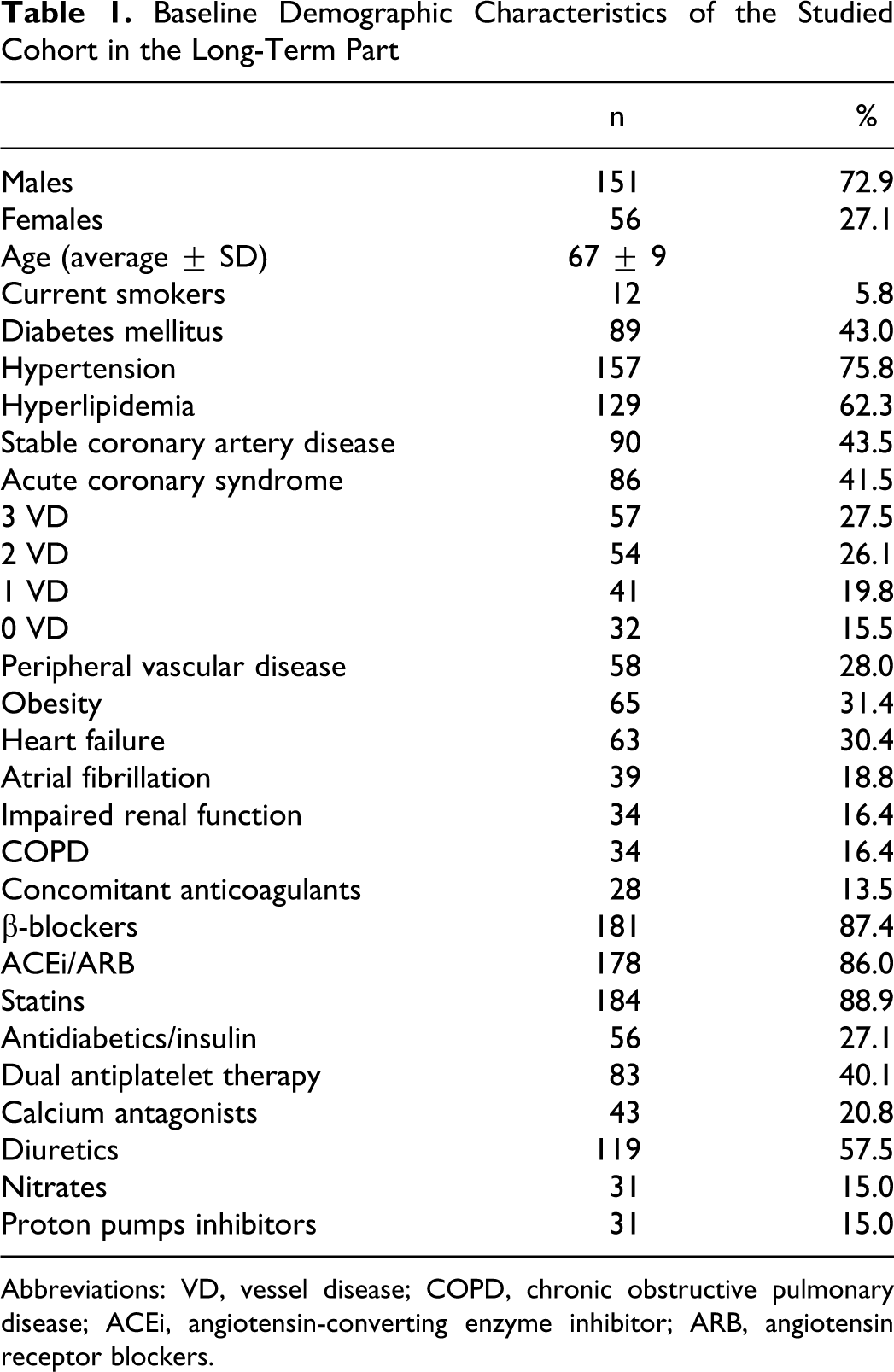
Abbreviations: VD, vessel disease; COPD, chronic obstructive pulmonary disease; ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blockers.
All the 207 consecutive patients were retested for platelet function. In all 171 (82.1%) patients had both measurements within the limits of adequate platelet suppression during the long-term part of the study. In all, 24 patients (11.6%) had inadequate platelet inhibition during the initial assessment, 11 patients (5.3%) during the control follow-up, and only 2 patients (1.0%) were consistently showing high RPR on both occasions. There was no linear correlation between both measurements (Figure 1 ) and also Bland-Altman plot showed typical diverging pattern (Figure 2 ).
