Abstract
This study was planned for searching possible changes of the total coagulation and fibrinolysis system in inflammatory bowel disease (IBD) in order to obtain some clues for explaining the relation between IBD and hypercoagulability. A total of 24 patients with ulcerative colitis, 12 patients with Crohn disease, and 20 healthy controls were studied. Platelets; prothrombin time (PT); partial thromboplastin time (PTT); fibrinogen;
Introduction
One of the etiopathogenetic hypotheses of inflammatory bowel disease (IBD) is small vessel thrombosis. 1 Multifocal intestinal infarction with vascular injury, focal arteritis, and fibrin deposition, in association with granulomatous or lymphocytic inflammation, are common findings in IBD.1–3 Additional indirect evidence that vascular thrombosis could be involved in IBD pathogenesis is provided by a large epidemiological study that found lower prevalence of IBD in congenital coagulation disorders (hemophilia or von Willebrand disease). 4 Successful treatment of some patients with IBD with heparin also supports this hypothesis. 5
Patients with IBD have a 3-fold increased risk of venous thrombosis compared with age, sex, and geographically matched controls. 6 Thrombosis is a major cause of morbidity and mortality in IBD. 7 The prevalence of systemic thromboembolism in IBD varies between 1% and 6.7% in clinical studies and rising up to 39% in some autopsy studies. 8 Although the risk may be increased with active disease, at least one third of the patients with IBD have inactive disease at the time of thrombosis. The risk seems to be specific to IBD as it is not seen in other inflammatory conditions such as rheumatoid arthritis or other chronic bowel disease such as celiac disease. 9 Despite the undoubted association between IBD and venous thrombosis, no consistent unifying etiology has been determined. There have been a few studies dealing with the total coagulation system in IBD, although there have been many reports on a part of the system. So, this study was planned for searching possible changes of the total coagulation and fibrinolysis system in patients with IBD in order to obtain some clues for explaining the relation between IBD and hypercoagulability.
Patients and Methods
Twenty-four patients with ulcerative colitis (UC) and 12 patients with Crohn disease (CD) together with 20 healthy controls were studied. All patients and control participants gave their informed consent to participate in the study, which was approved by the Hospital’s Scientific Committee. The diagnosis of IBD in all patients was confirmed by clinical, radiological, endoscopic, and histological examinations. The activity of patients with UC was determined by Rachmilewitz endoscopic index. 10 Harvey-Bradshaw simple index was used for determining the activity of patients with CD. 11 If the score was higher than 4, the disease was considered active. Twenty (14 UC and 6 CD) of the patients were active and 16 (10 UC and 6 CD) were inactive. Demographic characteristics of the groups were shown in Table 1 . Statistical difference was not present between the groups.
Demonstrating Demographic Characteristics of the Study Group
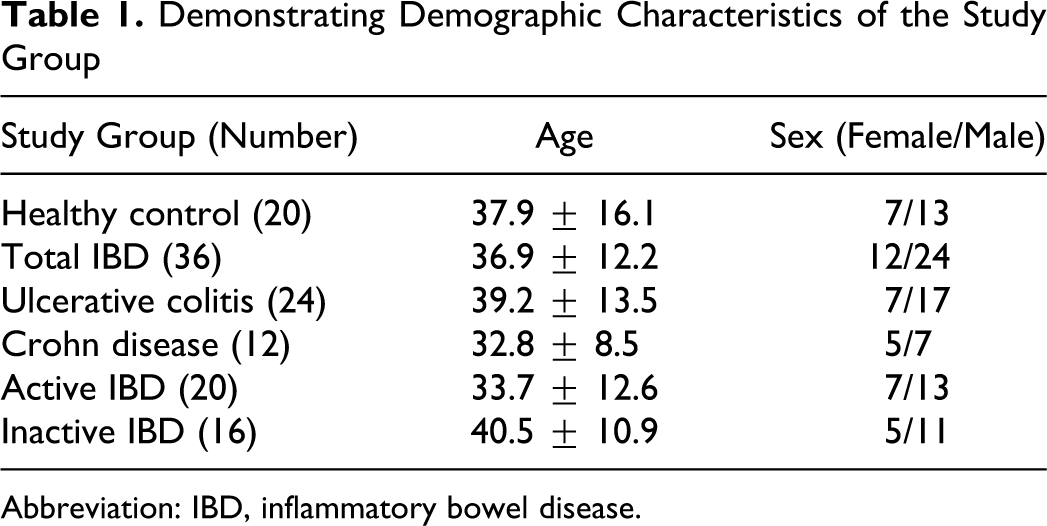
Abbreviation: IBD, inflammatory bowel disease.
