Abstract
The aim of the study was to assess the prevalence of HCV, HBV, and HIV infections among the patients with hemophilia. Patients with Hemophilia A and B were evaluated who visited hospital for factor replacement therapy. The viral markers tested in these patients included anti-HCV-Ab, HBsAg, and anti-HIV-Ab. Seroprevalence was compared from 5717 exchange healthy blood donors for same markers. A total of 173 multitransfused male hemophiliacs showed prevalence of 51.4% for HCV, 1.73% for HBV, and nil for HIV. In blood donors seroprevalence was 1.9% for HCV, 1.81% for HBV, while no HIV-positive case was detected. Prevalence of anti-HCV-Ab was significantly high in patients with hemophilia than normal donors (P = .0005). This study showed that HCV infection was more frequently identified than HBV and HIV infections in multitransfused hemophiliacs. The frequency of hepatitis C among blood donors is also higher than that of hepatitis B which is showing downward trend.
Keywords
Introduction
The dramatic bleeding, the effect of the disease on history through its presence in Queen Victoria's descendants, and the overwhelming role of therapeutic concentrates in the transmission of the acquired immunodeficiency syndrome (AIDS) have made hemophilia A the object of great medical, scientific, and public interest. Hemophilia, the commonest congenital coagulation disorder and a disease without ethnic or geographic limitations, its incidence approaches 20 per 100 000 male births.1,2 The mainstay of treatment in congenital bleeding disorders is replacement of the missing clotting factor. But one of the serious complications is transmission of viruses which have proved to increase morbidity and mortality. After the discovery of AIDS due to HIV transmission in 1982 in a hemophilia patient who had received transfusions leading to evolution of scientific development for safe factor products. Heat treatment and other methods to inactivate enveloped viruses in plasma-derived clotting factor concentrates became standard practice in the west, as people with hemophilia and other bleeding disorders were at very high risk of infection with plasma-borne viruses. 3 The concept of the “safety tripod” for safe products include selection of blood donors of an appropriate safety profile; testing of donations to ensure the exclusion of detectable transfusion transmissible pathogens; and treatment of the product or intermediates during manufacture to ensure elimination of any contaminating pathogens. 4
The problem of transfusion-transmitted infection is more serious in the developing countries with lower economic means like Pakistan, where factor replacement therapy in hemophilia and other bleeding disorder patients is based mainly on fresh frozen plasma and its components. Plasma or other blood products that they receive are not always appropriately screened for transmissible agents. They cannot easily obtain viral inactivated clotting products. Treatment with partially screened blood products has been associated with infections such as hepatitis B; C, and HIV. Transfusion-associated hepatitis was recognized in 1943, and it is now an important complication of hemophilia treatment in many countries. 5 Studies conducted in Pakistan on blood donors have shown prevalence of various infectious pathogens ranged as follows: hepatitis B 1.46% to 2.99%, with a downward trend over time, hepatitis C 3.01% to 4.99%, HIV 0% to 0.06%, respectively.6,7 Compared to neighbor India, hepatitis occurs with a similar frequency, but HIV is seen less common. Screening of blood products for hepatitis C started only about 10 to 12 years ago in Pakistan. Methodology for screening has been suboptimal because of the high cost of these tests. There is a high prevalence of HCV and HBV in general population and in blood donors. 8 Therefore, chronically transfused patients continue to have been exposed to substandard blood products as well as from donors in window period or who have low level of viraemia. Transfusion-transmitted infections (TTIs) continue to be a major confront for Blood transfusion organizations across the world and are directly proportionate to the prevalence of the infection in the blood donor community.1,2 The objective of this study was to determine the prevalence of these infections in our patients with hemophilia in relation to blood donors as it is an appropriate indicator of the risk of infection. Furthermore, a thorough understanding of the epidemiological characteristics of TTI in multitransfused patients may be of major assistance in clarification of important aspects of the transmission chain of these infections and so further improvement in the safety of the blood supply and estimates of the residual risk of transmission in the countries.