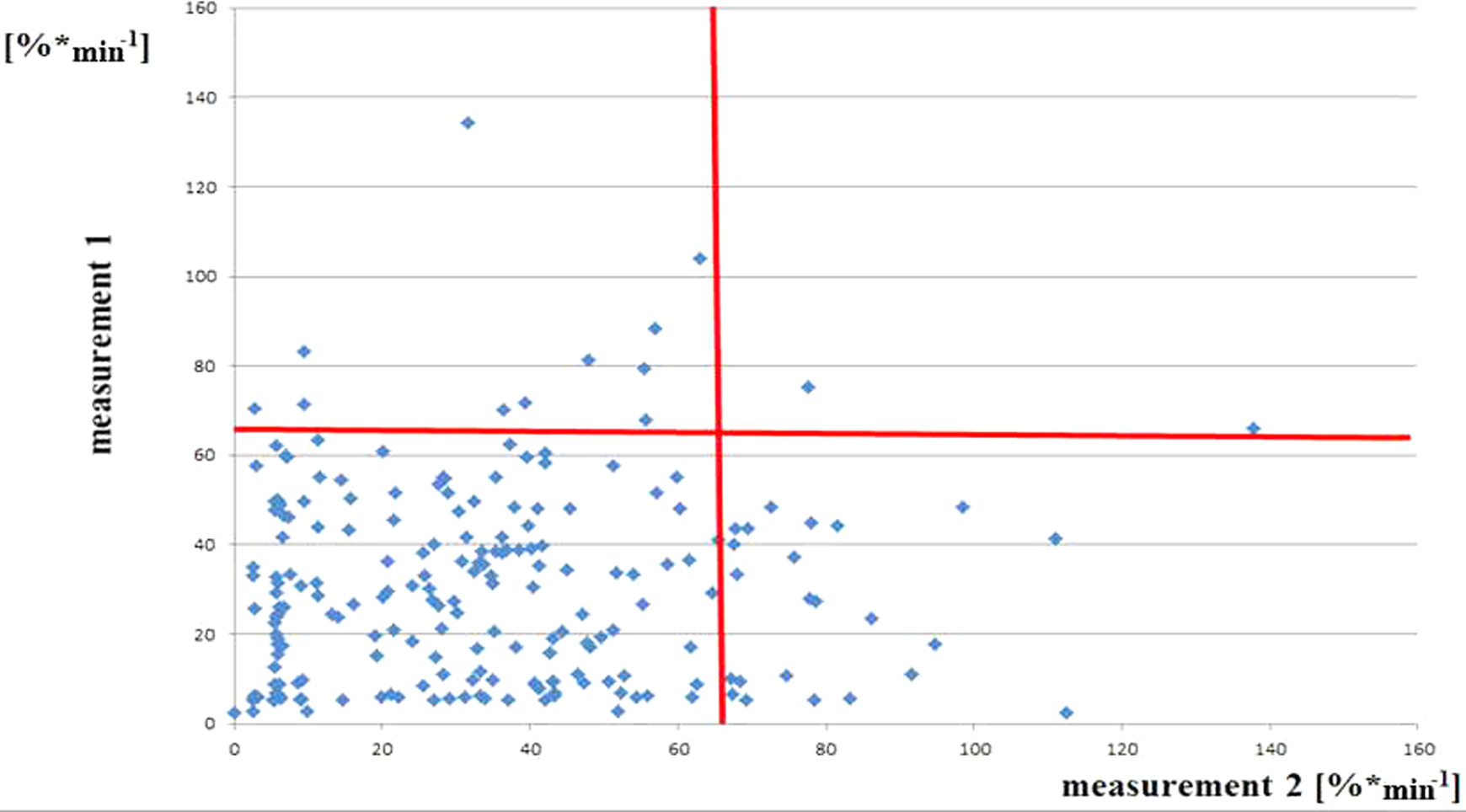
Relationship between the baseline (1) and the follow-up residual platelet reactivity (RPR) measurement (2) in the long-term part of the study.