Platelet count; prothrombin time (PT); activated partial thromboplastin time (aPTT); thrombin time (TT); fibrinogen;
All blood samples were drawn after fasting between 08:00 and 11:00
Platelet counts, aPTT, PT, TT, and fibrinogen were measured using conventional methods (Thromborel S, Pathromtin SL, BC thrombin Reagent, Multifibren U, BCT, Dade-Behring). The activities of CFs V, VII, VIII, IX, and XI and protein C were assayed in stages using a parallel line bioassay based on aPTT, PT, or TT (coagulometric method; CFs V, VII, VIII, IX, and XI deficient plasma, BCT, Dade-Behring, Marburg, Germany). The level of the
Statistical Analysis
All statistical tests were done using SPSS 13 software. Data were presented as mean ± standard deviation. For nonparametric values, Kruskal-Wallis test and for parametric values 1-way analysis of variance (ANOVA) and t test were used. P values lower than .05 were considered statistically significant.
Results
The results are given in Table 2
. First we compare IBD group with the healthy control group. Then we compare UC group with CD group and active IBD group with inactive IBD group. Also all these 4 groups were compared with the healthy control group. Activated partial thromboplastin time; protein C; CFs V, VIII, XI, and XIII; plasminogen; and antiplasmin were similar in all groups. Platelets (366 000 ± 139 000/μL, 225 000 ± 39 000/μL), fibrinogen (4.0 ± 1.1 g/L, 2.6 ± 0.4 g/L), FgDP (545 ± 181 ngFe/mL, 200 ± 61 ngFe/mL), vWF (203% ± 78%, 146% ± 49%), CF IX (104% ± 15%, 88% ± 10%), tPA (11.4 ± 6.3 ng/mL, 7.4 ± 1.4 ng/mL), PAI-1 (65 ± 27 ng/mL, 30 ± 12 ng/mL), and PF1 + 2 (11.1 ± 6.1 nmol/L, 1.1 ± 1.0 nmol/L) levels of IBD group were found significantly (P < .05) higher than control group. Similar differences were seen when UC, CD, and active IBD groups compared with control group except for vWF level in CD which was found similar to healthy control. When compared with control group, the