Patients and Methods
From August 2007 to March 2009, this study was conducted, approved by institutional ethics committee, and was done in accordance with declaration of Helsinki. Informed consent was obtained from all adult participants, parents, or legal guardians. This cross-sectional study was performed on patients diagnosed with hemophilia A and B of all types (mild, moderate, and severe) attending the bleeding disorder clinics and daycare of National institute of blood disease (NIBD) during the study period. Blood specimens were collected from a total of 173 patients. Serum samples were separated from the whole blood, aliquotted and stored at −70 C until tested. Blood donor’s questionnaires in Urdu and clinical laboratory data from blood donors were entered. Professional donors or those with a history of jaundice, recent fever, vaccination, dental procedure, surgery within 3 months were excluded. A total of 5717 exchange blood donors who were family members, relatives, friends of the patients, and a number of voluntary donors were selected. Peripheral venous blood sample, a portion of which was tested locally for complete blood counts (CBC) for anemia, blood films for malarial parasite(MP), and serum chemistries were taken after their acceptance as donors and performed daily on routine basis included HBV, HCV, HIV, and Syphilis. Demographic data of patients with hemophilia included age, duration, and number of blood transfusions. Complete blood counts in EDTA tubes was performed on XE-2100 hematology analyzer (Sysmex, Kobe, Japan). Malarial Parasite was screened by Leishman stain at pH 7.2 blood films 9 and Syphilis TP a qualitative immunoassay for the detection of antibodies to Treponema pallidum (TP), (Determine, Abbott, Japan). All sera were screened for HIV Ag-Ab (Bio-Rad, France), HBsAg (Bio-Rad, France), and anti-HCV (DiaSorin, Spain), with third-generation commercial ELISA kits on Evolis according to the manufacturer’s instructions. Serum samples of all hemophilic patients were also used for serum glutamic pyruvic transaminase (SGPT; normal limit was ≤43 μ/L in males) by spectrophotometric method, Randox on micro lab 200.
Statistical Analysis
Prevalence and its corresponding 95% confidence interval were calculated with SPSS software version 13.0 (SPSS Inc., Chicago, Illinois). Data comparisons were performed using the chi-square with Fisher exact test. The differences were considered significant if P < .05.
Results
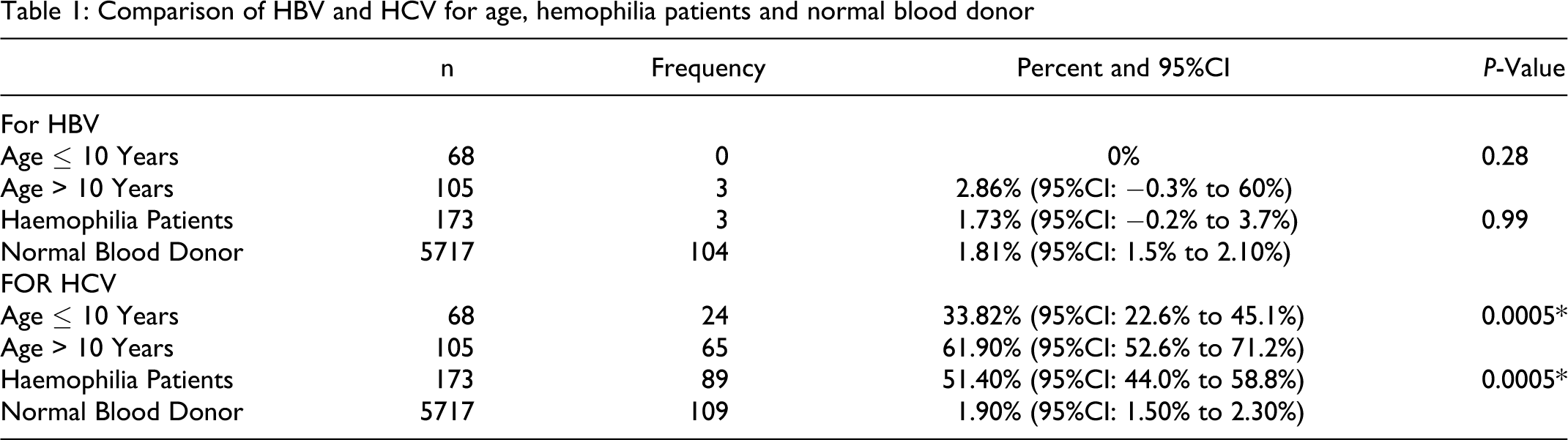
A total of 173 male patients with hemopihilia were screened. Median age was 14 years (age range 1 month to 54 years). Of these, 89 patients were reactive for HCV (51.4%). Only 3 out of 173 patients were reactive for HBV and none for HIV. Serum glutamic pyruvic transaminase was found raised in 21 HCV-positive patients (23.5%). Hepatomegaly was noted in 8% and splenomegaly in 4% among older participants. Exchange blood donor’s age ranged from 18 to 50 years, mean age 35 years. Majority donors were male; only 5 were females. In exchange blood donors it was 1.9% for HCV, 1.81% for HBV, while no HIV, 0.004% Syphilis, and 0.0003% malarial parasite was detected among them. Fresh frozen plasma (FFP) and its components were most frequently used; viral inactivated factor concentrate (plasma derived or recombinant) were used when available, Table 2 . Out of 68 patients with hemophilia under the age of 10 years, 24 (33.8%) are anti-HCV positive while 65 / 105 (61.9%) more than 10 years are anti-HCV positive P Value .005. The data shows highly significant P valve for HCV infection in the participants. There was no difference in HBV sero-positivity in blood donors and patients with hemophilia of both age groups, see Table 1 . The age cut-off 10 years was used arbitrarily as HCV screening started around 10 years back in Pakistan. No coinfection of any virus was detected in our subjects. Factors associated with increased positive HBV and HCV infection were greater severity of hemophilia, age more than 10 years, larger amounts of blood products use mostly consist of combination therapy and multicentre visits.