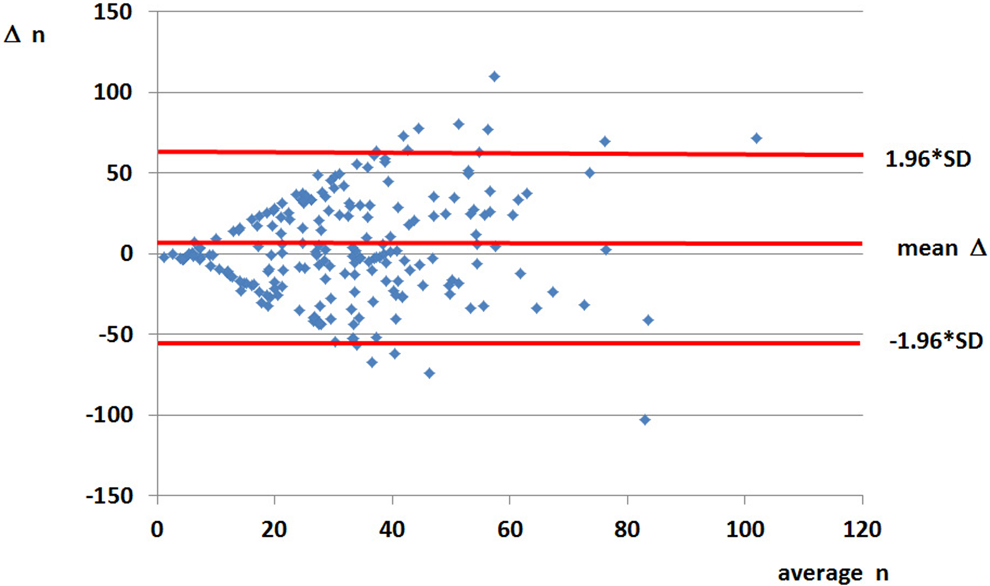
Bland-Altman plot of the residual platelet reactivity (RPR) values in the long-term part of the study showing typical diverging pattern.
Short-Term Reproducibility
Baseline demographic characteristics of the studied cohort are shown in Table 2 .
Baseline Demographic Characteristics of the Studied Cohort in Short-Term Part