Showing Results of the Study Groups a
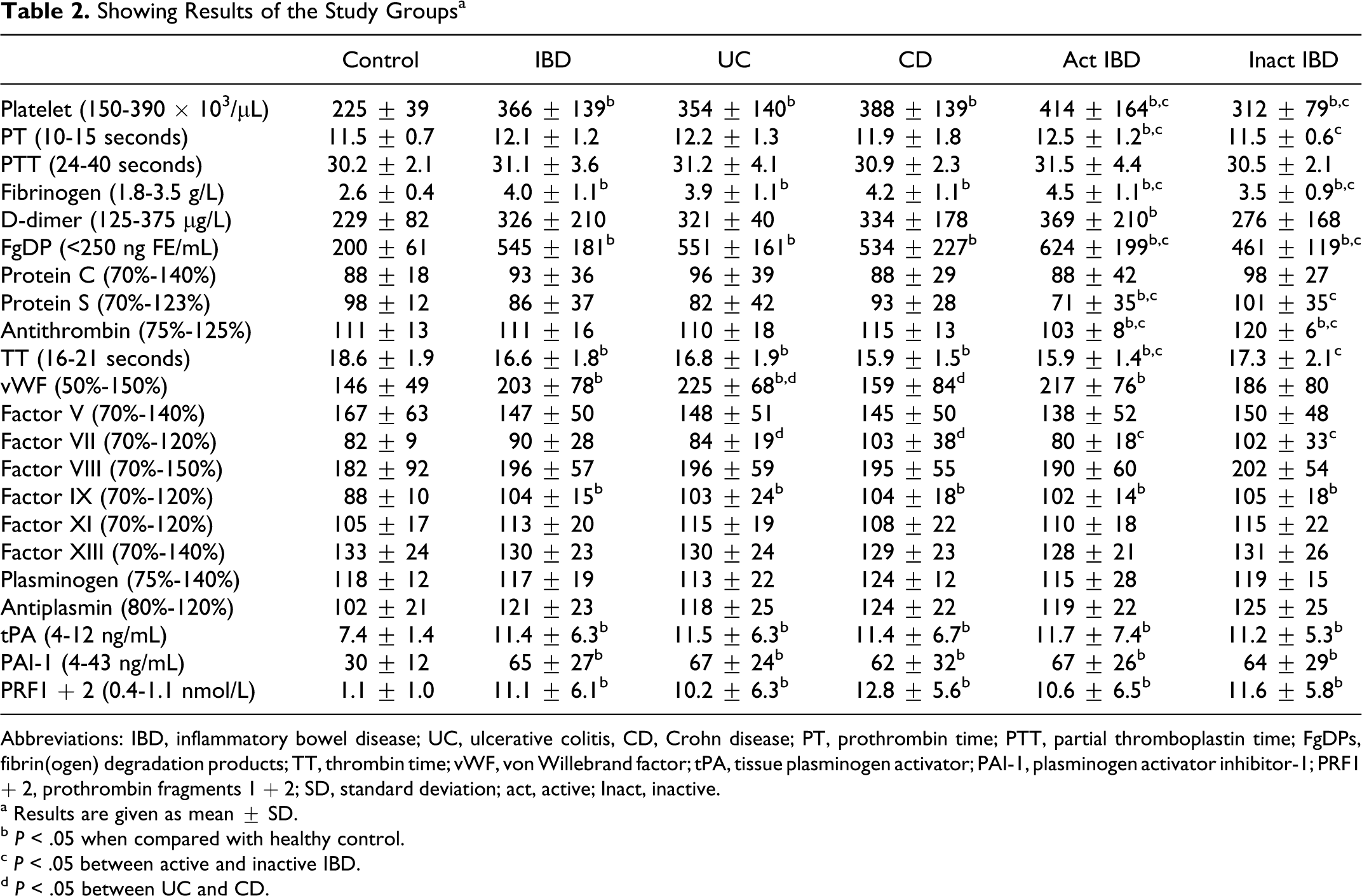
Abbreviations: IBD, inflammatory bowel disease; UC, ulcerative colitis, CD, Crohn disease; PT, prothrombin time; PTT, partial thromboplastin time; FgDPs, fibrin(ogen) degradation products; TT, thrombin time; vWF, von Willebrand factor; tPA, tissue plasminogen activator; PAI-1, plasminogen activator inhibitor-1; PRF1 + 2, prothrombin fragments 1 + 2; SD, standard deviation; act, active; Inact, inactive.
a Results are given as mean ± SD.
b P < .05 when compared with healthy control.
c P < .05 between active and inactive IBD.
d P < .05 between UC and CD.
Discussion
A growing amount of data highlights a tight mutual network in which inflammation, coagulation, and fibrinolysis play closely related roles. In both forms of IBD, a hypercoagulable state and a prothrombotic condition exist, whereas coagulation abnormalities are an intimate part of the IBD clinical picture. 12 Also, the fibrinolytic system has been widely investigated in IBD as a prothrombotic condition may also result from reduced fibrinolytic activity. Disease activity is a probable risk factor for thromboembolism.7,13 It is noteworthy however that one third of thromboembolic complications occurs in patients with quiescent disease. 7 Therefore, we compare the parameters of the active and inactive IBD groups with each other and with the healthy control. In the present study, we found that the parameters of the total coagulation–fibrinolysis system were higher in IBD group than in healthy control group, and this difference was present in all IBD subgroups regardless of the form (UC or CD) and the activity (active or inactive IBD) of the disease.