Comparison of HBV and HCV for age, hemophilia patients and normal blood donor
Treatment Given to Patients
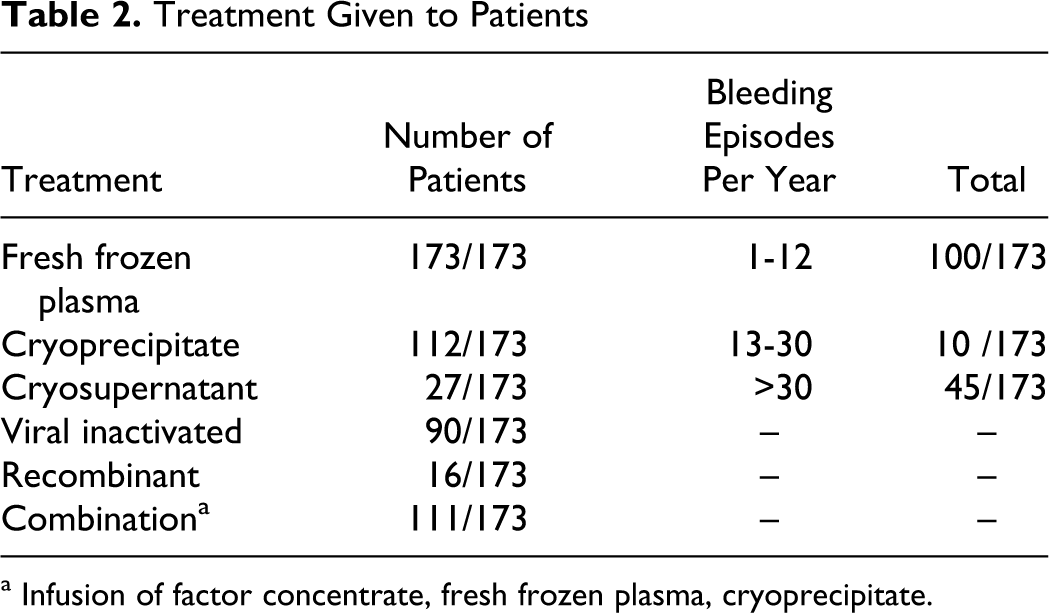
a Infusion of factor concentrate, fresh frozen plasma, cryoprecipitate.
Discussion
Eighty percent of the world’s hemophilia population lives in countries with limited medical or financial resources. They cannot easily obtain viral inactivated clotting products. Many patients are treated with locally supplied blood products and its components only. 10 Plasma or other blood products they receive are not always screened for transmissible agents whereas; the blood supply in the developed world is now safe. Therefore, transfusion transmitted infections continue to have a significant impact on patient management particularly in developing countries like, Pakistan. 11 Factors associated with high viral TTIs in countries with low human development index (HDI) 12 includes seroprevalence of these diseases in the general population, poorly organized blood donation system, and low sensitivity of pathogen-testing methods. Literature search supports the changing trends in HBV/HCV seroprevalence in blood donors and a low prevalence of HIV in Pakistani population. For HIV, a prevalence rate of 0.1% among the general population has been reported by the World Bank but reported that lack of awareness in Pakistan, and high-risk factors including low condom use and unscreened use of blood make the situation fertile for AIDS to become a major health issue.6,7,13 Our current data of blood bank shows that HCV infection prevalence is higher among our blood donors (1.9%). Thus, countries with a higher HCV prevalence in general population had a higher prevalence rate among multitransfused patients. Prevalence of HCV in blood donors from different countries of the developed world has been reported to be between 0.3 and 1.5%. 14 In Iran, the prevalence was 0.14%, 15 whereas from other countries such as Saudi Arabia 1.8%, 16 in India 0.25% to 0.9%, 17 Pakistan 3.01% to 4.99%.6,7 The prevalence rate among Pakistani blood donors seems to be much high. Mandatory anti-HCV screening was introduced to blood banks in Pakistan around year 2000. But still standard screening methods are accessible only in few blood banks of large cities.