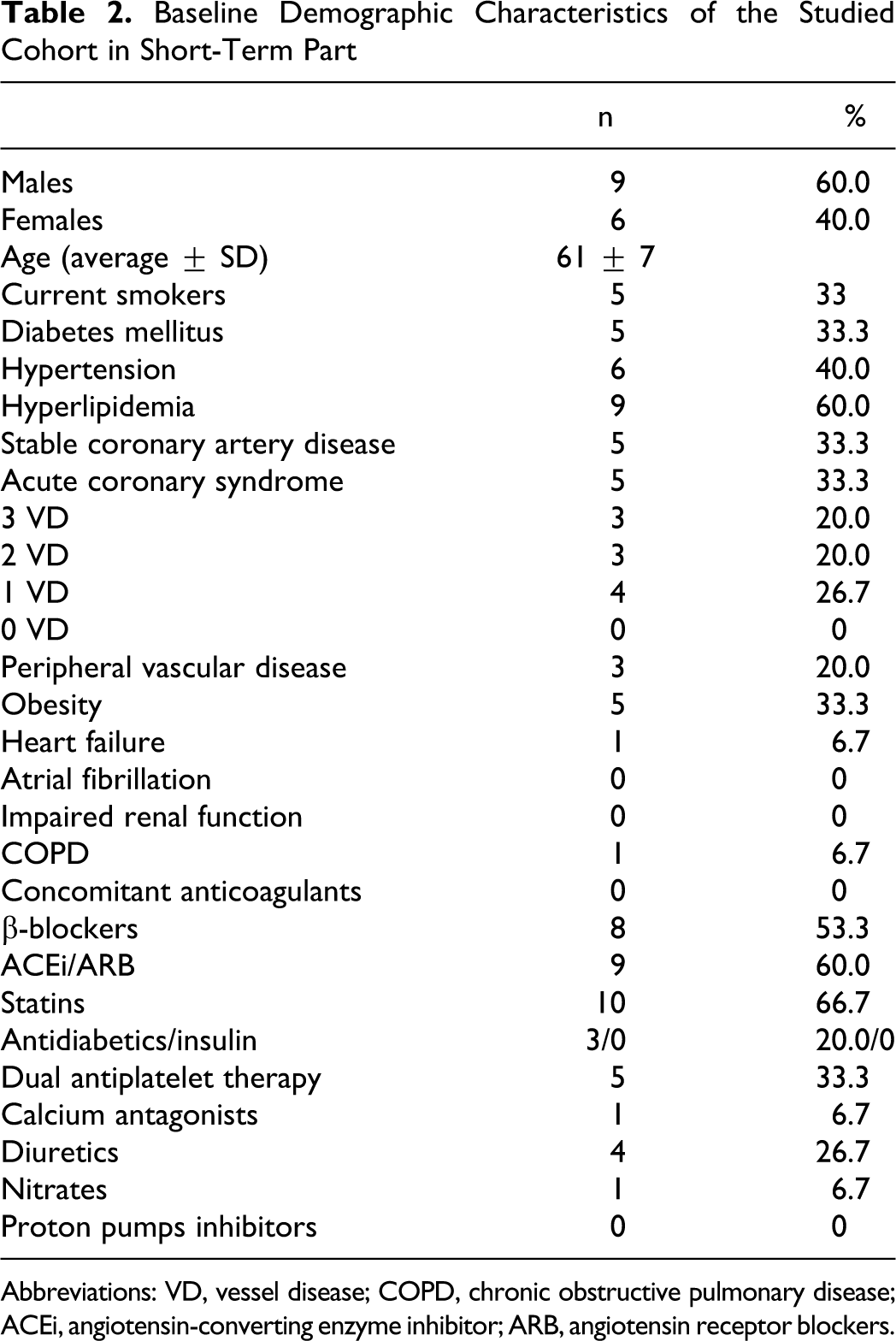
Abbreviations: VD, vessel disease; COPD, chronic obstructive pulmonary disease; ACEi, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blockers.
In contrast to the first part of the study, the STR of LTA was very good with ICC between 2 measurements 0.800 (Figure 3 ). The Bland-Altman plot for STR is shown in Figure 4 .