The hypercoagulable state in IBD is closely related to abnormal platelet function resulting in a high probability of microvascular thrombosis and microcirculatory dysfunction.14,15 On the other hand, vWF is a large glycoprotein that circulates in human plasma and is deposited in the vascular subendothelium. In response to endothelial injury, vWF also mediates platelet-to-platelet interactions and platelet adhesion to the subendothelium. 16 We found that the platelet counts of the all IBD groups (including inactive IBD group) were significantly higher than healthy control. Also platelet count of the active IBD group was higher than inactive group. Serum vWF levels of the total and active IBD groups and UC group were higher than healthy control. These findings were consistent with the literature. The elevated platelet and vWF levels in IBD groups could be caused by either vascular injury occurring secondary to bowel inflammation or an acute phase response to endothelial cell stimulation by mediators released during the inflammatory process.17,18 Interestingly, vWF level of our CD group was significantly lower than UC group and similar to the healthy control group. This unexpected finding may be due to the smallness of the CD group.
The coagulation system can be divided into extrinsic and intrinsic pathways. In the classical cascade model, coagulation is initiated via extrinsic and intrinsic pathways that are strictly linked and interdependent and maintained by the cascade activation of several factors. The intrinsic pathway is initiated when blood is exposed to a negatively charged surface, which activates CF XII, and the extrinsic pathway is initiated by tissue damage that exposes the tissue factor. 19 Hudson et al 20 reported that the mean CF VII level was significantly higher in patients with IBD than in normal participants. We cannot detect this difference in our study. Coagulation factor VII activities of our IBD groups were similar to healthy control. Activation of CFs XI and XII has been reported in <10% of patients with UC. 21 We found that CF XI activities of IBD groups were similar to healthy control group. Several investigators have reported that CFs V, VIII, IX, and X activities were elevated in IBD.21–23 We found that CF IX activities were higher in IBD groups, but CF V and VIII activities were not different from healthy control. Prothrombin time (which reflects intrinsic pathway) and aPTT (which reflects extrinsic pathway) levels of the groups were not different as stated in the literature. Fibrinogen levels of our all IBD groups were higher than healthy control group and active IBD group have significantly higher level of fibrinogen according to inactive group as reported in the literature. 21 As expected, TT of all IBD groups with the exception of inactive IBD group was found significantly shorter than the healthy control.
Deficiencies of antithrombin, protein C, and protein S are well-recognized causes of thrombotic complications related or not related to IBD.16,17,22 Beside the congenital deficiencies of these natural anticoagulant factors, it was reported that the antithrombin and protein S plasma levels were lower in patients with IBD. On the other hand, plasma protein C levels remained unchanged according to the literature. Our results show that the protein C levels of all groups were similar, and the antithrombin and protein S levels of active IBD group were lower than the inactive group and healthy control group.
Thrombin itself is impossible to quantitate and so the use of surrogate markers is necessary. PF1 + 2 is an index of in vivo thrombin generation; one molecule of PF1 + 2 is released with the generation of each thrombin molecule. The measurement of PF1 + 2 would be an excellent marker of thrombin generation. This is helped by the fact that PF1 + 2 is not generated in vivo by any other mechanism. It is well known that PF1 + 2 is a useful marker of the activation of blood coagulation in IBD.24–27 Moreover, the activation of PF1 + 2 is a risk factor for thromboembolic complications in IBD. Our present data also showed that mean PF1 + 2 levels of all IBD groups were significantly higher than healthy control group and suggested that thrombin generation might be an early event in IBD.
Plasminogen is an essential component of the fibrinolytic system, which is converted into plasmin by tPA and urokinase plasminogen activator and inhibited by PAI-1, PAI-2, and antiplasmin.28,29 Once formed, plasmin cleaves fibrin, generating soluble degradation products. We found that the plasminogen and antiplasmin levels of all our groups were similar, but tPA and PAI-1 levels of all the IBD groups were significantly higher than the healthy control, and these findings were concordant with the literature.12,28,30
The FgDP assay measure amounts of the fibrin and fibrinogen split products in the blood and directly indicate the level of activity of the fibrinolytic system. Plasma
When summarized, most of the procoagulants (platelets, fibrinogen, vWF, CF IX, and PAI-1) were found to be increased together with decreases in some anticoagulants (protein S and antithrombin) in IBD. Also the activation markers of coagulation (
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