According to World Health Organization (WHO), Pakistan falls in the low endemic area of HBV infection with prevalence of 3% infected population. Different studies present a quite variable image of the disease depending on the factors focused like sample size, objectives of the study, associated risk factors, population under study, diagnostic assays practiced, ethnicity, socio-economic status, and general population behavior. The low prevalence of HBV infection in our participants might be due to the introduction of hepatitis B vaccination, mass screening programmes, and stringent blood donor selection policy. The rate of positivity among blood donors is also reduced in other countries like Iran to 0.61% in 2005/2006, 17 in India the prevalence has remained fixed and ranged 1.7% to 2.2%, 16 in South East Asia and China it ranges from 1% to 15%. 18 The prevalence and infectivity could be under estimated for HBV. The study done by Muhammad Masroor Alam and et al on Pakistani population selected on random basis from all the 4 provinces show high prevalence rate of Hepatitis B virus infection and high incidence of acute and chronic liver diseases. 19 The study done by Bhatti FA on Pakistani blood donors have showed that one in 200 blood donors who are HBsAg-negative, anti-HBc-positive, however, have occult HBV infection, with likelihood of transmission of hepatitis B in recipients of blood components derived from them. 20 In the United States, anti-HBc is mandatory from 1986 to 1987 in view of high prevalence and to reduce the risk of transfusion-transmitted infections. Testing of all donated units for anti-HIV started since 1990. In Pakistan, the prevalence of HIV in high-risk subpopulations is 5% or higher. However, studies show low prevalence of HIV in blood donors similar to our results.6,7,20,21 The study done by Farid-ul-Hasnain and et al has shown alarming gaps in knowledge relating to HIV/AIDS and emphasizes the need to educate young adults and endow them with the appropriate information and skills to enable them to protect themselves from HIV/AIDS. 22
In our study significant number of Hepatitis C antibodies (51.4%) were detected in hemophilia patients with raised SGPT (23.5%) might be due to presence of chronic active hepatitis. Likewise, hepatomegaly was noted in 8% and splenomegaly in 4% among older participants. Current studies suggest that up to 5% of HCV-sero-positive patients develop cirrhosis or clinical manifestations of end-stage liver disease (ESLD) within 20 years, although the risk is considerably higher for the elderly patients. In the absence of HIV-1 infection, HCV is cleared from the blood, and spontaneous recovery occurs in approximately 20% of patients. The remaining 80% of HCV-sero-positive patients have persistent HCV viraemia and chronic hepatitis.10,23–25 No case of coinfection of Hepatitis C virus with HBV or with HIV-1 was found in our study. Lower HCV in younger patients may be a reflection of relatively less treatment days and rejection of HCV sero-positive donors once the screening is introduced.
Different studies on patients with hemophilia show seroprevalence of HCV 29.6% and HBsAg 4.9% from Zahedan, Southeast Iran, 26 other studies from Iran also show high prevalence of anti-HCV in hemophiliacs than that in other multitransfused patients. The rate of seropositivity ranges from 16% to 71.3% (mean 42.5%) in patients from various geographical regions. 15 From Indonesia, the prevalence of anti-HCV is reported to be 57% treated only with cryoprecipitate. 27 Whereas in some of the European countries such as Croatia and Slovenia where access to coagulation concentrates is only new, 15% of 120 Croatian and 15% of 166 Slovenian anti-HIV-positive hemophiliac patients are reported positive for anti-HCV. 28 Reports from Mexico showed the prevalence of 13.7% for HCV, 7% for HBV, and 1.7% for HIV. Patients with hemophilia had the highest prevalence for HCV and HIV infections. The majority of positive cases among patients with hemophilia reflect treatment with unscreened or not efficiently inactivated blood components in the past.
The percentage of HIV infection in multitransfused patients varies from country to country. The highest prevalence was reported from countries such as United States with 90% seroprevalence 29 and United Kingdom with 39%. 30 In the developing countries, as a result of scarcity of treatment as a whole and constant shortage of coagulation concentrates, the rate of HIV seropositivity is much lower. In India, HIV seropositivity of 4.4% to 12% has been reported in the hemophilia population.31,32 The results of HIV seropositivity for the Iranian hemophiliacs show low levels in various regions (0%-2.3%). 15
Conclusion
This study showed that HCV infection was more frequently identified than HBV and HIV infections in multiply transfused patients with hemophilia. The frequency of hepatitis C among blood donors is also higher than that of hepatitis B and showing downward trend. Thus, the risk of transmitting infection remains in developing countries where there is no ready access to virus inactivation concentrates and have considerably less HIV infection, although it does exist. Ensure implementation of blood donation system, standard screening methods, availability of viral inactivated factor concentrates continuous awareness and enhance safety measures. Prospective follow-up will be needed to see whether the abnormalities that we observed are forerunner of a major epidemic of liver disease and other residual risk of infection. Similarly, the preventive measures for HBV infection should continue, particularly the vaccination programme.
Footnotes
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
Dr. Tahir Shamsi (Medical Director) has received grant from Novo-Nordisk Hemophilia Foundation.