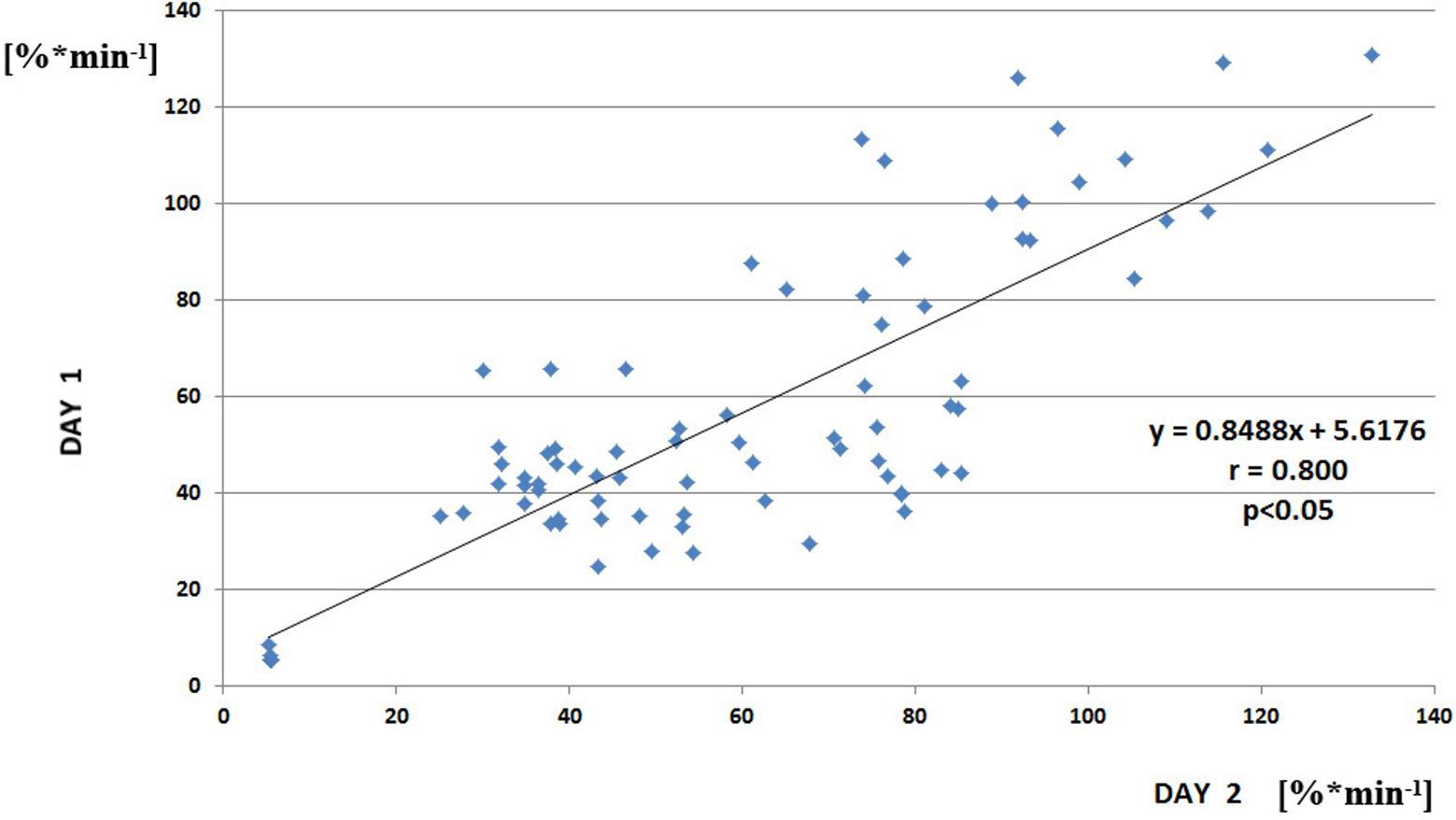
Correlation between day 1 and day 2 measurement of residual platelet reactivity (RPR) in the short-term part of the study.
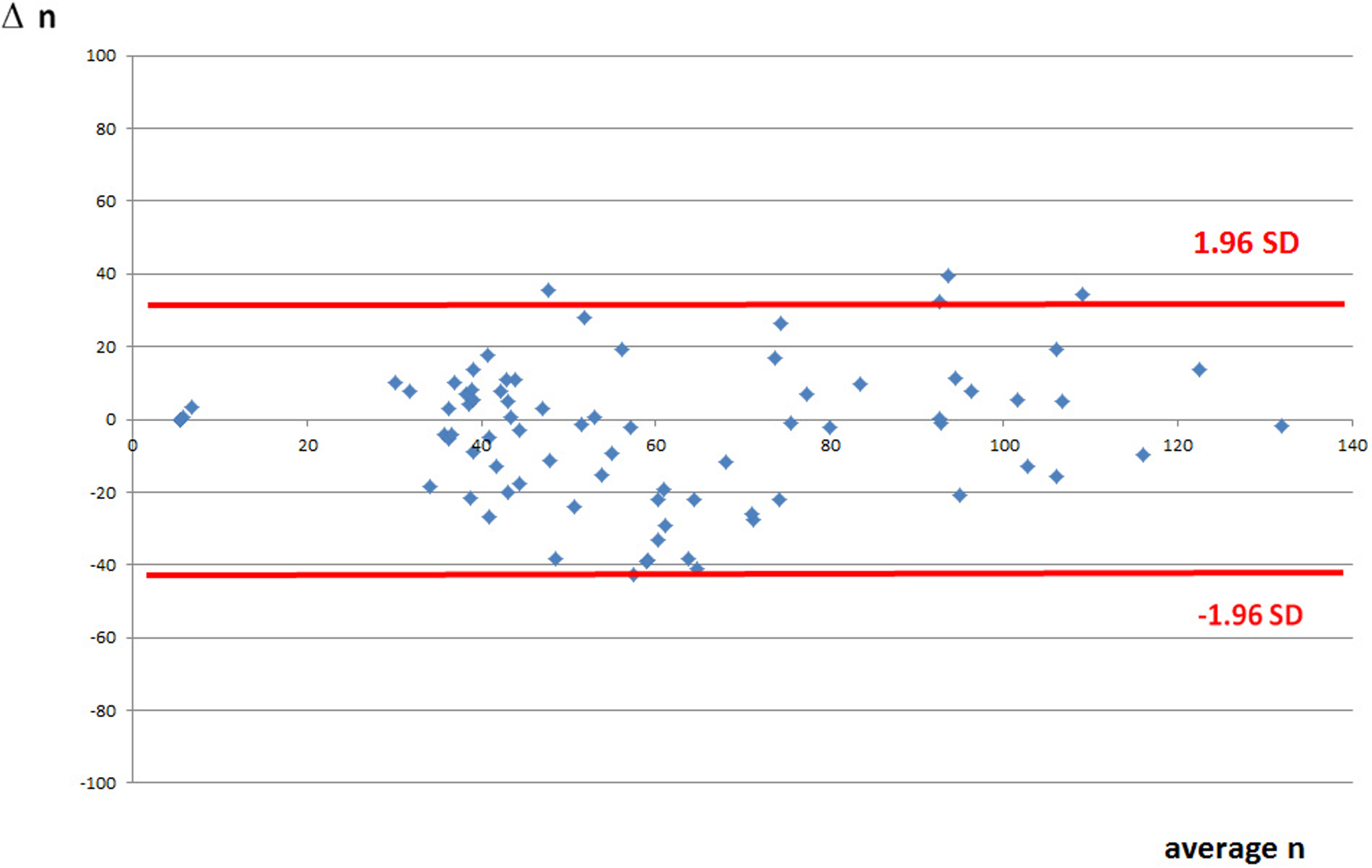
Bland-Altman plot of the residual platelet reactivity (RPR) values in the short-term part of the study showing most values within the limits of agreement.
Correlation Between LTA With CPG and ASPI Test With AA
Correlation between 2 methods of RPR measurement was assessed by LTA with CPG as an activator and the ASPI test on Multiplate analyser (Dynabyte) with AA as an activator in the subgroup of patients. Good correlation between these 2 methods was found (y = 0.7175x + 40.42, r = .7876, P < .05; Figure 5 ).
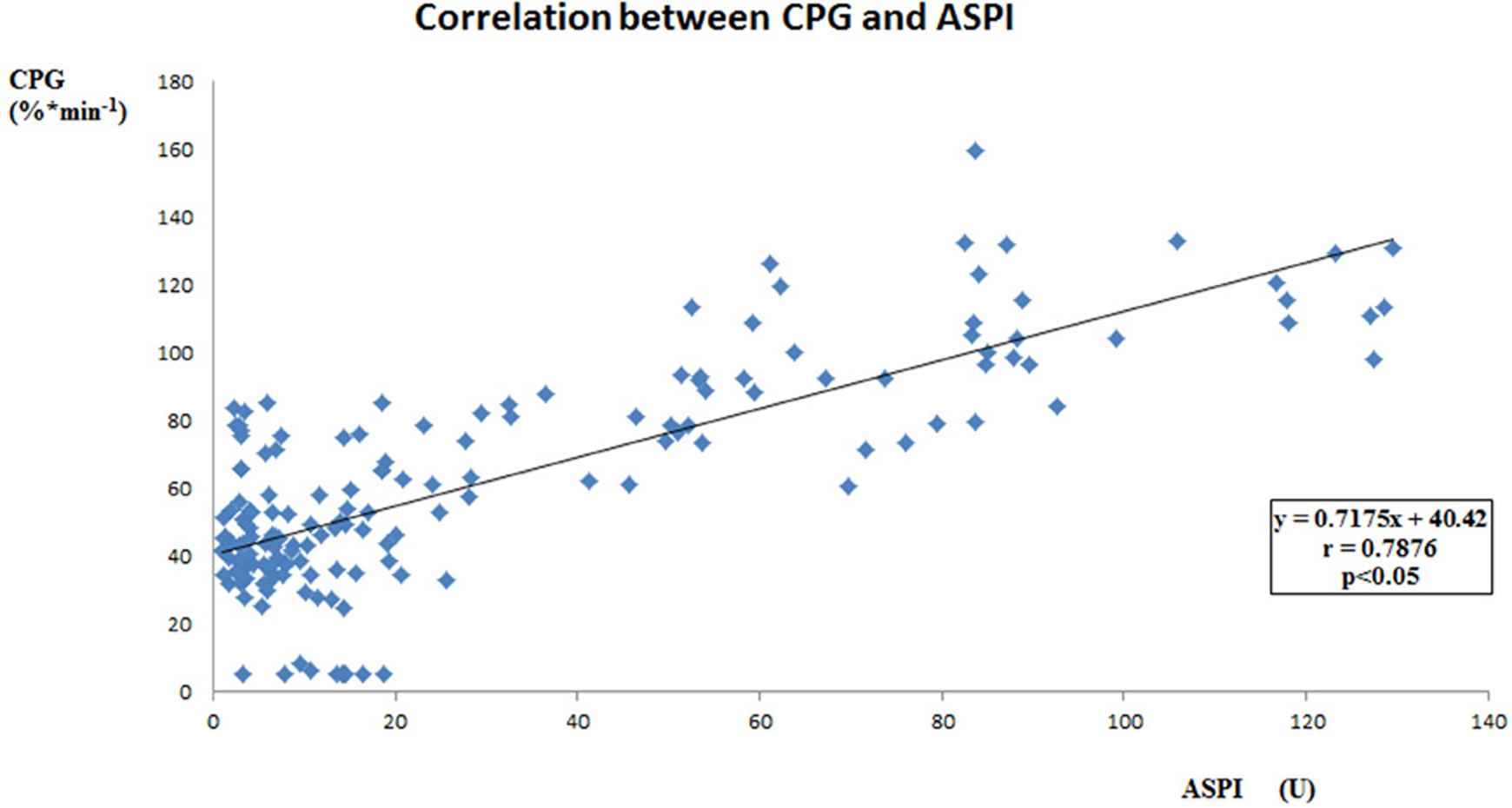
Correlation between 2 methods of residual platelet reactivity measurement in the short-term part of the study cohort. Cationic propyl gallate (CPG) was correlated to ASPI test with AA as assessed on Multiplate analyser (Dynabyte, Munich, Germany).
Discussion
Assessment of RPR in Patients on ASA Therapy
Light transmission aggregometry evaluates the luminosity of the PRP as aggregation occurs following platelet stimulation with specific (eg, AA) or nonspecific agonist (collagen, adenosine diphosphate, ristocetin, epinephrine, CPG, etc). Specific tests with AA-induced platelet aggregation depend on endogenous thromboxane formation and evaluate the ability of ASA to block the COX-1 pathway with consequent inhibition of TXA2 formation. Because a completely inhibited COX-1 reactivity can be bypassed by several other platelet activation pathways, less specific functional assays may detect high RPR even if COX-1 is completely blocked.10,11 The results are usually expressed either as maximal aggregation (AU), aggregation velocity (AU/min or % per min), and/or as area under the aggregation curve (AU*min). Substantial proportion of patients may exhibit increased RPR despite standard dosage of antiplatelet treatment. The problem is complex and has different implications for different antiplatelet drugs. Many factors determine the response to antiplatelet agents, some of them related to the drug itself and some to the characteristics of individual patients. Santilli et al, moreover, documented that the results of functional assays may not correspond to the serum TXB2 level, which may be suppressed by low-dose ASA in healthy participants even when function tests show increased RPR. 8 The nonlinearity of the relationship between inhibition of thromboxane production and inhibition of platelet function demonstrates the complexity of the problem and the need for precise assessment of the clinical and prognostic consequences of the tests used.
The role of inflammation in high RPA was also studied with inconsistent results. 12 Increased levels of several inflammatory markers like C-reactive protein (CRP), CD40 ligand, some interleukins, tumor necrosis factor, and some adhesion molecules are associated with adverse prognosis in patients with CAD. Possible association between elevated inflammatory markers and high RPA in patients treated with antiplatelet drugs has been speculated. 13 Platelets itself and some of their activators and products may be involved in inflammatory processes. 14 We also assessed the role of inflammation on high RPR, as assessed by LTA, in a smaller subgroup of 64 patients in our previous study. 15 Whereas we found no association between high RPR and matrix metalloproteinases 2, -3, -9, tissue inhibitor of metalloproteinases 2, monocyte chemoattractant protein 1, soluble CD40 ligand, high-sensitivity CRP, and endoglin plasma levels, the proportion of patients with high RPR was increased in those with increased plasma levels of tissue factor and decreased plasma level of tissue factor pathway inhibitor. 15
Several studies documented that only less than 5% of patients have inadequate inhibition by routinely used ASA dosages as assessed by COX-1-specific assays. 11 On the other hand, assays with less specific agonists also reflect pathways related to COX-1 independent effects of ASA and therefore may display increased RPR despite ASA administration due to enhanced platelet turnover, transient expression of COX-2 in newly formed platelets, or extraplatelet sources of TXA2.12,16 The relative frequency of inadequate response to ASA treatment demonstrated that noncompliance is the predominant cause of this phenomenon, 17 for these reasons we exercised maximal attention to secure reliable drug intake in all patients in our study at least 5 days before the assessment of platelet reactivity.
Reproducibility of the RPR Measurement
Reproducibility is defined as the consistency of a given test to measure the same results on different occasions. It should be quantified for all methods used for clinical decision making and should be reported in reliability studies. Minimal measurement error of given parameter is critically important also to the assessment of the efficacy of antiplatelet treatment. The main components of measurement error are systematic bias and random error due to biological variations. Both error components should be meaningfully quantified. 18 A commonly used statistical analysis for the reproducibility assessment is the ICC, with standard error of mean (SEM) calculated from the ICC. 19 Bland-Altman plot shows the measurement error schematically and helps to identify the presence of heteroscedastic or homoscedastic distribution.9,20
Our results show that sustained high RPR is very rare (only 1% of patients in our own series had consistently high RPR on both measurements during 10 ± 6 months of follow-up), on the other hand even during midterm follow-up, only 82.1% of our patients had both measurements within the limits of adequate platelet suppression. In all 35 patients (16.9%) had inadequate platelet inhibition during 1 assessment with the second being in the therapeutic range.
Ability to identify patients with insufficient platelet inhibition by antiplatelet drugs varies greatly according to the platelet function assay used.21–23 Harrison et al measured platelet reactivity by LTA, Platelet Function Analyser-100 (PFA-100), and VerifyNow-ASA and found no correlation between the initial test and retest at 1 year. 24 Theoretical differences among individual tests may play important role and may also explain the disparate results among the different tests. Although we documented good short-term intraindividual reproducibility in our study, the long-term comparison confirmed the results of several other studies showing high variability in longer time intervals. We were not able to identify any single factor in our database, which would explain the change in the individual response of the RPR during the follow-up. The test was highly reproducible if the samples are obtained during a short time period and most of different additional variables are excluded. On the other hand, during the long-term follow-up, not only the random fluctuation but also many different additional variables may influence the individual results. Most cited studies evaluate midterm or long-term variability, which is influenced by many additional factors with considerable fluctuation of individual results. To assess reproducibility of a medical test, it is essential to distinguish between random fluctuation and the role of different additional variables, which influence the test beyond random. In the case of the assessment of the platelet inhibition by ASA, it is not clear as yet whether routine administration of single dose of the drug is superior or at least noninferior to the therapy guided by a laboratory measurement of RPR with subsequent modification of antiplatelet treatment, if indicated. Moreover, it is not clear so far, whether 1 single measurement is sufficient or whether repeated assessments are necessary to control properly the chronic antiplatelet treatment. This problem becomes even more important in view of the contemporary multiple possibilities for antiplatelet drug management, including increased doses of ASA, clopidogrel, or administration of even more effective drugs such as prasugrel or ticagrelor.
Possible Limitations of Our Study
The aim of our study was to assess the STR and LTR of a platelet function test. We used a nonspecific assay (with CPG as an agonist) in our study. Although specific tests with AA-induced platelet aggregation evaluate the ability of ASA to block the COX-1 pathway with consequent inhibition of TXA2 formation, less specific functional assays may also evaluate several other platelet activation pathways and COX-1 independent effects of ASA. 16 Nevertheless, some authors postulate that when tests with less specific agonists are used, the platelet function may show normal results of laboratory testing, even despite sufficient pharmacodynamic action of an antiplatelet drug. 10
Our study was not powered to assess the impact of inadequate antiplatelet drug control on the occurrence of ischemic events. Moreover, during the follow-up the rate of events was very low. Our goal is to continue the long-term follow-up with regular laboratory measurements of the RPR, to categorize the group into quartiles according to the RPR level and to compare the incidence of predefined end points in these subgroups.
Conclusions
Our data show very good intraindividual reproducibility of RPR measurements, provided they were carried out during short-term interval, so that the influence of additional variables is minimized. On the other hand, during the long-term follow-up there is no correlation between individual measurements. We conclude that repeated examination of the effectivity of ASA may be necessary to assess and conduct the chronic antiplatelet treatment correctly. Bigger prospective trial would be, however, warranted to compare single routine dose of antiplatelet therapy against therapy tailored according to the laboratory assessment.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Internal Grant Agency, Ministry of Health, Czech Republic [grant number NR/9174-3].
